Abstract
Background:
To assess whether biomarker τ protein could differentiate between Alzheimer’s disease (AD) and vascular dementia (VaD).
Methods:
We conducted a comprehensive search to identify studies on τ protein, patients with AD, and patients with VaD. Cerebrospinal fluid (CSF) τ protein levels were compared to discriminate among patients with AD, healthy controls, and patients with VaD by a meta-analysis.
Results:
Patients with AD exhibit significantly higher CSF τ protein levels than healthy controls or patients with VaD in the Chinese population.
Conclusion:
Our findings suggested that CSF τ protein levels were found to be significantly associated with AD in the Chinese population. Measurement of τ protein could help in attenuating the strict distinction between AD and VaD.
Keywords
Introduction
Dementia is a clinical syndrome characterized by progressive cognitive impairment, 1 which is a rapidly growing health and socioeconomic problem worldwide, as it is estimated that 24 million people have dementia nowadays and that this will increase to 42 million by 2020 and 81 million by 2040. 2 Alzheimer’s disease (AD) and vascular dementia (VaD) are the 2 most common causes of dementia in the elderly patients, accounting together for more than 70% to 75% of cases. 3 Multiple genes and environmental factors underlie the etiology of AD, whereas VaD is defined as loss of cognitive function resulting from ischemic, hypoperfusive, or hemorrhagic brain lesions due to cerebrovascular disease or cardiovascular pathology. 4 However, the differential diagnosis between AD and VaD is not always easy in clinical practice because of lack of sensitivity and specificity of current clinical diagnostic criteria. 5 A most successful biomarker-based method of predicting AD has been the analysis of cerebrospinal fluid (CSF). In various biomarkers, τ protein is a microtubule-associated protein located in neuronal axons, which has been used in various configurations as a diagnostic marker of degenerative changes in the central nervous system. 6 In recent years, τ protein has been thought to be a general marker for neurodegeneration and a common factor in the pathology of various types of dementia. 7 Particularly, abnormal hyperphosphorylation of the microtubule-associated protein τ and its incorporation into neurofibrillary tangles (NFTs) are major hallmarks of the pathogenesis of AD. 8 So CSF τ protein has also been advocated as a diagnostic biomarker of AD. Several studies have demonstrated that CSF τ protein levels are increased in patients with AD9 –11 and may be of some help in the diagnostic workup of patients with AD. 12 –15 These give a prompt to investigate τ protein to explore its potential use for differential diagnosis of AD and VaD. Significantly, recent studies also found that CSF τ protein might be helpful to differentiate AD from VaD, 16 although some studies showed conflicting results on this biomarker for the discrimination between AD and VaD. 17,18 For instance, Wang et al found that there were no differences in CSF τ protein levels between VaD and AD. Likewise, Itoh et al also claimed that patients with VaD had CSF τ protein levels similar to those having AD, and CSF τ protein cannot discriminate AD from VaD. 19 Until now, the use of CSF τ protein in the differential diagnosis of AD and VaD was controversial. They remain difficult to differentiate in practice. To our knowledge, no previous meta-analysis article has compared the CSF biomarker for the differential diagnosis of AD and VaD. Therefore, we perform the present meta-analysis to assess whether measurement of CSF τ protein levels could be a clinically informative diagnostic adjunct that differentiates the 2 major types of dementia.
Materials and Methods
Identification of Studies
Eligible studies were identified by searching in the Cochrane Central Register of Controlled Trials (CENTRAL), PubMed, EMBASE, China National Knowledge Infrastructure (CNKI), and Chinese Biomedical Database (CBM) for relevant trials without language restrictions using the following mesh search terms: Cerebrospinal fluid,tau protein, Chinese population, Vascular Dementia, dementia, Alzheimer's Disease, and case-controlled trial. Terms were exploded whenever possible in each database. We fixed February 2013 as the cutoff date for inclusion of the studies.
Inclusion Criteria for Relevant Studies
We planned to include studies if they met all of the following criteria: (1) case–control design; (2) studies needed to include patients with clinical definite VaD and AD according to National Institute of Neurological Disorders and Stroke and Association Internationale pour la Recherché et l'Enseignement en Neurosciences (NINDS-AIREN) criteria 20 ; (3) healthy persons without dementia as controls; and (4) original articles containing independent data, and data were expressed as mean ± standard deviation. Studies would be excluded if they were no original data, no controls, review articles, and overlapping data.
Outcome Measures for This Review
Two independent reviewers obtained full manuscripts of all citations that were likely to meet the predefined selection criteria. They independently reviewed the articles and extracted the data from the included studies. Areas of disagreement or uncertainty were resolved by discussion and consultation with a third reviewer. When multiple articles were published from a single study, we selected the report that contained the most complete and relevant data on the association among CSF τ protein levels, patients with AD, and VaD. The study characteristics, number of patients and controls, and data of CSF τ protein levels were extracted from each selected article. Standardized mean difference (SMD) and 95% confidence interval (CI) were calculated to estimate the association among CSF τ protein levels, patients with AD, and VaD. The levels of τ protein in CSF were all measured using enzyme-linked immunosorbent assay in the included studies. We calculated statistical heterogeneity by an chi-square test on N − 1 degrees of freedom with P <.1 indicating significant heterogeneity. 21 To evaluate the heterogeneity, we also used the I2 test, taking values in the range 0% to 100%. 22 The I2 values of 25% may represent low, 50% modest, and 75% large heterogeneity, respectively. 23 When P ≥.1 or I2 ≤ 50% and indicated a lack of heterogeneity, the fixed effect model was employed to pool all studies for summary SMD estimation. Otherwise, the random effect model was chosen. Potential publication bias was examined using funnel plots. 24 A sensitivity analysis was also performed by repeating the meta-analysis and omitting each study at each iteration. 25 The calculations were performed using the Revman 5.1 Statistical software.
Result
A total of 139 studies were identified from the initial search, we identified 16 studies for the meta-analysis. 9 –11,14,18,26 –36 We excluded 123 studies for the following reasons: reviews or case reports, the publications dealt with other topics, duplication of data, using other diseases as control, or not available as full-text articles. All eligible studies considered were in case–control design. Selected characteristics of the included studies are summarized in Table 1. These studies represented data from 370 patients with VaD, 476 patients with AD, and 418 controls in the Chinese population. There was significant statistical heterogeneity among the studies, thus a random effect model was employed. Pooling all studies for summary SMD estimation, patients with AD have significant high CSF τ protein levels, compared to patients with VaD or controls (SMD = 1.06, 95% CI: 0.82-1.30; Figure 1; SMD = 1.94, 95% CI: 1.61-2.28; Figure 2, respectively). There was an evidence of an association between increased CSF τ protein levels and patients with AD. In addition, patients with VaD also showed significantly elevated τ protein levels compared with controls (SMD = 1.01, 95% CI: 0.78-1.25; Figure 3) but much lower as compared with those of AD. The results remained the same when we performed a secondary analysis by repeating the meta-analysis and omitting each study at each iteration. Publication bias may be acceptably low, because all the funnel plots on the correlation among patients with VaD, patients with AD, and controls for the included studies did not reveal obvious signs of publication bias (Figure 4).
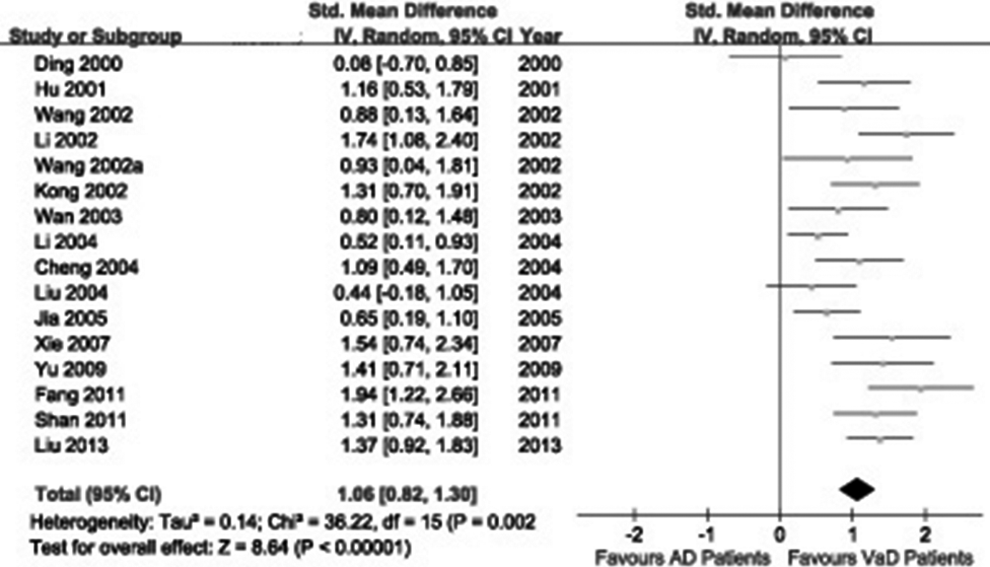
The cerebrospinal fluid (CSF) τ protein levels in patients with Alzheimer’s disease (AD) compared to patients with vascular dementia (VaD) in the Chinese population.
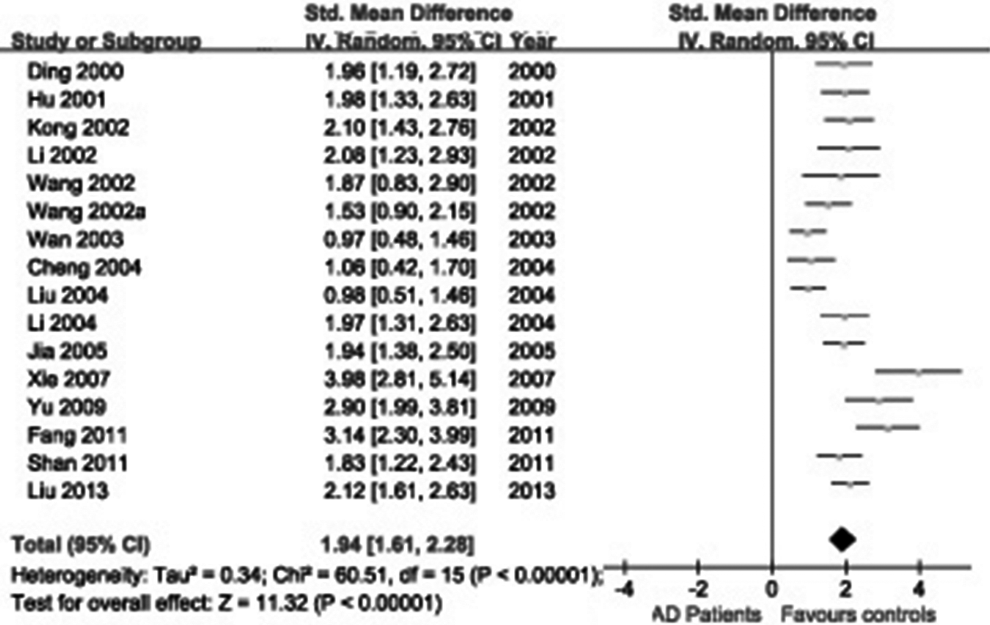
The cerebrospinal fluid (CSF) τ protein levels in patients with Alzheimer's disease (AD) compared to patients with healthy controls in the Chinese population.
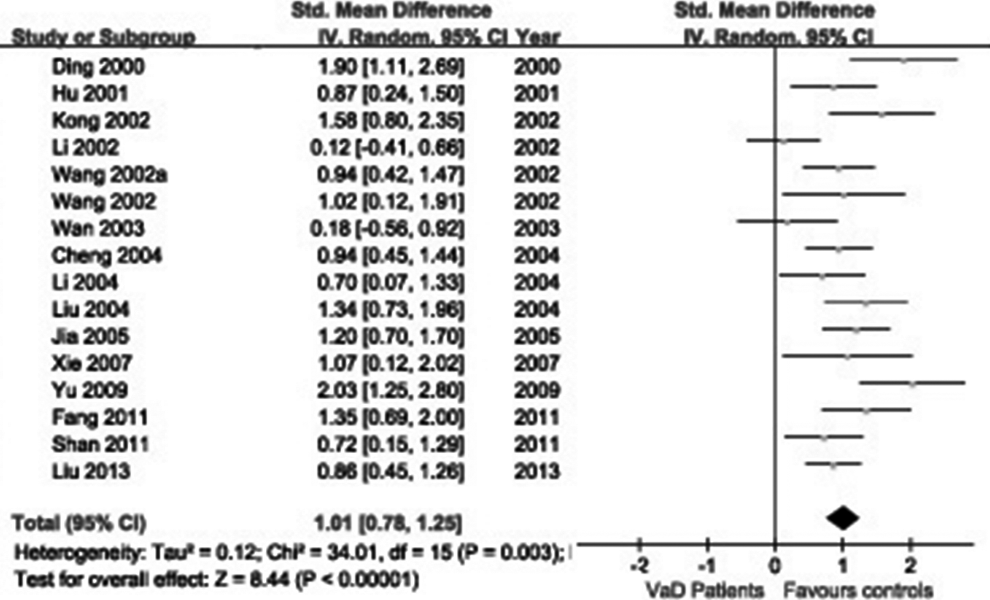
The cerebrospinal fluid (CSF) τ protein levels in patients with vascular dementia (VaD) compared to patients with healthy controls in the Chinese population.
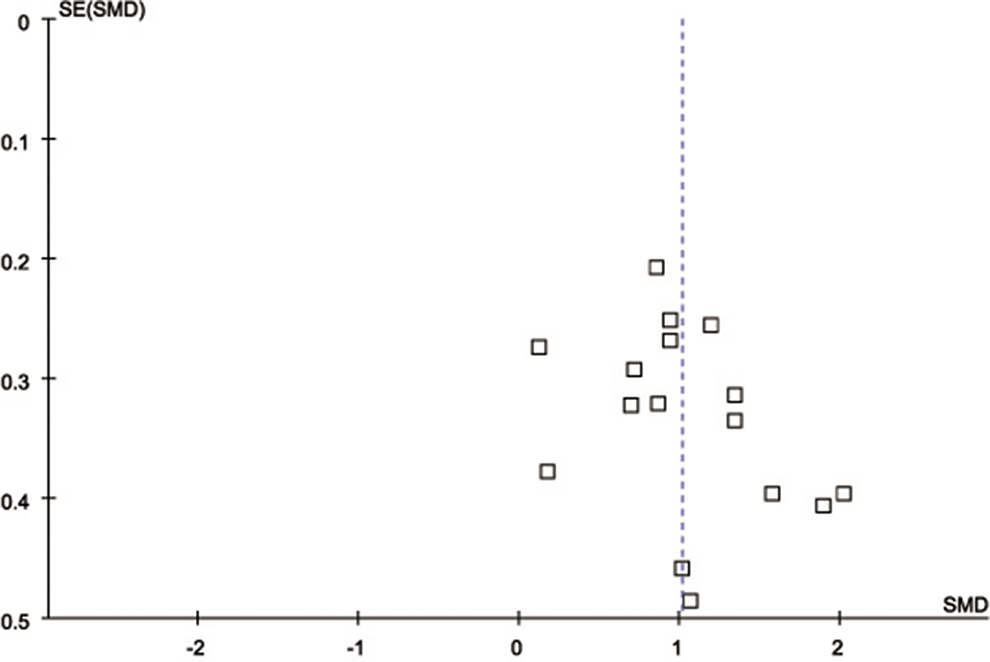
Funnel plots for the included studies.
The Characteristics of All Studies Included in the Meta-Analysis.
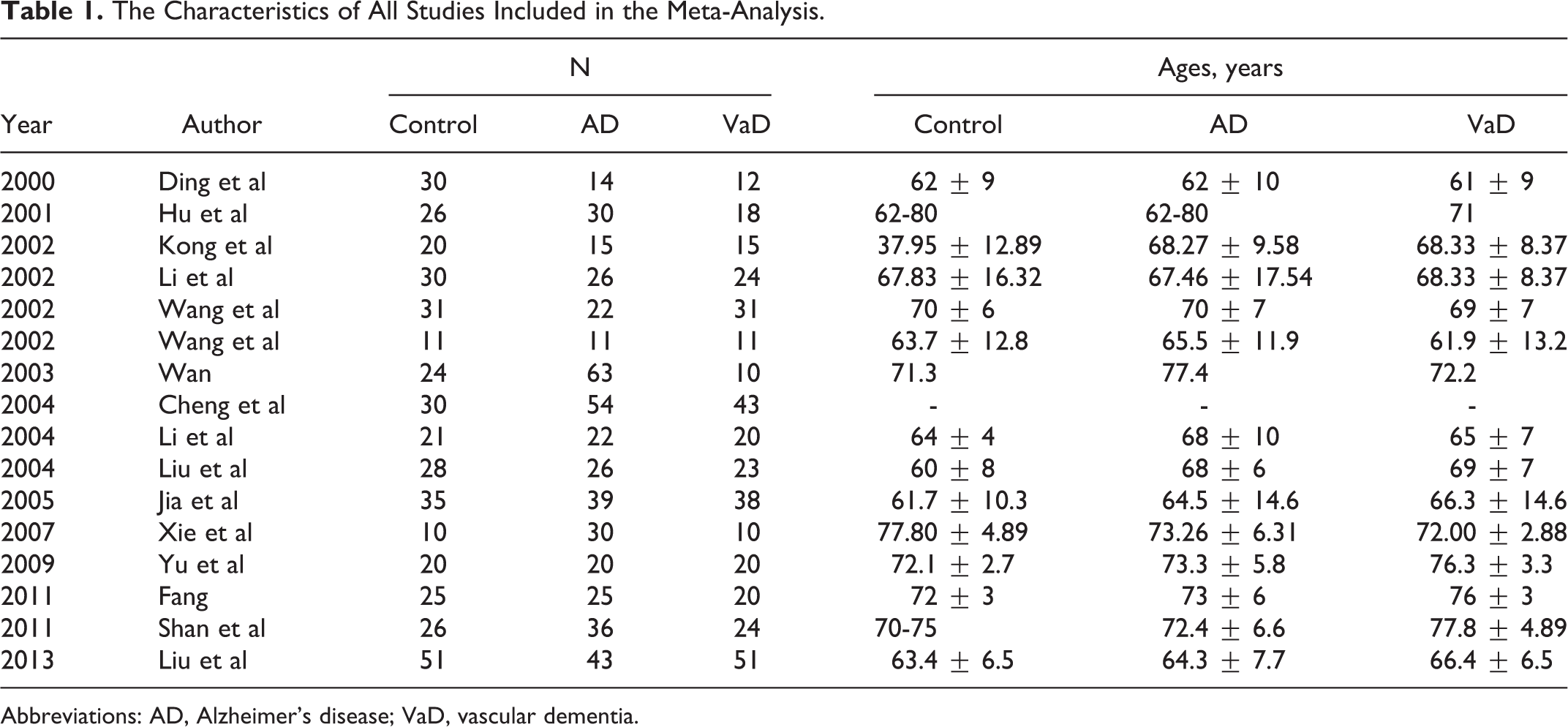
Abbreviations: AD, Alzheimer’s disease; VaD, vascular dementia.
Discussion
Alzheimer’s disease and VaD are now recognized to share some common risk factors and mechanisms including atherosclerosis, diabetes, and amyloid angiopathy, 37,38 while some experts even suggest a vascular contribution in the etiology of AD. 39 –41 Besides, VaD is potentially preventable and/or treatable, in contrast to the currently less treatable situation in AD. Thus, it is important to distinguish VaD more reliably and consistently from AD, although VaD is often difficult to differentiate from AD, because the sensitivity and specificity of clinical criteria are limited. So, in the search for an early diagnosis of AD, we should focus on evaluating biological markers as a complement to clinical assessments. Ancillary biomarkers might also be used to provide additional support for the differential diagnosis. One of the most currently investigated markers of AD is τ protein, a normal brain phosphoprotein that promotes the assembly and stability of neuronal axons by binding to microtubules. 42 To date, the pathogenetic role of τ protein in AD might be due to the following mechanism. The τ protein is a normal protein in axons that binds to microtubules and thereby promotes assembly and stability of microtubules. 42 In normal brain, τ protein is thought to have no specific structure, but in AD τ protein assumes an ordered conformationally altered aggregate form that interferes with its normal functions and may have additional toxic consequences. 43 In the AD brain, τ protein is abnormally hyperphosphorylated, and hyperphosphorylated τ (P-τ) is the main component of paired helical filaments, which form NFTs and neuritic plaques (NPs). 44 Both NFTs and NPs are hallmark neuropathologic lesions in AD. 43 Besides, P-τ starts intracellularly and is regulated by the balance between kinases and phosphatases and eventually leads to disassembly of microtubules and impaired axonal transport thereby compromising neuronal function. 15 Moreover, abnormal P-τ also undergoes several other abnormal posttranslational modifications. 45 The abnormal modifications of τ, especially hyperphosphorylation, appear to lead to neurofibrillary degeneration in AD. 46 All the above-mentioned evidences support a crucial role of τ protein in AD neurodegeneration. Recent studies have found that τ protein might be a valuable tool to discriminate AD from patients with VaD. For instance, Van Harten et al’s study found that increased CSF τ protein can be interpreted as evidence for the diagnosis of AD, with specificity exceeding 85% for comparisons with VaD. 1 Moreover, the results from Paraskevas et al’s study suggested that CSF τ protein offers significant additional information, over that of clinical criteria of AD, for the discrimination of AD from VaD. 47 In the same manner, Bibl et al’s findings also suggested that CSF τ protein could sufficiently discriminate probable AD and VaD from each other. 17 Presently, consistent evidence indicated that CSF τ protein levels are increased in patients with AD, 14,48 but the real diagnostic value of this index is still uncertain. Conversely, there are some studies with opposite results. Some of them found that patients with AD did not show any significant differences in CSF τ protein levels when compared to patients with VaD. 17–18,41 Patients with AD generally showed comparably elevated CSF τ protein levels when compared to patients with VaD. Furthermore, some patients with VaD do present with high or, sometimes, very high τ protein levels as well, which is supposedly conditioned by the coexistence of neuropathological alterations similar to those in AD. 49 –51 To date, the conclusions remain controversial. In our study, we have demonstrated that patients with AD have significantly high CSF τ protein levels, compared to patients with VaD or controls. The CSF τ protein levels were increased in patients with AD. This finding is in part consistent with previous studies analyzing CSF τ protein levels. 32 –35 These seem to suggest that the increased CSF τ protein levels indeed reflect aspects of the diseases themselves. So we could hypothesize that measurement of τ protein levels in the CSF seems useful to discriminate AD from VaD. However, high CSF τ protein levels have also been reported in patients with VaD in our study. This implied that increased τ protein levels support the diagnosis of AD, but they do not preclude a diagnosis of VaD. But, in face, the increase in CSF τ protein levels in patients with AD clearly exceeds the increase in VaD. Thus, CSF τ protein levels could help in attenuating the strict distinction between AD and VaD. On the basis of these findings, we could think increased CSF τ protein should serve as a potent biomarker for AD, even be a useful marker for differentiating AD from VaD. Despite the association between the levels of τ protein in CSF and underlying AD pathology, measures of biomarker accuracy for clinical diagnosis vary widely between studies. Such variability also underscores the need for the development of additional biomarkers that will, either on their own or in combination with more established markers, increase diagnostic accuracy. 52 Over the last decade, many biomarkers have shown statistical differences between AD and controls. The τ protein, β-amyloid 42 (Aβ42) and p-τ protein, indices of amyloid deposition, axonal damage, or tangle formation, respectively, have been suggested as biomarkers for the diagnosis of dementia disorders. 53 They also show potential to improve the diagnostic accuracy for AD. In patients with AD compared with controls, CSF concentration of Aβ42 has consistently been shown to be decreased, while P-τ protein levels are increased. 14 Furthermore, CSF analysis of amyloid Aβ42 and p-τ protein has also been advocated as diagnostics biomarker for AD. Smach et al’s study found that the combination of Aβ42 and τ protein at baseline yielded a sensitivity of 85.29% for the detection of AD, and the specificity was by 96.77% to differentiate controls. 54 Likewise, Ravaglia et al also noted that CSF levels of P-τ can be considered as a virtually specific biochemical marker of AD. 44 These imply that a combinative use of CSF levels of τ protein, P-τ protein, and Aβ42 might improve diagnostic accuracy of AD, even differentiate between AD and VaD. Results from recent studies also indicated that a combinative use of CSF biomarkers such as τ protein, P-τ, and Aβ42 could improve diagnostic accuracy of AD. 51,55 Moreover, Engelborghs et al showed that Aβ42, τ protein, and p-τ in CSF may serve as biomarkers in the diagnostic workup of patients with AD, even differentiate between AD and VaD. 12 A study in a large memory clinic cohort also found that CSF Aβ42, τ protein, and p-τ are useful in diagnosis of differential dementia. 56 Further studies are needed to explore their potential use for diagnosis, differential diagnosis of AD, and VaD. Interestingly, research progress during the last 2 decades has resulted in brain imaging, in particular magnetic resonance imaging (MRI) and positron emission tomography (PET), and CSF biomarkers that facilitate an accurate diagnosis of AD already during the MCI stage. 57 Brain imaging biomarkers include MRI measurements of hippocampal volume to gauge brain atrophy as well as PET measurements of fluorodeoxyglucose to assess glucose metabolism rate in brain cells (neurons and glial cells) in specific brain regions as well as global cortical retention of amyloid-binding ligands such as Pittsburgh compound B. 58 The PET imaging of Aβ in amyloid plaques has been obtained with a number of PET ligands including 18 2-(1-{6-[(2-[F-18]fluoroethyl)(methyl)amino]-2-naphthylethylidene)malononitrile (FDDNP), a compound that binds to Aβ and τ. 59,60 Small et al found that FDDNP-PET (PET was performed after injection of FDDNP) scanning can differentiate persons with MCI from those with AD and those with no cognitive impairment. 60 It is conceivable that the simultaneous use of multiple modalities will become a useful technique in the early diagnosis ofAD. 61 Neuroimaging markers are particularly useful in the diagnosis of AD. So we could think that the combination of these markers could help in the diagnosis of AD, even differential diagnosis of AD and VaD. The usefulness of longitudinal data needs to be explored.
Some limitations of this study should be discussed. First, the number of patients and controls, although well defined, is relatively small. We surveyed case–control studies related to τ protein, patients with AD, and patients with VaD. Using comprehensive search and review of the references, nonetheless, we could not make certain that all relevant studies had been included. Second, all eligible studies were published articles. It is possible that some relevant unpublished studies that may have met the inclusion criteria were missed. Thus, some inevitable publication bias may exist in the results, even though the publication bias was not detected with funnel plots presently. Third, we excluded some studies only as they did not provide enough data. This could have caused a selection and eliminating bias. Finally, our analyses did not consider the interaction of ethnicity and environmental factors, which could have confounded the ethnicity associations.
Conclusion
In conclusion, CSF τ protein levels were found to be significantly higher in patients with AD than in patients with VaD or controls in the Chinese population. Measurement of τ protein may significantly improve the differential diagnosis of AD and VaD. The combination of biomarkers, such as Aβ42, τ protein, and p-tau protein, may come close to fulfilling proposed criteria of a biological marker for AD.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
