Abstract
Objective:
We determine the association between dementia and the subsequent peptic ulcer disease (PUD).
Methods:
We identified patients with diagnosed dementia in the Taiwan National Health Insurance Research Database. A comparison cohort without dementia was frequency-matched by age, sex, and comorbidities, and the occurrence of PUD was evaluated in both cohorts.
Results:
The dementia and control cohort consisted of 6014 patients with dementia and 17 830 frequency-matched patients without dementia, respectively. The incidence of PUD (hazard ratio, 1.27; 95% confidence interval, 1.18-1.37; P < .001) was higher among patients with dementia. Cox models showed that being female, diabetes mellitus, chronic kidney disease, coronary artery disease, and chronic obstructive pulmonary disease were independent risk factors for PUD in patients with dementia.
Conclusion:
Dementia might increase the risk of developing PUD.
Introduction
Previous studies have reported that dementia impairs cognitive function, which has a negative effect on the quality of daily life of affected patients. 1,2 Furthermore, the behavioral and psychological symptoms of dementia, including mood and behavioral disturbances, could be a burden to patients. 3,4 In addition, previous studies have reported that patients with dementia typically have more comorbidities and associated complications compared to patients without dementia. 5 On the other hand, anxiety and schizophrenia are risk factors for peptic ulcer disease (PUD). 6,7
Peptic ulcer disease, including both gastric and peptic ulcers, is a gastrointestinal disease with high morbidity and mortality rates. 8 Although specific physical conditions and psychological distress have been associated with peptic ulcers, 9,10 limited evidence has been reported regarding the relationship between dementia and PUD. Therefore, we hypothesized that patients with a history of dementia are at an increased risk of subsequent PUD. To test our hypothesis, we examined the association between dementia and PUD by designing a nationwide population-based study.
Methods
Data Source
The Taiwan National Health Insurance (NHI) program, established in 1995, is a mandatory health insurance program that offers comprehensive medical care coverage, including outpatient, inpatient, emergency, and traditional Chinese medicine, to all residents of Taiwan (23.72 million people), with a coverage rate of more than 99% (http://www.nhi.gov.tw/english/index.aspx). The National Health Research Institute (NHRI) is responsible for managing all reimbursement claims data in the National Health Insurance Research Database (NHIRD), and patient confidentiality is maintained in accordance with the directives of the Bureau of the NHI. To protect the privacy of all persons registered in the program, the NHRI encrypts and converts the identification numbers of all NHIRD records before releasing the data for research. Data for this population-based retrospective cohort study were taken from the 2000 Longitudinal Health Insurance Database (LHID2000), a subset of the NHIRD. The LHID2000 contains claims data on 1 000 000 registered persons randomly selected from the 2000 Registry of Beneficiaries of the Taiwan NHI program. All clinical diagnoses are recorded according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes.
Sampled Participants
We collected data on patients aged 65 years who were diagnosed with dementia (ICD-9-CM Codes 290, 294.1, and 331.0-331.2) during the period of 2000 to 2010 as the dementia cohort. The date that patients had their dementia diagnosed was used as the index date. Patients with a history of PUD (ICD-9-CM Codes 531-533) before the index date, younger than 65 years of age, and those with incomplete age or sex information were excluded. Control patients without dementia or PUD at the baseline were randomly selected from the LHID2000. Three controls were frequency matched to each dementia case by age (every 5-year span), sex, and year of dementia diagnosis (subject to the same exclusion criteria).
Outcome and Comorbidity
Both the dementia and control cohorts were followed until a diagnosis of PUD was made or they were censored for loss to follow-up, withdrawal from the NHI program, or December 31, 2011. Comorbidities were cirrhosis (ICD-9-CM Code 571), hypertension (ICD-9-CM Codes 401-405), diabetes mellitus (DM; ICD-9-CM Code 250), chronic kidney disease (CKD; ICD-9-CM Code 585), coronary artery disease (CAD; ICD-9-CM Codes 410-414), alcohol-related illness (ICD-9-CM Codes 291, 303, 305, 571.0-571.3, 790.3, A215, and V11.3), chronic obstructive pulmonary disease (COPD; ICD-9-CM Codes 490-492, 494, and 496), anxiety (ICD-9-CM Code 300.0), depression (ICD-9-CM Codes 296.2, 296.3, 300.4, and 311), epilepsy (ICD-9-CM Code 345), cerebrovascular disease (ICD-9-CM Codes 430-438), gastroesophageal reflux disorder (GERD; ICD-9-CM Codes 530.11 and 530.81), and Parkinson’s disease (ICD-9-CM Code 332). All comorbidities were defined before the index date. In addition, the use of nonsteroidal anti-inflammatory drugs (NSAIDs) was analyzed in patients with dementia and in controls.
Data Availability Statement
All data and related metadata were deposited in an appropriate public repository. The data on the study population that were obtained from the NHIRD (http://w3.nhri.org.tw/nhird//date_01.html) are maintained in the NHIRD (http://nhird.nhri.org.tw/). The NHRI is a nonprofit foundation established by the government.
Ethics Statement
The NHIRD encrypts patient personal information to protect privacy and provides researchers with anonymous identification numbers associated with relevant claims information, including sex, date of birth, medical services received, and prescriptions. Patient consent is not required to access the NHIRD. This study was approved to fulfill the condition for exemption from the Institutional Review Board (IRB) of China Medical University (CMUH-104-REC2-115). The IRB specifically waived the consent requirement.
Statistical Analysis
Distribution of sex, age, and comorbidities were compared between the dementia and the control cohorts using the chi-square test for categorical variables and t test for continuous variables. The overall, sex-, age-, and comorbidity-specific incidence densities of PUD were measured for each cohort. Univariate and multivariate Cox proportion hazard regression models were employed to examine the effect of dementia on the risk of PUD, which are expressed as hazard ratios (HRs) with 95% confidence intervals (CIs). The multivariate model was controlled for age, sex, comorbidities (hypertension, DM, CKD, CAD, alcohol-related illness, COPD, depression, epilepsy, cerebrovascular disease, GERD, and Parkinson’s disease), and NSAIDs that differed significantly in the univariate model. Kaplan-Meier analysis was conducted to plot the cumulative incidence of PUD, and a log-rank test was used to test for differences in the incidence for PUD between the 2 cohorts. All statistical analyses were conducted using SAS version 9.4 (SAS Institute, Inc, Cary, North Carolina). The level of significance was set at P < .05 in 2-tailed tests.
Results
A total of 6014 patients with dementia were identified as eligible for this study, and 17 830 controls were frequency matched by age and sex (Table 1). Slightly more than half of the participants were women (52.2%) who were predominantly 75 to 84 years old (47.4%). The mean age of the dementia cohort was 78.6 years (standard deviation [SD] = 7.05) and that of the control cohort was 77.3 years (SD = 7.14), respectively. Compared with the controls, the prevalence of comorbidities of hypertension, DM, CKD, CAD, alcohol-related illness, COPD, anxiety, depression, epilepsy, cerebrovascular disease, GERD, and Parkinson disease was greater in the dementia cohort (all P < .001). The use of NSAIDs was more prevalent in the dementia cohort at the baseline, compared to the control cohort (P < .001).
Demographic Characteristics and Comorbidity in Patients With and Without Dementia.
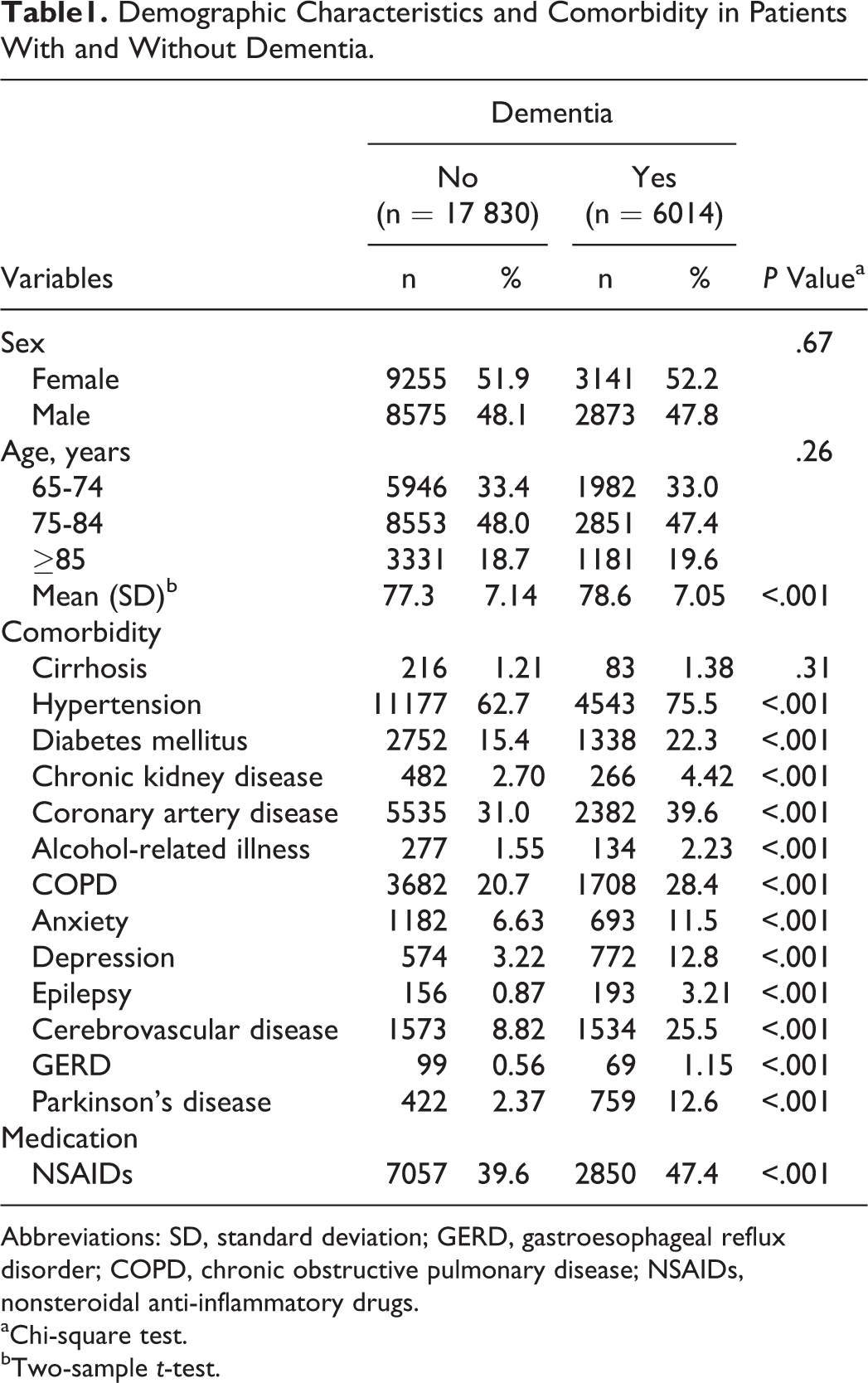
Abbreviations: SD, standard deviation; GERD, gastroesophageal reflux disorder; COPD, chronic obstructive pulmonary disease; NSAIDs, nonsteroidal anti-inflammatory drugs.
aChi-square test.
bTwo-sample t-test.
The cumulative incidence of PUD for the 2 cohorts (Figure 1) shows that the dementia incidence curve is significantly higher than that for the control cohort (logrank test P < .001). During the mean follow-up of 3.54 years for the dementia cohort and 4.36 years for the control cohort, the overall incidence of PUD (per 1000 person-years) was 52.6 and 36.8, respectively (Table 2).
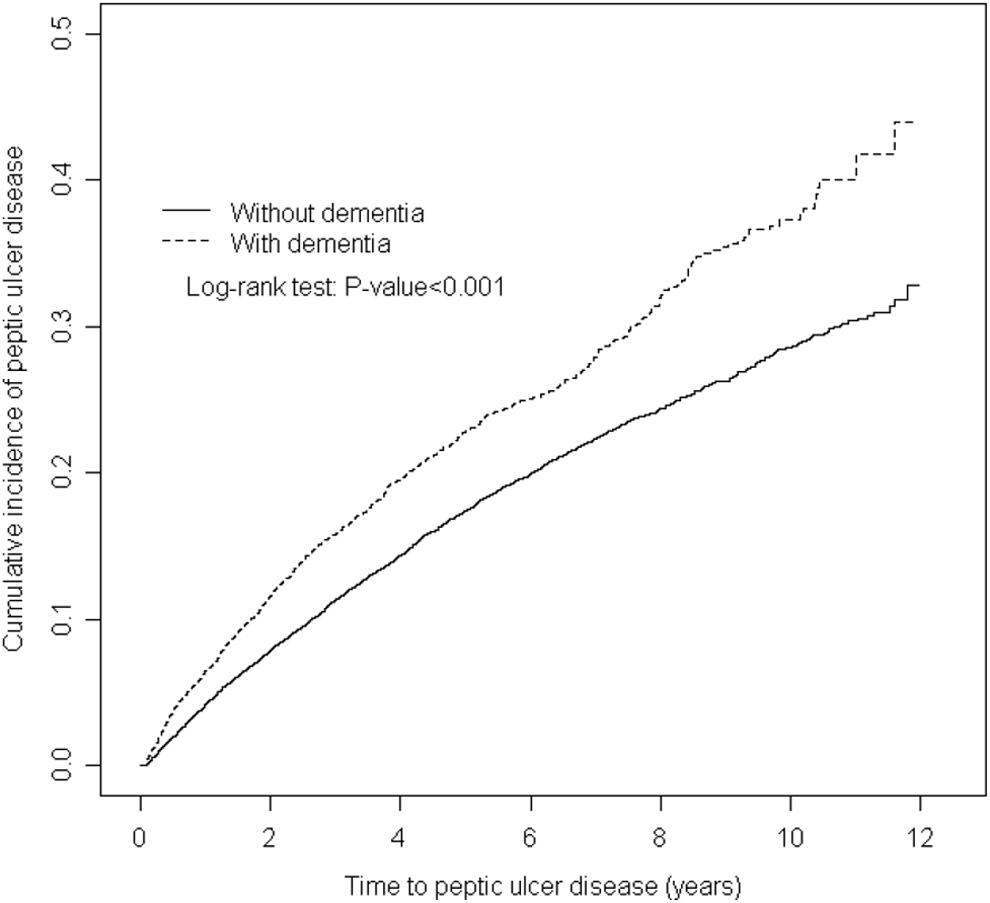
Cummulative incidence of peptic ulcer disease compared between patients with and without dementia.
Incidence and Hazard Ratios of Peptic Ulcer Disease for Dementia Cohort Compared to Nondementia Cohort by Demographic Characteristics and Comorbidities.
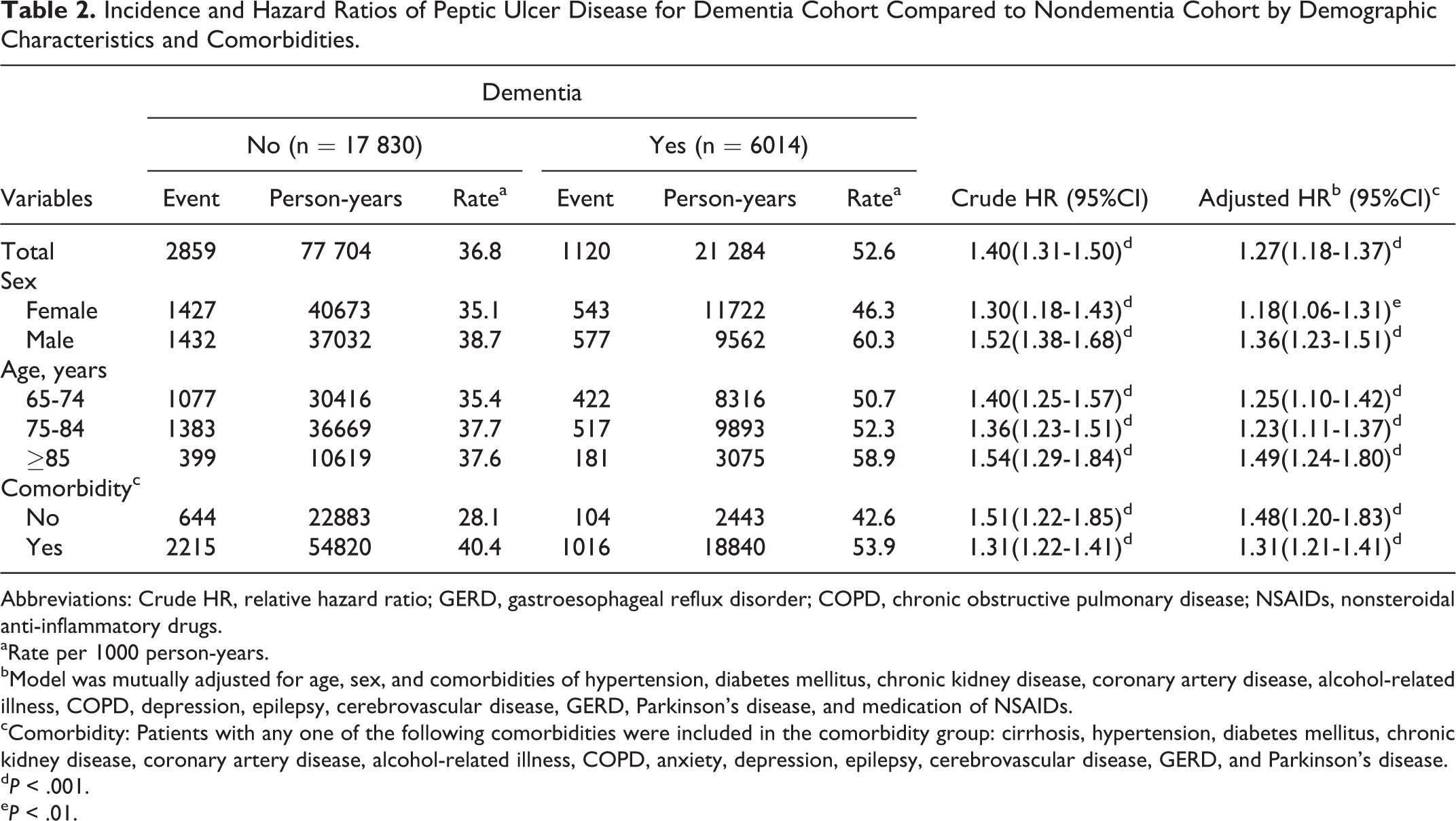
Abbreviations: Crude HR, relative hazard ratio; GERD, gastroesophageal reflux disorder; COPD, chronic obstructive pulmonary disease; NSAIDs, nonsteroidal anti-inflammatory drugs.
aRate per 1000 person-years.
bModel was mutually adjusted for age, sex, and comorbidities of hypertension, diabetes mellitus, chronic kidney disease, coronary artery disease, alcohol-related illness, COPD, depression, epilepsy, cerebrovascular disease, GERD, Parkinson’s disease, and medication of NSAIDs.
cComorbidity: Patients with any one of the following comorbidities were included in the comorbidity group: cirrhosis, hypertension, diabetes mellitus, chronic kidney disease, coronary artery disease, alcohol-related illness, COPD, anxiety, depression, epilepsy, cerebrovascular disease, GERD, and Parkinson’s disease.
dP < .001.
eP < .01.
After adjustment for age, sex, comorbidities (hypertension, DM, CKD, CAD, alcohol-related illness, COPD, depression, epilepsy, cerebrovascular disease, GERD, and Parkinson disease), and NSAIDs, the risk of developing PUD is higher in the dementia cohort (adjusted hazard ratio [HR], 1.27; 95% confidence interval [CI], 1.18-1.37). The incidence of PUD was greater in men in both cohorts. Patients with dementia exhibited a higher risk of PUD than that of the controls, regardless of sex. The age-specific relative risk between the dementia and the control cohorts was highest among patients aged 85 years or older (adjusted HR, 1.49; 95% CI, 1.24-1.80). The comorbidity-specific dementia cohort to control cohort adjusted HRs for PUD were 1.48 (95% CI, 1.20-1.83) and 1.31 (95% CI, 1.21-1.41) in patients without or with comorbidities, respectively.
Table 3 shows the results of the univariate and multivariate Cox proportion hazards regression models for the related risk variables contributing to PUD. The risk factors contributing to PUD included being female (adjusted HR, 1.13; 95% CI, 1.06-1.21) and having DM (adjusted HR, 1.09; 95% CI, 1.01-1.19), CKD (adjusted HR, 1.51; 95% CI, 1.28-1.77), CAD (adjusted HR, 1.10; 95% CI, 1.03-1.18), COPD (adjusted HR, 1.22; 95% CI, 1.13-1.31), or cerebrovascular disease (adjusted HR, 1.19; 95% CI, 1.08-1.31).
Cox Model With Hazard Ratios and 95% Confidence Intervals of Peptic Ulcer Disease Associated With Dementia and Covariates.
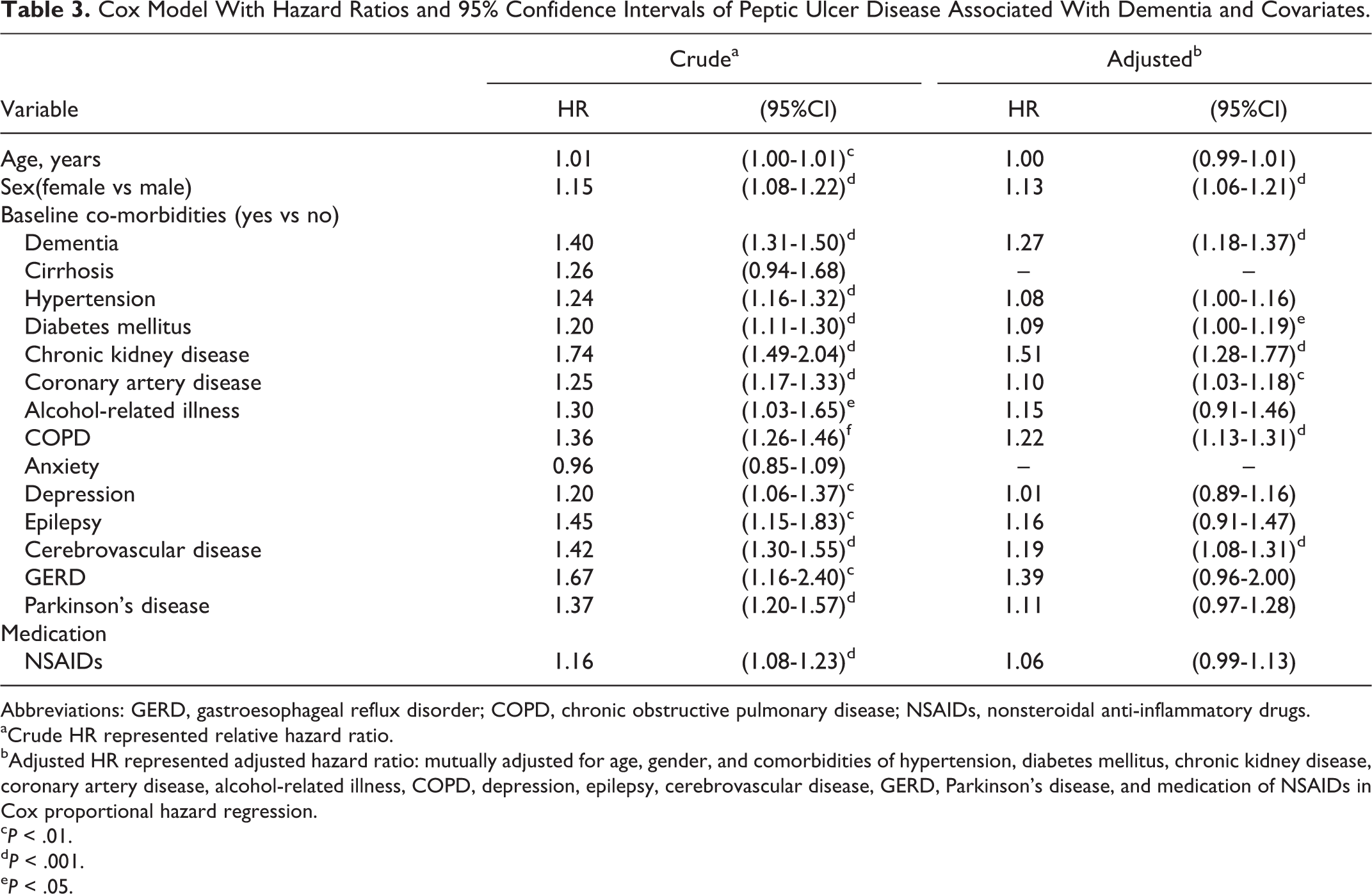
Abbreviations: GERD, gastroesophageal reflux disorder; COPD, chronic obstructive pulmonary disease; NSAIDs, nonsteroidal anti-inflammatory drugs.
aCrude HR represented relative hazard ratio.
bAdjusted HR represented adjusted hazard ratio: mutually adjusted for age, gender, and comorbidities of hypertension, diabetes mellitus, chronic kidney disease, coronary artery disease, alcohol-related illness, COPD, depression, epilepsy, cerebrovascular disease, GERD, Parkinson’s disease, and medication of NSAIDs in Cox proportional hazard regression.
cP < .01.
dP < .001.
eP < .05.
Discussion
This is the first retrospective study using a matched cohort and a long-term follow-up period (10-year) to examine the association between dementia and PUD. The major finding of our study was the discovery of a higher incidence of subsequent PUD among patients with dementia. In addition to the patients’ sex, the comorbidities DM, CKD, CAD, COPD, and cerebrovascular disease may be risk factors.
This study determined that patients with dementia were at a higher risk of subsequent PUD. We hypothesized that the possible mechanism may be associated with the hypothalamus–pituitary–adrenal (HPA) axis and glucocorticoid resistance. Hippocampal disinhibition and damage was reported to result in the disruption of HPA axis in patients with dementia. 11,12 In addition, a previous study reported that dementia was connected with depression through chronic neuroinflammation. 13 The inflammation may induce glucocorticoid resistance under chronic conditions. 14 Although glucocorticoids showed some gastroprotective effects under acute stress conditions, 15 an animal model exhibited the opposite effect under chronic stress condition. 16 Thus, dementia may increase the risk of PUD.
Our study showed that women with dementia are at a higher risk of subsequent PUD, which might be associated with sex hormones. Although estrogen reportedly has a gastroprotective effect in premenopausal women, 17 elderly menopausal women were exposed to the risk of developing peptic ulcers. Therefore, women with dementia may have a higher risk of subsequent PUD.
In our analysis of the risk factors associated with subsequent PUD in patients with dementia, we found that DM, CKD, CAD, COPD, and cerebrovascular disease were associated with an increased risk of subsequent PUD in patients with dementia, which is in agreement with the findings of other studies. 18 –22 However, the risk was greater in the patients with dementia than in those without dementia. We asserted that the mechanism may be associated with the interaction between chronic inflammation and disruption of the HPA axis. Previous studies have observed chronic inflammation in patients with dementia, DM, CKD, CAD, COPD, and cerebrovascular disease. 23 –25 The immune reaction associated with proinflammatory cytokines could induce neuroinflammation. 26 In addition, chronic inflammation has been shown to disturb the HPA axis and induce hypercortisolemia through the proinflammatory response and the effects of neuroinflammation. 27 –29 A previous study reported that HPA axis dysfunction increases the risk of PUD under chronic stress conditions. 16 Consequently, patients with dementia and comorbid DM, CKD, CAD, COPD, and cerebrovascular disease may be at an increased risk of subsequent PUD.
To our knowledge, we are the first to conduct the retrospective study to examine dementia as a risk factor for PUD. We adopted a population-based study with adequate controls for comorbidities, thus further strengthening our study. However, several limitations that are inherent to the use of claims databases were encountered while conducting this study. First, the diagnosis basis of dementia was according to the ICD-9-CM codes. Thus, the severity of dementia as a risk factor for subsequent PUD was not explored. Second, the causal relationship was examined according to the chronological order of diagnosis between these two diseases. However, the possibility that PUD causes dementia cannot be excluded. Moreover, many demographic variables, such as socioeconomic status, daily lives, and family history, were unavailable. These variables might have provided useful information for the study of dementia and PUD. Furthermore, although Helicobacter pylori infection was a well-known etiology of PUD, 30 H pylori infection was confirmed by clinical examinations such as urea breath tests and upper endoscopy, after the diagnosis of PUD. 31 Therefore, it was hard to differentiate whether PUD resulted from H pylori infection or dementia in our study. Finally, the data derived from a retrospective cohort study is generally considered as lower methodological quality than that from the prospectively randomized clinical trial. The main reason is retrospective cohort study subjecting to have many biases that are lack of the necessary adjustments or some unmeasured or unknown confounding factors.
In conclusion, the findings of this study suggest that dementia increases the risk of developing PUD. Based on our data, we suggest that women with dementia should receive closer observation, particularly those with the comorbidities such as DM, CKD, CAD, COPD, and cerebrovascular disease. Further prospective clinical study on the relationship between dementia and PUD is warranted.
Footnotes
Authors’ Contribution
Chih-Chao Hsu, Yi-Chao Hsu, Chia-Hung Kao contributed to conception/design; Chia-Hung Kao contributed to provision of study materials; Yi-Chao Hsu, Chih-Chao Hsu, Cheng-Li Lin, Chia-Hung Kao contributed to collection and/or assembly of data; All authors; contributed to data analysis and interpretation, manuscript writing, and Final approval of the manuscript. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. No additional external funding received for this study. Chih-Chao Hsu and Yi-Chao Hsu contributed equally to this work.
Authors’ Note
Kuang-Hsi Chang is now affiliated to Management Office for Health Data, China Medical University Hospital, Taichung, Taiwan and Department of Public Health, China Medical University, Taichung, Taiwan. Chang-Yin Lee is now affiliated to College of Medicine, The School of Chinese Medicine for Post Baccalaureate, I-Shou University (Yancho Campus), Kaohsiung, Taiwan, and Department of Chinese Medicine, E-DA Hospital, Kaohsiung, Taiwan. Lee-Won Chong is now affiliated to Division of Hepatology and Gastroenterology, Department of Internal Medicine, Shin Kong Wu Ho-Su Memorial Hospital, Taipei, Taiwan. Cheng-Li Lin is now affiliated to Management Office for Health Data, China Medical University Hospital, Taichung, Taiwan and College of Medicine, China Medical University, Taichung, Taiwan.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported in part by Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence (MOHW105-TDU-B-212-133019); China Medical University Hospital, Academia Sinica Taiwan Biobank, Stroke Biosignature Project (BM10501010037); NRPB Stroke Clinical Trial Consortium (MOST 104-2325-B-039 -005); Taiwan Ministry of Science and Technology (MOST103-2314-B-715- 001-MY2, and MOST104-2314-B-715-003-MY3); Mackay Medical College (MMC 1012A10, RD1010061, RD1020038, RD1020047, RD1012B13, RD1031B05, RD1030053, RD1030076, RD1040109), and Mackay Memorial Hospital (MMH-MM-10304, MMH-MM-10405); Tseng-Lien Lin Foundation, Taichung, Taiwan; Taiwan Brain Disease Foundation, Taipei, Taiwan; Katsuzo and Kiyo Aoshima Memorial Funds, Japan; and CMU under the Aim for Top University Plan of the Ministry of Education, Taiwan.
