Abstract
Although the Anosognosia Questionnaire-Dementia (AQ-D) is one of the main instruments for assessing awareness in Alzheimer’s disease (AD), the normative data were until now limited to people from Argentina and Japan. This study aims to validate this instrument in an European context, in particular in an Italian sample. In a multicenter project (Verona, Padova, and Trapani), 130 patients with AD and their caregivers participated in the study. Psychometric characteristics of AQ-D are confirmed indicating that the scale permits the early identification of anosognosia and the correct care management of patients. Indeed, anosognosia results to be present also in patients with very mild AD (moderate: 44.44%; mild: 47.17%; and very mild: 23.73%). Moreover, the results indicate that deficits in awareness may vary in severity and that different types of anosognosia may be identified.
Keywords
Introduction
Exactly a century ago, the term anosognosia was coined to indicate a phenomenon involving an unawareness of disability. 1 It initially referred to the incapacity of some patients to identify their own paralysis, but then rapidly extended to other neuropsychological disorders such as amnesia, aphasia, spatial neglect, and so on. 2 In Alzheimer’s disease (AD), anosognosia refers to a lack of awareness concerning impairments in daily activities or neuropsychological deficits, in particular memory deficits. 3 More precisely, in this context, awareness is considered as “a reasonable or realistic perception or appraisal of a given aspect of one’s situation, functioning or performance, or of the resulting implications, which may be expressed explicitly or implicitly.”(pp. 396) 4 Awareness has been considered as operating on 4 levels of increasing complexity but reciprocally interacting sensory registration, performance monitoring, evaluative judgment, and metarepresentation. 4 Various neurological conditions can effect 1 or more of these awareness levels. 4
In addition, clinical and neuropsychological investigations have revealed that anosognosia is a multifaceted and heterogeneous phenomenon, the expressions of which may change 5 –9 as may the degree of severity. 10,11 In AD, in addition to unawareness of deficits concerning abilities in daily life activities (ADLs), specific forms of amnestic anosognosia, executive anosognosia, and unawareness for affective disorders have been identified. 9,12 Furthermore, a form of anosognosia known as affective anosognosia, which specifically involves changes in mood and interpersonal reactions, has been reported. 9
Research on functional neuroanatomy has identified the crucial role of cingulofrontal and parietotemporal networks in the awareness of deficits, 13 in particular in the right hemisphere. Hypometabolism and reduced blood flow have been found to be associated with a lack of awareness of memory deficits and anosognosia in AD, in particular in the right inferior frontal gyrus, 14 in the right prefrontal and orbitofrontal cortex, 15,16 in the anterior cingulate gyri and medial and orbital frontal areas, 17 and in medial temporal structures. 18
Anosognosia impacts a patient’s capacity to maintain autonomy in ADL, his or her readiness to engage in assessment and treatment, relationships with relatives and caregivers and in general his or her emotional and behavioral responses to the environment. For these reasons, over recent years, researchers have become increasingly interested in knowing more about the multiple features of awareness and in instruments, which are able to identify at an early stage the presence of signs of anosognosia.
To date, the assessment of anosognosia in AD has been carried out in various different ways. Clinical interviews 19 help the examiner to identify severe disorders but are not sufficient when symptoms are unclear or when these are only specific to some functions. Comparisons of self-ratings in task performances with objective test scores may also be useful as an index of performance monitoring. 20 Nevertheless, this method usually investigates awareness in neuropsychological tests and does not permit an evaluation of the patient’s perception of his or her functional difficulties in ADLs. 21 Finally, comparisons of self-ratings with informant ratings on a parallel measure with in-depth interviews may help in the analysis of awareness as related to day-to-day activities. This method measures the awareness levels of evaluative judgments and metarepresentations. 4,17 The information provided by caregivers represents a general reference point. It is important, however, to take into consideration any individual psychological factors relating to the caregivers as well as their perception of the patients’ condition. When these variables are controlled, patient–caregiver discrepancy scores turn out to be a greatly informative method. 22,23 Among the tools available for assessing anosognosia in AD, the most commonly used, especially in research, is the Anosognosia Questionnaire for Dementia (AQ-D). 22 This is a patient–caregiver discrepancy scale that includes a cognitive and functional assessment of the patient taking into consideration any changes in behavior. In addition, the scale makes it possible to discriminate between patients with minimal alterations in awareness and patients who clearly present with anosognosia. 24
It has been validated in Spanish on a large sample of patients in Argentina, 22,21 and its reliability and validity have also been confirmed in a Japanese population. 25 In addition, it has been utilized to compare the frequency of deficits in awareness in patients with mild cognitive impairment and AD 3 and to investigate the role of neuropsychiatric symptoms in anosognosia. 14,26 Finally, this scale has been used to investigate the potential correlations between anosognosia and clinical competence. 27
The present study aims to develop the Italian validation of the AQ-D and to analyze the correlations between anosognosia and the severity of mental deterioration, which is for the most part still unexplored. It was possible to do this by comparing the AQ-D scores of 3 groups of patients, respectively, affected by very mild, mild, and moderate AD. In addition, we explored the possibility of identifying various degrees of anosognosia as well as investigating the earliest markers of awareness disorders. This is particularly interesting, as our preliminary data 27 indicate that signs of unawareness may be present even at the initial stages of the illness and may impact the functional use of the cognitive strategies and environmental aids necessary to deal with the requests of the daily life. Finally, the potential effects of neuropsychological and neuropsychiatric variables in anosognosia were investigated.
Materials and Methods
Participants
Patients
A total of 130 patients were recruited in 3 Centers for Alzheimer’s Disease, in Verona, Padova, and Trapani. They were affected by mild to moderate AD according to McKhann’s criteria 28 or prodromal AD according to the criteria of Dubois. 29 In the latter, “prodromal AD” refers to individuals presenting with specific cognitive symptoms (not involving limitations to ADLs) in addition to positive CSF or neuroimaging biomarkers consistent with AD pathology. 29 Only 1 of our patients in the very mild group agreed to be subjected to cerebrospinal fluid analysis, but magnetic resonance imaging (MRI) and positron emission tomography (PET) were carried out for all the patients. These excluded the presence of atrophy or hypometabolism in frontotemporal cortex normally connected with frontotemporal dementia. This was also excluded by clinical and neuropsychological assessments. Unfortunately, the duration of illness was not available because of the difficulties in identifying the onset of the very early symptoms of mental deterioration.
In order to include the patients in the study, they received a comprehensive neurological evaluation as well as blood tests (thyroid function, homocysteine, vitamin B12, and folic acid dosage), brain MRI and/or PET, neuropsychological tests (Battery for Mental Deterioration 30 ), and functional assessment (instrumental activities of daily living [IADL] scale and index of independence in activities of daily living [ADL]). 31,32
The inclusion criteria were absence of behavioral disorders, a Mini-Mental State Examination (MMSE) 33 score ≥15 of 30, age between 65 and 85 years, education ≥5 years, and preserved verbal comprehension. Patients with a history of head injury, psychiatric disorders, neurological diseases, or severe sensorial deficits were excluded. On the basis of the degree of impairment, the patients were stratified in 3 classes of MMSE values, 20 (15-19.50 = moderate AD; 19.51-24.50 = mild AD; and 24.51-30 = very mild—including “prodromal”—AD). Demographic and clinical data are shown in Table 1. In all, 32.31% (n = 42) of the patients were treated with inhibitors of acetylcholinesterase or memantine.
The Patients’ Clinical and Demographical Data are Separately Reported for the 3 Groups.a
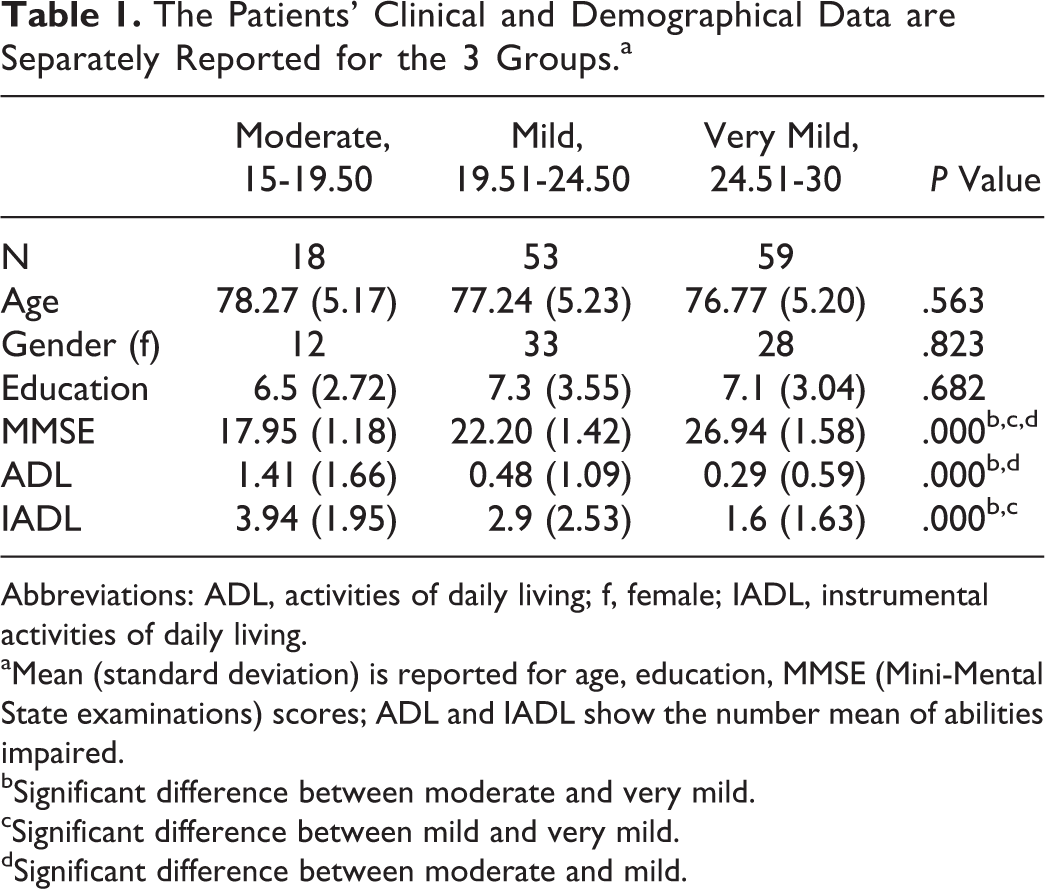
Abbreviations: ADL, activities of daily living; f, female; IADL, instrumental activities of daily living.
aMean (standard deviation) is reported for age, education, MMSE (Mini-Mental State examinations) scores; ADL and IADL show the number mean of abilities impaired.
bSignificant difference between moderate and very mild.
cSignificant difference between mild and very mild.
dSignificant difference between moderate and mild.
Caregivers
The caregiver of each patient was also recruited. This was defined as the relative currently responsible for the participant or a person who is in regular professional contact (more than twice a week) with him or her. Of the caregivers, 70% were females, mainly spouses (51.5%), 42.3% were daughters or sons, and only 6.2% were professional caretakers. Of the caregivers belonging to the family, 30.8% were employed, but 22.3% of them had changed their work schedule or type of job due to their caregiving schedule. In all, 6.2% were unemployed, 20.8% were housewives, and 42.3% were pensioners. The duration of caregiving up to that point was on average 15.47 months (range: 2-72 months) and the time dedicated to assistance was on average 6 h/d (range: 1-24 hours; very mild = 4.62; mild = 6.28; and moderate = 9.78).
All the patients agreed to participate in the study, which was approved by the local ethics committee (AOUI Pt n. 2238/2012).
Procedure
Neuropsychological assessment
All the patients underwent a comprehensive neuropsychological examination to assess cognitive general functions (Raven matrix 34 , attentional matrix, cognitive estimation, and verbal judgments 35 ), memory (auditory verbal learning test, 36 story recall, and verbal span 35 ), executive functions (trail making test, 37 phonemic and semantic fluency, 35 and reversed span 38 ), and constructive apraxia (drawing from copy and with planning 35 ).
The scores of the 3 groups are reported in Table 2.
The Results of the 3 Groups in the Assessment of Cognitive Functions and Mood.a
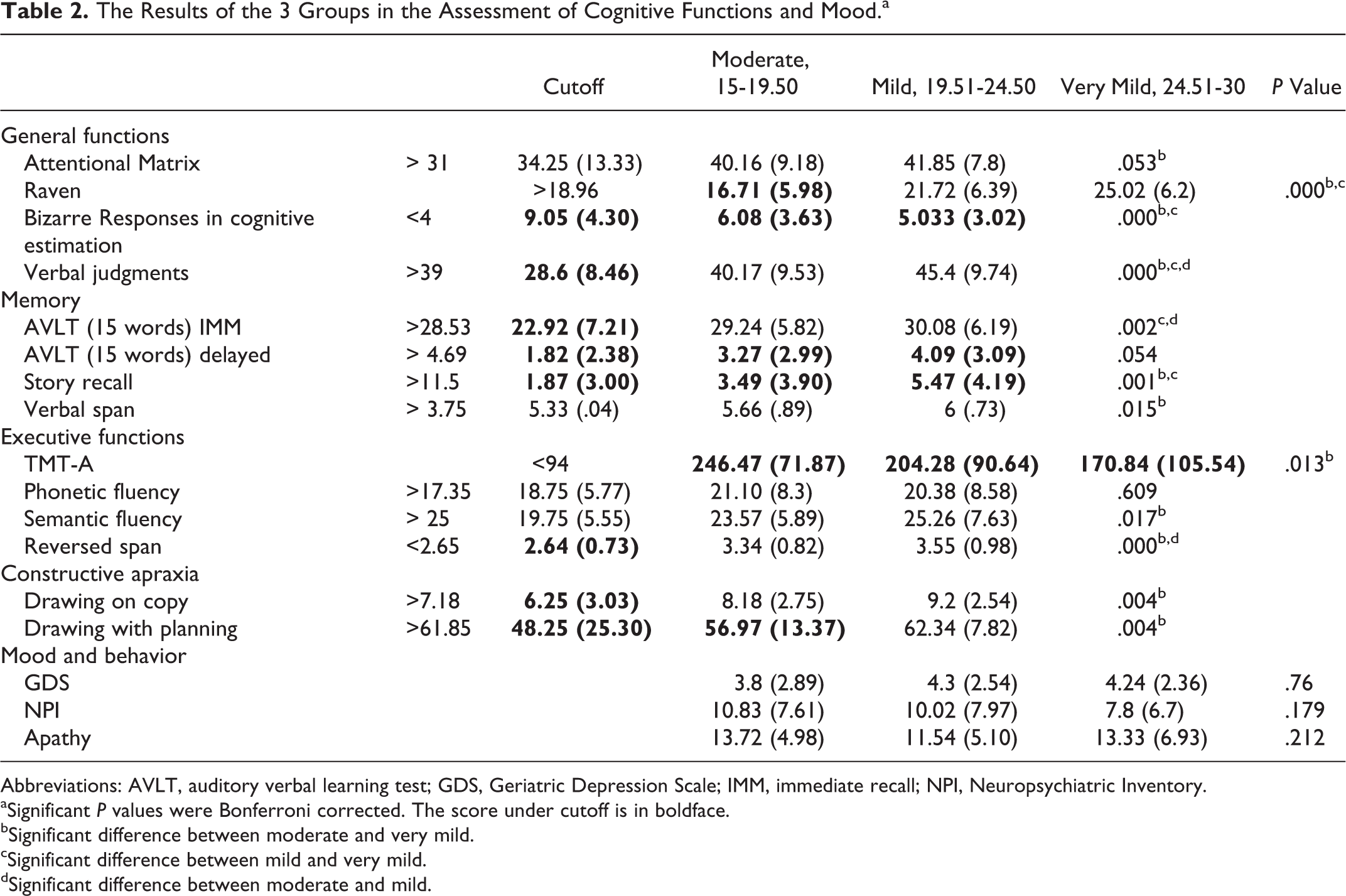
Abbreviations: AVLT, auditory verbal learning test; GDS, Geriatric Depression Scale; IMM, immediate recall; NPI, Neuropsychiatric Inventory.
aSignificant P values were Bonferroni corrected. The score under cutoff is in boldface.
bSignificant difference between moderate and very mild.
cSignificant difference between mild and very mild.
dSignificant difference between moderate and mild.
Psychiatric examination
In order to investigate the presence of affective and behavioral disorders, the Geriatric Depression Scale (GDS 39 ), the Starkstein’s Apathy Scale, 40 and the Neuropsychiatric Inventory (NPI) 41 were administered (Table 2).
Anosognosia assessment
The AQ-D was administered by 4 examiners (NC, FF, VS, and PE). This scale 21,22 consists of 30 questions with the aim of assessing awareness in 2 domains, that is, intellectual functioning (IF) and behavior (BEH). The scores relating to 2 factors in 2 specific subscales provide information regarding awareness concerning IF, that is, basic ADL (bADLs) and IADLs. Scores for awareness concerning changes in the patient’s BEH are taken from 2 other subscales, namely, the depression and disinhibition subscales. Each answer is rated on a 4-point scale (0 = never; 3= always) in 2 parallel versions. Form A is answered by the patient, when necessary with the examiner’s help, and form B (identical to form A but with questions in the third person) is answered by the caregiver who is not given any information about the patient’s responses. Information regarding levels of awareness is taken from the final differential score (ie, the caregiver’s score minus the patient’s score). A positive final AQ-D score indicates that the caregiver has rated the patient as more impaired than the patient has rated himself or herself. Vice versa, a negative final AQ-D score indicates that the patient has overestimated his or her deficits. After preliminary contacts with the authors (Starkstein) requesting permission, the scale was translated from English to Italian by 2 different researchers. A back-translation was then carried out by a native speaker of English in order to ascertain that the Italian version was the semantic equivalent of the published version in English. 22
In order to verify the concurrent validity of the scale in the Italian population, 3 neurologists (GG, AC, and GT) who have experience of diagnoses of dementia classified the patient as aware or unaware basing on the Reed 4-point interview. This is a scale usually employed by health practitioners in order to rate the level of awareness in people affected by AD. The scale considers 4 levels of self-awareness in which the patient (1) easily admits his or her memory deficits (score 1), (2) admits to (sometimes inconsistently) small amount of memory disorders (score 2), (3) is not aware of any impairments (score 3), and (4) angrily insists that no cognitive problem does exist (score 4). 19 Patients were considered anosognosic when realized a score ≥2.
Caregiver examination
Since the caregivers served as informants, the Hospital Anxiety and Depression Scale (HADS) 42 for anxiety and depression and the Caregiver Burden Inventory (CBI) 43 were administered in order to check for any emotional or affective conditions. The scores are shown in Table 3.
Demographical and Clinical Data for the Caregivers.a

Abbreviations: CBI, Caregiver Burden Inventory; HADS, Hospital Anxiety and Depression Scale.
aSignificant P values were Bonferroni corrected.
bSignificant difference between moderate and very mild.
cSignificant difference between moderate and mild.
dSignificant difference between mild and very mild.
Statistical Analysis
Comparison between groups
Statistical analyses were performed using SPSS and STATA software (significant value at P < .05). Comparisons between the 3 groups (corresponding to the 3 classes of MMSE: 15-19.50, moderate AD; 19.51-24.50, mild AD; and 24.51-30, very mild AD) with a gender variable were made using chi-square tests. Univariate 1-way analyses of variance (ANOVAs; Bonferroni corrected) were employed to compare the groups for age, standard of education, bADL, and IADL.
Validation of AQ-D in the Italian version
The internal consistency of the AQ-D scale was estimated by determining Cronbach’s α coefficient. The interrater agreement between examiners was measured by means of the Cohen’s κ coefficient. The Keiser-Meyer-Olkin (KMO) measure was calculated to evaluate sampling adequacy in order to carry out an exploratory factor analysis. Values of KMO equal or above 0.60 are suggested as necessary in order to perform and interpret satisfactorily a factor analysis solution. 44 A principal component factor analysis using varimax rotation was performed on the 30 items of the AQ-D. The model included factors with an eigenvalue >1. An item was considered to load onto a factor if its factor loading score exceeded 0.30. Examination of scree plot was also employed to determine the number of factors. Finally, we calculated the congruence coefficients for the components after varimax rotation between the present study and the results of the previous validations. 21,25
A receiver–operating characteristics (ROC) curve was constructed (referring to the clinical diagnosis of presence or absence of anosognosia—Reed’s score ≥2) to analyze sensitivity and specificity for the possible scores on the AQ-D items concerning the IADL domain. This was done in order to identify clinically significant anosognosia and confirm the original cutoff 21 in the Italian population.
For concurrent validity between AQ-D scores and clinical diagnoses of anosognosia, an ANOVA was carried out with group as the independent variable (anosognosic vs nonanosognosic patients 18 ) and the weighted scores (the discrepancy between caregiver and patient) of the Starkstein’s 4 domains of anosognosia (IF, BHE, bADL, and IADL) as the dependent variable (ie, the total score for each domain divided by the number of items in the domain).
In order to verify the consistency of caregiver judgments, the AQ-D form B and the scores of the MMSE test and the Reed’s interview were inserted in 2 Pearson’s correlation analyses. In addition, a logistic regression was carried out to estimate the potential effects of caregiver’s affective variables in the form B scores.
Finally, differences in frequency of anosognosia between the 3 classes of MMSE were analyzed by means of chi-square tests.
Anosognosia and clinical variables
Pearson’s coefficient (r) was employed to evaluate any association between awareness (AQ-D) and age, MMSE, neuropsychological tests, and psychiatric disorders (GDS, apathy scale, and NPI). The logistic regression was utilized to evaluate any association between the dichotomized awareness status (awareness/unawareness) and MMSE (3 levels, reference: 15-19.50), controlling for gender (reference: male), age, education, ADL (reference ADL <3), and IADL (male, 5 functions: reference IADL<3; female, 8 functions: reference IADL <5). Results were summarized as odds ratios (with a 95% confidence interval [CI]). Furthermore, for each of the 3 groups, t tests were employed to compare the MMSE scores of aware and unaware patients.
Results
Comparison Between Groups
As shown in Table 1, the 3 groups did not differ for gender, age, and education. As expected, they significantly differed in the ADL and IADL scores (ADL: F 2 = 8.22, P = .000 and IADL: F 2 = 11.27, P = .000). The Bonferroni post hoc test showed that ADL scores were significantly lower in the moderate group than in the mild (P = .004) and very mild (P = .000) groups, most probably due to the loss of autonomy typical in advanced stages of the disease. In contrast, in the IADL (which assesses abilities in more complex functions), there was a significant difference between the very mild and the mild AD groups (P = .003) and the very mild and moderate (P = .000) groups with better performance in the first group.
Validation of AQ-D Italian Version
Cronbach’s α coefficient confirmed the excellent internal consistency of the AQ-D (patient responses = .90 and caregiver responses = .91). The KMO analysis revealed a value of 0.78, indicating that correlation matrix was suitable for factors analysis. The exploratory principal component analysis as well as the examination of scree plot confirmed the data of previous validations. 21,25 Four factors in this study explain the 62.6% of variance in the data (1) IADL, corresponding to anosognosia for memory and executive functions deficit, (2) bADL, (3) depression and apathy, and (4) disinhibited behavior.
The factor congruence coefficients between the present results and those of the study carried out by Starkstein et al 21 were: factor 1: 0.654; factor 2: 0.632; factor 3: 0.764; and factor 4: 0.88. The congruence between the 2 studies was therefore high, and the factors observed in the present study replicated the original factor model.
The only differences concern item 29 (less interest in favorite activities), which in our sample was included in factor 1, item 4 (problems with understanding conversations), which is included in factor 3 and item 25 (more irritable), included in factor 4 (Table 4).
Factor Analysis for 130 Patients with AD and Frequency of Caregiver/Patient discrepancy for each item and group.a
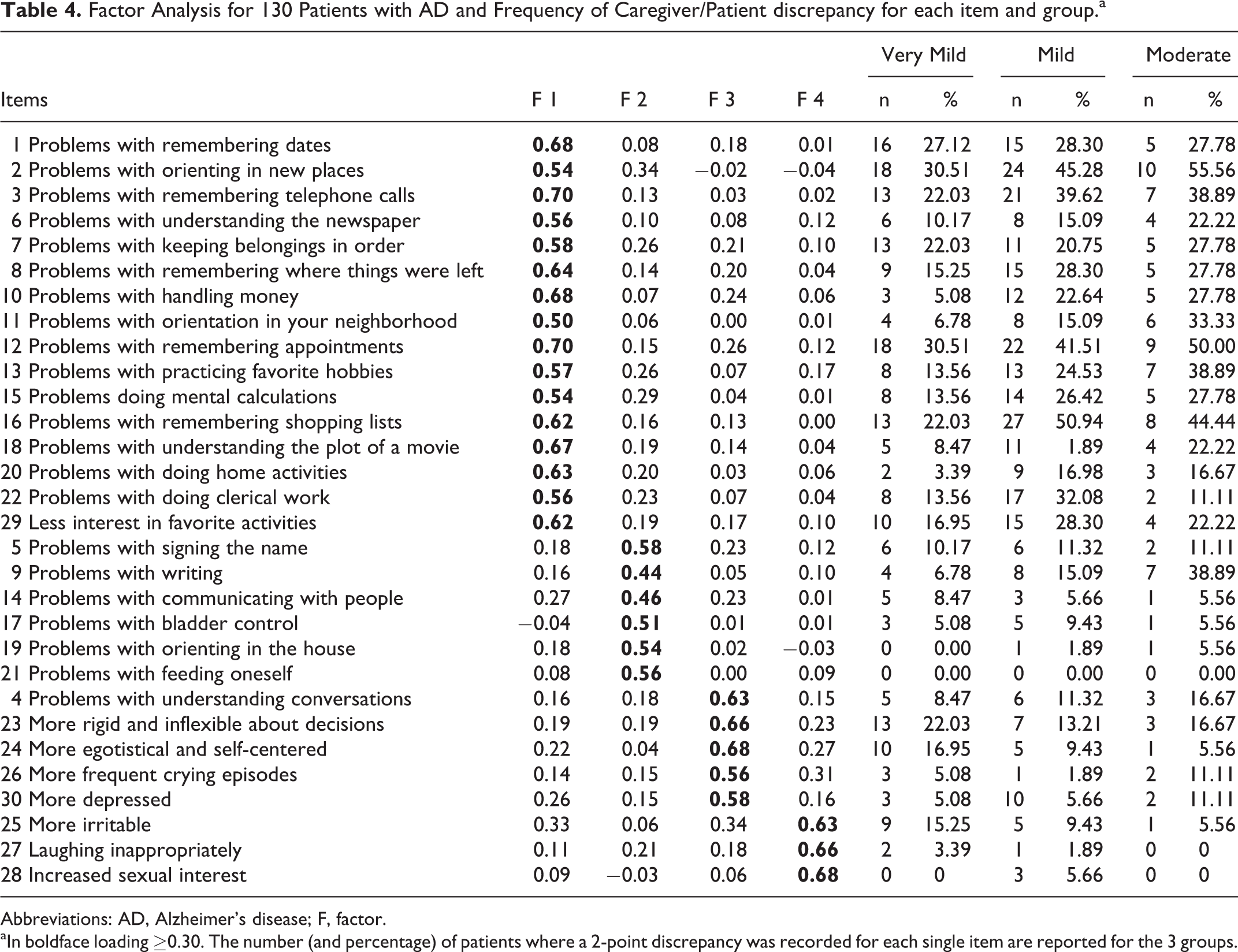
Abbreviations: AD, Alzheimer’s disease; F, factor.
aIn boldface loading ≥0.30. The number (and percentage) of patients where a 2-point discrepancy was recorded for each single item are reported for the 3 groups.
In accordance with the original version of the test, we established that a difference of 1 point would not be clinically relevant for the diagnosis of anosognosia. 21 Thus, a caregiver–patient discrepancy was considered to be present whenever the difference in the respective AQ-D items was at least 2 points (ie, the patient evaluates a deficit as being never present and the caregiver evaluates the same deficit as often or always present, or the patient scores a deficit as rarely present and the caregiver as always present). The ROC statistics were calculated with the clinical diagnosis of anosognosia (presence or absence of deficit in awareness) as the classification variable and the number of IADL items (factor 1) with a 2-point caregiver/patient discrepancy as the criterion. The area under the ROC curve was 0.971 (95% CI, 0.945-0.997), indicating high accuracy. A score ≥4 showed a specificity of 93.98% and a sensitivity of 89.36% for the clinical diagnosis of anosognosia. This confirmed the results of the original version of the scale. 21
The interrater agreement between the examiners was very high for all the items (all Cohen’s κ coefficients K >.882, P = .000).
The concurrent validity of the AQ-D scores and the clinical diagnosis of anosognosia was significant in terms of both the AQ-D total score, F 1,128 = 102.66, P = .000, and the IADL score, F 1,128 = 153.23, P = .000, that is, the score considered in the calculation of the cutoff. In addition, there is significant concurrent validity between the clinical diagnoses and the AQ-D scores for depression, F 1,128 = 14.05, P = .000, but this does not apply to the scores relating to bADL and disinhibition.
The consistency of caregiver judgments is confirmed by the correlation between the AQ-D/form B score (a measure of the caregiver’s perception of deficit) and the MMSE (R = −.33, P = .01). Although the AQ-D scores and the degree of anxiety and depression of the caregiver were associated (HADS anxiety: r = .32; P = .000; HADS depression: r = .33; P = .000), the multiple linear regression analysis indicates that the latter does not influence the AQ-D scores (HADS anxiety: β = .11, t = .84, P = .403; HADS depression: β = .14, t = 1.02, P = .306).
Only the caregivers’ scores in the CBI influence the results in the AQ-D (β = .23; t = 1.85; P = .046). Nevertheless, their judgments correlate with clinical diagnoses of anosognosia, confirming the consistency of the evaluations provided by the caregivers. In addition, multiple linear regression shows that CBI scores are determined by the severity of illness of the patient as assessed by the MMSE (β = −.196; t = −2.59; P = .011) and the time taken for daily assistance (β = .428; t = 5.31; P = .000).
The Diagnosis of Anosognosia
If one considers a discrepancy in ≥4 items as the cutoff, anosognosia is present in all 3 groups, with a general frequency of 36.15% (47 of 130). The deficit correlates with the interval between the expression of the first symptoms and the interview (r = .306, P = .001). This frequency changes in relation to the various different stages of the disease and is thus very similar in the moderate (44.44%, 8 of 18) and mild AD (47.17%, 25 of 53) groups and lower in the very mild AD group (23.73%, 14 of 59). Although this difference is significant, (mild versus very mild, χ2(1) = 6.76, P = .009), this confirms that anosognosia may represent a symptom that is present right from the early stages of the illness. 26 The reduction in awareness of the 3 groups does not seem to correlate completely with cognitive deficits, since in the comparison of MMSE scores between unaware and aware patients, no differences were found in the very mild (t = 0.503, df = 57, P = .617) and moderate (t = 0.558, df = 16, P = .584) groups. In contrast, unaware patients in the mild group have MMSE scores that are significantly lower than aware patients (t = 3.578, df = 51, P = .001). This is probably due to the more accentuated heterogeneity between the patients of this group in comparison to the other groups. In fact, in this “halfway” group, there were patients sited very close to the extremes of the score range (19.5-24.5). Of course, these patients varied for clinical and neuropsychological conditions.
The total number of discrepant items is widely variable (range 4-19). This suggests the possibility of identifying various degrees of anosognosia (Table 5).
Range of Discrepancy in AQ-D Scores in Unaware (Upper) and Aware (Lower) Patients.a
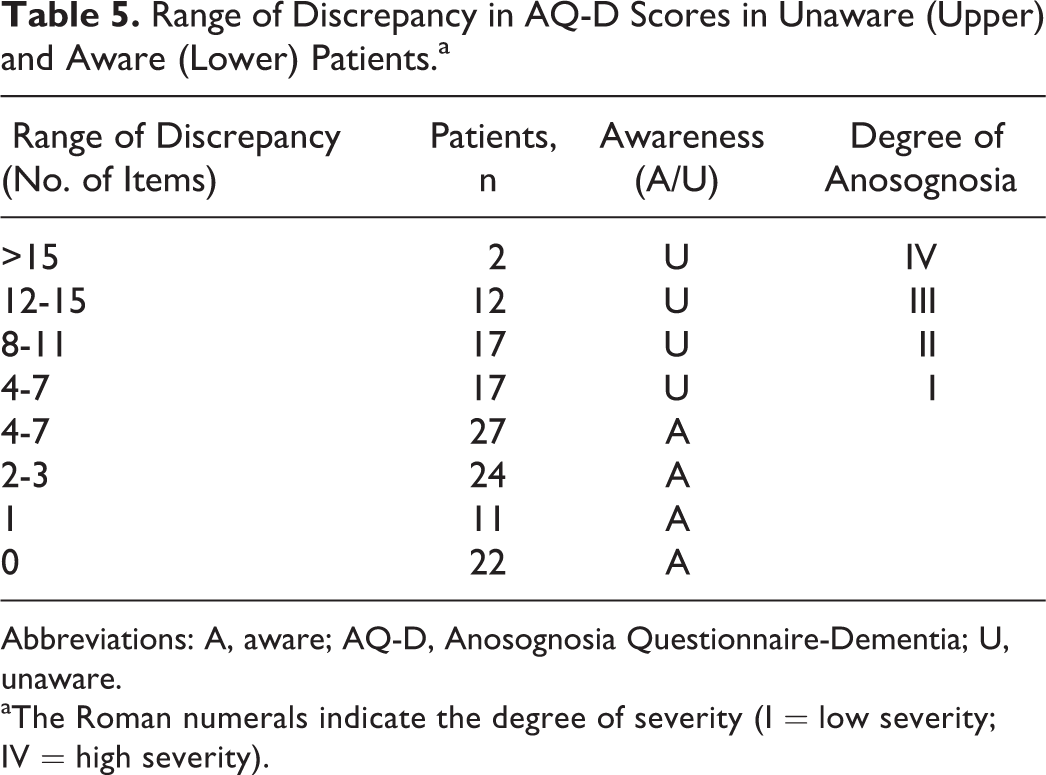
Abbreviations: A, aware; AQ-D, Anosognosia Questionnaire-Dementia; U, unaware.
aThe Roman numerals indicate the degree of severity (I = low severity; IV = high severity).
In line with our hypothesis regarding the degree of severity of anosognosia (4 levels: I, II, III, and IV) as reported in Table 5, it is also interesting to note that the comparisons between patients with different degrees of anosognosia fail to reveal any association with MMSE (all t test P > .05). This would suggest that although the general diagnosis of anosognosia (based on the IADL items) is correlated with the degree of mental deterioration, the total number of functions in which patients do not recognize their deficits does not depend on the severity of the illness in general.
A qualitative analysis of the individual items (Table 4) shows that from the earliest stages of the disease awareness deficits mainly involve disorders in memory abilities (factor 1). In addition, in all 3 groups, our patients do not seem to be aware of their reduced interest in their favorite activities (item 29).
In mild and moderate AD, the patients are unaware of their disorders in executive functions. Only in the moderate stage of the illness does anosognosia involve deficits in understanding the plot of a movie as well as episodes of crying and depression. Symptoms of “affective” anosognosia are typically present in very mild AD, in particular involving rigid and inflexible behavior with respect to decision making (item 23), egotistical and self-centered behaviors, and irritability. These disappear in later stages of the illness.
A qualitative analysis of the results concerning aware patients indicates that a certain degree of discrepancy between patients’ and caregivers’ judgments may also be present, although 4 items of the IADL are not involved thus meaning that anosognosia cannot be diagnosed in these cases (Table 5).
Furthermore, 32.53% (27 of 83) of the aware patients tend to overestimate their deficits, judging their disabilities more severely than the caregivers. The range of these overestimations is 1 to 7 and correlates with depression (r = .252, P = .007). Anosognosic patients also occasionally overestimate their deficits but only in a few situations (19.15%, 9 of 47, range: 1-3), in particular with regard to items involving behavior factors (items 23-30).
Anosognosia and Clinical Variables
The logistic regression model confirmed that only the variable MMSE (24.51-30; 19.51-24.50) is a significant predictor of the expected outcome, that is, to have awareness of cognitive deficits (odds ratio = 6.26; P = .003 in the MMSE range 24.51-30 and odds ratio = 4.365; P = .015 in MMSE range 19.51-24.50). Other variables (gender, age, education, IADL, and ADL) were not significant predictors of outcome (aware vs unaware). Considering the whole sample, also the neuropsychological and behavioral variables, are not predictive of anosognosia.
Nevertheless, in order to avoid floor effects on neuropsychological tests in later stages of dementia, we controlled for potential correlations between these variables and anosognosia exclusively in the mild and very mild groups. This analysis indicates a correlation between anosognosia and cognitive estimation (r = .2, P = .037), verbal judgments (r = −.193, P = .048), and NPI (r = .372, P = .000).
Discussion
The validation procedure demonstrated that the Italian version of the AQ-D maintains the psychometric characteristics of the original version 21 and the Japanese versions as well as the abridged version. 25 Good internal and external reliabilities of the scale and a satisfactory concurrent validity were confirmed with the following 4 factors originally identified by Starkstein and colleagues: (1) IADL, (2) bADL, (3) apathy, and (4) depression and disinhibition. The 2-point discrepancy cutoff in at least 4 IADL items provides a good level of specificity and sensitivity to the scale.
An analysis of the patients’ scores indicated that AQ-D is more informative with respect to assessments based on clinical observation or discrepancies between self-ratings of task performance and objective results. This is due to the fact that it allows measurements not only of the degree of severity but also of the qualitative aspects of anosognosia.
Although in our sample the frequency of anosognosia is lower than in other studies, in the cases where it varies from 80% 43 to 42%, 3 the data confirm our previous results in patients with a similar general cognitive level. 27 Evidence that deficits of awareness may be present from the earliest stages of the illness is also supported. Indeed, while the mild and moderate groups do not differ in terms of the frequency of anosognosia, it is also present in patients with very mild deterioration although less frequently.
The number of items with a discrepancy between patients’ and caregivers’ judgments is variable, ranging from 4 to 15. This indicates that it is possible to identify various different degrees of severity in anosognosia and that these are only partially correlated with the severity of the dementia. Indeed, although the scores in the AQ-D globally correlate with the MMSE, if we consider each of the 4 groups with varying degrees of severity in AQ-D scores individually (Table 5), the patients do not differ from each other in terms of the MMSE.
A qualitative comparison of the scores of the 3 groups suggests the hypothesis that anosognosic symptoms are progressive. In effect, deficits in awareness seem to involve the memory in the first stages and only later involve executive functions. This is in line with previous evidence of various specific types of anosognosia, 5 –8 in particular amnestic and executive anosognosia. 12 Our sample suggests the possibility of a progression in anosognosia, which would start with amnestic anosognosia followed by executive anosognosia. This is only partially explained by the progression of the symptoms. In fact, although the 3 groups show mean scores under cutoff in memory tests, this is also true for executive functions (Table 2) and cognitive estimation. Thus, symptoms of deficits in executive functions may be present from the first stages of the illness, without executive anosognosia.
In addition, a sort of affective anosognosia 9 may be identified in the earliest stages and we found a patient–caregiver discrepancy in items linked to mood. 45 Interestingly, these discrepancies became fewer or disappear in the later stages of the illness. It is possible that the first behavioral changes are immediately noticed and recorded by caregivers, but these changes are not realized by patients who in some way or another are not able to alter their beliefs about themselves. 17,10,46,47 However, the hypothesis that patients develop depressive reactions in the first phases of the disease seems to be excluded in our sample by the absence of depression in anosognosic patients. On the contrary, overestimation (and not anosognosia) is correlated with depression in aware patients. These data expand the debate on the possible link between affective and mood disorders and awareness 48 and confirm previous results indicating that patients with more symptoms of depression show greater consciousness of their cognitive problems. 18,49
Thus, a patient’s difficulty in recognizing changes in behavior and mood might also be considered a prodromal symptom of anosognosia in AD, 5 as confirmed by the correlation between AQ-D and NPI.
In contrast to previous studies, 23 we did not find any correlations with executive functions, such as mental flexibility and inhibition of dominant response. Nevertheless, in the mild and very mild groups, the AQ-D scores correlate with verbal judgments and cognitive estimations, confirming that this interview investigates metacognitive aspects of awareness. Moreover, this correlation between anosognosia and deficits in the ability to analyze and evaluate situations (not specifically self-related) might support the hypothesis that in patients with AD the deficit of awareness may be at least in part exacerbated by general deficits in cognitive estimation and judgment. 5
Although the anxiety and/or depression of the caregivers is correlated with their judgment (but on average below the cutoff for diagnosis), the only measure that impacts the AQ-D scores is the CBI. In other words, the more stressed the caregivers are, the more negative their judgment is. Crucially, the CBI scores also correlate with objective measurements of the level of engagement in assistance (severity of illness, general duration of assistance, and daily time dedicated to assistance). In addition, the form B scores (answered by caregivers) correlate with clinical judgments and with MMSE scores. Due to the aforementioned factors, we consider the caregivers’ scores to be reliable. Nevertheless, this result indicates that it is necessary to associate the AQ-D with a measurement of any stress relating to the caregiver.
This last issue is particularly interesting in the case of aware patients’ responses. As for anosognosic patients, there is extensive variability (range 0-7). Discrepancies concern problems with orienting in new places (item 2); remembering appointments (item 12), remembering things they have to buy when they go shopping (item 16); taking care of their checkbook, accounts, payments (item 22); and being more rigid and inflexible about decisions (item 23). There is a high degree of discrepancy also in the 3 groups of patients for items 2, 12, and 16 and for item 23 in the very mild group. Our suggestion is that these items may in some way represent “sentinel items” and as such would be useful in terms of identifying potential signals of anosognosia and devising specific programs. Further longitudinal investigations are needed to better understand qualitative aspects and the progression of anosognosia.
Conclusions
In conclusion, the present validation demonstrates that the Italian version of the AQ-D is both reliable and valid for a large number of patients with AD. In addition, the questionnaire has revealed that deficits in awareness are present from the earliest stages of AD and that it is possible to find various different degrees of severity of anosognosia with different cognitive domains affected. Finally, the hypothesis that sentinel items do exist may have a great impact on clinical practice.
Footnotes
Acknowledgments
We would like to thank A. Bonazzi and G. Tripi for their help with patient recruitment and testing and for their useful comments regarding the first version of the questionnaire; we also thank G. Bisoffi and O. Bortolami for their help with methodology and statistics; S. Floreani for the back translation of the questionnaire; and Francesco Nicodemo for his help with editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Fondazione Cariverona (Neuroscience Project Disabilità cognitive e comportamentali nelle demenze e nelle psicosi—Prot. N. 1855). VM is funded by the Italian Ministry of Health (Project Code RF-2010-2312912).
