Abstract
Alzheimer’s disease (AD) is classified as a neurodegenerative disease, impacting on brain integrity and functioning, resulting in a progressive deterioration of cognitive capabilities. Epigenetic changes can be acquired over the life span and mediate environmental effects on gene expression. DNA-methyltransferase 3A (DNMT3A) plays an important role in the development of embryogenesis and the generation of aberrant methylation late-onset AD (LOAD). In this study, the rs1550117 polymorphism of DNMT3A was determined by polymerase chain reaction/restriction fragment length polymorphism and confirmed by sequencing. The results showed that AA genotype carriers had a 2.08-fold risk of developing LOAD in comparison with GG genotype carriers (odds ratio [OR] = 2.08, 95% confidence interval [CI]: 1.03-4.21, P = .038) and had a 2.05-fold risk for LOAD compared with GG+GA genotype carriers (OR = 2.05, 95% CI: 1.03-4.11, P = .038), indicating that the DNMT3A polymorphism supports a major role in the pathogenesis of LOAD and can be used as a stratification marker to predict an individual’s susceptibility to LOAD.
Introduction
Alzheimer’s disease (AD) is a neurodegenerative disorder characterized by the presence of β-amyloid in the senile plaques, intracellular aggregates of tau protein in the neurofibrillary tangles, and progressive neuronal loss. 1 Alzheimer’s disease is mainly characterized by memory loss, with disoriented behavior and impairments in language, comprehension, and spatial skills able to interfere with the quality of life and normal daily activities. Neuropsychiatric symptoms, such as depression, psychosis, and agitation, are also frequent in people with AD and are a common precipitant of institutional care. 2 Up to date, the etiology of AD is still completely unknown, but it is extensively accepted that the abnormal DNA methylation was involved in the pathogenesis.
The genetic susceptibility contributed to the late-onset AD (LOAD) seems to be more complicated compared with early-onset AD (EOAD). Until recently, the variant of the apolipoprotein E (APOE) gene was the most important identified genetic association for LOAD, 3,4 and only the ∊4 allele of APOE gene has been identified as a major genetic susceptibility factor to this disorder. 5,6 However, only 30% to 50% of all cases with AD bear the APOE∊4 allele, and not all APOE∊4 carriers develop this disease, 3 suggesting that there are additional risk loci influencing the susceptibility of LOAD that remain to be discovered.
Epigenetic modification of chromatin, including DNA methylation at the sites of CpG dinucleotides, is a key regulator of gene expression, growth, and differentiation in virtually all tissues, including brain. 7 -9 Changes in methylation status at selected genomic loci may affect social cognition, 10 learning and memory, 11 and stress-related behaviors 12 and is believed to contribute to dysregulated gene expression in a range of adult-onset neuropsychiatric disorders, including autism, schizophrenia, depression, and AD. 4,13 -16 Finally, there is strong evidence that aberrant methylation of tumor suppressor genes contributes to the molecular pathology of a subset of astrogliomas and other types of brain cancers. 17,18
DNA methylation is established and maintained by DNA methyltransferases (DNMTs). In humans, 3 enzymes, DNMT1, DNMT3A, and DNMT3B, are known to have DNMT activity. DNA-methyltransferase 3A and DMMT3B are responsible for de novo methylation and modification of unmethylated DNA, whereas DNMT1 is required to maintain DNA methylation. 19,20 DNA-methyltransferase 3A, one of the 2 human de novo DNAMTs, was involved in imprinting of germ cells and normal embryonic development. 21,22 Moreover, DNMT3A contributes to the differentiation of neural progenitors and hematopoietic stem cells. 23,24 DNA-methyltransferase 3A in human cerebral cortex is expressed primarily in neurons, which is in agreement with similar findings in mice. 25 Furthermore, the DNMT3A was expressed across all ages, including a subset of neurons residing in layers III and V of the mature cortex. Therefore, DNA methylation is dynamically regulated in the human cerebral cortex throughout the life span and involves differentiated neurons. 26 Polymorphisms of the DNMT3A gene may regulate gene expression, affect its enzymatic activity, and potentially contribute to susceptibility to cancer. It has recently been found that the DNMT3A polymorphism rs1550117 alters the promoter activity and risk of gastric cancer and colorectal cancer. 27,28 However, to our best knowledge, few studies have reported on the possible association of LOAD susceptibility with DNMT3A. In the present study, we want to determine whether the rs1550117 polymorphism in DNMT3A promoter contributes to susceptibility of LOAD.
Methods
Participants
This study including 300 patients with sporadic LOAD (age at onset ≥ 65 years) and 320 age- and gender-matched healthy controls was approved by the hospital-based ethics committee. The informed consent was obtained from each participant or from a guardian. All patients with LOAD and controls were recruited from the Department of Neurology at Wenzhou hospital of Traditional Chinese Medicine from June 2009 to May 2014. The detailed clinical evaluations including medical history, neuropsychological test, a physical examination, and screening laboratory tests were established by at least 2 neurologists according to the criteria of the National Institute of Neurological and Communicative Disorders and Stroke/Alzheimer’s Disease and Related Disorders Association. 29 Family history of dementia was ascertained excluding all the participants with even one relative who developed AD or other dementias. The control group was taken neurological and medical examinations, which showed that they were free of any symptoms suggestive of cognitive decline. Cognitive performance was measured by the Mini-Mental State Examination (MMSE) test. The characteristics of the participants were shown in Table 1.
Clinical Characteristics of the Participants.
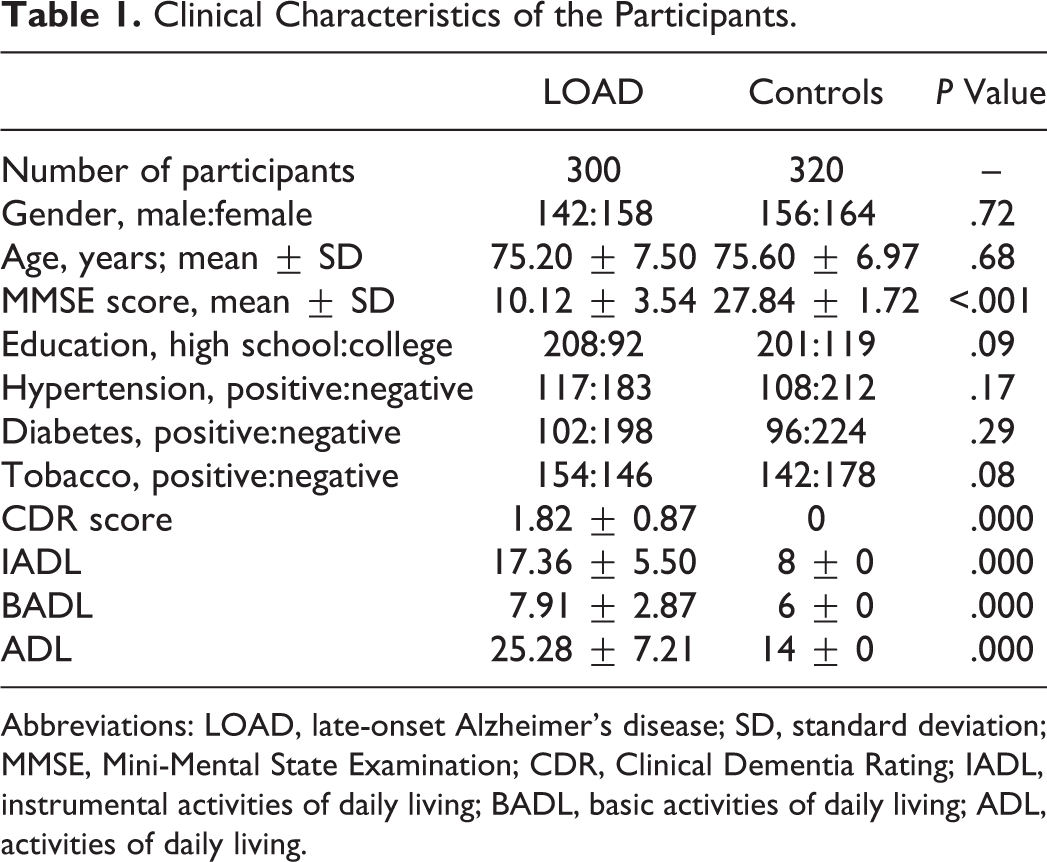
Abbreviations: LOAD, late-onset Alzheimer’s disease; SD, standard deviation; MMSE, Mini-Mental State Examination; CDR, Clinical Dementia Rating; IADL, instrumental activities of daily living; BADL, basic activities of daily living; ADL, activities of daily living.
Extraction of Genomic DNA
Peripheral blood samples were collected in vacuum tubes with 5% EDTA. Genomic DNA was extracted using DNA Purification Kit (Tiangen Biotech, Beijing) according to the instruction of the protocol.
DNMT3A Genotyping by Polymerase Chain Reaction–Restriction Fragment Length Polymorphism
The genotyping of the DNMT3A rs1550117 polymorphism was performed by polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLP). The forward primer 5′-ACACACCGCCCTCACCCCTT-3′ and the reverse primer 5′-TCCAGCAATCCCTGCCCACA-3′ were used for PCR. The 20-μL PCR mixture contained 100 to 150 ng of genomic DNA and 10 μL 2 × PCR mix (Tiangen Biotech). For PCR amplification, an initial denaturation at 94°C for 5 minutes was followed by 36 cycles at 94°C for 30 seconds, at 66°C for 30 seconds, at 72°C for 30 seconds, and a final extension at 72°C for 8 minutes. TaaI (Fermentas Co, Glen Burnie, Maryland) was used to detect this A-G transition at 65°C for 5 minutes. The wild-type G allele had 3 bands (153, 94, and 87 bp), while the variant A allele had 4 bands (247, 153, 94, and 87 bp). To confirm the genotyping results, 10% PCR-amplified DNA samples were examined by DNA sequence, and the results were 100% concordant.
Statistical Analysis
SPSS16.0 (SPSS Inc, Chicago, Illinois) was used for statistics analysis. Patients and controls were compared using the Student t test for continuous variables and the chi-square (χ2) test for categorical variables. The genotype distribution and the genotype frequencies were analyzed by the χ2 test. The standard goodness-of-fit test was used to test the Hardy-Weinberg equilibrium. A P value <.05 was considered to be statistically significant.
Results
The clinical parameters of patients including patients with LOAD and healthy controls were shown in Table 1. The rs1550117 polymorphism of DNMT3A gene was genotyped in 300 patients with LOAD and in 320 age- and gender-matched healthy controls. Significantly, MMSE score in patients with LOAD was lower than that of the controls (P < .001). All genotype distributions of the DNMT3A (rs1550117) were in the Hardy-Weinberg equilibrium both in LOAD and in control groups (P > .05).
The DNMT3A promoter rs1550117 polymorphism in patients with LOAD and controls was detected by PCR-RFLP. The genotyping was confirmed by DNA sequencing, and the results of the PCR-RFLP genotyping and sequencing analysis were 100% concordant. The genotypic and allelic frequencies of DNMT3A rs1550117 are summarized in Table 2. The distributions of rs1550117 genotypes in the patients with LOAD were GG, 51.3%; AG, 31.7%; and AA, 8.0%. The A allele frequency was 23.9%, while the frequency for the controls was 63.8%, 32.2%, and 4.0%, respectively, and 20.2% for the A allele. AA genotype carriers had a 2.08-fold risk for developing LOAD in comparison with GG genotype carriers (odds ratio [OR] = 2.08, 95% confidence interval [CI]: 1.03-4.21, P = .038) and had a 2.05-fold risk for LOAD compared with GG+GA genotype carriers (OR = 2.05, 95% CI: 1.03-4.11, P = .038). The patients with LOAD were then stratified by gender. No significantly different frequencies of rs1550117 were observed in patients with LOAD (data not shown).
Distribution of DNMT3A rs1550117 Polymorphism Genotypes and Allele Frequencies in Patients With LOAD and Healthy Controls.
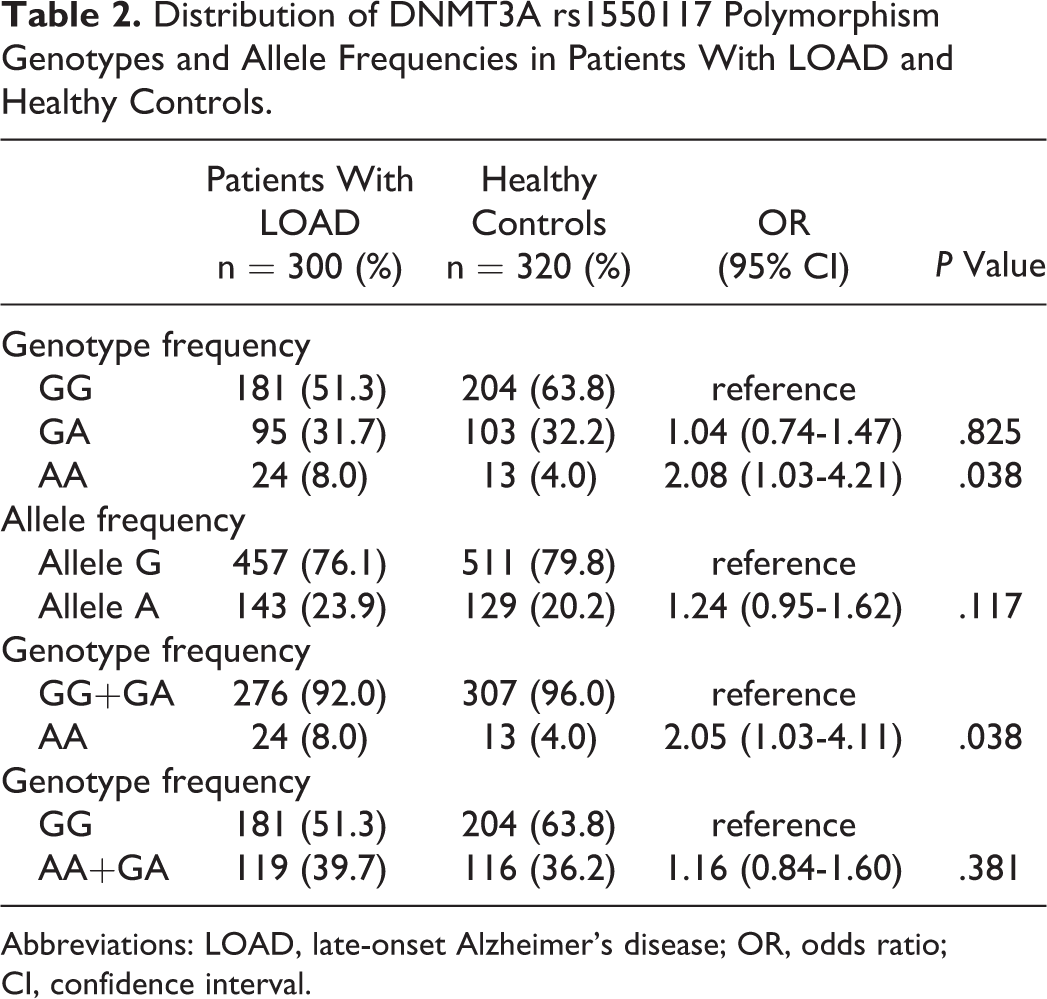
Abbreviations: LOAD, late-onset Alzheimer’s disease; OR, odds ratio; CI, confidence interval.
Discussion
Alzheimer’s disease is a highly prevalent, progressive, and fatal neurodegenerative disease associated with aging. Termed LOAD, the majority of cases with AD are sporadic and symptoms manifest after age 65. Despite enormous research efforts, the etiology of AD remains obscure and puzzling. Evidence for the role of epigenetics in AD pathogenesis is found in human studies of various tissues, animal models, and cell culture. 30,31
Abnormal DNA methylation is thought to be a major early event in the development of AD, 32 -34 where DNMTs, DNMT1, DNMT3A, and DNMT3B have been identified as DNA methylation functional enzymes in eukaryotic cells. DNMT1,which is often referred to as the maintenance methyltransferase, is responsible for maintaining preexisting methylation patterns during DNA replication. DNMT3A and DNMT3B are considered to be de novo DNA methyltranferase, which are critical in the dynamic DNA methylation process during embryogenesis and pathogenesis. It had been reported that the DNMT3B polymorphism did not involve in the pathogenesis of patients with LOAD. 35 But, to the best of our knowledge, little is known about the association of DNMT3A and the genetic susceptibility to LOAD. Therefore, we investigated the effect of DNMT3A rs1550117 polymorphism and risk of LOAD in a hospital-based, case–control study in a Chinese population.
In this study, the result showed that distributions of rs1550117 genotypes in the patients with LOAD were GG, 51.3%; AG, 31.7%; and AA, 8.0%. The A allele frequency was 23.9%, while the frequency for the controls was 63.8%, 32.2%, 4.0%, respectively, and 20.2% for the A allele. There were significant differences in AA genotype carriers; they had a 2.08-fold risk of developing LOAD in comparison with GG genotype carriers (OR = 2.08, 95% CI: 1.03-4.21, P = .038) and had a 2.05-fold risk of LOAD compared with GG+GA genotype carriers (OR = 2.05, 95% CI: 1.03-4.11, P = .038). No significantly different frequencies of rs1550117 were observed in patients with LOAD when the patients with LOAD were stratified by gender. These results suggest that DNMT3A rs1550117 polymorphism may be associated with AD.
In conclusion, this is the first report to investigate the association of a single-nucleotide polymorphism in DNMT3A with genetic susceptibility to LOAD. These results suggest that DNMT3A rs1550117 polymorphism may be associated with AD itself in Chinese patients, assuming that the DNMT3A gene may have effects on pathogenesis of AD and can be used as a stratification marker to predict an individual’s susceptibility to LOAD. This association requires to be confirmed in more large cohorts and in diverse ancestry populations, since genetic variations vary among the populations of different ethnic and geographical origin.
Footnotes
This article was accepted under the editorship of the former Editor-in-Chief, Carol F. Lippa.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants from the Technology Bureau of Wenzhou city (2013S0064).
