Abstract
Alzheimer’s disease (AD) is a progressive neurodegenerative disease that involves numerous cellular and biochemical mechanisms resulting in synaptic alterations and extensive neuronal loss. It is primarily characterized by impairment of memory, associated frequently with mood disorders. Continuous studies have shown that insula may be an important target of AD, but neuropathological alterations have not been described extensively. In the present study, we attempted to describe the morphometric and morphological changes of the spines of Reil insula in AD in comparison with normal aging using a silver impregnation technique. We classified spines into 3 types: (1) long neck, (2) short stubby, and (3) other types; and we measured and correlated the length of them in normal controls and in individuals with AD using ImageJ application. Statistical analysis was based on the Student t test on the basis of 360 cells in SPSS v.17.0, and significance was taken as P < .05.
Keywords
Alzheimer’s disease (AD) is a multifarious progressive neurodegenerative disorder involving a number of cellular and biochemical mechanisms resulting in synaptic alterations, 1,2 accumulation of neurofibrillary tangles (NFTs), extracellular deposition of Aβ peptide, and extensive selective neuronal loss. Alzheimer’s disease invades insidiously, affecting millions of humans, leading to an unavoidable tragic outcome, which nowadays became a serious medical challenge for aging population, inducing at the same time many ethical and socioeconomic issues. Alzheimer’s disease is mostly characterized primarily by loss or impairment of memory, associated frequently with behavioral and mood disorders, disorientation, and loss of professional skills, which gradually decline as the disease advances with individuals to become bed bound and reliant to around-the-clock care. 3
The neuropathological background of AD is constituted by neuronal loss, 4 extensive synaptic alterations, 5,6 abnormalities in mitochondrial trafficking and morphology, 1,5 -8,9 and tau pathology, 10 resulting in neurofibrillary degeneration in the form of NFTs, 11 and eventually neurodegeneration 12 and inflammatory responses, 13 leading to the production and release of pro-inflammatory cytokines (interleukin [IL-1β], tumor necrosis factor type α [TNF-α], and Interferon -γ [IFN-γ]) associated with blood–brain barrier disruption. 14 In the advanced stages, the disease is characterized by numerous extracellular extensive deposits of polymers of Aβ peptide in the form of neuritic plaques 15,16 in the neocortex, the hippocampus, and many subcortical structures, which are involved in cognitive function. 1,7
Macroscopically, cerebral atrophy is the essential finding in the majority of cases of AD, being primarily most impressed in the temporal mesolimbic areas and secondary in the frontal, fronto-orbital, and parietal cortex. 9 Deposition of senile plaques and NFTs is obvious under a light microscope in the entorhinal cortex and hippocampus before further spreading in other cortical areas such as the acoustic and visual corteces, the frontal cortex, the temporal cortex, and the cerebellum. 9,17,18
Entering into the pathophysiological spectrum, it is found that AD is associated with reduced levels of acetylcholine, noradrenaline, serotonin, and corticotrophin-releasing factors, as well as levels of glutamate. 19 Cognitive decline may be the result of the degeneration of cholinergic neurons in the basal–forebrain–cholinergic system that projects to the neocortex, hippocampus, and other brain areas. 19,20
The insular cortex plays an important role in regulatory mechanisms ranging from visceral control and sensation to covert judgments regarding inner well-being 21 -23 (Figure 1). Main causes of death in AD include myocardial infarction, heart failure, and pulmonary infections. 24,25 Numerous studies have revealed that all of these failures of visceral organs may be inflamed by autonomic dysfunction, 26 -28 and insular pathology may be the key to this disequilibrium.
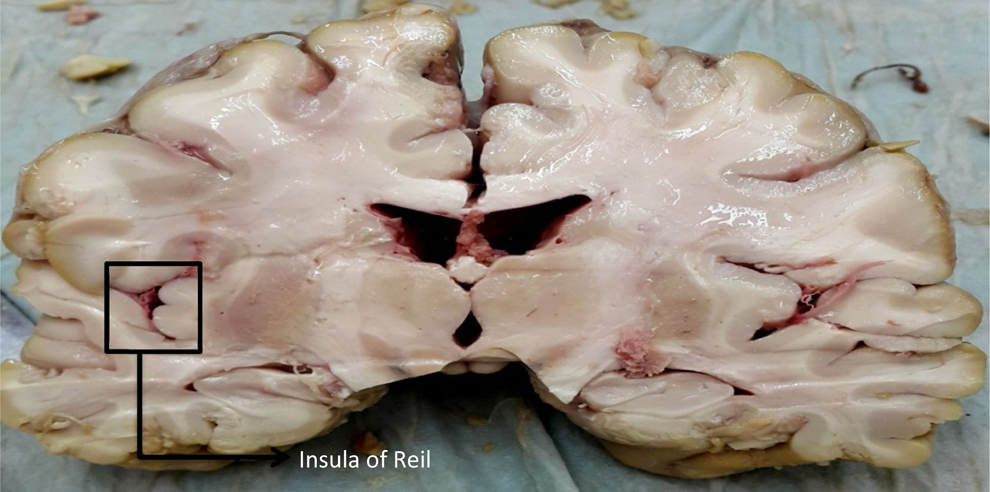
Coronal section of brain from a 69-year-old normal individual illustrating the position of Reil insula.
Many authors have suggested that the insula is implicated in AD. Neuroimaging studies have revealed atrophy of the insular cortex, 29 -31 whereas other studies using the positron emission tomography (PET) scan have shown changes in insular blood flow, 32 monoaminooxidase-B (MAO-B) enzyme or concentrations, 30 muscarinic receptor densities, 33 metabolic activity impairment in FDG-PET scan studies, 34 high activity in monoamine oxidase-B enzyme, 30,35 and decreased cytoplasmic RNA and total protein content in pyramidal neurons of the insular cortex. 36 Insula undergoes substantial pathological alterations in AD, and continuous studies have shown that insula may be an important target urging scientists to enlighten the neuropathological alterations in the insula.
Spines are small specialized postsynaptic dendritic protrusions, where neurons develop synaptic contacts in order to receive and consolidate information. 37 Normally, 3 types of dendritic spines have been described, that is, mushroom-like spines, which are characterized by large head and thin neck, stubby spines, which have a large head but there is no obvious constriction between head and neck, and thin spines, which are slender, filopodia-like protrusions with smaller head and narrow neck. Spines contain specialized proteins, including scaffolding and signaling proteins and ion channels. 38,39 Spine size and morphology depends on the anatomical region of the brain. Thus, longer spines may exist in brain regions where target axons are located away from dendrites such as in the reticular nucleus of thalamus and in gelatinous substance (substantia gelatinosa) of the dorsal horn of the spinal cord. 40 Dynamic alterations in spine morphology affect functional characteristics. For instance, long-term potentiation increases spine head size and helps accommodate higher receptor numbers, while shortening and widening of spine necks leads to a decrease in the electrical resistance of the spine neck, thereby resulting in an increase in glutamate receptors and in larger excitatory postsynaptic potentials. 41
Homeostasis of physiologic spines is disturbed in a number of neuropsychiatric disorders, 42 while the most common example is loss of dendritic spines, which is encountered in most neurodegenerative disorders. Pathologically activated N-methyl-D-aspartate (NMDA) receptors during excitotoxicity 43,44 or upheaval of intraneuronal transportation both may lead to spine loss and would be considered as neuron-autonomous causes. In the same way, a decrease in local protein synthesis at the spine may alter spine densities and morphology. 45 Cases for extraneuronally caused spine loss are trauma or inflammation. Loss of spines usually occurs in trauma, which is mediated by calcineurin, followed by overgrowth of spines. 46 Inflammation may promote the production of IL-1β, which antagonizes the action of BDNF, leading to spine loss. 47 Furthermore, TNFα leads to phosphorylation and upregulation of NMDA-AMPA receptors, 48,49 which subsequently induces excitotoxicity, 48,50 resulting eventually in spine loss. 51
On the other hand, pathological mechanisms can lead not only to spine loss but also an increased stability or density of spines. It is found that patients with fragile X syndrome, which appear with mental retardation, have elevated spine numbers. 52 Additionally, some neuropsychiatric diseases are characterized by elevated spine density in specific brain regions. 42
Individuals with AD, even in the initial stages of the disease, had significantly fewer synapses in the inferior temporal gyrus, which plays an important role in verbal fluency, 53 in the CA1 region of the hippocampus, 54 in the dentate gyrus, 55 and in the posterior cingulate gyrus, which is a cortical region affected primarily in the onset of AD. 56 Recent postmortem studies using intracellular injections of Lucifer yellow in brains of patients with AD revealed that intraneuronal tau accumulation is associated with progressive alteration in dendritic spines. 57
In the present study, we attempted to describe the morphometric and morphological changes of the spines of Reil insula in AD in comparison with normal aging using a silver impregnation technique.
Materials and Methods
Participants
Postmortem tissue samples were obtained from 8 neurologically normal individuals and 7 with AD, all of them aged 52 to 75 years. All brains were immersed in 10% neutral-buffered formalin for at least 25 days.
All brains were examined for their medical history, and all the appropriate information was collected for their physical history. In normal controls, no NFTs or senile plaques were observed, whereas AD brains were classified according to Braak and Braak classification and CERAD score 58 (Table 1). Histologically, AD brains were classified according to those of National Institutes of Health/American Association of Retired Persons. 59
Demographic Features and Braak and Braak Staging of the Brains Used in the Present Study.
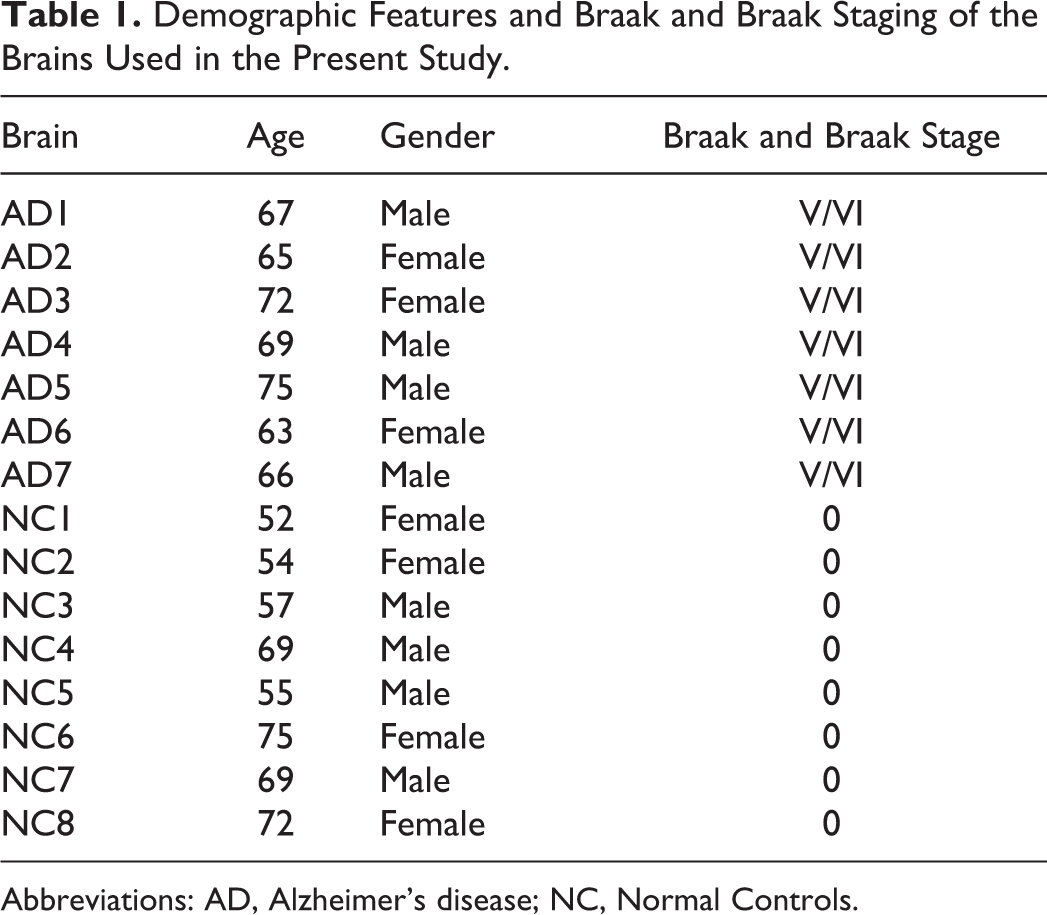
Abbreviations: AD, Alzheimer’s disease; NC, Normal Controls.
Tissue Selection and Processing
All brains were immersed in 10% neutral-buffered formalin at room temperature at least for 40 days before staining. A tissue block from Reil insula was excised (2 cm × 2 cm ×2 cm). Tissue block was coded in order to prevent experimental bias and was used for silver impregnation techniques. Processed tissue was serially sectioned at 120 μm with a vibratome, in a way that the preparation was vertical to the pial surface and perpendicular to the long axis of the gyrus.
Golgi Method
For silver impregnation technique, specimens were immersed in a solution of potassium dichromate (7 g of potassium dichromate and 20 mL of formaldehyde solution 37% in 300 mL of still water) at room temperature in a photoprotective environment. They remained in that solution for 7 days and then they were immersed in an aqueous solution of 1% silver nitrate, where they remained for another 7 days in a photoprotective environment.
After the period of fixation, specimens were embedded in low-melting-point paraffin and cut in thick sections at a range of 100 to 120 μm. After a process of dehydration in graded alcohol solutions, specimens were mounted in Permount, between 2 cover slips, and studied in a Carl Zeiss Axiolab Photomicroscope.
Cell Selection Criteria
Neurons examined for quantitative alterations met the criteria set forth by Jacobs et al, 60 which request uniform staining of neuronal processes, absence of precipitated debris, good contrast between cells and background, and relatively uniform tissue thickness.
Spine Counts
Spine counts were carried out at 360 pictures, which were taken using a Carl Zeiss AxioCam HR, at the standard magnification of ×1000, on a Zeiss Axiostar Plus photomicroscope. Visible spines were counted on the 3 segments of the dendritic field. The first segment, 20 to 30 μm in length, was located on the first-order dendrite, the second segment, 20 to 30 μm in length, on second-order dendrite, and the third one, 40 to 50 μm, along the tertiary dendrite.
We classified spines into 3 types: (1) long neck, (2) short stubby, and (3) other types, and we measured and correlated the length of them in normal controls and in individuals with AD. Overall, spinal density was counted in basal and horizontal dendrites as well as the number of spines in first-, second-, third-, and fourth-order dendrites in normal control individuals compared to those in Alzheimer’s brains.
Dendritic spine density was estimated by counting the number of the dendritic spines on 100 μm of dendritic branches on 10 randomly distributed secondary, tertiary, and quaternary dendritic branches of the apical and the basal dendrites. Thus, we estimated the total number of spines per dendritic length per neuron, as indicative of the dendritic spine density. The densities of dendritic spines as well as their size were estimated manually with the aid of ImageJ application. For our research, we studied and counted 10 slides from each brain.
Statistical Analysis
Statistical analysis was based on the Student t test on the basis of 360 cells in SPSS v.17.0. The significance was taken as P < .05. To ensure that autolysis time did not affect dendritic measurements, 2-tailed Pearson product correlations were performed between all dependent measures and autolysis time.
Results
In normal control brains, we observed that long neck spines represent the majority of this type (85%), while short stubby spines were significantly lower (10%) and in a smaller amount were other types (5%). On the other hand, there was differentiation in spines from individuals with AD, with short stubby spines to predominate against long neck type spines, short stubby 70% and long neck 20%, respectively (Figure 2, Tables 1 and 2, Figures 3 and 4).
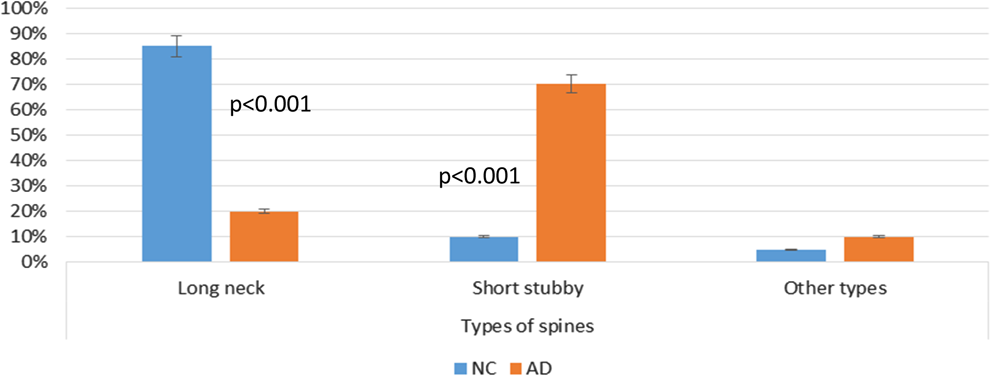
Spinal types comparing normal control and individuals with Alzheimer’s disease (AD). Statistical significance was P < .001.
Spinal Types Comparing Normal Control and Individuals With AD.
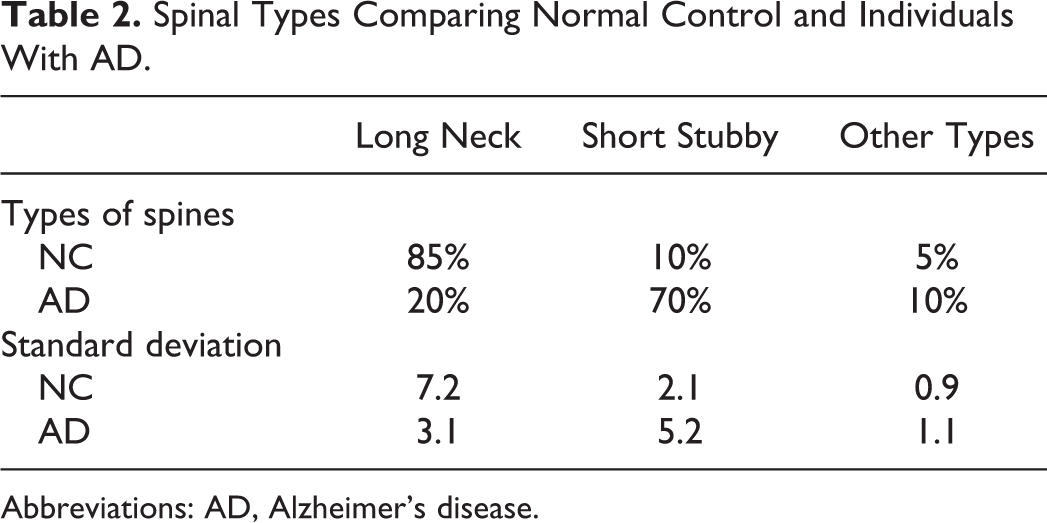
Abbreviations: AD, Alzheimer’s disease.
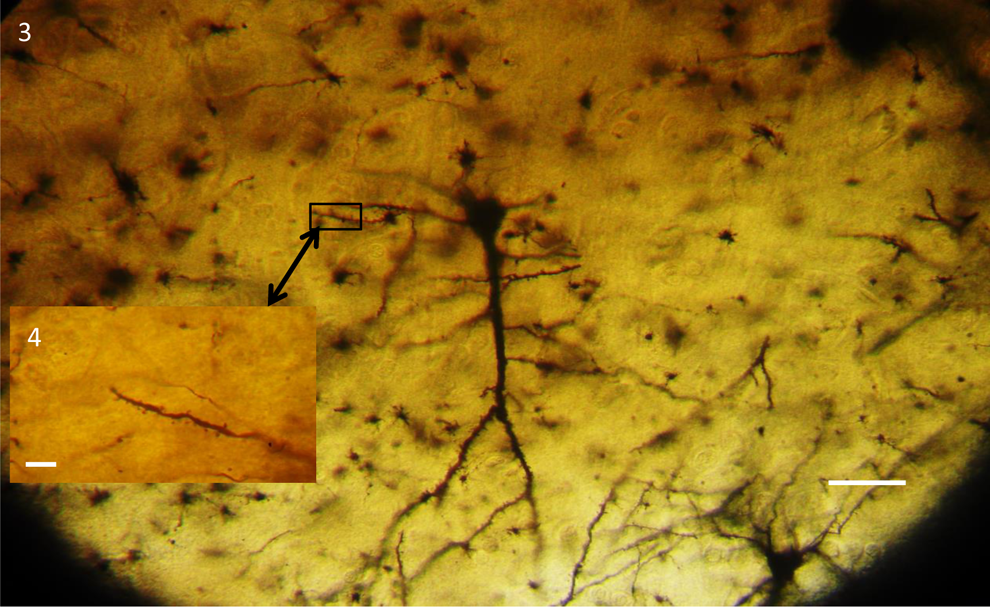
Pyramidal neuron from normal control brain showing long neck spines in apical dendrite. Golgi staining method. Scale bar 5 × 104 and 700 μm, respectively.
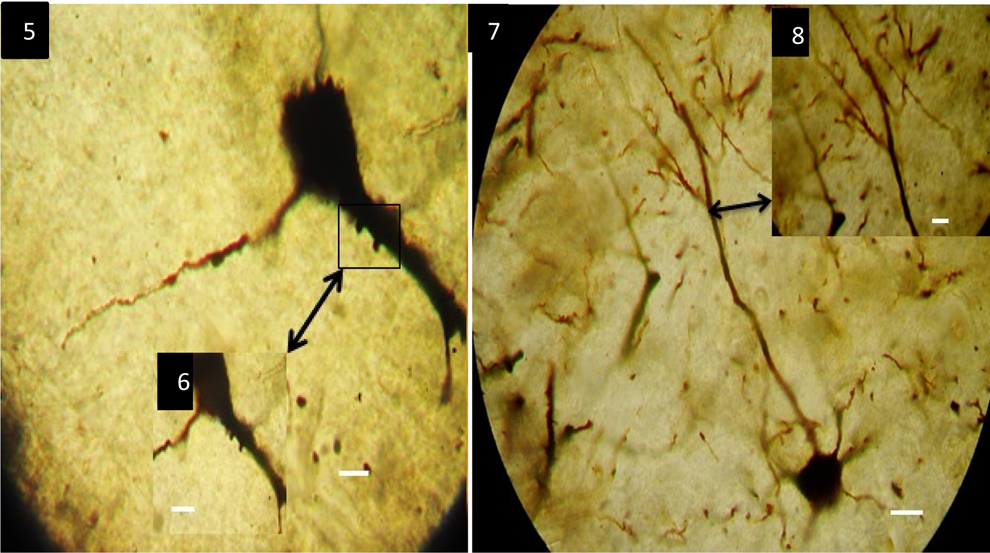
Pyramidal neurons of the insula from individual with Alzheimer’s disease (AD) showing primarily few short stubby spines and secondly significant reduction of spines in basal and especially in distal dendrites. Golgi staining method. Scale bar 3.5 × 103, 2.5 × 103, and 75 × 103, and 6.5 × 103 μm, respectively.
Spinal length between the 2 groups was statistically significant, showing a decrease in the length of individuals with AD in one-third compared to normal control brains. The mean value of spine length was 0.4 μm in AD brains and was 1.2 μm in normal control individuals, respectively (Figure 5).
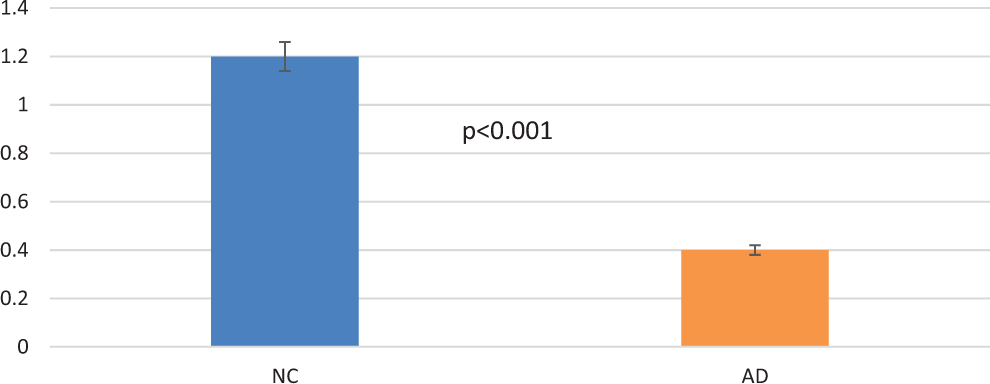
Spinal length between 2 groups showing remarkable reduction in Alzheimer’s disease (AD) brains compared to normal controls. Statistical significance was P < .001.
Moreover, in our study, we revealed a tremendous decrease in the number of dendritic spines and especially in the distal spines in apical and basal dendrites. Impressive finding was that a large number of tertiary and quaternary dendritic branches were apparently denuded of dendritic spines in both basal and horizontal dendrites. In distal branches of basal dendrites, the mean number of spines in AD brains was almost the half of those from normal control brains in quaternary dendritic branches, and there was no significant difference in the other branches (Figure 6). In horizontal dendrites and especially in tertiary dendritic branches, there was a 36.3% reduction in the number of the dendritic spines, and in quaternary branches, there was 50% reduction (Figure 7).
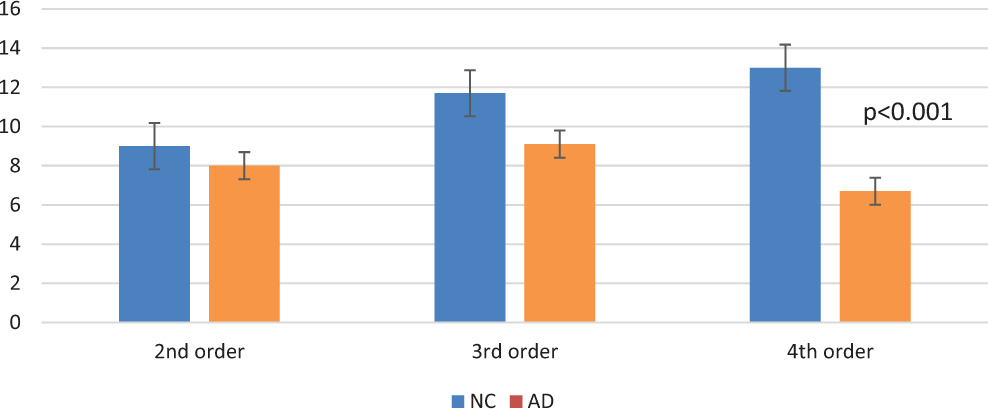
Number of dendritic spines in basal dendrites showing the significant reduction of spines in distal branches. There was statistical significance of P < .001 in fourth-order branches between Normal Controls (NC) and Alzheimer’s disease (AD).
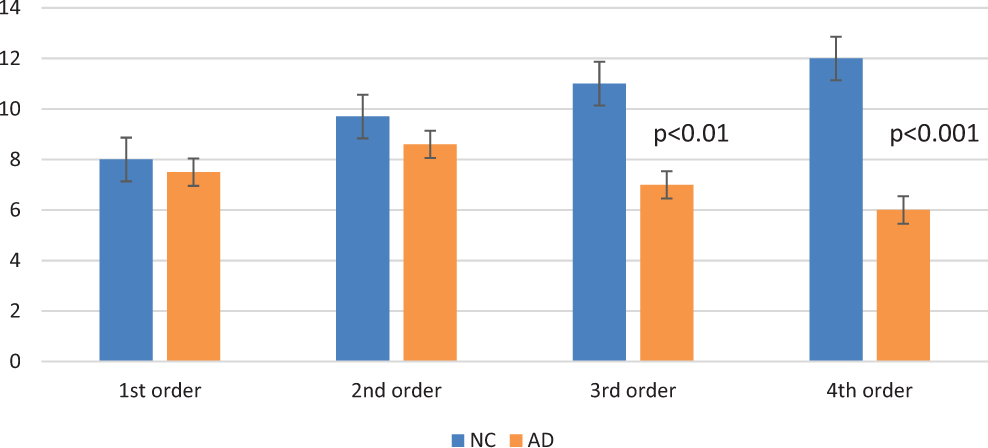
Number of dendritic spines in horizontal dendrites. The reduction in dendritic spines in tertiary and quaternary dendritic branches in Alzheimer’s disease (AD) brains is remarkable. There was a statistically significant reduction in third- and fourth-order apical dendrites in individuals with AD of P < .01 and P < .001, respectively.
Concerning the overall density of dendritic spines, there was a statistically significant difference in normal control and AD brains in horizontal dendrites, showing a 42.5% reduction, as well as there was a 34.5% reduction in dendritic spines in AD brains as compared to normal controls (Figure 8)
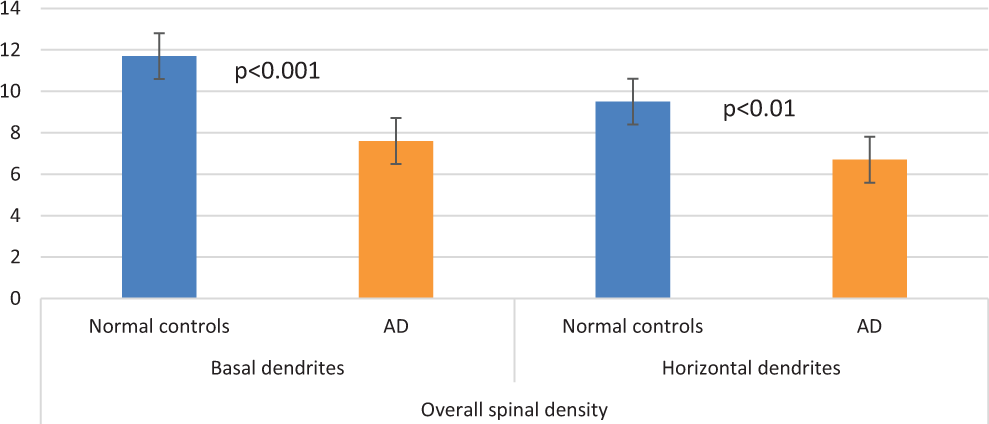
Overall, spinal density in normal controls and in brains with Alzheimer’s disease (AD) in horizontal and basal dendrites of Reil insula. There is a statistically significant reduction in P < .001 and P < .01 of spinal density in AD brains in both basal and apical dendrites.
Discussion
It is unequivocal that the insula is pathologically involved in AD, since extensive neuroimaging studies have revealed atrophy of the insula. 29 -31 In addition, changes in insular blood flow, 32 concentrations of activated MAO-B, 30 changes in biogenic amines and their metabolites, 33 deterioration of metabolic activity in FDG PET scan studies, 34 high activity in monoamine oxidase-B enzyme, 30,35 and decreased neuronal RNA and protein content in pyramidal neurons of the insular cortex 36 advocate for the participation of the insula in the vulnerability due to AD. Thus, insula undergoes substantial pathological alterations in AD, and continuous studies have shown that insula may be an important target urging scientists to enlighten the neuropathological changes within the insula.
It is believed that insula cortex is a protrusive target of AD, and in many cases, insula is the brain region which is highly loaded with NFTs and NPs. 57 These results show that behavioral function subserved by insula is impaired in AD, and insular dysfunction is part of Alzheimer’s clinical phenomenology.
Moreover, insula plays an important role in the sense of taste 61 and this function is impaired in ADs, 62,63 since insula includes neurons that respond specifically to gustatory stimuli. 64,65 Consequently, insular pathology can trigger gustatory dysfunction leading to impairment in recognizing and experiencing disgust. 66,67 Concerning disgust in AD, caregivers witness that patients often develop abnormal behaviors such as producing and eating unpalatable food that most healthy people would find disgusting. 68 This inability to recognize and experience food emotion may be another crucial deficit of AD based on neuronal alterations in insula.
The loss of dendritic spines leads to a substantial decrease in the synaptic contacts and a significant impairment of the pyramidal–interneuronal connectivity, 69 as well as of the connections of the cells of the insula with the neurons of other cortical and subcortical areas, which could be implicated in the modulation of the incoming information. 70 Severe changes are potentially related to the disruption of the cytoskeleton and its associated signal transduction proteins described by English et al. 71
Soluble Aβ peptide and Aβ oligomers stagger neuroplasticity, resulting in failure of synaptic stabilization 72,73 and loss of dendritic spines and synapses in AD brains. Hyperphosphorylated tau protein may also cause noxious effects in neuroplasticity and may underlie its role in the etiology of AD. 74,75
The loss of distal dendrites and dendritic spines leads to a substantial decrease in the synaptic area and synaptic contacts of the pyramidal neurons, and this could contribute to the cognitive decline of AD. 76
Consideration of Methodology
Today, Golgi techniques still remain virtually unique for depiction of neurons and estimation of dendritic morphology in a biplanar or stereoscopic way, viewed by conventional light microscopy, equipped with a computerized image analysis system. Golgi techniques contributed to broadening our views on neuronal development, maturation, dendritic and synaptic plasticity, senescence, and degenerating processes. 77
Conclusion
Loss of dendritic spines in AD is intimately related to synaptic dysfunction and loss of memory, cognition, and professional skills, functions that complete the human being in its wholeness. Today, it is very difficult to understand the complexity of pathophysiology of AD and the mechanisms, which lead to synaptic loss or dysfunction. In this study, we try to reveal that these changes lead to a substantial decrease in the synaptic contacts and impairment of the pyramidal–interneuronal connectivity, as well as partial loss of contact with other cortical and subcortical areas, which might be related to impairment of regulatory mechanisms of insular cortex, ranging from visceral control and sensation to covert judgments, regarding inner well-being.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
