Abstract
In this study, the importance of plasma viscosity (PV) as a biomarker in differential diagnosis of dementia subtypes especially Alzheimer’s disease (AD) and vascular dementia (VaD) was investigated. Our study recruited 45 patients with AD, 35 patients with VaD, and control participants. Individuals with inflammatory disease, infection, heart, liver, renal failure, and with high erythrocyte sedimentation rate and C-reactive protein levels were excluded from the study. The cases underwent comprehensive geriatric assessment. The PV measurements were performed with Brookfield DV-II viscometer. The PV measurements of AD, VaD, and control groups were 1.61 ± 0.08, 1.70 ± 0.06, and 1.48 ± 0.06 mPa S, respectively. The PV levels of the dementia group were significantly higher than the control group (
Introduction
Dementia characterized by the impairment of cognitive functions, such as memory, learning, orientation, language functions, perception, decision making, and planning, is the loss of intellectual and social abilities to a level that would hinder daily functioning of the individual. Alzheimer’s disease (AD) is the most common cause of all dementia cases affecting 50% to 70% of this population. 1 Vascular dementia (VaD) is accepted as the second most common cause according to several authors. The VaD is a form of cognitive impairment developing due to ischemic brain damage caused by cerebrovascular disease, infarction, and leukoaerosis. 2 The VaD constitutes 10% to 25% of all dementia cases. 3 Interestingly, AD and VaD have common risk factors such as hypertension, hypercholesterolemia, hyperhomocysteinemia diabetes mellitus, and apolipoprotein E. 4,5
As the treatment and follow-up procedures for AD and VaD are different in clinical practice, various guidelines and tests have been developed for differential diagnosis. Despite such efforts, as these 2 types of dementia have overlapping etiopathogenesis and findings, imaging techniques such as magnetic resonance imaging (MRI) can even be insufficient for differential diagnosis at times. 6 Techniques such as cerebrospinal fluid studies, single photon emission computed tomography, and positron emission tomography are invasive and expensive. 7,8 Hence, the need for noninvasive, practical, and cheap biochemical markers for AD and VaD is obvious.
Certain studies stated that the pathophysiologies of AD and VaD could be ethiologically linked. Inflammation of the neurons and the endothelium is the most important ethiological factor. In population-based studies, increased serum high-sensitivity C-reactive protein (hs-CRP) concentrations have been associated with poor memory, poor global cognitive performance, and VaD and AD. 9 –12
Changes in the rheological system expressing the blood and plasma viscosity (PV) are known to play important roles in the pathogenesis of atherosclerosis and several other diseases. Among the hemorheological parameters, PV was demonstrated as an independent risk factor for several diseases and primarily for atherosclerosis. 13,14 In a study that was performed, in the long-term follow-up of patients having basal PV elevations, the loss of cognitive function was higher when compared to those with normal PV. 15 There are studies showing a positive correlation between PV, cerebrovascular disease, and VaD. 16
With this study, we aimed to demonstrate whether PV can be used as a biomarker in clinical practice for the differential diagnosis of dementia subtypes especially AD and VaD.
Methods
Participants of the Study
Our cross-sectional study recruited 45 patients with AD and 35 patients with VaD over the age of 65 years admitting to geriatrics outpatient unit together with 62 age- and sex-matched individuals without dementia as a control group. Those with dementia had recently been diagnosed and had not received any treatment for dementia. For all the patients covered in the study, comprehensive geriatric assessment was performed in addition to having a detailed history and physical examination.
Patients with collagen tissue diseases, vasculitis, diabetes mellitus, infectious diseases, malignancies, hematological diseases associated with hyperviscosity, liver, kidney (those with glomerular filtration rates of 60 mL/min per 1.73 m2 or below), and heart failure, patients with erythrocyte sedimentation rate (ESR) 40 mm/h and above, CRP and fibrinogen elevations, and those using anticoagulants such as coumadin or agents such as pentoxifylline and/or trimetazidine were excluded from the study.
Information about the cases was obtained by face-to-face interviews with the patients and their close ones as well as using the hospital files. Data pertaining to age, sex, height, body weight, education level, personal history, existing treatment, and comorbidities of the patients were recorded.
Comprehensive Geriatric Assessment and Cognitive Assessment
A medical history was obtained and a physical examination was performed; a comprehensive geriatric assessment including activities of daily living ([ADLs]
17
lower scores indicate less ability), instrumental activities of daily living ([IADLs]
18
lower scores indicate poorer ability), Mini-Mental State Examination (MMSE),
19
clock drawing test,
20
and Yesavage Geriatric Depression Scale (GDS)
21
was performed. We made the initial diagnosis of dementia based on
Laboratory Tests
Blood samples were obtained using venipuncture of the antecubital vein after an overnight fast and analyzed immediately. For both the groups, complete blood count, liver and renal function tests, serum electrolytes, lipid profile, thyroid function tests, ESR, CRP, vitamin B12, folic acid, fibrinogen, lipoprotein a, and homocysteine levels were measured. The CRP levels were measured with rate nephelometry and plasma proteins were measured by the Biuret method. Fibrinogen measurement was performed by radial immunodiffusion. Turbidimetric immunoassay was used to determine the lipoprotein a levels. Quantitative measurements of homocysteine levels were performed using a fluorescence polarization immunoassay. Serum folic acid and vitamin B12 were measured using electrochemiluminescence immunoassay.
Plasma Viscosity Sampling
A blood sample was placed into a tube containing EDTA anticoagulant for viscosity analysis. Measurement of PV was performed using Brookfield DV-II viscometer (Brookfield, Stoughton, Massachusetts). One milliliter of plasma sample was separated by centrifugation at 3000 rpm for 10 minutes and used for viscosity measurement. Viscosity was measured at shear rates of 100, 20, and 5/s at 37°C. The sample was analyzed in duplicates and the mean viscosity was determined. The intraassay coefficient of variation for the viscometer was 0.33%. In the method that we used, PV was expressed in millipascal second (mPa S).
The hospital ethics committee approved the study protocol and all patients gave informed consents. Informed consent was taken from relatives when the patient with dementia could not give an informed consent.
Statistical Analysis
SPSS (version 15.0; SPSS Inc, Chicago, Illinois) statistical package program was used for the evaluation of data. The results were expressed as mean ± standard deviation (mean ± SD) and median (minimum–maximum).
Results
The mean ages of AD, VaD, and control groups were calculated as 76.4 ± 7.8, 77.2 ± 7.7, and 75.5 ± 4.8 years, respectively (Table 1). The groups had similar age and sex features. Other demographical features are expressed in Table 1.
Demographical Features of the Patients.
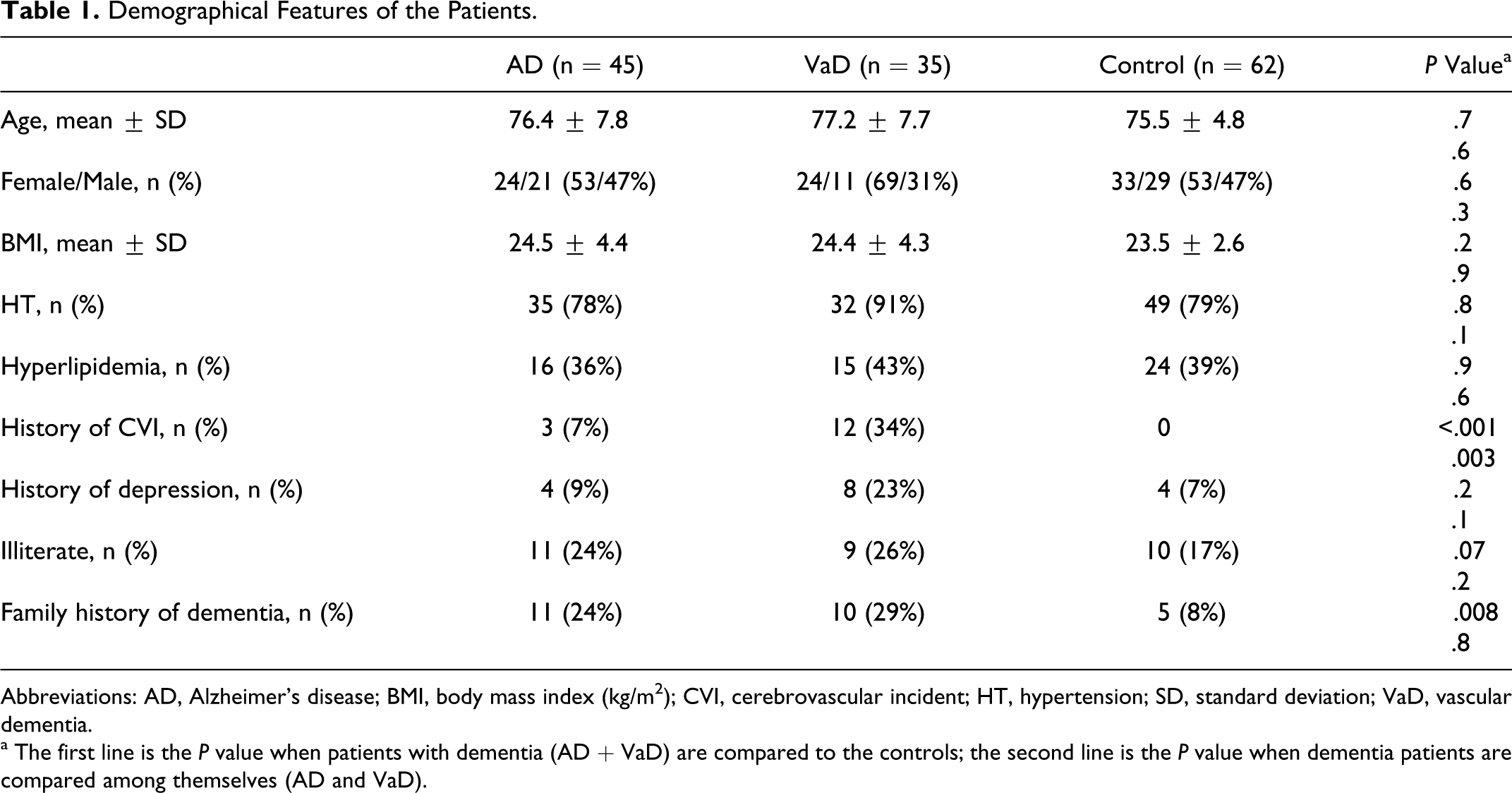
Abbreviations: AD, Alzheimer’s disease; BMI, body mass index (kg/m2); CVI, cerebrovascular incident; HT, hypertension; SD, standard deviation; VaD, vascular dementia.
a The first line is the
For the AD, VaD, and control groups included in the study, mean ± SD, median, and
The PV values for AD, VaD, and control groups were 1.61 ± 0.08, 1.70 ± 0.06, and 1.48 ± 0.06 mPa S, respectively. The PV levels of patients with dementia were significantly higher than the controls (
Geriatric Evaluation Tests of the Cases and Laboratory Results.
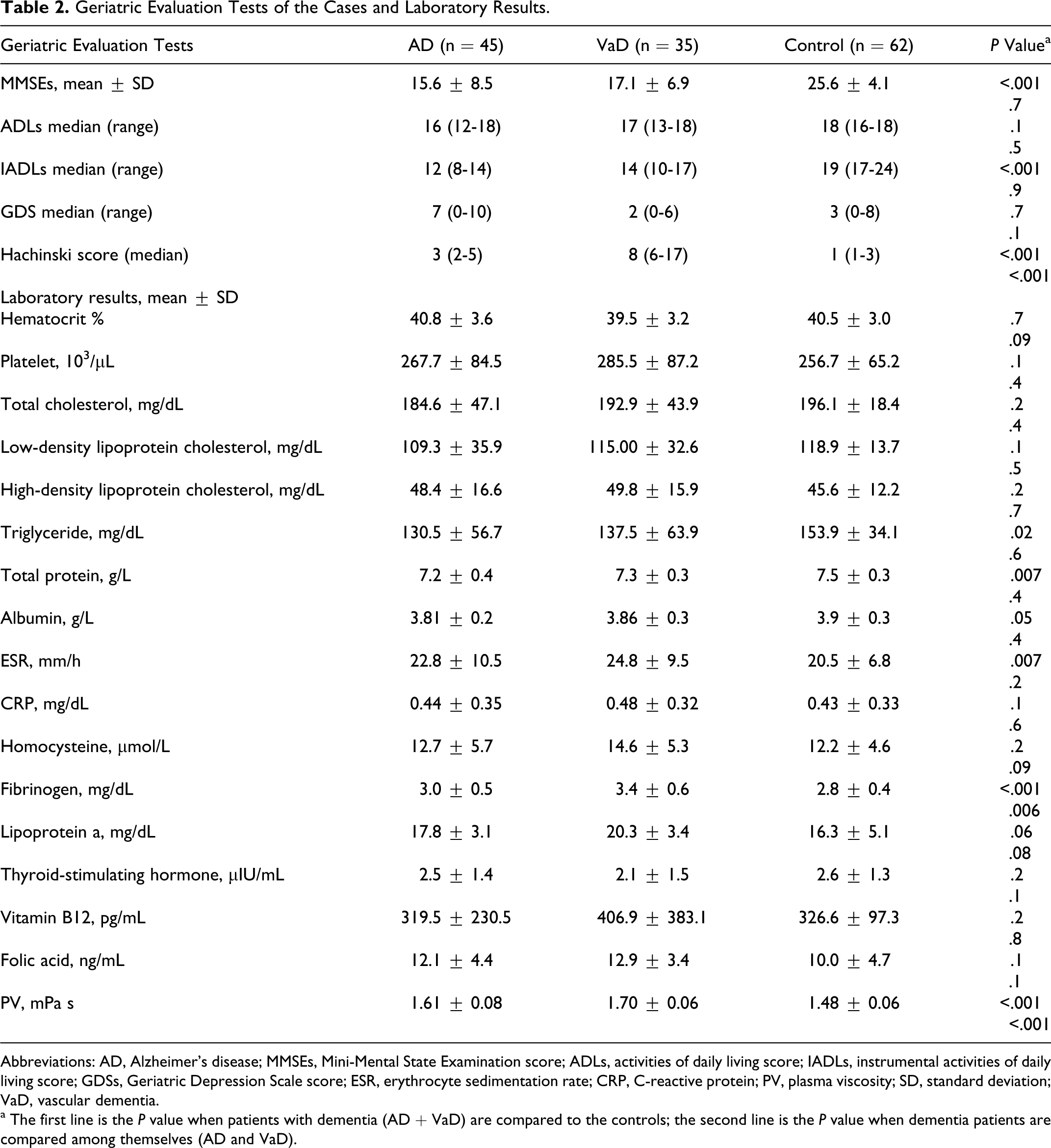
Abbreviations: AD, Alzheimer’s disease; MMSEs, Mini-Mental State Examination score; ADLs, activities of daily living score; IADLs, instrumental activities of daily living score; GDSs, Geriatric Depression Scale score; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; PV, plasma viscosity; SD, standard deviation; VaD, vascular dementia.
a The first line is the
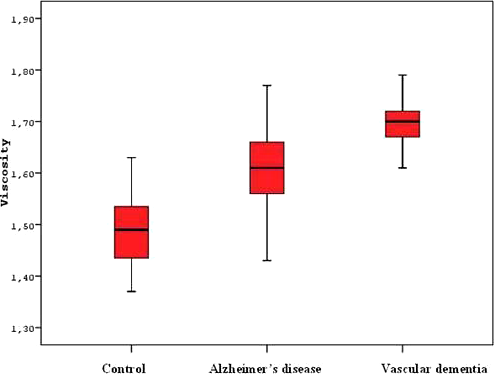
Comparison of PV in AD, VaD, and controls
The PV levels were in negative correlation with MMSE, ADLs, and IADLs scores statistically (
Those variables with significant
Univariate and Multivariate Analysis Results of Biochemical Parameters
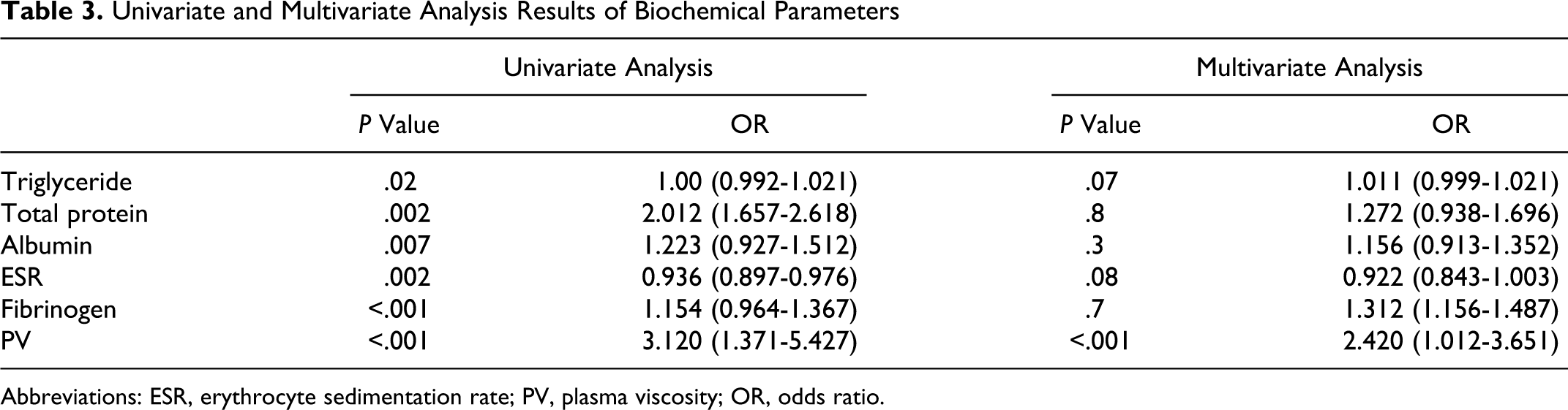
Abbreviations: ESR, erythrocyte sedimentation rate; PV, plasma viscosity; OR, odds ratio.
Based on the CDR scores, 56% of the patients were at early stage (n = 45), 25% were at intermediate stage (n = 20), and 19% (n = 15) were at advanced stage. Means of PV and its SDs were calculated based on CDR stages (1.65 ± 0.08, 1.64 ± 0.08, and 1.68±0.11, respectively). When
In ROC analysis, PV values higher than 1.54 have 91% sensitivity and 82% specificity for differentiating cases with dementia from nondementia, and the value of area under curve is 0.944 (Figure 2). The PV values higher than 1.66 have a 77% sensitivity and 76% specificity for differentiating AD from VaD, and the value of area under curve is 0.805 (
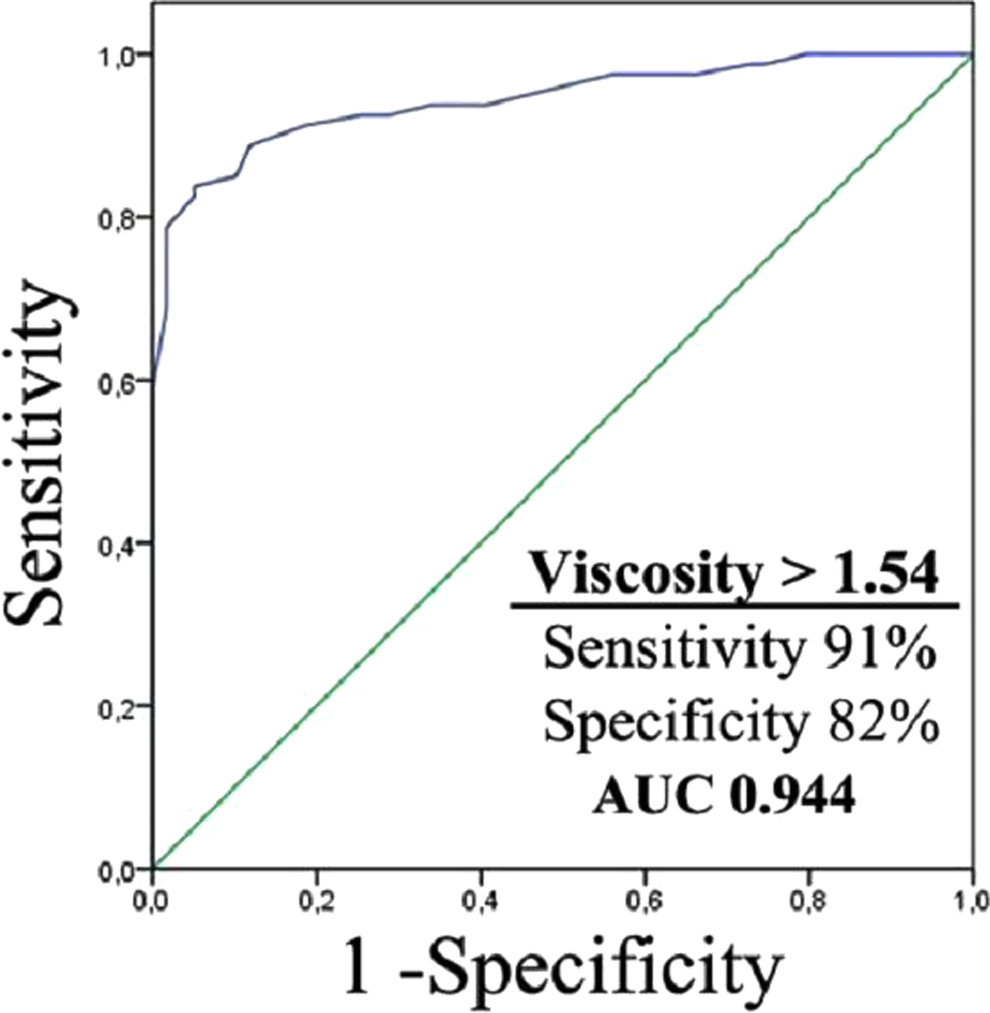
ROC curve of PV in patients with dementia
Discussion
In the elderly population, AD is the most common cause of dementia with a frequency of 50% to 70%. The VaD is responsible for 10% to 25% of all dementia cases. 3 We definitely need noninvasive, practical, and cheap biochemical markers for the diagnosis and differential diagnosis of dementia.
In this study, we tested the PV measurement for the patients with dementia and for controls without dementia. We identified that elevations of PV was an independent risk factor for dementia (Table 3). In patients with dementia, PV was higher than controls without dementia. Furthermore, PV of patients with VaD were higher than the patients with AD (Table 2, Figure 1). In our study, there was no correlation with the clinical severity of dementia and PV.
The sensitivity and specificity of the cutoff values found for differentiating cases with dementia from nondementia was higher than the values found for differentiating VaD from AD, but both the results were statistically significant. These results showed that measurement of PV can be useful in diagnosis and differential diagnosis of dementia in clinical practice.
Together with aging, we see a decrease in cognitive functions; AD, VaD, and other degenerative dementia are influenced by different ethiopatogenetic factors.
Inflammatory mechanisms can be responsible for cognitive impairment and dementia. 26 The CRP has been found in and around β amyloid plaques in the brains of the patients with dementia. 27 Systemic low-grade inflammation and increased serum concentrations of hs-CRP have been associated with impaired cognition and an increased risk of VaD and AD in some follow-up studies. 9,11,28,29
Inflammation-related decreases in cerebral vascular perfusion and neuronal degeneration that results from this seem to be a common pathophysiological process for AD and VaD. It is obvious that several factors are responsible for this pathophysiology. Changes seen in rheological system is one of these. Hemorheological disturbances result in several changes both in the vascular wall and in the blood that circulates within this structure. As a result, microcirculation decreases in these tissues. 30 Decreases in the microcirculation lead to hypoxia. Hypoxia influences a wide range of mitogenic and fibrogenic effects and modifies the expression of genes involved in angiogenesis and capillary permeability, vasomotor response, glycolysis, matrix metabolism, and cell survival. 31 Lowe identified that in determining the blood flow in cerebral microcirculation, PV had a more important role than hematocrit and related blood viscosity. 32
Marioni et al conducted a study with 5-year follow-up with 2312 participants at the age of 50 to 80. They investigated the effects of CRP, fibrinogen, hematocrit, and PV on cognitive functions. Baseline PV was negatively correlated with the follow-up test scores for general cognitive ability. 15 In our study, at the low scores of MMSE which showed cognitive function, PV was identified to be high. This negative correlation was statistically significant.
In a study by Schneider et al, PV was shown to increase significantly in subcortical arteriosclerotic encephalopathy (Binswangers disease) and this increase hampered the microcirculation leading to progressive degeneration of cerebral white matter. 33 Similar to the results of this study, our study had higher PV values in VaD group compared to AD and control groups (Table 2, Figure 1).
Based on Hachinski ischemic staging, having a score of 7 or above favors VaD. 25 In our study, patients with VaD had a median Hachinski staging score of 8. Having a significant correlation between PV and Hachinski ischemic score was in accordance with the significant correlation between PV and VaD.
In our study, there was no significant correlation between stages showing the clinical severity of dementia and PV. This result supported the fact that high levels of PV could play a role in the pathogeneses of AD and especially of VaD; yet after the appearance of dementia symptoms, it does not play an active role in the progression of the disease. In order to identify the importance of PV in the progression of dementia, we need studies with the participation of larger number of patients in a controlled manner with long-term follow-up. These studies will also shed light into understanding the role of treatments geared at decreasing PV in dementia.
Changes in PV are associated with the changes in serum proteins mainly seen as decreases in albumin or decreases and increases in globulins. 34 Individuals having plasma protein abnormalities that might influence PV were not included in the study. Despite this intervention, total protein and albumin levels were low in the dementia group. In multivariate analysis, PV was shown to be high in dementia independent of the total protein and albumin levels (Table 3).
Fibrinogen is the most important protein responsible for PV. 35 Oijen et al demonstrated the association of high fibrinogen levels with AD and VaD. 36 Chang et al conducted a study in 21 patients with AD and 23 control participants; the blood viscosity and fibrinogen were found to be higher in this disease group than the control group. 37 The PV is influenced by fibrinogen to a significant degree, as we know that several other pathologies could influence the fibrinogen levels, we excluded the patients having high fibrinogen measurements from our study. Despite the fact that fibrinogen was within the normal range in our study, it was higher in VaD group than AD and control groups. In multivariate analysis, PV was shown to be high in dementia and VaD independent of fibrinogen (Table 3).
The PV was not affected by the changes in blood cells (anemia and polycythemia), age, or gender like ESR. 38 Although PV is known as acute inflammatory marker, some studies showed that PV can be elevated in some chronic diseases like CRP 39 ,40 The elevation of PV in dementia and especially in VaD can be interpreted, like elevation of CRP in coronary heart disease.
The potential limitation of this study should also be addressed. Sectional studies like ours are inadequate in identifying causality relationships when compared to longitudinal studies. In order to understand whether elevated PV is the cause or the result of dementia, we need long-term prospective studies with higher patient participation. Furthermore, the demographical data of our study group were obtained from the patients themselves or their close ones. Hence, the possible error related to demographical data should also be taken into account.
We used accepted guidelines and appropriate laboratory tests for diagnosis of dementia, but the differential diagnosis of dementia is a hard job. The definite diagnosis of dementia can only be made by pathological evaluation of the brain tissue. However, data from the clinical studies showed that autopsy findings in AD cases can confirm clinical diagnosis in a ratio of 80% to 90%. 41,42
Conclusion
This study showed that, PV which can be measured in peripheral blood samples is a simple and cheap biomarker that can support the clinical and imaging findings of dementia. This is true both for diagnosis of dementia and for the differential diagnosis of AD and VaD which are the 2 most commonly observed forms of dementia. However, this finding should be tested and supported in larger case series including patients with other types of dementia in a prospective study design.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
