Abstract
Patients with amnestic mild cognitive impairment (aMCI) are at risk for developing Alzheimer’s disease. Due to their prominent memory impairment, structural magnetic resonance imaging (MRI) often focuses on the hippocampal region. However, recent positron-emission tomography data suggest that within a network of frontal and temporal changes, patients with aMCI show metabolic alterations in the precuneus, a key region for higher cognitive functions. Using high-resolution MRI and whole-brain cortical thickness analyses in 28 patients with aMCI and 25 healthy individuals, we wanted to investigate whether structural changes in the precuneus would be associated with cortical thickness reductions in frontal and temporal brain regions in patients with aMCI. In contrast to healthy people, patients with aMCI showed an association of cortical thinning in the precuneus with predominantly left-hemispheric thickness reductions in medial temporal and frontal cortices. Our data highlight structural neuronal network characteristics among patients with aMCI.
Introduction
In the early course of developing Alzheimer’s disease, patients experience prodromal stages with subjective memory impairment and subsequent cognitive deficits, which can be measured by neuropsychological examinations. 1 In these late prodromal disease stages, known as mild cognitive impairment (MCI), everyday activities and functions are still relatively preserved. 1 However, an MCI syndrome can occur in a clinically heterogeneous group of patients. Depending on the neurocognitive profile, an amnestic subtype with predominant memory impairment (aMCI) and a non-aMCI subtype can be differentiated, whereas both entities can be further classified as single- or multidomain. 2,3 The aMCI represents the most frequent subtype. 4 The risk of conversion to Alzheimer’s disease is higher for patients with aMCI when compared to other MCI subtypes. 5 -7 Patients with MCI show annual conversion rates to dementia of up to 12%, 1 but not all patients with MCI experience a cognitive decline. A considerable percentage of patients with MCI remains stable or even reverses to normal cognition. 8,9
Since there are no causal treatment options for neurodegenerative disorders, an accurate characterization of the MCI spectrum is particularly important to predict the clinical course of cognitive impairments and, ultimately, to develop early intervention strategies for specific patient populations. Besides neurocognitive testing and cerebrospinal fluid (CSF) biomarkers, morphometric and metabolic neuroimaging can contribute to a more accurate identification of patients at risk for Alzheimer’s disease. Due to the predominant memory impairment in patients with aMCI, magnetic resonance imaging (MRI) studies often focus on structural and functional changes of the hippocampal region. 10 -13 Increasing evidence from positron-emission tomography (PET) neuroimaging suggests that brain regions outside the medial temporal lobe, specifically the precuneus, show metabolic alterations in patients with aMCI. 14 -16 The precuneus is a region of the posteromedial parietal lobe. 17 It is functionally involved in visuospatial imagery, episodic memory retrieval, self-processing, and consciousness, with a variety of connections to higher associative cortical and subcortical structures. 17 Patients with aMCI show a reduced cortical thickness in the precuneus compared to the non-aMCI subtype. 18 Positron-emission tomography studies demonstrate hypometabolic changes as well as a high susceptibility to amyloid deposition in the precuneus of patients with aMCI. 14 -16 These changes are embedded into a network of metabolic alterations not only in medial temporal but also in frontal brain regions. 14,15,18 Using high-resolution MRI and whole-brain cortical thickness analyses, our aim was to investigate whether these network characteristics can be detected in structural brain imaging data as well. We hypothesized that cortical thickness reductions in the precuneus of patients with aMCI would be associated with cortical thinning in medial temporal or frontal brain regions. We also hypothesized that we would not find such an association among cognitively healthy participants.
Methods
Participants
Fifty-three participants (28 patients with aMCI, mean age: 70.5 ± 6.6 years; 25 healthy controls, mean age: 66.7 ± 7.4 years) participated in this study. We recruited these individuals through advertisements and through our memory clinic. All participants were selected from a cohort of 130 people who underwent neuropsychological testing and MRI scans aimed at investigating Alzheimer’s disease risk factors. We obtained written informed consent and the university’s ethics committee approved the study. All patients with aMCI met standard clinical criteria. 1 Inclusion criteria for patients with aMCI were memory complaints and objective memory impairment. In the Consortium to Establish a Register for Alzheimer’s Disease-Neuropsychological Battery, patients with aMCI were required to score at least 1 standard deviation (SD) below age- and education-adjusted norms in the word list recall subtest. Patients were also required to perform within age- and education-adjusted norms in other cognitive domains (visuoconstructive abilities, language, executive functions) and to present with intact functional activities (patient and spouse/relatives reported no impairment in daily functioning). Control participants (CTLs) presented without subjective and objective cognitive impairments. In both groups, we only included right-handed participants between the ages of 50 and 80 with a school education of at least 8 years. Study exclusion criteria were psychiatric or neurological disorders other than the MCI, a systemic disease affecting brain functioning, current psychotropic medication, and MRI contraindications, such as metallic implants or cardiac pacemakers.
Procedures
Magnetic resonance imaging scanning was performed on a GE Signa HDxt (General Electric Health Care, Waukesha, Wisconsin) 3 T whole-brain MRI scanner. We acquired a T1-weighted magnetization-prepared rapid acquisition gradient-echo scans (repetition time: 2300 milliseconds, echo time: 2.93 milliseconds, voxel size: 1mm 3 , 160 slices, field of view (FOV): 256 mm). Reconstruction of cortical surfaces and thickness measurements were performed using the FreeSurfer toolkit (http://www.surfer.nmr.mgh.harvard.edu). For a detailed description of these methods, see references. 19 -21 Briefly, cortical thickness data were obtained using automated cortical parcellation techniques within the FreeSurfer toolkit. 22 The boundaries of specific brain regions, such as the precuneus, are based on the Destrieux anatomical atlas. 23 Thickness was measured by reconstructing grey and white matter boundaries and subsequent distance measures at each point (vertex) across the cortex. The method uses intensity and continuity information from the 3-dimensional volume in segmentation and deformation procedures to create representations of cortical thicknesses. The maps are produced using spatial intensity gradients across tissue classes and are not restricted to the original data’s voxel resolution. Therefore, submillimeter differences between groups can be detected. 20 The resulting thickness maps were smoothed using a Gaussian kernel of 10 mm full width at half-maximum and averaged across participants with a nonrigid high-dimensional spherical averaging method to align cortical folding patterns. Cortical thickness measures can be mapped on an “inflated” brain surface for visualization without interference from cortical folding. This procedure has been validated by histological and manual measurements. 24,25
FreeSurfer statistical analyses include a general linear model to control the effect of selected variables, such as precuneus cortical thickness, on regional thickness across the whole brain. We estimated the associations between precuneus thickness and whole-brain thickness using the “Query, Design, Execute, Contrast (QDEC)” application included in the FreeSurfer distribution. This application allows calculating and visualizing group statistics on brain morphometry data. We first estimated the association between precuneus thickness and whole-brain thickness for each group separately. Direct comparisons were subsequently computed with all participants in the same statistical model and “group” (aMCI/control) as a fixed factor. Although not statistically significant, there was a trend for older age among patients with aMCI when compared to controls. We, therefore, used age as a covariate in all analyses. We applied a false discovery rate correction for multiple comparisons at a threshold of P < .05. False discovery rate correction is an effective solution to the threshold selection problem in the analysis of neuroimaging data, as it provides an interpretable and adaptive criterion with higher power than other methods for multiple comparisons (eg, Bonferroni). 26
Results
Our participants did not significantly differ in mean age, gender distribution, or educational status. As expected, performance in the Mini-Mental State Examination (MMSE) was better in CTLs when compared to patients with aMCI (Table 1). There was no correlation between the MMSE score and left/right hemispheric precuneus thickness in both participant groups.
Demographic Characteristics and MMSE Scores.
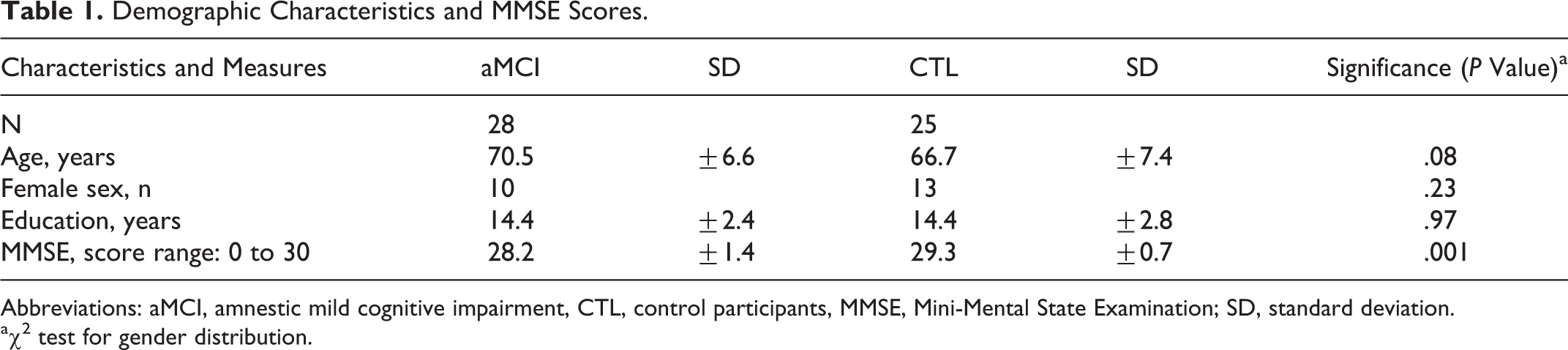
Abbreviations: aMCI, amnestic mild cognitive impairment, CTL, control participants, MMSE, Mini-Mental State Examination; SD, standard deviation.
aχ2 test for gender distribution.
Cortical thickness in the precuneus was significantly reduced among patients with aMCI (left hemisphere—aMCI: 2.11 mm, CTL: 2.26 mm, P = .008; right hemisphere—aMCI: 2.11 mm, CTL: 2.25 mm, P = .004). We then investigated whether precuneus cortical thickness would be associated with cortical thinning elsewhere in the brain, separately for each group. Although control participants did not show such an association, patients with aMCI exhibited widespread cortical thinning across all cortices. When we directly compared this association between the groups, we found a predominantly left-lateralized effect in frontal and temporal cortices (Figure 1). Finally, we investigated whether structural differences in medial temporal regions, such as the hippocampus, would be associated with similar network effects across the brain. We did not find such associations in both groups. There was no significant difference in hippocampal thickness between both groups.
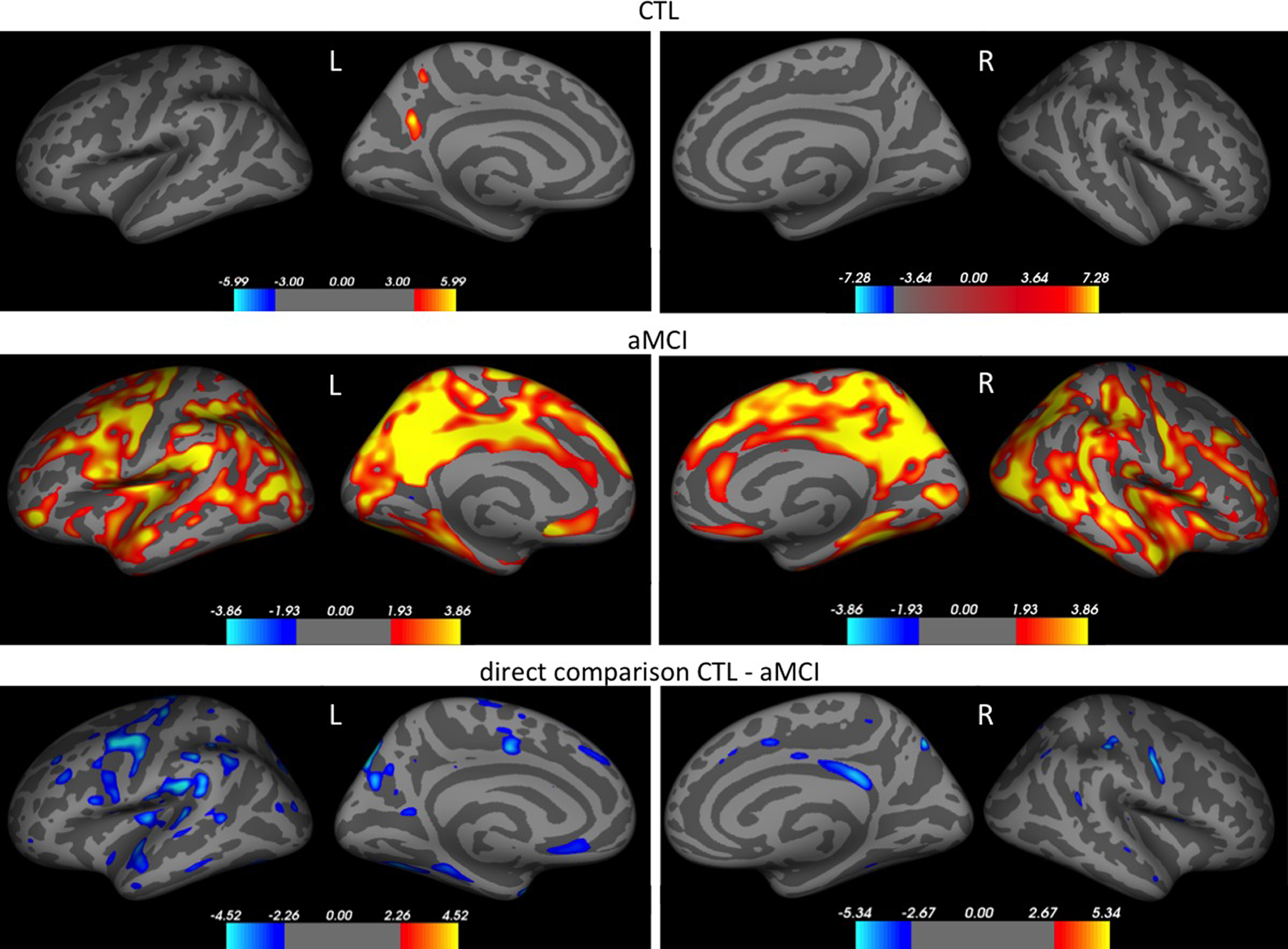
Correlation between precuneus thickness and whole-brain cortical thickness. Colors highlight areas with a significant correlation (for separate analyses of participants with amnestic mild cognitive impairment [aMCI] and CTL) and areas with significant correlation differences (for direct group comparison). Red color represents a positive correlation; blue color a negative correlation. Color bars: colors indicate statistical significance, unit: P value presented as −log10(P) with an FDR correction of P < .05.
Discussion
In this study, we show an association of precuneus cortical thinning with thickness reductions in predominantly left-lateralized frontal and temporal cortices of patients with aMCI. Among cognitively healthy individuals, cortical thickness of the precuneus was not associated with similar thickness changes in frontal and temporal regions. The hippocampal cortical thickness was not associated with cortical thinning elsewhere in the brain in both groups.
Although the majority of structural MRI studies focuses on the hippocampal region changes of patients with MCI, 10,11,13 Csukly and colleagues show reduced cortical thickness in the precuneus of patients with aMCI when compared to participants having the non-aMCI subtype. 18 The precuneus plays an essential role in a variety of highly integrated brain functions, such as visuospatial processing, episodic memory retrieval, and correlates of self-consciousness. 17 It has diverse reciprocal connections with frontal, temporal, and parietal cortices. 17 In aMCI development, precuneus function changes may have pathophysiological relevance, reflected by cortical thinning, hypometabolic alterations, and early amyloid deposition in this region. 14 -16
In the search for biological characteristics, decreased hippocampal volume is the most robust MRI marker for an imminent conversion of MCI to dementia. 10 However, PET data suggest that metabolic changes in patients with MCI are more widespread compared to structural MRI findings, which could indicate functional alterations preceding brain atrophy. 14,15 Particularly, interesting PET findings in patients with aMCI are metabolic and molecular changes outside the medial temporal lobe, where the earliest Alzheimer’s disease-associated neuropathological changes are known to arise. 27 Positron-emission tomography data highlight an involvement of the precuneus in the early course of a neurodegenerative process—within a wider network of metabolic abnormalities in frontal and temporal brain regions among patients with MCI. 15,28 Coutinho and colleagues hypothesize that precuneus hypometabolism would be more specific for patients with aMCI, whereas additional frontal hypometabolism is associated with the non-aMCI subtype. 16 Although we cannot directly compare our findings with PET data, our results suggest structural network changes in patients with aMCI involve the precuneus, frontal, and temporal brain regions. Additional structural MRI investigations of non-aMCI patients would be necessary to differentiate brain changes across MCI subtypes. Using structural MRI, Csukly and colleagues also revealed thinning of the entorhinal cortex, the fusiform gyrus, the precuneus, and the isthmus of the cingulate gyrus in patients with aMCI when compared to non-aMCI patients and healthy controls. 18 Yang and colleagues found reduced regional temporal and frontal volume reductions among others when comparing patients with aMCI to healthy controls, whereas no significant volumetric differences could be detected between non-aMCI patients and controls. 29 In line with our results and existing PET data, this suggests that brain structure changes in patients with aMCI are less localized than it could be assumed given the characteristic memory impairment.
Study limitations include the unavailability of CSF or PET data for our study participants. Our sample size was small, which could have prevented the detection of additional effects on cortical thickness. Future research could integrate CSF data as well as structural and functional neuroimaging to define MCI subtypes based on clinical and biological characteristics. However, we highlight structural network changes associated with cortical thinning in the precuneus of patients with aMCI that point to a potentially valuable biomarker, which is also accessible for most clinicians when compared to metabolic neuroimaging.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Linn has received research support from B. Braun Stiftung, Friedrich-Baur-Stiftung, and from the Förderprogram für Forschung und Lehre and travel expenses and/or honoria for educational lectures from Bayer Healthcare, Phillips Healthcare, and Bracco. Dr Donix has received research support from the Roland Ernst Stiftung, a consulting fee from Trommsdorff, and a lecture fee from Mundipharma.
