Abstract
An integrative literature review was performed to identify the challenges in current cognitive screening. The aim of the review was to serve as an evaluative resource to guide clinicians in the selection of the best available cognitive screening measures for early assessment of mild cognitive impairment (MCI) in people with chronic diseases. The review classified the available cognitive screening measures according to purpose, time to administer, and cognitive domains assessed as: 1) simple/ brief cognitive screening measures, 2) disease specific screening measures, 3) domain specific screening measures, 4) self-administered screening measures, and 5) technology-based screening measures. There is no single optimal cognitive measure for all patient populations and settings. Although disease specific cognitive screening measures are optimal, there is a lack of validated screening measures for many chronic diseases. Technology-based screening measure is a promising avenue for increasing the accessibility of cognitive screening. Future work should focus on translating available screening measures to mobile technology format to enhance the utility in busy primary care settings. Early cognitive screening in persons with chronic disease should enhance appropriate referrals for detailed neurocognitive examination and cognitive interventions to preserve and or minimize cognitive decline.
Introduction
The United States Census Bureau predicts that the population of adults older than 65 years of age will double and the population older than 85 years of age will triple by 2060. 1 The prevalence of Alzheimer’s disease (AD) is also increasing at an alarming rate. By 2050, the number of people with AD aged 65 and older may nearly triple, from 5 million to as many as 16 million. 2 The annual rate of conversion to dementia in people with mild cognitive impairment (MCI) ranged from 6% to 25% with an average of 10% per year, which is much higher than the dementia incidence rate of 1% to 2% seen in the general population. 3,4 Although MCI is not an inevitable consequence of aging, age is a major risk factor and the risk is greater in people with chronic diseases with profound consequences. 5 The longitudinal Einstein Aging Study reported an overall prevalence of dementia at 4.9%, with amnestic MCI at 11.6% and nonamnestic MCI at 9.9% among community-dwelling older adults. 6 Alarmingly, the prevalence of MCI among older adults with chronic comorbid conditions is reported to be much higher; 28% in persons with heart failure, 26% in persons with chronic obstructive pulmonary disease (COPD), 23% in individuals with cancer, and 14% in persons with diabetes. 7 Given the increased prevalence of MCI among persons with chronic diseases, in this article, we have focused on identifying the etiological heterogeneity for MCI in chronic diseases, challenges in current cognitive neuropsychological evaluation, general review of available cognitive screening measures, and emphasis on utilizing technology-based screening for early assessment.
Mild cognitive impairment is a clinical syndrome that commonly arises as a result of neurodegenerative pathology, and in chronic diseases MCI indicates early cognitive decline beyond the normal range according to respective age and level of education. 8 It is believed that by the time AD is diagnosed, sufficient neuronal injury has occurred to the extent that reversal of the disease is unlikely. 9 Mild cognitive impairment in chronic diseases may result from hypoperfusion or reduced cerebral blood flow, 10 neuronal cell death, 11 focal brain abnormalities ranging from multiple cortical or subcortical infarcts to small vessel disease with white matter lesions and lacunar infarcts, 12,13 cortical atrophy, 14 and gray matter reductions, 15 suggesting an etiological heterogeneity.
Despite recent advances in the identification of MCI-related biomarkers, neuropsychological assessment remains a critical component of evaluation to ensure that cognitive function correlates with biomarker abnormalities to assist in detecting and tracking progression of MCI to early AD. 16 In addition, the long-term progression from MCI to dementia or AD (ie, 3 years) may be predicted by the presence of abnormal levels of brain amyloid neurodegeneration seen by magnetic resonance imaging and positron emission tomography scan. 16
Why Screen for Cognitive Impairment in Chronic Diseases?
Persons with chronic diseases such as heart failure, stroke, COPD, HIV, diabetes, multiple sclerosis, and Parkinson’s disease have higher incidence of MCI that affects daily self-care practices. Persons with chronic diseases are required to detect changes in their physical condition that require them to take action and implement a treatment strategy as prescribed by their physician. 17,18 Changes in memory, judgment, and the inability to complete usual activities can result in poor disease management, impact medication adherence, and ultimately have a negative effect on quality of life. 19 Poor performance on cognitive screening tests suggests the presence of cognitive deficits and provides a rationale for comprehensive follow-up of neurocognitive assessment. 20
The rationale for early cognitive screening in persons with chronic diseases is to make referrals appropriately for detailed neurocognitive examination and cognitive interventions to preserve and or minimize memory decline. 17,18 Although over 50 cognitive screening instruments are available, evidence remains unclear on an ideal cognitive screening tool for use in persons with chronic diseases. Most research studies have used 4 to 16 neurocognitive batteries to determine incidence and risk of cognitive impairment and/or to describe common cognitive domains affected in various chronic diseases. 21,22 A plethora of studies among varied populations have documented that the Mini-Mental Status Examination (MMSE), the commonly used screening instrument, is not sensitive to identify subtle cognitive changes seen in MCI related to chronic diseases. 23 In persons with memory complaints seen in primary care, cognitive screening with MMSE did not identify those who require further examination for dementia, thus defeating the intended purpose of early referral. 24 Despite clear limitations, the MMSE has proved resilient in the clinical arena for more than 30 years.
The common elements that are most predictive of progression of MCI are related to poor performance in neuropsychological testing at baseline and the number of different cognitive domains that are impaired. 25 Current challenges in the neuropsychological evaluation of MCI include test selection, the availability of normative databases, the effect of different base rates of MCI and AD in different settings, establishing cutoff points for cognitive impairment, and developing measures more sensitive to specific chronic diseases, while having sufficient specificity to distinguish between etiologically different chronic conditions. 25 In addition, cognitive complaints experienced by older adults are overtly masked by the presence of chronic diseases. 26 Because, MCI associated with chronic disease is subtle and is often not identified by clinicians until the person displays an inability to carry out everyday activities that are often reported by family members and/or caregivers. 27 Such subtle cognitive impairment challenges the persons’ ability to learn and remember self-care management for chronic disease conditions such as heart failure, 28 diabetes, 29 COPD, 30 hypertension, 31 and multiple sclerosis. 32 Evidence also indicates that MCI associated with chronic diseases is associated with poor medication adherence, 33,34 poor quality of life, 35 –37 and increased mortality rate. 38,39 A prospective study of patients with heart failure (N = 577) reported an association with poor self-care adherence and memory impairment. 28 Evidence also indicates increased readmissions rates on people who have MCI associated with chronic diseases such as heart failure and thus the associated cost. 28
Current Standard of Cognitive Screening in Persons With Chronic Diseases
The clinical expert consensus guidelines recently recommended early identification of MCI in order to optimize medical management, improve self-care for chronic diseases, offer better understanding of symptoms associated with chronic disease conditions, maximize decision-making autonomy, and planning for the future. 40 A report from the Einstein aging study showed that 50% to 66% of persons with chronic diseases seen in primary care offices were found to have dementia, yet no such diagnosis was documented in the medical record. 6
The gold standard for assessment of cognitive abilities necessitates systematic and detailed evaluation of a broad range of cognitive processes using multiple neuropsychological batteries. 41 Such detailed examination is not always feasible for use by clinicians in busy outpatient clinic settings due to cost and time. 20,42 Although multiple cognitive screening instruments are available, it is unlikely that a “one size fits all” approach is ideal for cognitive screening due to inconsistencies in the selection and use of screening measures as well as a lack of robust evidence to support the many screening measures available for use in clinical practice. 43
Therefore, the aim of this integrative literature review was to examine evidence on available cognitive screening measures to serve as an evaluative resource to guide clinicians in the selection of the best available cognitive screening measures and provide rationale for the use of novel technology-based cognitive screening that would be both feasible and valid for early cognitive screening among persons with chronic diseases.
Data Source and Results
The Cochrane Database of Systematic Reviews followed by PubMed, Medline, EMBASE, CINAHL, and PsychINFO databases were searched using the subject terms “cognition disorders,” “cognitive impairment,” “cognitive dysfunction,” “memory,” “attention,” “screening,” “screening tool,” and combined them with names of multiple chronic diseases. The searches were then combined with AND, limits to the search were placed for humans and English language. The goal was to identify available disease-specific cognitive screening measures as well as general cognitive screening measures used among these chronic disease conditions. Additional articles were identified by hand searches from reference lists and articles included in systematic reviews and from authors who have published in this field. In addition, the initial studies for cognitive screening measures identified were gathered to identify their psychometrics.
The review classified the cognitive screening measures and grouped them based on the time to administer the measure, mode of administration, domains of cognition assessed, and specific purpose of the measures. From the 240 articles identified, the measures were categorized as (1) nonspecific simple/brief cognitive screening measures for a total of 29 measures, 44 –71 (2) disease-specific cognitive screening measures consisting of 14 disease-specific cognitive screening measures for 6 chronic disease conditions (ie, Parkinson’s disease, HIV, stroke, multiple sclerosis, cancer, and schizophrenia), 72 –85 (3) 17 domain-specific cognitive screening measures, 86 –102 (4) 12 self-reported and telephone-based screening measures, 103 –113 and (5) 7 technology-based screening measures utilizing the Internet or mobile apps. 114 –120 These measures are presented in Tables 1 to 5. Note these are not all exhaustive lists.
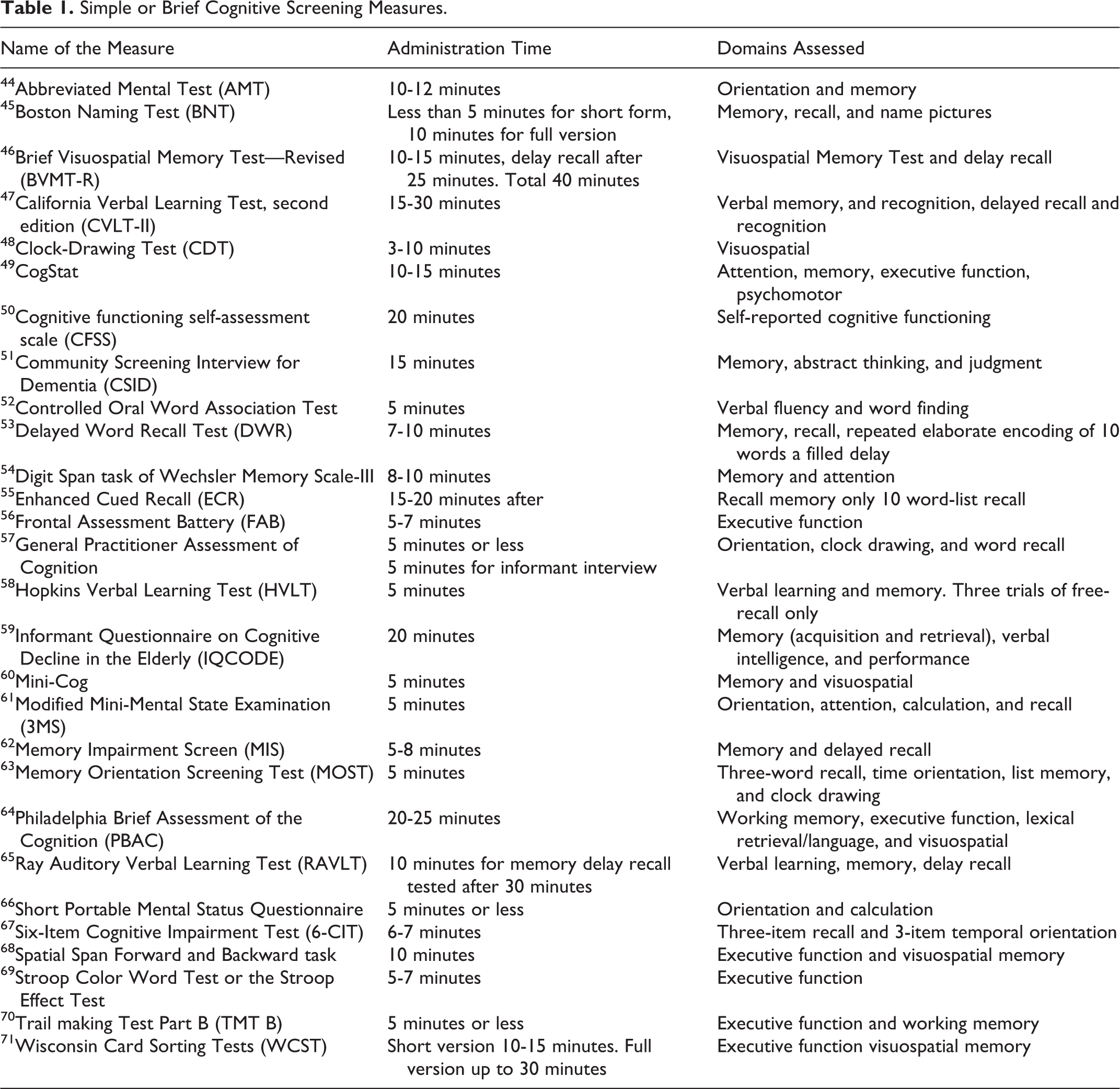
Simple or Brief Cognitive Screening Measures.
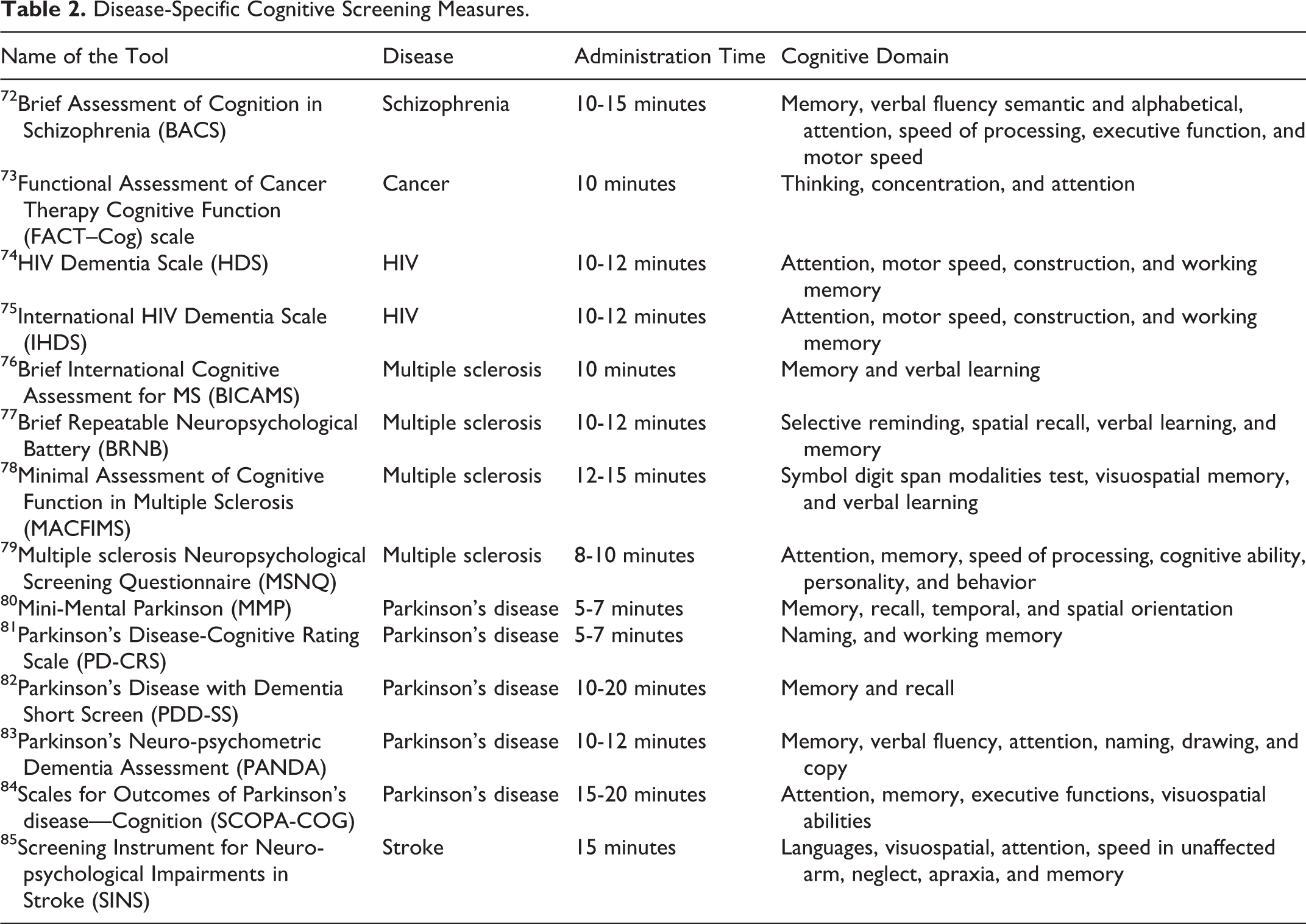
Disease-Specific Cognitive Screening Measures.
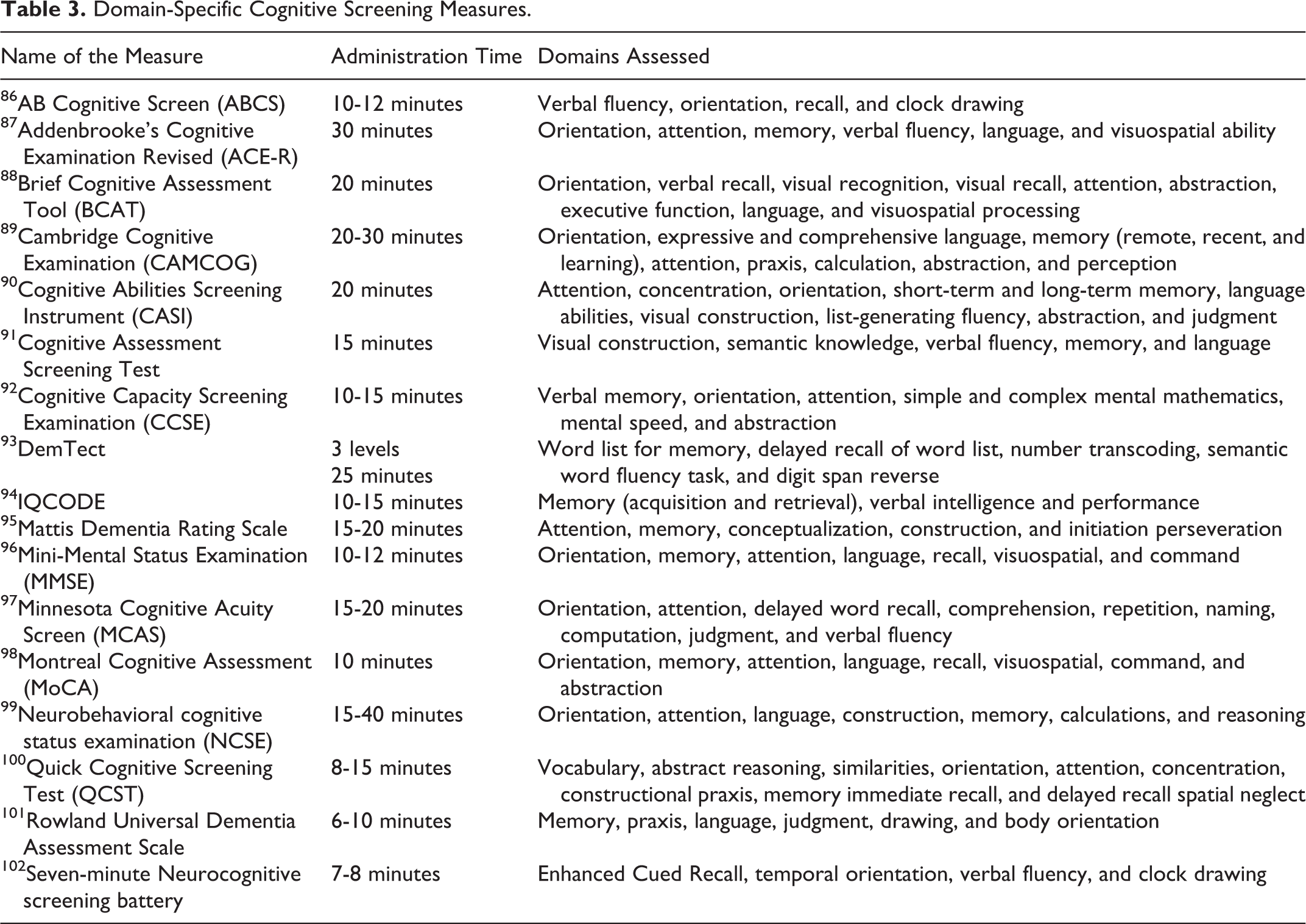
Domain-Specific Cognitive Screening Measures.
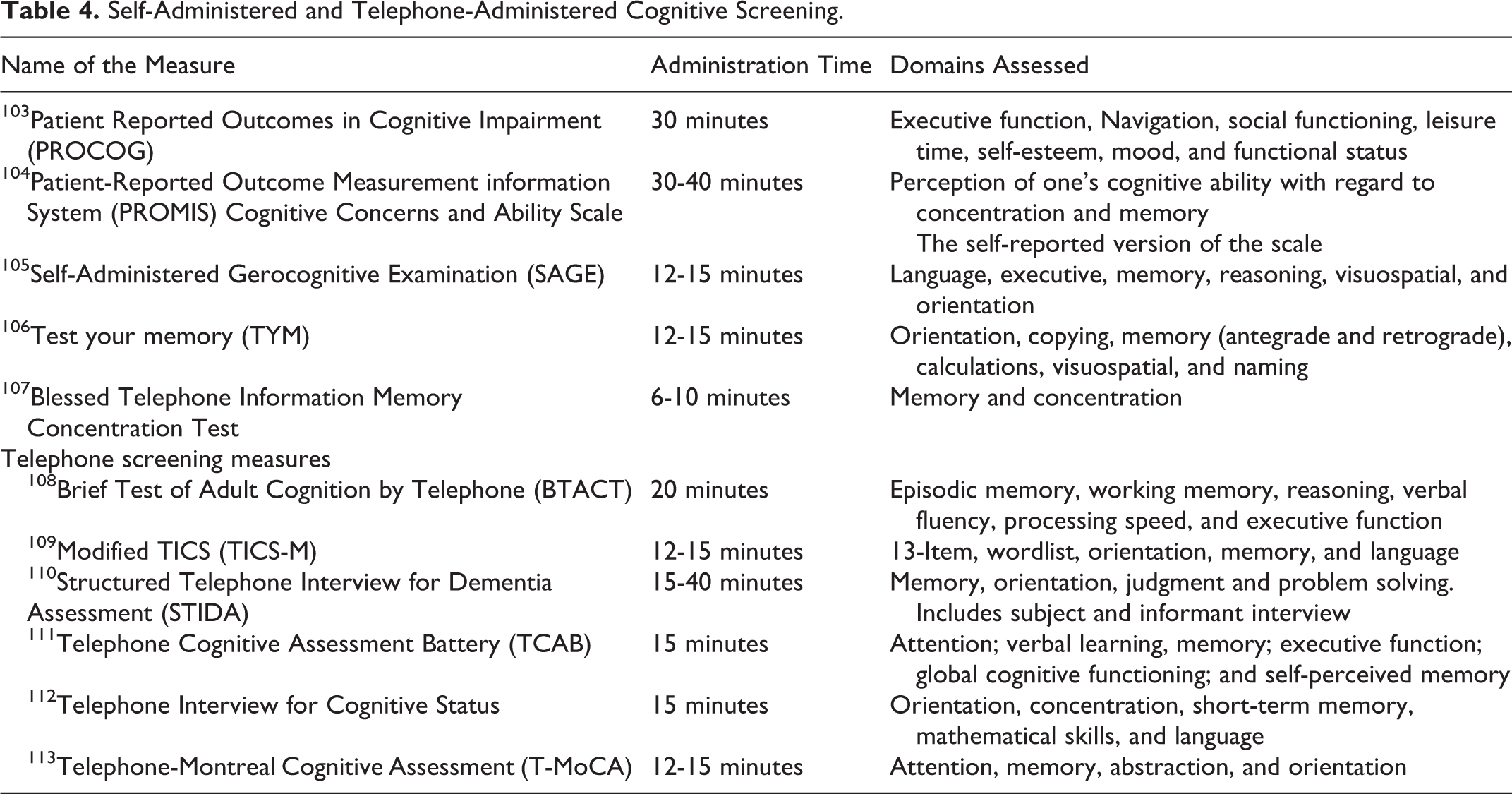
Self-Administered and Telephone-Administered Cognitive Screening.
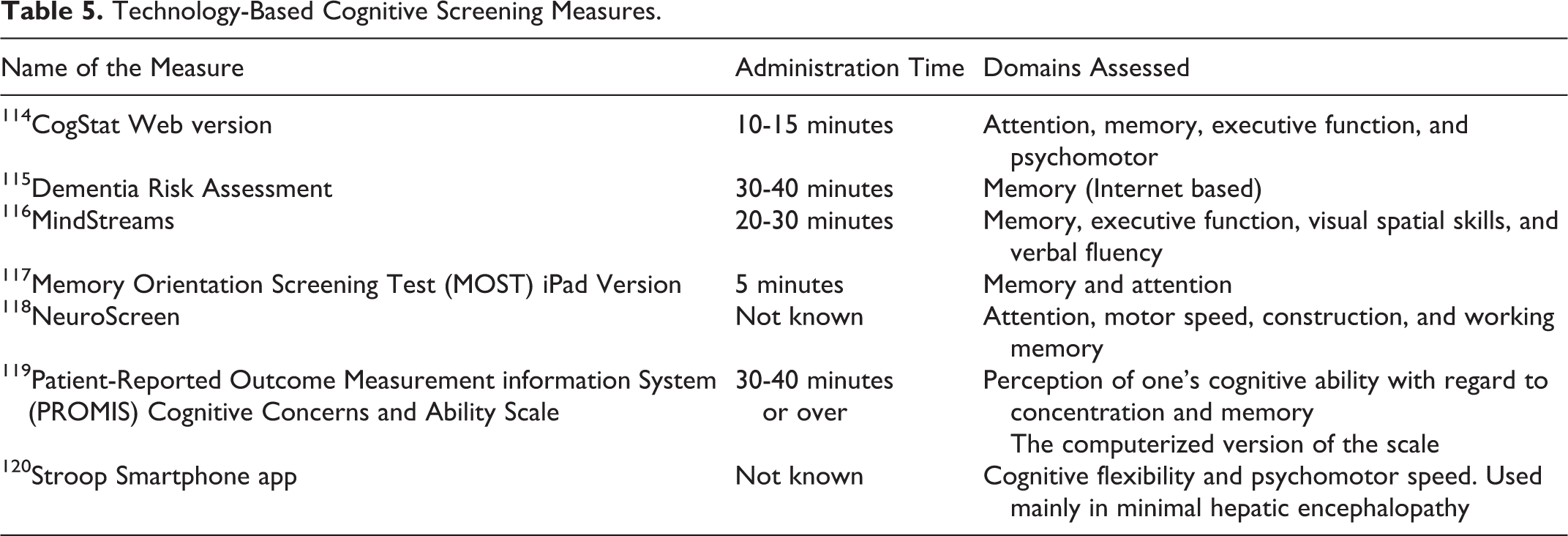
Technology-Based Cognitive Screening Measures.
Discussion of Results
Utility of Simple or Brief Cognitive Screening Measures
Although brief, the general practitioner assessment of cognition (GPCOG), 57 Mini-Cog, 60 memory impairment screen, 62 and memory orientation screening test (MOST) 63 are simple yet structured measures that are valid and suitable for assessment of cognitive function in persons with chronic diseases. Each measure has the unique benefits that they are easy to administer by clinicians or nurses, take less than 5 minutes to administer, and have been validated in the primary care or community setting. The GPCOG has patient and informant (family or caregiver) components that can be used alone or together to increase specificity and sensitivity. 57 The Mini-Cog has been validated in population-based studies and in community–dwelling older adults heterogeneous with respect to language, culture, and education. 60 Kansagara and Freeman reviewed 6 brief cognitive screening measures that could serve as possible alternatives to the MMSE for use by the US Department of Veterans Affairs. 121 The review provided evidence that the Mini-Cog has the shortest administration time (2-4 minutes) and has been studied in a large population sample as well as in multiethnic samples compared to other brief cognitive measures. 121 This was supported in a review that assessed the clinical utility of the GPCOG and Mini-Cog in which both were found to be equally high in sensitivity and specificity. 122 Similarly, the MOST offers an accurate assessment of cognition and could be used in the primary care setting. 123 These brief cognitive screening measures may be ideal to screen cases of dementia from clinics or the community as a first step measure but are considered not appropriate for use in a primary care setting to screen for MCI in persons with chronic diseases. 122 Although controversial, the utility of routinely asking about memory problems with patients and family members followed by a brief cognitive assessment method in patients with a positive response offers a platform for early referral and follow-up for a detailed neuropsychological examination (see Table 1).
Disease-Specific Cognitive Screening Measures
Results from this review identified validated disease-specific cognitive screening measures for only 6 chronic disease conditions, namely, Parkinson’s disease (5 measures), 80 –84 multiple sclerosis (4 measures), 76 –79 with 2 measures for use in HIV, 74,75 and 1 measure each for use in stroke, 85 cancer, 73 and schizophrenia, 72 all of which were developed in recent years. The evolution of new disease-specific cognitive screening measures in recent years clearly indicates the need for a detailed synthesis of available evidence for use by clinicians and researchers. Strengthening the evidence by utilizing existing screening measures works to strengthen the validity and reliability of the currently available screening measures rather than the design of new measures. When the MMSE was compared with the disease-specific screening measures, the MMSE could not achieve the required 80% sensitivity at any cutoff score in persons with Parkinson’s disease. 84 This example illustrates that when possible clinicians should use validated disease-specific screening measures for accurate measurement of cognitive domains affected by specific chronic diseases. This warrants further examination of cognitive domain-specific cognitive screening measures compared with disease-specific cognitive measures to further increase their utility and validity for early assessments. Table 2 provides a list of the most commonly used disease-specific cognitive screening measures. This is, to our knowledge, a comprehensive list of the available cognitive screening measures specific for chronic diseases.
Utility of Cognitive Domain-Specific Screening Measures
Although there is no single cognitive screening measure that is considered to be the gold standard, domain-specific cognitive screening measures that assess 5 or more cognitive domains are considered the second best. Most domain-specific cognitive measures take more than 10 minutes to administer. The commonly used tests for screening MCI are the MMSE, 96 Montreal Cognitive Assessment (MoCA), 98 Mattis Dementia Rating Scale, 95 Addenbrooke’s Cognitive Examination Revised (ACE-R), 87 and the Neurobehavioral cognitive status examination. 99 These measures are often used to validate simple/brief screening measures as well as disease-specific cognitive measures. The MoCA has shown better sensitivity than the MMSE, takes a short duration of 10 to 12 minutes for administration, and has a wide application in routine clinical practice. 98 The MMSE lacks sensitivity in identifying subtle cognitive symptoms of MCI associated with chronic diseases such as in Parkinson’s disease, 124 heart failure, 125 and stroke. 126 Similarly, compared to ACE-R, the MMSE demonstrated inferior accuracy. 127 In addition, the MMSE is not available in the public domain anymore and charging a fee for clinical use has become an issue because of the MMSE copyright. Domain-specific cognitive screening measures offer clinicians’ processes to better describe patients’ cognitive symptom profile for further assessment and to make appropriate referrals; therefore, when disease-specific screening measures are not available, providers should consider using domain-specific cognitive screening measures such as the MoCA or ACE-R.
Self-Administered Cognitive Screening
The self-administered screening measures are designed to rapidly screen large numbers of individuals in the community or practice at the same time, they require no setup of a computer, minimal operator time to administer, test a reasonable range of cognitive functions, and are sensitive to mild AD. 128 The test your memory self-administered measure indicated a sensitivity of 93% and specificity of 86% in the diagnosis of AD but has not been tested on patients with MCI and in chronic disease condition. 106 Similarly, the Self-Administered Gerocognitive Examination (SAGE), a pen and paper examination, encourages patients to complete the self-administered test and take the completed test to your primary care physician for interpretation and management. 105 However, SAGE requires that individuals must be literate and have adequate vision and writing skills to answer the questions, thus it demands active patient participation and provider training to score the measure, which may be challenging. 105 In addition, the Patient Reported Outcomes in Cognitive Impairment (PROCOG) has a self-administered detailed screen, which includes 55 items for a total score of 220. 103 The PROCOG as well as the Patient-Reported Outcome Measurement information System Cognitive Concerns and Ability Scale takes more than 30 minutes to complete and can be very exhaustive for patients. 103,104 Often cognitively impaired persons shy away from being tested and may not fill out the self-administered test if they take more than 10 minutes.
More recently, cognitive screening measures have been tested for validity when administered via telephone. 107 –113 Currently, several cognitive training intervention trials have utilized telephone screening to determine eligibility. However, these measures are not widely tested for their utility in the primary care setting.
Utilizing Technology for Screening, the Future of Cognitive Screening
In the last 3 decades, many validated cognitive assessment measures have been adapted into an alternative and attractive strategy to in-person cognitive assessments by utilizing Web version screening measures that can be self-administered such as the CogStat.
115
The validated NeuroScreen
119
was compared with the Samsung Galaxy Note® smartphone assessing the same cognitive domains that showed good correlation (
Online screening is becoming more popular among community-dwelling adults. Recently, community-dwelling older adults (≥60 years) with subjective memory complaints (n = 30) and no subjective memory complaints (n = 30) participated in an online screening program containing the Cognitive Symptom Checklist and the informant Questionnaire on Cognitive Decline in the Elderly shorten version.
130
All 100% of participants completed the online assessment without assistance, including 1 woman who had no previous experience in using computers indicating the feasibility of technology-based screening.
130
Recently, the paper version of the MOST was compared with a computerized (iPad app) version among 98 older adults and demonstrated an intertest correlation of .92 (
Discussion for Clinical Practice
Recognizing that there is no single optimal screening measure, clinicians may often be left in limbo to select the appropriate screening measure and the best mode for administration. Our review indicates the utility of disease-specific cognitive screening measures as first choice where available and/or an alternative domain-specific sensitive measure such as the MoCA or structured brief screening measures including GPCOG or Mini-Cog. The GPCOG is recommended as the next best, since it had both a patient and an informant questionnaire. 107 Combining an informant questionnaire with other simple or domain-specific measures improved accuracy and diagnostic utility. 106 The GPCOG is a 1-page scale that can be administered in 5 minutes for a total score of 9. A score of 0 to 4 indicates the need for detailed assessment and a score 5 to 15 warrants the need to get the informant interview that includes 6 questions for a score of 6, and a score of 0 to 3 warrants detailed cognitive assessment. 106 The MoCA, a domain-specific validated measure showed excellent sensitivity and specificity among multiple chronic diseases. 124 In addition, incorporating a simple screening measure such as the MOST within an annual wellness visit may provide an objective measure of cognition that will enable providers to develop a better personalized cognitive screening and a more accurate assessment for monitoring memory change over time. 63 –117,123
The Centers for Medicare and Medicaid Services elected not to recommend a specific screening measure but encourages providers to use the algorithm for assessment of cognition and use structured cognitive assessment tools for both patients and informants utilizing GPCOG or an alternative measure such as the MoCA. 131 Unfortunately, evidence suggests that up to 81% of patients who met the criteria for MCI have never received a screening and had no documented diagnosis for MCI. 40 The first step in diagnosing MCI or dementia in persons with chronic diseases is to develop a best practice to screen all patients during annual wellness visits to primary care clinics. The individual practices need to develop a protocol for annual cognitive screening.
Patients waiting to be seen by the provider may complete cognitive screening using computer-based tests or hand-held devices such as a smartphone or tablet. Early detection and documentation of early cognitive impairment is vital in improving medical care and patient outcomes. Diagnosis and documentation of early cognitive impairment could inform all clinicians involved in patient care to determine treatment options that may require aspects of self-care and adjustments to accommodate cognitive decline. 40
Conclusion and Clinical Recommendation
In summary, clinicians should routinely screen for MCI in their patients, especially in patients with chronic diseases. When possible, the choice of test used for screening should be disease specific. However, there is a lack of validated cognitive screening measures for many chronic diseases. There is a need for simple measures that take less than 10 minutes such as the GPCOG, Mini-Cog, and MOST that require little training for health care personnel to administer. The Pew Internet survey reported that 90% of American adults own a mobile phone, 58% of those are smartphones, and 52% use social networking. 132 Mobile technology is transforming clinical practice for health care providers and offers powerful tools that are ultraportable and easy to use. Hence, smartphone-based cognitive screening tests may offer solutions in increasing the number of patients screened and treated for MCI in the early phases of cognitive decline. Future work should focus on translating available validated screening measures to a mobile technology format to enhance the utility of these screening tools in the busy primary care setting. More research is needed at the primary care setting to utilize technology-based smartphone apps to screen for early cognitive changes and offer clinical recommendation to halt the progression of MCI to AD among persons with chronic disease.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
