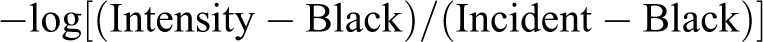Abstract
Primary progressive aphasia (PPA) is characterized by progressive loss of language, unassociated with generalized cognitive loss and with no uniform pathological abnormality. To address whether the issue of PPA is associated with synaptic loss, we compared immunoreactivity in Broca’s area with that in the adjacent gyrus from a case of progressive, nonfluent PPA using antibodies against synaptophysin. Using semiautomated methods, we assessed serial 0.10 mm2 diameter areas from the cortical surface to the white matter in both the case and the control. In PPA, synaptophysin immunoreactivity was significantly less in Broca’s area than in the adjacent area (optical densities of 0.16 and 0.24). The reduced synaptophysin immunoreactivity was significantly greater in the upper cortex than in the regions of cortex adjacent to the white matter (mean optical densities of 0.14 and 0.18). These data suggest that synaptic loss may be a factor underlying the language deficits in PPA.
Keywords
Introduction
Primary progressive aphasia (PPA) is characterized by a progressive loss of language function, unassociated with a more generalized loss of cognition. 1 Although once considered rare, there is now increased awareness and better methods to diagnose this condition. 2 –5 Postmortem studies demonstrate variable neuropathological diagnoses. Some cases, particularly those with progressive, nonfluent dementia, show features of Alzheimer’s disease (AD), including β-amyloid plaques and neurofibrillary tangles. 6 Other PPA cases have tau pathology only and meet pathologic criteria for Pick’s disease or other tauopathies including corticobasal degeneration or progressive supranuclear palsy. 6,7 Additionally, some PPA cases have only ubiquitinated inclusions or neuronal loss without distinctive histopathologic features. 8 Given the lack of specific pathological protein aggregates, strategies for protein-directed interventions for PPA are problematic.
Many central nervous system (CNS) degenerative diseases are characterized by synaptic loss and degeneration of the distal axon. 7,9 –11 Synaptic loss was initially studied in AD. Data suggest that synaptic proteins including synaptophysin and synapsin 1 are the major pathological hallmarks of AD. 7,9 –11 Synaptic failure impairs the ability of neurons to communicate, and it underlies the cognitive deficits these individuals experience. Synaptic loss in degenerative diseases is important because it is a better correlate of cognitive loss than neuritic degeneration, Aβ, or neuronal loss. 9,12
Many hypotheses regarding the mechanism by which the abnormal aggregates lead to disease implicate the synapse. The amyloid precursor protein (APP), tau, and α-synuclein are all neuronal proteins that normally occur in synapses. β-Amyloid oligopeptides also localize in synaptic regions. 13,14 Oligomeric Aβ targets synaptic spines 15,16 and may lead to reduced long-term potentiation and synaptic abnormalities. 13,14
Synapse loss occurs early in the disease course. Defects in perforant path input from both the entorhinal cortex and the dentate gyrus result in early memory losses that precede amyloid plaque buildup. 17 Synaptic loss is observed in frontotemporal dementia, of which PPA is a major subtype. However, to our knowledge, synaptic loss has not been assessed in the targeted regions in PPA. The following clinical case supports the notion that synaptic loss is present in progressive nonfluent dementia.
Methods
Clinical Case
The case was a 66-year-old male who noted dysfluent speech. Serial neuropsychological tests confirmed that he had a progressive nonfluent aphasia. Symptoms progressed over 3 years until he was completely unable to say even single words. He died 4 years after symptom onset. Tau-positive aggregates were present in neurons. No Pick bodies were observed. No other pathology was present to suggest other disorders; β-amyloid and α-synuclein immunoreactivity were largely absent except for rare diffuse amyloid plaques. The comparison case was a cognitively normal control case who died at the age of 68 years, without relevant clinical or pathological diagnoses. Both cases had a postmortem delay of less than 8 hours.
Histological Methods
A formalin-fixed, paraffin-embedded tissue block was sampled at 2 rostrocaudal levels to include Broca’s area and the adjacent gyrus. The tissue was stained for synaptophysin immunohistochemistry (SNP88; BioGenex; Neat) using previously published methods involving microwave enhancement. 10
Optical Density Measurements
Optical density data were converted from intensity collected for all slides with a black and white camera (Photometric Sensys) mounted on a light microscope (Olympus BX60) and sent to a computer equipped with Image-Pro Plus (version 3.0; Media Cybernetics, Bethesda, MD). Serial measurements were collected from the cortical surface inward to the white matter along premeasured, equidistant points at ×10 magnification. Sample areas were 0.10 mm2. All intensity measurements were converted into optical density using the formula:
with black being the intensity measured with no light passing through the microscope and incident being the intensity measured on the slide where there was no tissue. Statistical analyses were performed by 2-tailed t tests.
Results
The PPA case with antisynaptophysin immunostaining showed uniform immunostaining in most regions of the cortex, including the gyrus adjacent to Broca’s area. In contrast, there was a clear-cut reduction in immunostaining in Broca’s area. Also, tissue staining was lighter in the lamina near the cortical surface, suggesting that synaptic losses are greater in the upper cortical layers.
Overall synaptophysin immunoreactivity was less in Broca’s area than in the adjacent brain region with a mean optical density of 0.16 (standard deviation [SD] 0.015) and 0.24 (SD 0.016), respectively (P < .001). When measuring optical densities from the cortical surface toward the white matter in Broca’s area, much of the loss of synaptophysin immunoreactivity was in the upper third of the cortical layers relative to the lower third regions of cortex (mean optical densities of 0.14 [SD 0.021] and 0.18 [SD 0.019], respectively; P < .001).
Graphs demonstrate optical density measurements in Broca’s area compared with the adjacent gyrus. Figure 1 shows the mean optical density values at 2 sampled levels in Broca’s area and the adjacent gyrus. Note that the optical densities are consistently lower, indicating more light transmission and fewer synapses, in Broca’s area relative to the comparison area.
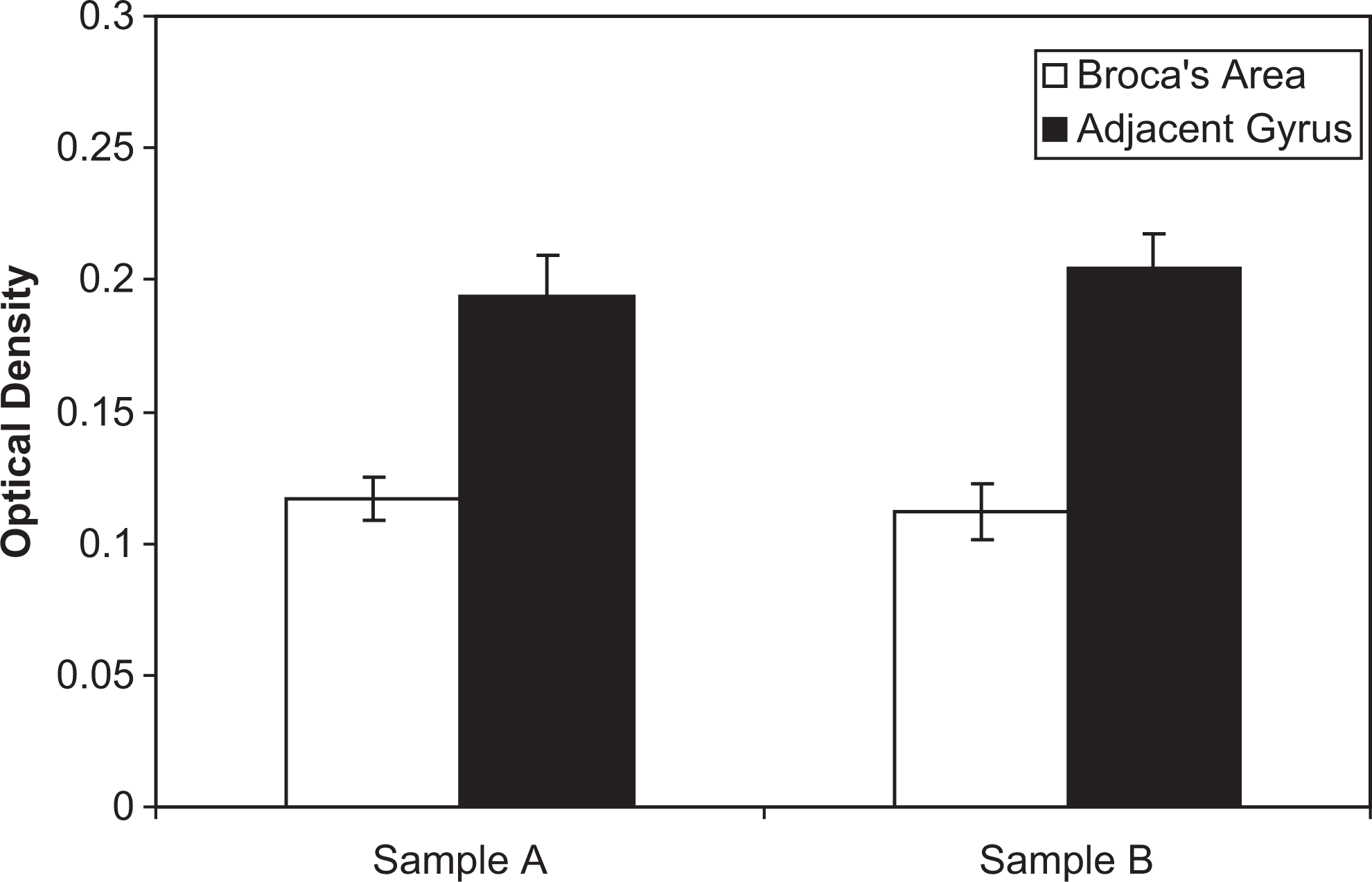
Mean optical density values at the 2 sampled levels in Broca's area and the adjacent gyrus. The optical densities are consistently lower in Broca's area relative to the comparison area, indicating more light transmission and fewer synapses.
Figure 2 shows the difference in optical density in the more superficial layers of Broca’s area (left bars) compared with deeper cortical layers. In Broca’s area from a comparable control case, the immunostaining is more uniform.
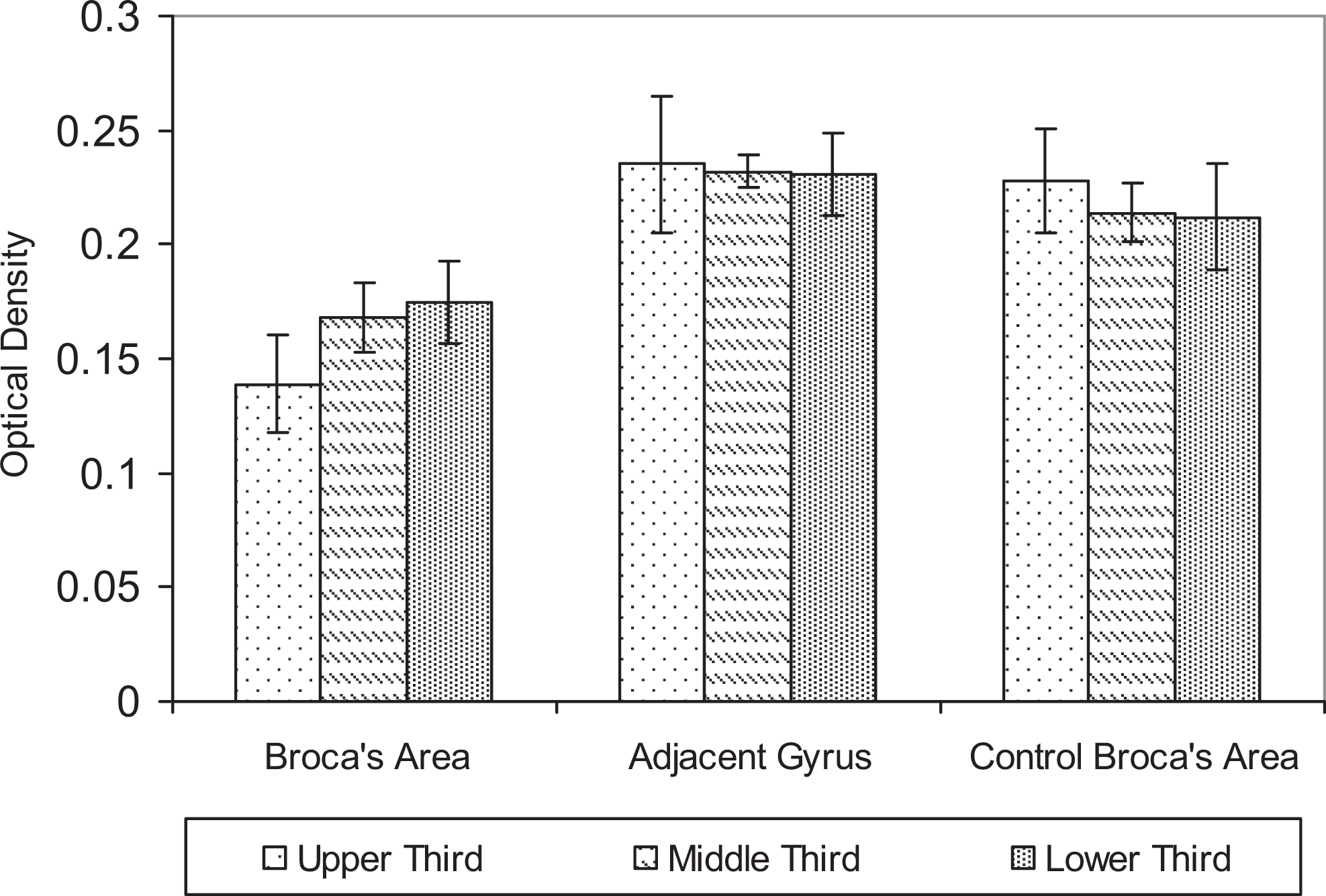
Mean optical density in the more superficial layers (upper third) compared with deeper cortical layers (lower third). Control areas have more uniform staining than the Broca's area of the case.
Discussion
We demonstrate a relative loss of synaptophysin immunoreactivity in Broca’s area in a case with progressive, nonfluent aphasia relative to immunoreactivity in an adjacent gyrus. This finding was not seen in a cognitively normal age-matched control. Similar findings were observed using another synaptic transmitter-specific protein, synapsin 1 (Research Diagnostics, Inc North Las Vegas, NV). This loss was more prominent in the superficial cortical layers. These findings suggest that the loss of immunoreactivity reflects a reduction in synapses rather than just a reduction in 1 synaptic protein. Thus, the language deficit this patient experienced is related, at least in part, to a loss of synaptic input to Broca’s area. It raises the possibility that symptoms are influenced by loss of input to Broca’s area rather than exclusively a loss of neurons within this region.
A review of cortical connections is relevant to our premise that deafferentation to Broca’s area is a part of this participant’s symptoms. The deeper cortical layers (V and VI) provide major output from Broca’s area to the thalamus (VI) and other brain regions (V). The more superficial cortical layers receive input from other brain regions, with cortical layers III and IV receiving the major input from the thalamus (most noncortical input to cortical regions is channeled through the thalamus). This implies that some of the patient’s problems may relate to loss of axonal tracts to Broca’s area rather than loss of cell bodies within (or long axons exiting) this region.
Much of the current drug development for the degenerative dementias focuses on protein processing since CNS diseases are the result of abnormalities in CNS proteins. One difficulty with these treatments for PPA is that the underlying substrate involves several pathological protein aggregates. Thus, treatment strategies cannot target 1 abnormal protein. Furthermore, there is no reliable biomarker to identify the protein abnormality present in an individual with PPA.
Current therapies provide relief from symptoms in FTD/PPA by increasing neurotransmitter levels, modulating neurotransmitter systems, or providing behavioral strategies. 18 Most help is with behavioral symptoms that may co-occur; there is little to offer patients for the language losses in PPA. It would be premature to suggest that strategies that prevent synaptic loss would help those with PPA. However, most other degenerative diseases show synaptic loss and dysfunction before protein aggregation occurs, 19,20 so there is interest in developing agents that can slow or prevent these losses. It is possible that interventions with growth factors or cytokines such as tumor necrosis factor-α will play a role in maintaining synaptic connectivity.
One weakness of the current study is that it is a single case report. Cases of PPA coming for autopsy are rare, and examination of speech-specific areas is not performed commonly. Further studies with PPA cases with different language deficits are needed to define the role of synaptic loss in these disorders.
Footnotes
Authors’ Notes
Appropriate consent was obtained from the participants for tissue collection and research purposes.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by the Julius Newman Neurodegenerative Disease Research Fund.