Abstract
Urinary Alzheimer-associated neural thread protein (AD7c-NTP) is a potential biomarker of Alzheimer disease (AD) or mild cognitive impairment (MCI). It is still unclear whether the urinary levels of AD7c-NTP are different between patients with amnestic MCI (aMCI) and nonamnestic MCI (naMCI). The present study aimed to explore the differences in urinary levels of AD7c-NTP between patients with aMCI and naMCI. Forty-six patients with MCI were divided into aMCI group (n = 23) and naMCI group (n = 23). The mean level of urinary AD7c-NTP in the aMCI group (32.75 ± 10.0 µg/mL) was significantly higher than that in the naMCI group (25.34 ± 9.0 µg/mL; P = .011). As far as we know, the present study is the first to show that individuals with aMCI have higher levels of urinary AD7c-NTP than those with naMCI, suggesting that urinary AD7c-NTP may be a potential biomarker to help identify patients with aMCI and naMCI.
Keywords
Introduction
Mild cognitive impairment (MCI) is a heterogeneous clinical concept that represents a transitional state between normal cognitive aging and dementia. 1 Mild cognitive impairment with memory domain impairment is called amnestic MCI (aMCI), while patients with nonmemory domain impairment are considered as having nonamnestic MCI (naMCI). 2 Amnestic MCI is said to have a high likelihood of conversion to Alzheimer disease (AD), and individuals with naMCI are considered to have a higher likelihood of conversion to various types of non-AD dementia. 1,2 Recent advances generated from AD biomarker studies, such as amyloid positron emission tomography (PET) as well as cerebrospinal fluid (CSF) amyloid β 1-42 fragment (Aß1-42), total tau (T-tau), and phosphorylated tau (P-tau) make it possible to detect AD much earlier than previously possible, when activities of daily living are nearly preserved. 3 A National Institute on Aging-Alzheimer’s Association (NIA-AA) international working group proposed the concept of MCI due to AD to categorize these group of patients. 1,4 Patients with MCI due to AD are at a high risk of converting to AD. 1,4 The NIA-AA working group suggested that effective biomarkers can assist the early and accurate detection of MCI due to AD. 1,3,4 Various biomarkers have been discussed for the early and accurate diagnosis of MCI with high likelihood of conversion to AD. Amyloid PET as well as CSF Aß1-42, T-tau, and P-tau protein reflect the neuropathological changes associated with AD, such as senile plaque and neurofibrillary tangles. 1,3,4 However, these biomarkers are of limited utility for a few reasons, including the high cost and radiation exposure associated with PET as well as the wide variability and invasive lumbar puncture procedure for CSF biomarker studies.
Increased urinary levels of Alzheimer-associated neural thread protein (AD7c-NTP), a 41-kD membrane-associated phosphoprotein, have been reported in patients with AD and MCI. 5 -9 These findings suggest that urinary AD7c-NTP could be a useful biomarker for identifying patients with MCI who have a high likelihood of conversion to AD. However, it remains unclear whether the levels of urinary AD7c-NTP among patients with aMCI are different from those with naMCI. The present study aimed to determine whether the levels of urinary AD7c-NTP could assist to distinguish patients with aMCI from those with naMCI.
Methods
Participants
This study examined 104 patients diagnosed with MCI at the Memory Disorder Clinic in the Department of Neurology at Myongji Hospital, in Goyang, South Korea. This study was approved by the institutional review board of the Myongji Hospital, and written informed consent was obtained from each participant. Based on the clinical and neuropsychological evaluation, eligible participants were divided into aMCI and naMCI groups. Diagnostic categorization of the participants was done using NIA-AA international working group criteria. 1 All of the enrolled participants were asked to complete a questionnaire using standardized methods. The diagnostic evaluation included a detailed medical history, physical and neurological examinations, standardized cognitive domain-specific comprehensive neuropsychological tests, magnetic resonance imaging (MRI), and blood tests.
The inclusion criteria were as follows: age 45 years or older; diagnosed according to the NIA-AA international working group MCI criteria; brain MRI findings available within 6 months of diagnosis; and a Clinical Dementia Rating (CDR) scale score of 0 or 0.5.
The exclusion criteria were as follows: a history of significant hearing or visual impairment; a history of neurological or psychiatric illness, such as Parkinson’s disease, seizure disorder, head trauma, schizophrenia, mental retardation, major depression or mania; the concomitant use of psychotropic medications; or a history of significant alcohol and other substance abuse. Patients with major territorial stroke other than ischemia were also excluded.
Cognitive Assessment and Neuropsychological Tests
For cognitive and functional independence screening, we used the Korean version of the Mini-Mental Status Examination (K-MMSE) and Seoul instrumental activities of daily living (SIADL). 10,11 All patients underwent neuropsychological tests using a standardized neuropsychological battery called the Seoul neuropsychological screening battery (SNSB). The SNSB contains tests for language, visuospatial ability, memory, and frontal executive function. 12 The neuropsychological tests were as follows: the short form of the Korean version of the Boston naming test (SK-BNT), Rey–Osterrieth complex figure test (RCFT) copying, Seoul verbal learning test (SVLT) 20-minute delayed recall of 12 items, the controlled oral word association test (COWAT), and the Korean version of the color word Stroop test (K-CWST). We investigated possible problem behaviors using the neuropsychiatric inventory (NPI). Additionally, we used the Hachinski ischemic scale (HIS), CDR, CDR-sum of boxes (SOB), global deterioration scale (GDS), and the geriatric depression scale (Ger DS). We evaluated the 4 different cognitive domains—memory, language, visuospatial ability, and frontal executive function—for each participant. We determined the presence or absence of cognitive impairment in each participant based on the participants’ clinical history and SNSB performance. Cognitive decline was determined as an education- and age-adjusted score below the 16th percentile on the SNSB. The domain assessment parameters were as follows: the SVLT delayed recall score for memory, the SK-BNT score for language, the RCFT copy score for visuospatial ability, and phonemic COWAT or K-CWST color reading time for frontal executive function. Clinical diagnoses were made without reference to urinary AD7c-NTP levels. The participants who scored below the 16th percentile in terms of education- and age-adjusted SVLT delayed recall were assigned to the aMCI group, and the remaining participants were assigned to the naMCI group.
Magnetic Resonance Imaging and Assessment of White Matter Hyperintensities
The brain MRI scans included axial T1-, T2-weighted, gradient echo, fluid-attenuated inversion recovery (FLAIR), and T1-weighted coronal slices. The white matter hyperintensities (WMHs) on the T2 or FLAIR axial images were rated as minimal, moderate, and severe using the scale previously developed by the Clinical Research Center for Dementia of South Korea study. 13
Urine Specimen Collection and Determination of Urinary AD7c-NTP Levels
We used urinary AD7c-NTP kits according to a standard protocol described by Levy et al. 14 A first morning urine sample of 30 to 50 mL was obtained from each participant in a clean urine collection cup. Urine samples were blindly coded without any knowledge of participants’ identities, clinical information, or diagnosis.
Exclusion criteria for urine sample collection were as follows: evidence of sample contamination or a urinary tract infection (urinary glucose, protein, cells, bacteria, crystalluria, hematuria, pyuria, urine with ketones, nitrites, bilirubin, or urobilinogen, abnormal specific gravity, or abnormal pH), not a first morning void, urinary creatinine concentration greater than 225 mg/dL, urinary creatinine concentration less than 50 mg/dL, or urine that was frozen before processing.
Patients who did not provide a sample that met criteria for urinary AD7c-NTP testing were excluded from the final statistical analysis. All urinary procedures were done in a central laboratory (Neodin, Seoul, South Korea). The analyses were performed using urinary AD7c-NTP assay kits (AlzheimAlert) purchased from Nymox Pharmaceutical Corp (Quebec, Canada). The measurement unit of this kit is expressed by the microgram unit
We used the competitive affinity of an enzyme-linked immunosorbent assay for the detection of AD7c-NTP after centrifugation and filtration. A microtiter plate was coated with a bispecific receptor antibody that has high affinity for rabbit immunoglobulin G (IgG) and AD7c-NTP. Wells in the AD7c-NTP kit microtiter plate were filled with 200 μL of a standard solution or the treated samples of patients with aMCI and naMCI. Alkaline phosphatase was added to the plate, which was incubated overnight at 4°C and washed 6 times. Then, 150 µL of paranitrophenol phosphate was added to each well, and the plate was incubated for 1 hour at room temperature and then washed 6 times. The AD7c-NTP in each participant’s urine samples competed with rabbit IgG. In the presence of AD7c-NTP, rabbit IgG binding decreases in proportion to the amount of AD7c-NTP in the samples. The labeled IgG conjugates with alkaline phosphatase, which reacts with paranitrophenol phosphate to produce an optical density (OD) signal. The OD signal was detected using a plate reader at 405 nm. In the absence of AD7c-NTP, there is no competition, and the absorbance is high (high OD signal). In the presence of AD7c-NTP, there is competition that produces less rabbit IgG alkaline phosphatase binding, and lower absorbance (low OD signal, supplemental material). 14
Statistical Analysis
The comparison of demographic, clinical, and neuropsychological variables was performed using the independent-sample t test, Mann-Whitney U test, analysis of variance with Tukey post hoc analyses, the χ2 test, Fisher exact test, or Pearson correlation. The sensitivity (percentage of the aMCI group with a positive test result) and specificity (percentage of the naMCI group with a negative test result) of urinary AD7c-NTP were calculated using receiver–operator characteristic (ROC) curve analysis. Positive-predictive values (probability that a patient is actually diagnosed as aMCI when the test is positive) and negative-predictive values (probability that a patient is not diagnosed as aMCI when the test is negative) were calculated. The association between urinary AD7c-NTP levels and demographic, cognitive, and neuropsychological variables were assessed using Pearson correlation. Data analysis was carried out using Statistical Package for Social Sciences for Windows version 22.0 (IBM Corp, Armonk, New York). Data are expressed as mean ± standard deviation in continuous variables or number (percentage) in categorical variables. All the tests were 2-tailed, and values of P < .05 were regarded as statistically significant.
Results
Participant Demographics and Clinical Characteristics
One hundred four participants qualified for initial inclusion. However, 58 (55.8%) participants were excluded. Of these, 41 were excluded because of inappropriate urine samples (contaminated, proteinuria, hematuria, or an abnormal creatinine level), and of these 41 participants, 23 were excluded because of a low creatinine level (<50 mg/dL), 3 were excluded because of a high creatinine level (>250 mg/dL), and 15 were excluded for other reasons such as refusing urine sample delivery (n = 13) or withdrawing or refusing consent (n = 2). Finally, a total of 46 participants were recruited for this study and divided into an aMCI (n = 23) group and an naMCI group (n = 23; Figure 1). There were no significant differences in age, sex, years of education, disease duration, height, or weight between the 2 groups (Table 1). The demographic and clinical characteristics of all participants are summarized in Table 1. Minimal, moderate, and severe WMHs were observed in 29 (64.4%), 12 (26.7%), and 4 (8.9%) patients with MCI, respectively. No significant WMH difference was noted between the aMCI and the naMCI group (P = .390).

Enrollment of the participants. aMCL indicates amnestic mild cognitive impairment; naMCI, nonamnestic mild cognitive impairment.
The Demographics and Clinical Characteristics of all Participants.a
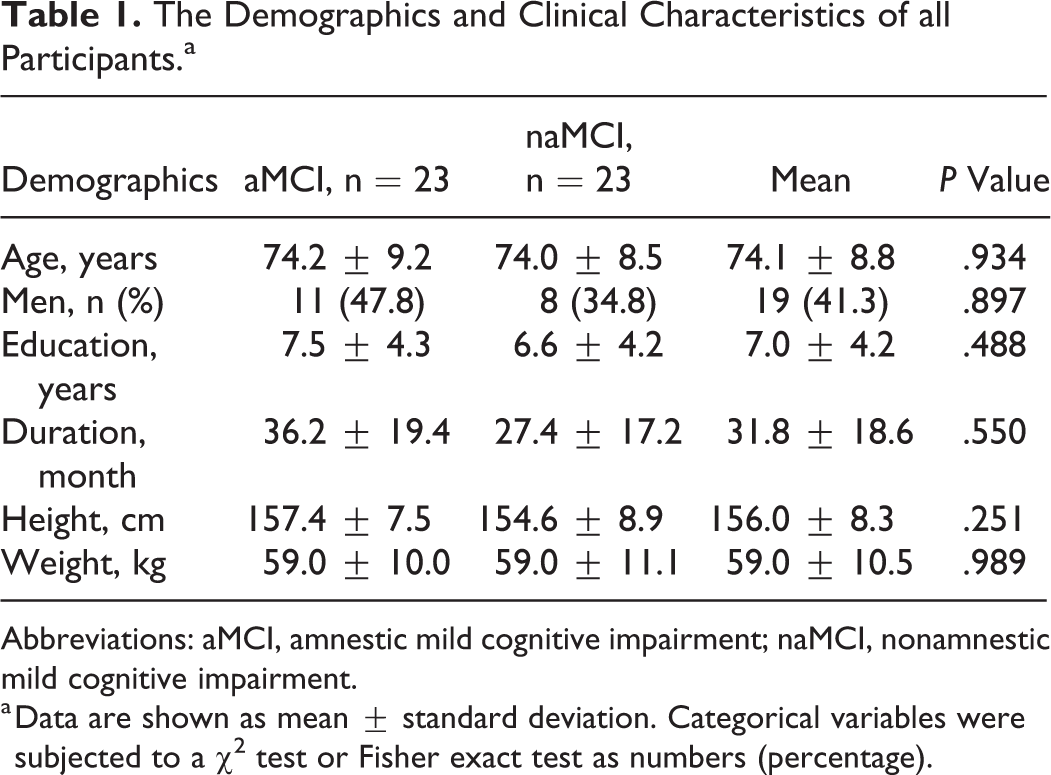
Abbreviations: aMCI, amnestic mild cognitive impairment; naMCI, nonamnestic mild cognitive impairment.
a Data are shown as mean ± standard deviation. Categorical variables were subjected to a χ2 test or Fisher exact test as numbers (percentage).
General Cognitive Profiles and Neuropsychological Evaluation
Between the 2 groups, there was no significant difference in terms of general cognitive profiles and functional abilities, such as K-MMSE, SIADL, CDR, CDR-SOB GDS, Ger DS, Sum of NPI score, and HIS. The mean K-MMSE scores of the patients with aMCI and naMCI were 21.9 ± 4.3 and 22.9 ± 3.5, respectively (P = .397). The mean SIADL scores of all participants were 5.6 ± 2.0 (5.7 ± 3.4 of aMCI and 5.5 ± 2.2 of naMCI; P = .321). The differences in the neuropsychological test scores between the aMCI and the naMCI groups are summarized in Table 2. There were no significant differences in neuropsychological test profiles between the aMCI and the naMCI groups, except SVLT delayed recall score (P = .002).
General Cognitive, Functional, and Neuropsychological Profiles of Patients With Amnestic Mild Cognitive Impairment and Nonamnestic Mild Cognitive Impairment.a

Abbreviations: aMCI, amnestic mild cognitive impairment; naMCI, nonamnestic mild cognitive impairment; K-MMSE, Korean version of the mini-mental status examination; SIADL, Seoul instrumental activities of daily living; CDR, clinical dementia rating; CDR-SOB, clinical dementia rating-sum of boxes; GDS, global deterioration scale; GerDS, geriatric depression scale; K-CWST, Korean version of the color word Stroop test; NPI, neuropsychiatric inventory; HIS, Hachinski ischemic scale; SK-BNT, short Korean version of the Boston naming test; RCFT, Rey complex figure test; SVLT, Seoul verbal learning test; COWAT, controlled oral word association test; K-SWST. Korean version of the color word Stroop test.
a Data are shown as mean ± standard deviation. Categorical variables were subjected to a χ2 test or Fisher exact test as numbers (percentage).
Urinary AD7c-NTP Measurements
The mean urinary AD7c-NTP level for the aMCI group (32.7 ± 10.0 µg/mL) was higher than of the naMCI group (25.3 ± 9.0 µg/mL, P = .011). The box plot in Figure 2 shows the distribution of urinary AD7c-NTP measurements for aMCI and naMCI groups. Urinary AD7c-NTP level was similar for men (mean, 31.6 ± 7.7 µg/mL) and women (mean, 27.3 ± 11.4 µg/mL, P = .158) in the 2 groups.

Comparison of the level of the urine Alzheimer-associated neural thread protein (AD7c-NTP) between patients with amnestic mild cognitive impairment (aMCI) and nonamnestic mild cognitive impairment (naMCl). The mean values of urine AD7c-NTP are 32.7 ± 10.0 µg/mL in aMC1 group (n = 23) and 25.3 ± 9.0 µg/mL in naMCI group (n = 23).
An optimized threshold of 26.9 µg/mL was derived from ROC curve analysis maximizing the area under the curve (Figure 3). The area under the curve was 68.5%, and the standard error was 7.9%. The sensitivity and specificity of urinary AD7c-NTP were found to be 73.9% and 60.9%, respectively, for the diagnosis of aMCI. There were 73.9% (17/23) of participants with aMCI who had elevated urinary AD7c-NTP levels, compared to 39.1% (9/23) of participants with naMCI who had elevated urinary AD7c-NTP. The likelihood ratio for a positive test result (>26.9 µg/mL) was calculated using the probability of test-positive aMCI (17/23) and naMCI (9/23), and the likelihood ratio for a negative test result (<26.9 µg/mL) was calculated using the probability of test-negative aMCI (6/23) and naMCI (14/23). The positive and negative likelihood ratios of the urinary AD7c NTP test were 1.89 and 0.43, respectively.

Receiver–operating characteristic (ROC) curve depicts sensitivity and 1 minus specificity level of urinary Alzheimer-associated neural thread protein (AD7c-NTP) for discrimination between amnestic mild cognitive impairment (aMCI) and nonamnestic mild cognitive impairment (naMCI). The area under the curve (AUC) is 0.69 (95% confidence interval [CI]: 0.53-0.84) with sensitivity 73.9% and sensitivity 60.9%.
To evaluate the effect of CDR score on urinary AD7c-NTP level, we compared the urinary AD7c-NTP levels of participants who had CDR scores of 0 with those of participants who had CDR scores of 0.5. The mean urinary AD7c-NTP level for CDR 0 patients with MCI (n = 4) was 17.3 ± 4.2 µg/mL, in contrast to a mean urinary AD7c-NTP level of 30.2 0.2 ± 9.8 µg/mL among patients with MCI with CDR scores of 0.5 (n = 42). Figure 4 shows the distributions of urinary AD7c-NTP results for CDR 0 and CDR 0.5 patients with MCI (P = .010).
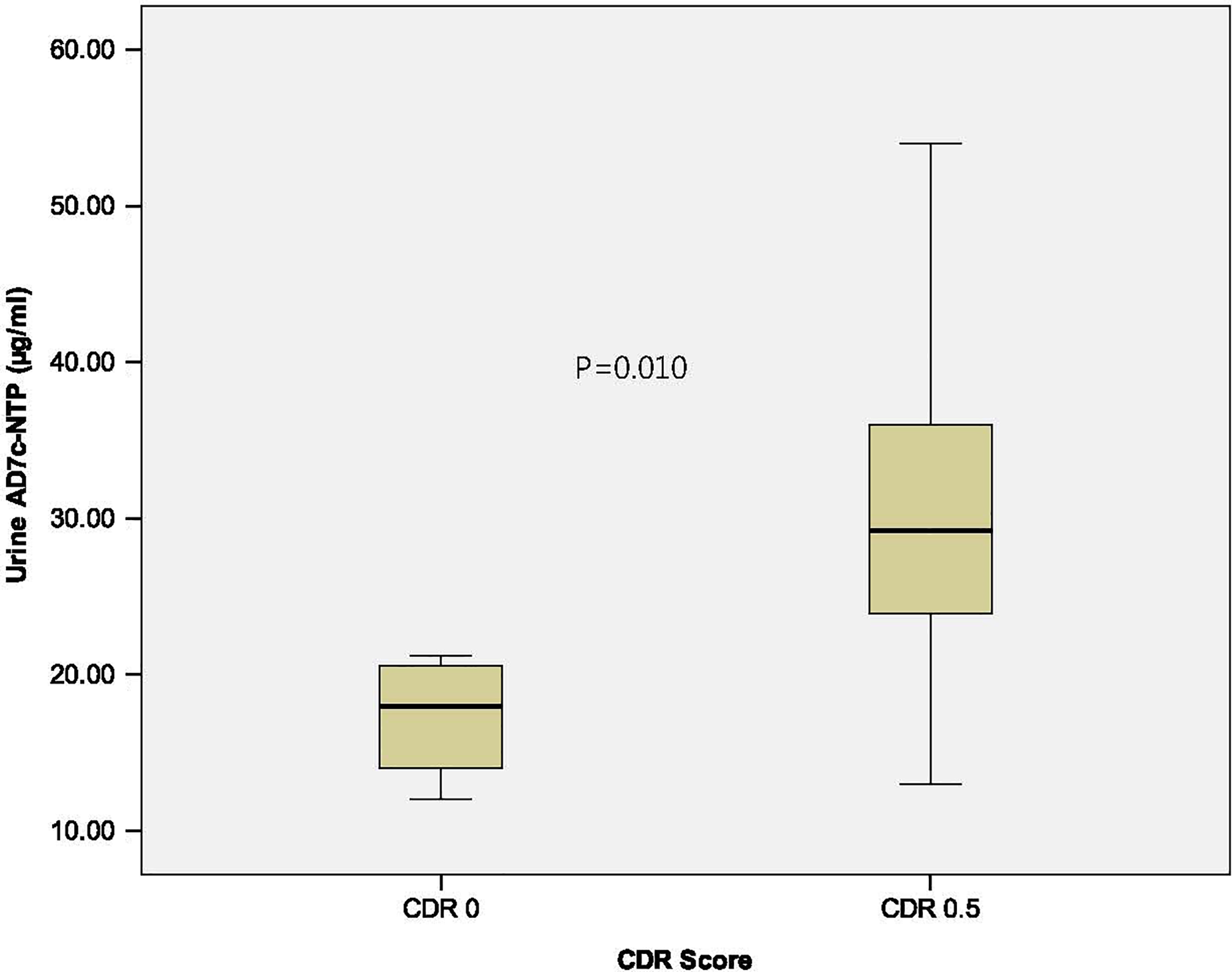
Comparison of the level of the urinary Alzheimer-associated neural thread protein (AD7c-NTP) between patients with clinical dementia rating (CDR) 0 and CDR 0.5. The mean values of urine AD7c-NTP are 17.3 ± 4.2 µg/mL in CDR 0 group (n = 4) and 30.2 ± 9.8 µg/mL in CDR 0.5 group (n = 42).
There was no significant difference in the urinary AD7c-NTP level among the minimal (n = 29, 28.4 ± 9.6 µg/mL), moderate (n = 12, 32.7 ± 10.7 µg/mL), and severe (n = 5, 26.5 ± 10.0 mg/mL, P = .381) WMHs groups. The CDR score 0 and 0.5 (r = .362, P = .013), and K-CWSP color reading time (r = .356, P = .016) showed significant correlation with the urinary AD7c-NTP level.
Discussion
The present study aimed to determine whether urinary AD7c-NTP levels could assist to distinguish patients with aMCI from those with naMCI. It is often unclear whether a patient diagnosed with MCI is at risk of dementia conversion. 1,3 Alzheimer-associated neural thread protein is closely linked with the tau protein and is positively associated with P-tau accumulation in the brain and CSF in patients with AD. 15 Recently, it has been reported that AD7c-NTP is elevated in the urine of patients with MCI as well as those with AD. 5 -9 The urinary AD7c-NTP has been shown to have a high sensitivity and specificity for the diagnosis in AD and MCI. 9,16 -18 Several studies have suggested that urinary AD7c-NTP is a useful biomarker and may be widely used to screen the risk of elderly individuals with MCI and AD. 15 -20 However, to the best of our knowledge, no previous urinary AD7c-NTP level studies tried to distinguish patients with aMCI from patients with naMCI using urinary AD7c-NTP as a biomarker. Moreover, most previous studies regarding the urinary AD7c-NTP levels of the patients with MCI were performed with relatively brief neuropsychological tests that are not cognitive domain-specific. 5 -9 More comprehensive cognitive domain-specific standardized neuropsychological tests are required to separate patients with MCI into amnestic and nonamnestic groups. Our study was performed through a neuropsychological screening battery of different cognitive domains to divide patients with MCI into amnestic and nonamnestic groups.
We demonstrated that urinary AD7c-NTP was elevated among patients with aMCI (32.7 ± 10.0 µg/mL) relative to patients with naMCI (25.3 ± 9.0 µg/mL). The overall mean concentration of urinary AD7c-NTP in patients with MCI (29.0 ± 10.1 µg/mL) was a little high compared to that found in an earlier study. 5 The cutoff value (26.9 µg/mL) was also higher than previous studies. 5,6,21,22
Earlier studies have found significant correlations between urinary AD7c-NTP concentration and MMSE score, Blessed dementia scale score, and NPI agitation score. 9,23,24 In the present study, urinary AD7c-NTP level showed a significant correlation with CDR score 0 and 0.5 (r = .362, P = .013) and K-CWSP color reading time (r = .356, P = .016) in patients with MCI. Whether urinary AD7c-NTP level could reflect disease severity is still unclear. It was previously reported that urinary AD7c-NTP levels increase in proportion to disease severity. 7,25 Overexpression of AD7c-NTP, which leads to neuritic sprouting and cell death, is associated with disease severity in AD. 25 In contrast, mean urinary AD7c-NTP level among CDR 2.0 patients has been reported to be lower than that among CDR 1.0 patients, with the explanation being that severely injured neurons are incapable of compensatory processes, such as neuritic sprouting or synaptic formation. 6 In the present study, increased urinary AD7c-NTP was observed among CDR 0.5 patients relative to CDR 0 patients. The increased urinary AD7c-NTP level among CDR 0.5 patients suggests that neuritic sprouting is progressing during the early course of AD. This finding suggests AD7c-NTP increases in MCI or very early stages of AD, which is compatible with a recent biomarker study related to β-amyloid. 3 The K-CWSP test, the processing of the specific stimulus, impedes the simultaneous processing of a second stimulus and represents the inhibitory interface of frontal executive function. 26 This finding suggests that frontal inhibitory processing speed may be influenced by the levels of urinary level of AD7c-NTP in patients with MCI.
There is some controversy as to whether urinary AD7c-NTP level is affected by some epidemiological factors or other neurological and non-neurological diseases. Recent evidence suggested that urinary AD7c-NTP level is affected by gender and aging in the general population. 27 Female and older individuals have higher levels of urinary AD7c-NTP, suggesting a higher incidence of AD in those groups. However, the present study did not reveal any significant differences among the strata of various epidemiological factors, including gender and age in patients with MCI. Previous studies have reported that levels of urinary AD7c-NTP could be affected by cerebral vascular changes. 5 However, several studies, including ours, observed no significant effect on urinary AD7c-NTP by different neurological and non-neurological diseases, drugs, and WMHs. However, inconsistent results have reported in association with depression, and further research is required. 5,15
Compared to a previous study, our results showed a similar sensitivity (73.9%) but a lower specificity (60.9%), presumably because of the small number of participants in our study as well as our use of an naMCI control group instead of normal controls. Other studies have found that the elevation in urinary AD7c-NTP, associated with the diagnosis of AD, has a sensitivity and specificity of approximately 70% to 90% and 80% to 90%, respectively. 5,6,22,23
In our study, there was a statistically significant gender difference among the excluded participants. Of the excluded participants, 70.7% were female. The preponderance of females in the excluded cases was mainly due to the higher frequency of contaminated urine samples in this group. We initially recruited 104 patients with MCI, but 58 (55.8%) participants were excluded because of inappropriate urine samples, refusal to provide urine samples, and withdrawal from the study. Previous urinary AD7c-NTP studies had similar dropout ratios. 5,6 Alzheimer-associated neural thread protein requires that the urine should be sterile and free of particulate debris. Elderly people more commonly have asymptomatic bacteriuria, hematuria, proteinuria, and urinary incontinence. Urinary creatinine concentration less than 50 mg/dL, associated with excessive urinary dilution due to nonfirst morning urine samples, may cause false negatives. In elderly patients, nighttime urinary incontinence is a common cause of low creatinine concentrations. In contrast, a urinary creatinine concentration greater than 225 mg/dL is associated with nonspecific excessive solute concentration, which can lead to false-positive results. 5,6,14 This at least partly explains the high exclusion rates of urinary AD7c-NTP studies, including ours.
Among the MCI categories, it is difficult to decipher what proportion of the MCI is true MCI due to AD because of its heterogeneity. 28 The diagnosis of MCI who have a high risk of AD progression is often difficult in usual clinical settings, despite good history taking, neurological examinations, brain MRI, neuropsychological, and laboratory tests. The urinary AD7c-NTP assay might be helpful for the detection of patients with aMCI who have high probability of AD progression. Ideal biomarkers have been suggested to have (1) the ability to detect the pathological change, (2) to be validated by pathologically confirmed cases, (3) the ability to detect a disease early in its course, (4) reliability, (5) noninvasiveness, (6) technical simplicity, and (7) inexpensiveness. 3,5,22 Urinary AD7c-NTP detection is a noninvasive, simple to perform, relatively inexpensive, and validated pathologically confirmed biomarker. 20,23
There are several limitations to our study. First, the small sample size—due to single institutional recruitment and the large number of excluded participants—limits the power of the findings. Second, as mentioned earlier, the use of naMCI as a control group resulted in a low specificity. Third, the absence of a diagnostic gold standard comparison, such as amyloid PET, could influence our results. Additionally, the cross-sectional design made it difficult to explore the sequential changes of AD7c-NTP levels in patients with MCI. A longitudinal study is required to determine how accurately the urinary AD7c-NTP level can predict conversion of patients with aMCI to AD.
Our study demonstrated a statistically significant difference in mean urinary AD7c-NTP level between individuals with aMCI and those with naMCI. We also found that urinary AD7c-NTP level correlated with CDR score of 0 and 0.5, and K-CWSP color reading time in patients with MCI. The present study showed that the mean level of urinary AD7c-NTP in the aMCI group was higher than that in the naMCI group, suggesting that urinary AD7c-NTP maybe a useful biomarker for distinguishing aMCI from naMCI.
Supplemental Material
Supplemental Material, CRCD_NPT_total_-_Supplementary - Comparison of Urinary Alzheimer-Associated Neural Thread Protein (AD7c-NTP) Levels Between Patients With Amnestic and Nonamnestic Mild Cognitive Impairment
Supplemental Material, CRCD_NPT_total_-_Supplementary for Comparison of Urinary Alzheimer-Associated Neural Thread Protein (AD7c-NTP) Levels Between Patients With Amnestic and Nonamnestic Mild Cognitive Impairment by Bon D. Ku, Hyeyun Kim, Yong Kyun Kim and Han Uk Ryu in American Journal of Alzheimer's Disease & Other Dementias
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
