Abstract
This study provides empirical evidence on whether polypharmacy and potentially inappropriate prescription medications (PIRx, as defined by the 2003 Beers criteria) increase the likelihood of functional decline among community-dwelling older adults with dementia. Data were from the National Alzheimer’s Coordinating Center, Uniform Data Set (9/2005-9/2009). Study sample included 1994 community-dwelling participants aged ≥65 with dementia at baseline. Results showed that participants having ≥5 medications were more likely to have functional decline than participants having <5 medications. However, the increased likelihood was only apparent in participants who did not have PIRx. Instead of magnifying the associated risk as hypothesized, PIRx appeared to have a protective effect albeit marginally statistically significant. Therefore, increased medication burden may be associated with functional decline in community-dwelling older adults with dementia who are not prescribed with PIRx. More research is needed to understand which classes of medications have the most deleterious effect on this population.
Introduction
Ensuring appropriate medication use continues to be a major focus in health services research particularly among older adult populations. 1 Optimal prescribing in community-dwelling older adults with dementia may be even more important as memory loss, executive dysfunction, and underlying functional impairment can lead to problems following medication instructions, greater susceptibility to side effects, and difficulties in identifying and reporting adverse drug events. 2 Prior epidemiological studies on medication utilization in this population have examined the use of presumptive inappropriate medications, such as the Beers medications 3,4; coadministrations of cholinesterase inhibitors and anticholinergics 5 ; underuse of medications to treat comorbid conditions 6 ; and potential drug–drug and drug–disease interactions. 7 However, few studies have empirically examined the adverse health outcomes associated with such prescribing patterns in persons with dementia. 8,9
A high number of prescription medications (or polypharmacy) represents a significant risk factor in cognitively intact older adults for adverse health events, including hypoglycemia, fractures, hospitalization, and death. 10 Furthermore, potentially inappropriate prescription medications (PIRx) have been linked with poorer health outcomes including adverse drug reactions, hospitalization, and falls. 11,12 Perhaps the most cited explicit definition of PIRx, the Beers list has been developed by a national expert consensus panel and contains medications that generally are considered inappropriate for all older adults regardless of comorbidities. 13 Beers-defined PIRx use in cognitively intact older adults has been associated with the onset of cognitive decline, as well as psychological conditions including depression and malaise. 14
Community-dwelling older adults with dementia may be at a higher risk for polypharmacy than those without dementia, and high number of prescription medications is significantly associated with PIRx. 3 Given the potential adverse health events associated with polypharmacy and PIRx, 12,15 we aim to investigate the relationship between polypharmacy and Beers-defined PIRx with functional decline—a key, independent health-related risk factor for nursing home placement—among community-dwelling older adults with dementia.
Specifically, this study examines (1) whether polypharmacy is associated with functional decline among community-dwelling older adults; and (2) how this association may be modified by PIRx. We hypothesize that polypharmacy would increase the odds of functional decline, and PIRx would magnify this association. Findings of this study will have significant policy relevance because polypharmacy and adapted versions of the Beers list are currently employed as indicators of prescription quality by various national organizations, including the National Committee on Quality Assurance and the Centers for Medicaid and Medicare Services. Understanding the potential risk of functional decline related to medication use will further inform policy makers and clinicians in their evaluation of a high number of prescription medications and employment of the Beers list as a presumptive measure of prescribing quality in community-dwelling older adults with dementia.
Methods
Data and Study Sample
Institutional Review Board approval for this study was obtained from the lead investigator’s (D.T.L.) institution. This retrospective cohort study analyzed data from the National Alzheimer’s Coordinating Center (NACC), Uniform Data Set (UDS). 16 The UDS contains demographic and clinical data from participants with and without dementia enrolled in 1 of 29 Alzheimer’s Disease Centers (ADCs) across the United States funded by the National Institute on Aging (NIA). Since 2005, research-trained clinicians and staff followed standard protocols to collect data from respondents (patients and accompanying key informants) during their initial and follow-up ADC visits. All respondents (cognitively healthy controls and patients with dementia or mild cognitive impairment) are required to have an accompanying key informant (a family member or friend) to provide information about the participant, including medications taken.
At each ADC visit, data collected from respondents included, but were not limited to, patients’ age, sex, race/ethnicity, education, marital status, and living arrangement. The names of all prescription medications used by the patients within the last 2 weeks of the ADC visit were self-reported by the respondents. Based on in-person clinical assessments, trained ADC clinicians and staff systematically evaluated each patient’s dementia status (presence and severity of dementia according to the Clinical Dementia Rating [CDR] scale 17 ); functional status (a single item about the general level of dependence with activities of daily living); and a predetermined list of comorbid conditions. (See “Measures” section for further details.)
This study examined participants’ first and second ADC visits captured in the UDS between September 2005 and September 2009, restricting the analysis to participants aged 65 and older who were community dwelling (ie, living in an independent family residence or retirement community). To identify participants with dementia at baseline, we included persons evaluated by an ADC clinician as meeting “the standard clinical criteria for dementia” 16 and having CDR global scores ranging from 0.5 to 3.0 (very-mild-to-severe dementia). Participants without any prescription medication data were not included in the analysis because they represented an undistinguishable, heterogeneous group—those with missing data (don’t know or refused to answer) or those not taking any prescription medications—therefore, making interpretation difficult. At the final stage, we excluded a small portion of persons with missing values in any of their sociodemographic variables. The remaining 1994 persons with baseline dementia were included in our study (Table 1).
Sample Size According to Inclusion Criteria (Stepwise)
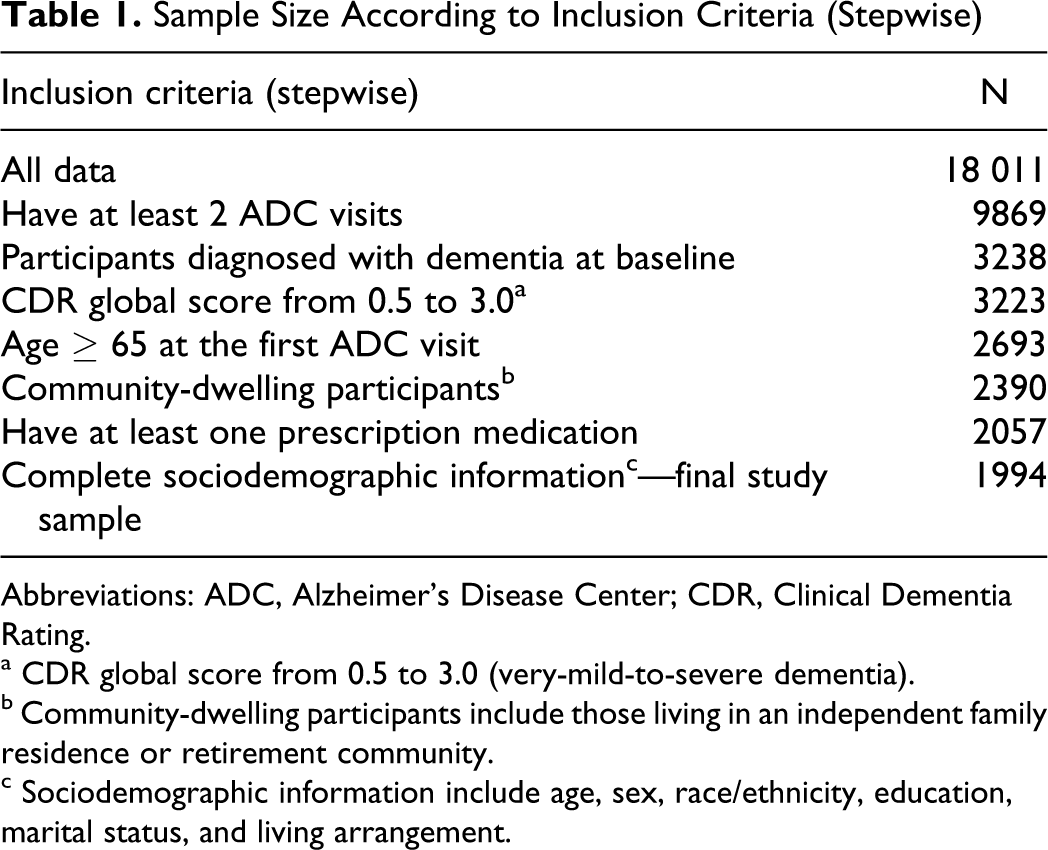
Abbreviations: ADC, Alzheimer’s Disease Center; CDR, Clinical Dementia Rating.
a CDR global score from 0.5 to 3.0 (very-mild-to-severe dementia).
b Community-dwelling participants include those living in an independent family residence or retirement community.
c Sociodemographic information include age, sex, race/ethnicity, education, marital status, and living arrangement.
Participants without baseline dementia
For comparison purposes, we also assessed whether the associations between polypharmacy and PIRx with functional status varied by the presence or absence of baseline dementia. Among 1988 persons who were clinically evaluated as having “normal cognition” and had a CDR global score of 0 (no dementia), our analysis found that 98.5% were able to live independently and the majority (97.4%) had the same functional status at both ADC visits. Due to the lack of variation in functional status between ADC visits, sufficient information for participants with normal cognition was not available to reliably estimate regression parameters. We therefore did not report these findings; however, data are available upon request.
Measures
Dependent variables: functional status
To indicate functional status at the second visit, we compared functional abilities between the 2 visits using the following response categories, regardless of living arrangement, (1) was able to live independently; (2) required assistance with complex activities of daily living such as paying bills, shopping, driving, and cooking; (3) required assistance with basic activities of daily living such as eating, dressing, and bathing; or (4) was completely dependent on others. 18 Ratings were based on the impact of cognitive impairment rather than physical impairment on functional skills. Functional decline was defined as any increase in functional dependence between the visits.
Independent variable: polypharmacy
The total number of prescription medications, excluding vitamins, herbal supplements, and topical agents, was counted per participant. Polypharmacy was defined with a dichotomous variable (yes/no) as having 5 or more prescription medications at the first ADC visit because prior research found that 5 or more medications substantially increased the risk of serious adverse drug events, including hospitalization and death. 19
To assess the robustness of this definition of polypharmacy, we constructed an additional version of polypharmacy with a cutoff relative to the percentage distribution of total prescription medications in the study sample. We found that at the 85th percentile, the total number of prescription medications was 7; therefore, we also defined polypharmacy as having 7 or more medications at the first ADC visit.
Independent variable: PIRx
The 2003 Beers list 13 attempts to identify medications that are inappropriate because their potential health risks outweigh therapeutic benefits or because safer alternatives are available. Beers medications have 3 categories of PIRx for older adults: (1) medications that generally should be avoided in all patients; (2) medications at a specified dose that should not be exceeded; and (3) medications that should be avoided in patients with specified comorbid conditions. 13 Because of the data constraints of the UDS (eg, incomplete information about drug dosage and duration, and participants’ comorbid conditions), our analysis was limited to 44 agents/drug classes of the Beers medications that generally should be avoided in all older patients, irrespective of their health conditions or indications. A person-level dichotomous variable (yes/no) was constructed to indicate whether the participants had any PIRx by matching the Multum drug codes (http://www.multum.com) of each medication prescribed at the first ADC visit with the Multum codes corresponding to each of the Beers medications.
Other characteristics
Other baseline participant characteristics are listed in Table 2. Age was calculated using date of birth and date of the first ADC visit. Race/ethnicity was combined into 1 variable based on separate questions regarding race and Hispanic/Latino ethnicity. The number of comoribid conditions per participant was based on affirmative responses to predetermined questions in the UDS about active coexisting diagnoses and recent procedures (range: 0-7 comorbid conditions): hypertension, hypercholesterolemia, thyroid disease, diabetes, incontinence (urinary or bowel), cardiovascular disease (angioplasty, cardiac bypass procedure, congestive heart failure, or other cardiovascular diseases), and cerebrovascular disease (stroke, transient ischemic attack, or other cerebrovascular diseases). Additionally, based on the month and year of ADC visits, the time lapse in years between the first and second visits was calculated.
Statistical Analysis
Baseline characteristics and functional status were tabulated. Categorical and continuous variables were summarized as percentages and mean ± 1 standard deviation (SD), respectively. To quantify the independent association between PIRx and polypharmacy with subsequent functional status at follow-up visit, an ordinal logistic regression model was developed (model 1). Potential confounders that were controlled for in the model included the patient’s age, gender, race/ethnicity, number of comorbid conditions, CDR global score, time between visits, and baseline functional status. Then, to determine whether PIRx modified the relationship between polypharmacy and subsequent functional status, we included an interaction term between polypharmacy and PIRx (ie, having fewer than 5 medications with and without PIRx; and having 5 or more medications with and without PIRx; model 2). In both models, robust or sandwich standard errors were calculated to account for the within ADC institutional correlations. Estimated coefficients and 95% confidence intervals were exponentiated to reflect the odds ratio of having a higher functional status score among those participants receiving PIRx (and 5 or more medications) versus those who did not. Proportional odds assumptions were graphically assessed. 20 Furthermore, to assess the robustness of our key findings, we performed sensitivity analysis using a different cutoff to define polypharmacy (having 7 or more medications) and re-estimated both models. All analyses were performed using R-version 2.10.0. 21
Baseline Characteristics of Study Sample (n = 1994) a
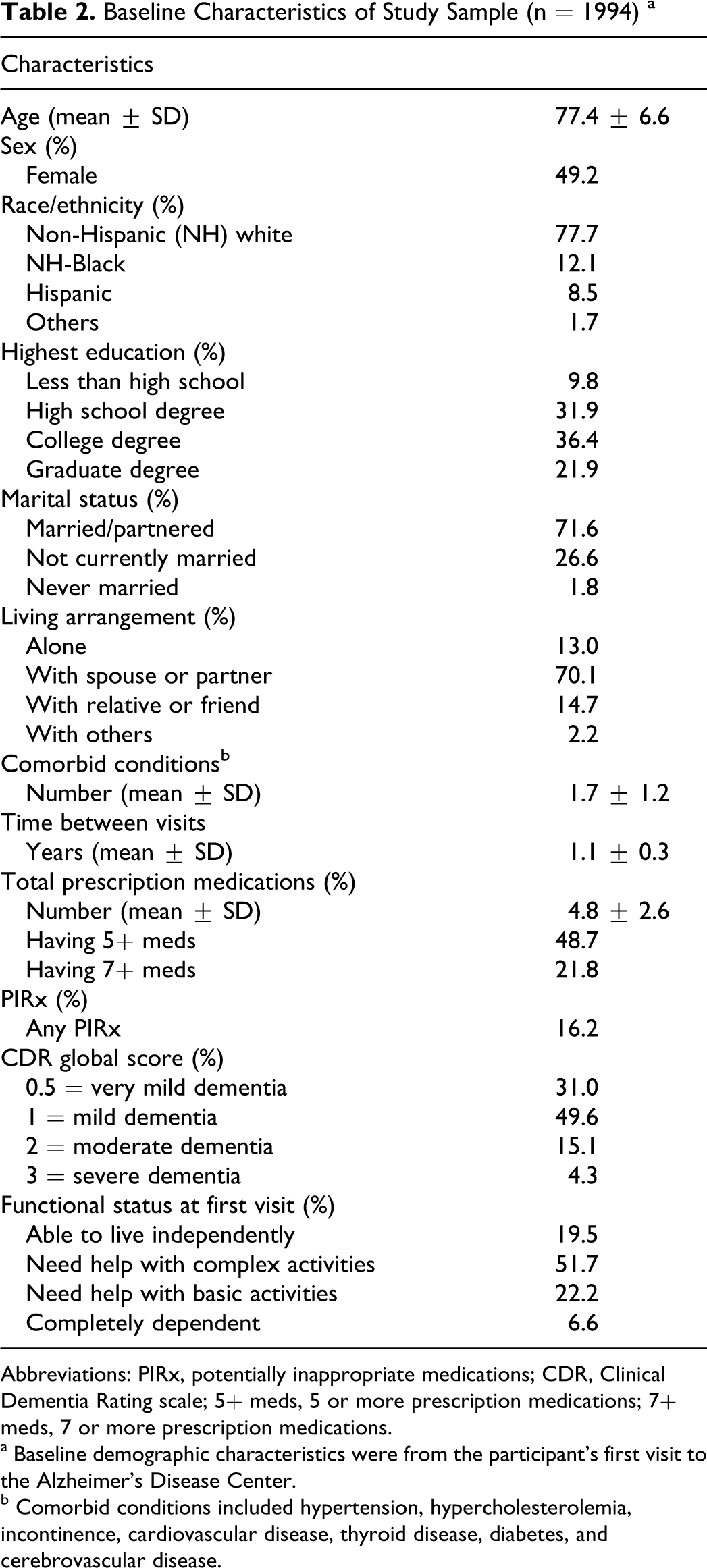
Abbreviations: PIRx, potentially inappropriate medications; CDR, Clinical Dementia Rating scale; 5+ meds, 5 or more prescription medications; 7+ meds, 7 or more prescription medications.
a Baseline demographic characteristics were from the participant’s first visit to the Alzheimer’s Disease Center.
b Comorbid conditions included hypertension, hypercholesterolemia, incontinence, cardiovascular disease, thyroid disease, diabetes, and cerebrovascular disease.
Results
Table 2 shows baseline characteristics of the study sample. The average age was about 77 years. Most participants were male, non-Hispanic White, married, living with at least 1 other person, and had at least a college degree. At the first ADC visit, the average number of prescription medications in the study sample was 4.8 (SD = 2.6). About 49% had 5+ prescription medications and 22% had 7+ medications in the first ADC visits. Comparable percentages were observed in the second visits: 48% had 5+ medications and 22% had 7+ medications (available upon request). Similar proportion of participants had Beers-defined PIRx in both visits: 16% in the first and 18% in the second ADC visit. The 5 most commonly reported PIRx used were conjugated estrogens, nifedipine, oxybutynin, fluoxetine, and doxazosin (data available upon request). These most common PIRx remained unchanged between the first and second visits.
Most participants had very mild or mild dementia and needed assistance with complex or basic activities of daily living. Overall, about 59% (1173 of 1994) had the same functional status at both ADC visits. By functional status, 43% of those who were able to live independently at the first ADC visit were also able to live independently at the second visit; 61% of those who required assistance with complex activities of daily living at the first visit had the same functional status at the second visit; 62% of those who required assistance with basic activities of daily living at the first visit had the same functional status at the second visit; and 77% of those who were completely dependent on others at the first visit were also completely dependent on others at the second visit (Table 3). Similar patterns in functional decline between ADC visits were observed among participants with PIRx and those with polypharmacy (5+ or 7+ prescription medications)—data available upon request.
Functional Status of Study Sample at Visits 1 and 2 to the Alzheimer’s Disease Centers
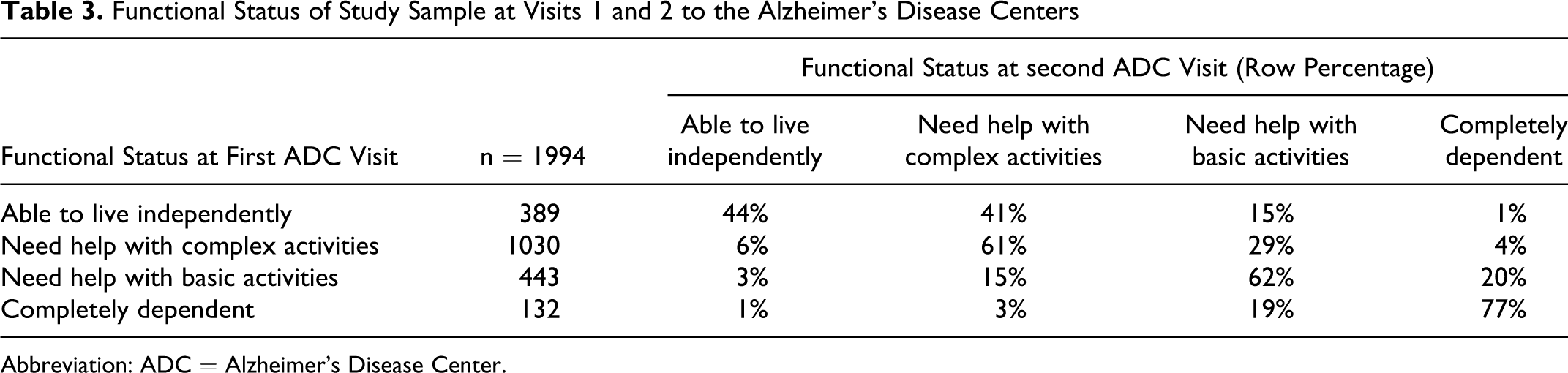
Abbreviation: ADC = Alzheimer’s Disease Center.
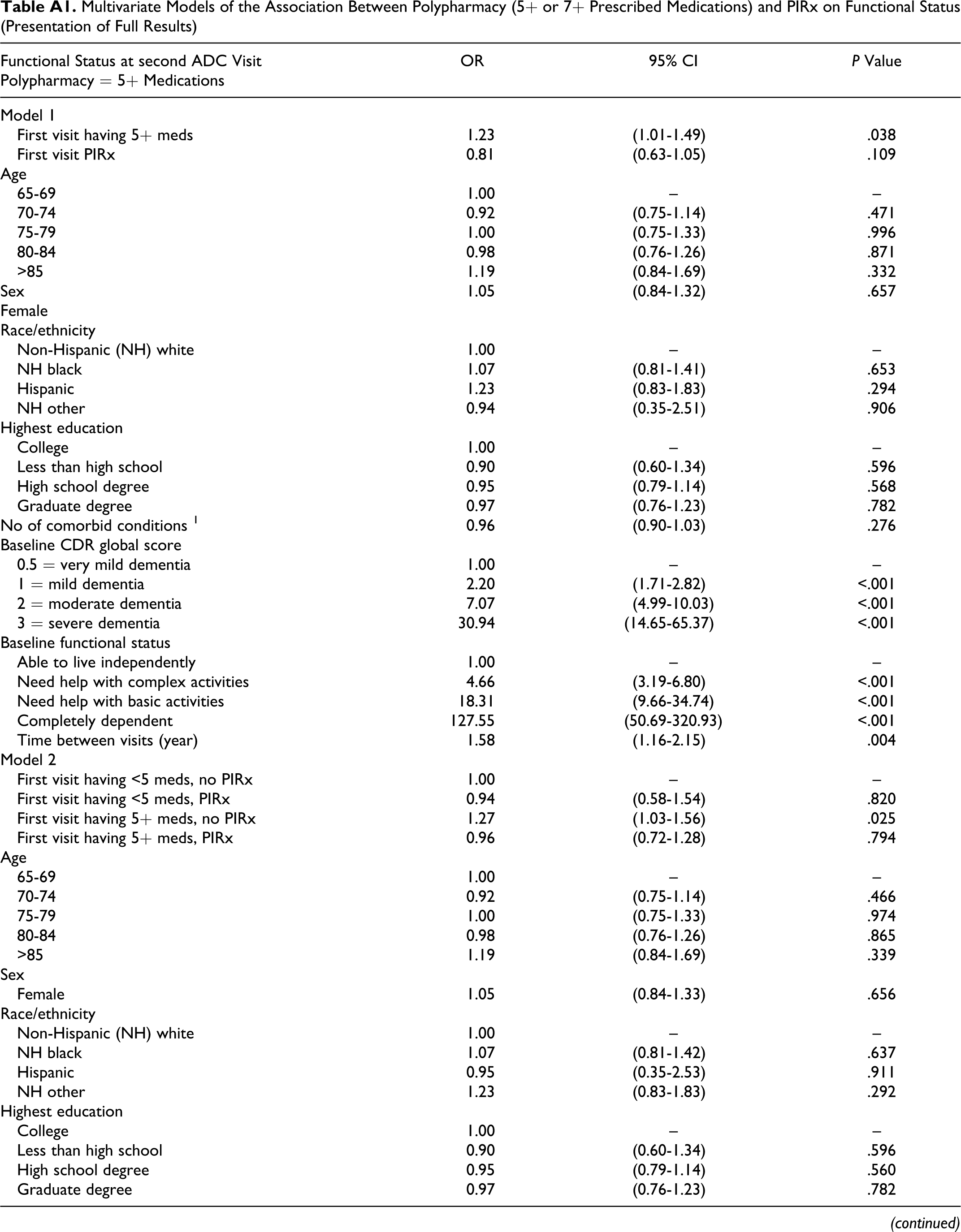
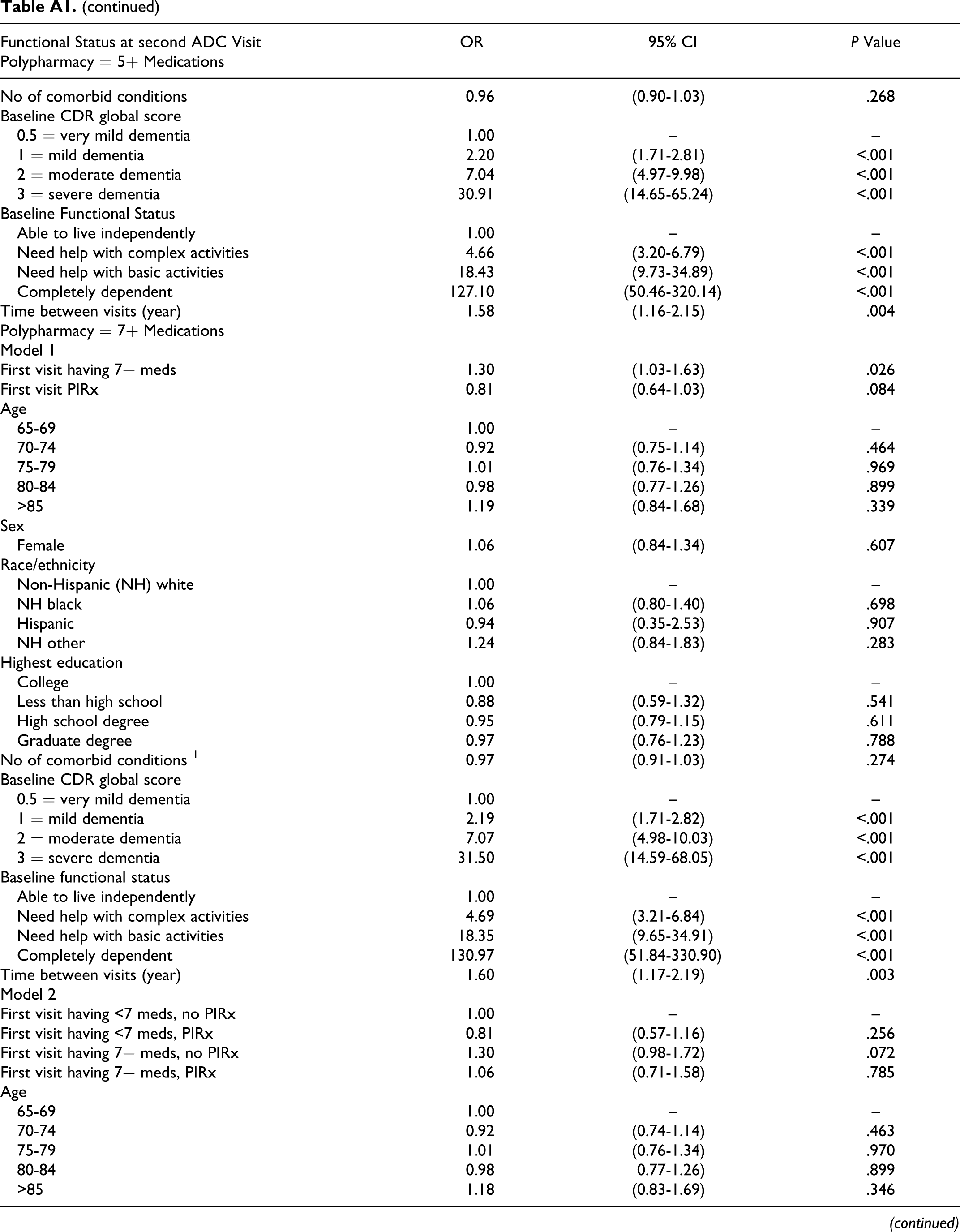
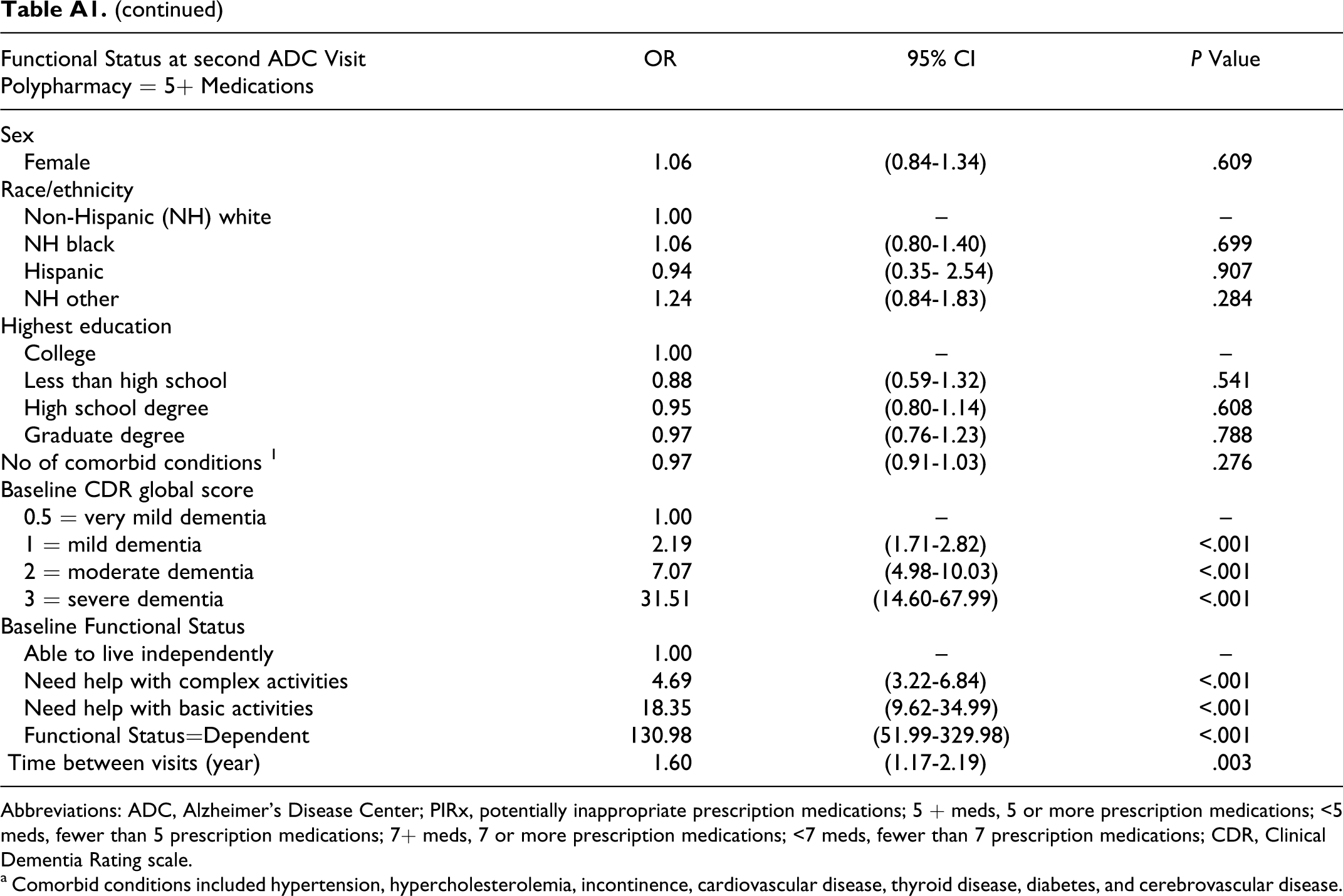
Table 4 displays the key results of the multivariate regression analysis by polypharmacy definition (5+ or 7+ prescribed medications). Model 1 shows that participants having 5 or more medications at the first ADC visit had higher odds (odds ratio [OR] = 1.23, 95% confidence interval [CI] = 1.01-1.49) of having a lower functional status at the second visit than participants having fewer than 5 medications, controlling for PIRx, baseline functional status, and other factors. According to model 2, among participants with no baseline PIRx, those with 5 or more medications had higher odds (OR = 1.27, 95% CI = 1.03-1.56) of having lower functional status in the next visit than those with fewer than 5 medications. However, compared to those participants having fewer than 5 medications that were not PIRx, participants with PIRx regardless of polypharmacy had statistically similar odds of having lower subsequent functional status (having <5 medications: OR = 0.94, 95% CI = 0.58-1.54; having 5+ medications: OR = 0.96, 95% CI = 0.72-1.28). Similar patterns of association were observed when polypharmacy was redefined as having 7 or more medications. Appendix A contains the results of all the other variables included in the regression models.
Multivariate Models of the Association Between Polypharmacy (5+ or 7+ Prescribed Medications) and PIRx on Functional Status (Presentation of Key Results) a
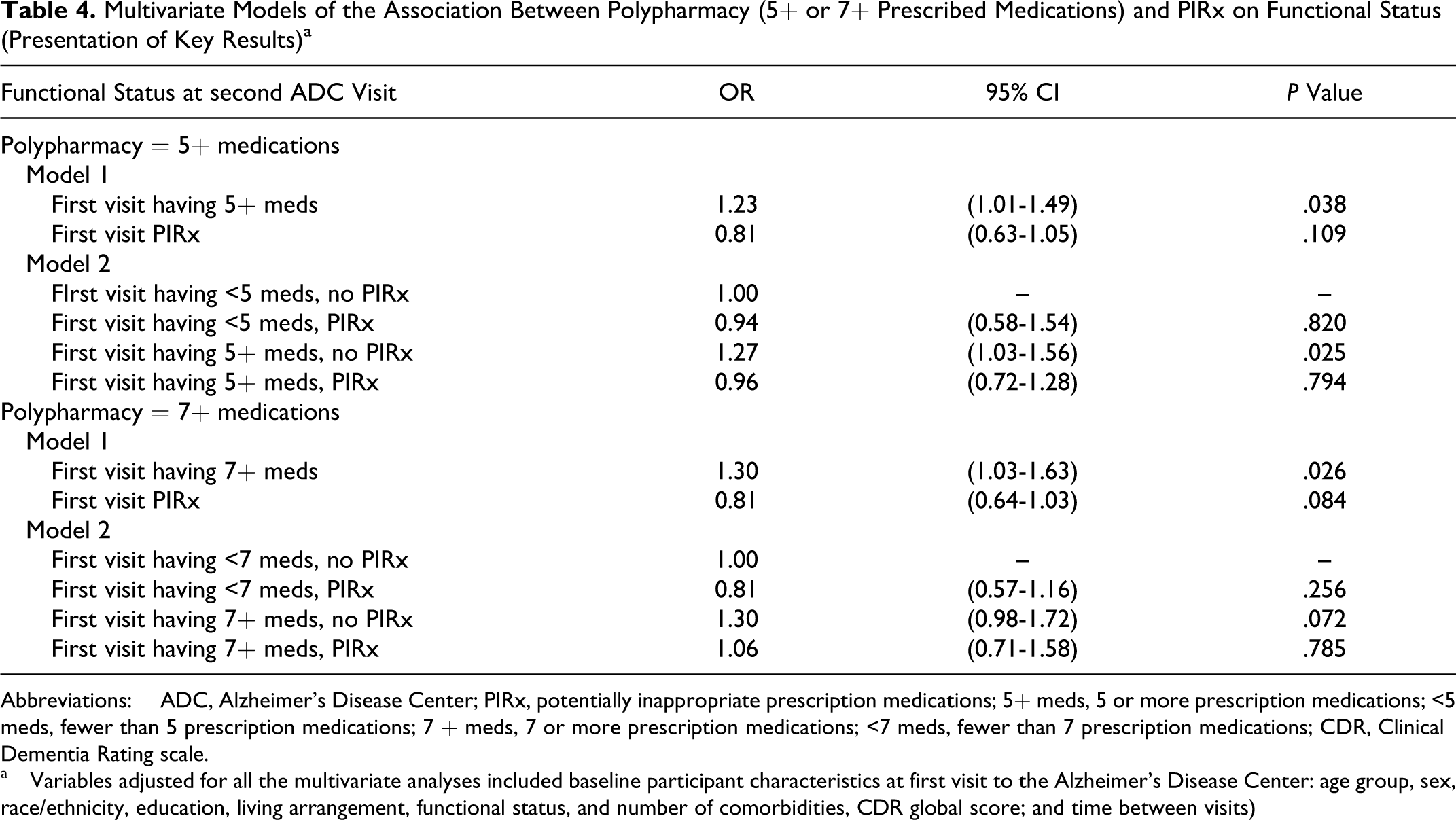
Abbreviations: ADC, Alzheimer’s Disease Center; PIRx, potentially inappropriate prescription medications; 5+ meds, 5 or more prescription medications; <5 meds, fewer than 5 prescription medications; 7 + meds, 7 or more prescription medications; <7 meds, fewer than 7 prescription medications; CDR, Clinical Dementia Rating scale.
a Variables adjusted for all the multivariate analyses included baseline participant characteristics at first visit to the Alzheimer’s Disease Center: age group, sex, race/ethnicity, education, living arrangement, functional status, and number of comorbidities, CDR global score; and time between visits)
Discussion
Necessary and proper use of medications are important health concerns among older adults, especially those with dementia, because of their increased potential for medication error, sensitivity to drug effects, and significant risk for nursing home placement. 22 Previously, we found that community-dwelling older adults with dementia generally had more prescription medications than those without dementia. 3 Employing the 2003 Beers list 13 as a presumptive measure of inappropriate medication use in older adults, we also found that polypharmacy was associated with elevated risk of PIRx, regardless of presence or absence of dementia. In this study, we used the UDS data to analyze the first and second ADC visits of community-dwelling older adults and empirically examined whether polypharmacy and PIRx in the initial visit were associated with functional decline—a significant risk factor for nursing home placement. Our hypothesis was partially supported.
Consistent with our hypothesis, we find that participants with baseline dementia having 5 or more medications are more likely to have lower functional status in the subsequent visit than those participants having fewer than 5 medications. However, the association with lower subsequent functional status exists only among participants with 5 or more medications that do not include PIRx. Participants with PIRx, regardless of having fewer or greater than 5 medications, have statistically similar odds of having lower functional status in the subsequent visit. The magnitude of these associations is similar when polypharmacy is redefined as having 7 or more medications.
In accordance to the growing body of literature, 23 our study suggests that the mere count of total prescription medications, in and of itself, may not necessarily indicate poor prescribing quality or predict worse functional outcome in older adults with dementia because participants in this study with high number of medications are not more likely to have lower functional status if they have PIRx. Clinical efforts to reduce the prevalence of polypharmacy in this population therefore should entail a judicious process of weighing the risks against benefits of each medication prescribed according to the older patient’s goal of care.
Furthermore, PIRx does not magnify the associated risk between having 5 or more (or 7 or more) medications and subsequent functional status. These findings are counterintuitive because many Beers medications—including anxiolytics, antipsychotics, antidepressants, analgesics, antihistamines, stimulants, sedatives, and hypnotics—have known adverse psychotropic effects. 24 With regard to increasing the risk for functional decline, our study raises potential questions about using the Beers list as a presumptive measure of medication inappropriateness in community-dwelling older adults with dementia. Future research should examine the impact of Beers medications on other health indicators, as well as investigate other criteria 25 or more specific sets of medications-to-avoid in this population. For example, estrogens have been found to increase the risk of the development and progression of dementia. 26 Other anticholinergic and sedative medications currently not included in the Beers criteria should be examined in this population due to their documented association with cognitive and function decline. 27
This study has additional limitations. First, despite using the first and second ADC visits from UDS, observed relationships between polypharmacy and PIRx with dementia progression and functional decline cannot be assumed as causal. Without follow-up medication data in the analysis, it is possible that changes in polypharmacy or PIRx may be significant risk factors of functional decline. Even though the average time between ADC visits is over 1 year, our findings indicate that the 5 most commonly reportedly PIRx remain the same between the first and second ADC visits. Furthermore, the proportion of participants with polypharmacy and PIRx were similar between the first and second ADC visits. These findings suggest that discontinuation of medications or changes in PIRx between ADC visits may account for a small proportion of the study sample. Although we controlled for a number of potential confounders including sociodemographic characteristics and health measures, our findings may reflect some unobserved factors. For example, this study examines medication prescriptions but not participants’ adherence to medications that may differ between participants with and without dementia between ADC visits. In addition, despite controlling for a number of health measures, people who receive more medications may have greater severity in their health conditions that could adversely affect their likelihood of functional impairment (rather than the medications they were taking). Second, although the UDS is one of the most comprehensive data sets currently available for studying patients with dementia across the United States, the UDS does not contain data from a random sample of patients; therefore, these findings cannot be interpreted as nationally representative. Recruitment procedures into ADCs may bias toward more-educated and higher-income older adults who received tertiary care from NIA-funded centers. More years of education has been associated with a lower likelihood of developing dementia. 28,29 Third, data on medication use are reported by patients and key informants; consequently, there is potential bias toward underestimating medication utilization. Finally, over-the-counter medications were not accounted for in this study due to data quality but might be common among participants with fewer prescription medications. For example, older adults with dementia may prefer taking over-the-counter medications, such as diphenhydramine for its sedative effects to prevent restlessness particularly at night. Our estimates of PIRx would be conservative by not accounting for Beers medications that are available over-the-counter. Similarly, because the data set does not contain dosage information, our estimates of PIRx did not include Beers-defined medications with dosage specifications (eg, use of some of the benzodiazepines).
Prescribing practices with older adults involve a complex, evidence-based decision-making process in which clinicians must balance risks and benefits to achieve optimal health outcomes. Using national data from the UDS, this study is the first to our knowledge to empirically examine the potential adverse health outcomes associated with high number of prescription medications and Beers-defined PIRx in older adults with dementia. Although polypharmacy increases the risk of PIRx, our study provides little evidence that PIRx contributes to the observed association between polypharmacy and lower functional status among participants with dementia. Our results will need to be replicated in other more generalizable populations. In summary, the necessity of high number of prescription medications should be further examined in community-dwelling older adults with dementia. Often employed as indicators of prescription quality, the Beers list needs to be further investigated in regard to its adverse health effects in this population.
Footnotes
Appendix A
Multivariate Models of the Association Between Polypharmacy (5+ or 7+ Prescribed Medications) and PIRx on Functional Status (Presentation of Full Results)
| Functional Status at second ADC Visit | OR | 95% CI | P Value |
|---|---|---|---|
| Polypharmacy = 5+ Medications | |||
| Model 1 | |||
| First visit having 5+ meds | 1.23 | (1.01-1.49) | .038 |
| First visit PIRx | 0.81 | (0.63-1.05) | .109 |
| Age | |||
| 65-69 | 1.00 | – | – |
| 70-74 | 0.92 | (0.75-1.14) | .471 |
| 75-79 | 1.00 | (0.75-1.33) | .996 |
| 80-84 | 0.98 | (0.76-1.26) | .871 |
| >85 | 1.19 | (0.84-1.69) | .332 |
| Sex Female | 1.05 | (0.84-1.32) | .657 |
| Race/ethnicity | |||
| Non-Hispanic (NH) white | 1.00 | – | – |
| NH black | 1.07 | (0.81-1.41) | .653 |
| Hispanic | 1.23 | (0.83-1.83) | .294 |
| NH other | 0.94 | (0.35-2.51) | .906 |
| Highest education | |||
| College | 1.00 | – | – |
| Less than high school | 0.90 | (0.60-1.34) | .596 |
| High school degree | 0.95 | (0.79-1.14) | .568 |
| Graduate degree | 0.97 | (0.76-1.23) | .782 |
| No of comorbid conditions 1 | 0.96 | (0.90-1.03) | .276 |
| Baseline CDR global score | |||
| 0.5 = very mild dementia | 1.00 | – | – |
| 1 = mild dementia | 2.20 | (1.71-2.82) | <.001 |
| 2 = moderate dementia | 7.07 | (4.99-10.03) | <.001 |
| 3 = severe dementia | 30.94 | (14.65-65.37) | <.001 |
| Baseline functional status | |||
| Able to live independently | 1.00 | – | – |
| Need help with complex activities | 4.66 | (3.19-6.80) | <.001 |
| Need help with basic activities | 18.31 | (9.66-34.74) | <.001 |
| Completely dependent | 127.55 | (50.69-320.93) | <.001 |
| Time between visits (year) | 1.58 | (1.16-2.15) | .004 |
| Model 2 | |||
| First visit having <5 meds, no PIRx | 1.00 | – | – |
| First visit having <5 meds, PIRx | 0.94 | (0.58-1.54) | .820 |
| First visit having 5+ meds, no PIRx | 1.27 | (1.03-1.56) | .025 |
| First visit having 5+ meds, PIRx | 0.96 | (0.72-1.28) | .794 |
| Age | |||
| 65-69 | 1.00 | – | – |
| 70-74 | 0.92 | (0.75-1.14) | .466 |
| 75-79 | 1.00 | (0.75-1.33) | .974 |
| 80-84 | 0.98 | (0.76-1.26) | .865 |
| >85 | 1.19 | (0.84-1.69) | .339 |
| Sex | |||
| Female | 1.05 | (0.84-1.33) | .656 |
| Race/ethnicity | |||
| Non-Hispanic (NH) white | 1.00 | – | – |
| NH black | 1.07 | (0.81-1.42) | .637 |
| Hispanic | 0.95 | (0.35-2.53) | .911 |
| NH other | 1.23 | (0.83-1.83) | .292 |
| Highest education | |||
| College | 1.00 | – | – |
| Less than high school | 0.90 | (0.60-1.34) | .596 |
| High school degree | 0.95 | (0.79-1.14) | .560 |
| Graduate degree | 0.97 | (0.76-1.23) | .782 |
| No of comorbid conditions | 0.96 | (0.90-1.03) | .268 |
| Baseline CDR global score | |||
| 0.5 = very mild dementia | 1.00 | – | – |
| 1 = mild dementia | 2.20 | (1.71-2.81) | <.001 |
| 2 = moderate dementia | 7.04 | (4.97-9.98) | <.001 |
| 3 = severe dementia | 30.91 | (14.65-65.24) | <.001 |
| Baseline Functional Status | |||
| Able to live independently | 1.00 | – | – |
| Need help with complex activities | 4.66 | (3.20-6.79) | <.001 |
| Need help with basic activities | 18.43 | (9.73-34.89) | <.001 |
| Completely dependent | 127.10 | (50.46-320.14) | <.001 |
| Time between visits (year) | 1.58 | (1.16-2.15) | .004 |
| Polypharmacy = 7+ Medications | |||
| Model 1 | |||
| First visit having 7+ meds | 1.30 | (1.03-1.63) | .026 |
| First visit PIRx | 0.81 | (0.64-1.03) | .084 |
| Age | |||
| 65-69 | 1.00 | – | – |
| 70-74 | 0.92 | (0.75-1.14) | .464 |
| 75-79 | 1.01 | (0.76-1.34) | .969 |
| 80-84 | 0.98 | (0.77-1.26) | .899 |
| >85 | 1.19 | (0.84-1.68) | .339 |
| Sex | |||
| Female | 1.06 | (0.84-1.34) | .607 |
| Race/ethnicity | |||
| Non-Hispanic (NH) white | 1.00 | – | – |
| NH black | 1.06 | (0.80-1.40) | .698 |
| Hispanic | 0.94 | (0.35-2.53) | .907 |
| NH other | 1.24 | (0.84-1.83) | .283 |
| Highest education | |||
| College | 1.00 | – | – |
| Less than high school | 0.88 | (0.59-1.32) | .541 |
| High school degree | 0.95 | (0.79-1.15) | .611 |
| Graduate degree | 0.97 | (0.76-1.23) | .788 |
| No of comorbid conditions 1 | 0.97 | (0.91-1.03) | .274 |
| Baseline CDR global score | |||
| 0.5 = very mild dementia | 1.00 | – | – |
| 1 = mild dementia | 2.19 | (1.71-2.82) | <.001 |
| 2 = moderate dementia | 7.07 | (4.98-10.03) | <.001 |
| 3 = severe dementia | 31.50 | (14.59-68.05) | <.001 |
| Baseline functional status | |||
| Able to live independently | 1.00 | – | – |
| Need help with complex activities | 4.69 | (3.21-6.84) | <.001 |
| Need help with basic activities | 18.35 | (9.65-34.91) | <.001 |
| Completely dependent | 130.97 | (51.84-330.90) | <.001 |
| Time between visits (year) | 1.60 | (1.17-2.19) | .003 |
| Model 2 | |||
| First visit having <7 meds, no PIRx | 1.00 | – | – |
| First visit having <7 meds, PIRx | 0.81 | (0.57-1.16) | .256 |
| First visit having 7+ meds, no PIRx | 1.30 | (0.98-1.72) | .072 |
| First visit having 7+ meds, PIRx | 1.06 | (0.71-1.58) | .785 |
| Age | |||
| 65-69 | 1.00 | – | – |
| 70-74 | 0.92 | (0.74-1.14) | .463 |
| 75-79 | 1.01 | (0.76-1.34) | .970 |
| 80-84 | 0.98 | 0.77-1.26) | .899 |
| >85 | 1.18 | (0.83-1.69) | .346 |
| Sex | |||
| Female | 1.06 | (0.84-1.34) | .609 |
| Race/ethnicity | |||
| Non-Hispanic (NH) white | 1.00 | – | – |
| NH black | 1.06 | (0.80-1.40) | .699 |
| Hispanic | 0.94 | (0.35- 2.54) | .907 |
| NH other | 1.24 | (0.84-1.83) | .284 |
| Highest education | |||
| College | 1.00 | – | – |
| Less than high school | 0.88 | (0.59-1.32) | .541 |
| High school degree | 0.95 | (0.80-1.14) | .608 |
| Graduate degree | 0.97 | (0.76-1.23) | .788 |
| No of comorbid conditions 1 | 0.97 | (0.91-1.03) | .276 |
| Baseline CDR global score | |||
| 0.5 = very mild dementia | 1.00 | – | – |
| 1 = mild dementia | 2.19 | (1.71-2.82) | <.001 |
| 2 = moderate dementia | 7.07 | (4.98-10.03) | <.001 |
| 3 = severe dementia | 31.51 | (14.60-67.99) | <.001 |
| Baseline Functional Status | |||
| Able to live independently | 1.00 | – | – |
| Need help with complex activities | 4.69 | (3.22-6.84) | <.001 |
| Need help with basic activities | 18.35 | (9.62-34.99) | <.001 |
| Functional Status=Dependent | 130.98 | (51.99-329.98) | <.001 |
| Time between visits (year) | 1.60 | (1.17-2.19) | .003 |
Abbreviations: ADC, Alzheimer’s Disease Center; PIRx, potentially inappropriate prescription medications; 5 + meds, 5 or more prescription medications; <5 meds, fewer than 5 prescription medications; 7+ meds, 7 or more prescription medications; <7 meds, fewer than 7 prescription medications; CDR, Clinical Dementia Rating scale.
a Comorbid conditions included hypertension, hypercholesterolemia, incontinence, cardiovascular disease, thyroid disease, diabetes, and cerebrovascular disease.
Acknowledgments
The authors thank Mr Andrew Harris for his contribution to the early stages of this manuscript.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The data for this study—the Uniform Data Set from the National Alzheimer's Coordinating Center (NACC)—was supported by the National Institute on Aging Grant (U01AG016976) to the NACC. During this study, Dr Lau was supported in part by a K01 research award (K01AG027295) from the National Institute on Aging. Mr Mercaldo was supported in part by the National Alzheimer's Coordinating Center under a research grant from the National Institute on Aging (U01AG016976). Dr Shega was supported by a career development award sponsored by the National Palliative Care Research Center and the National Institute on Aging (K23AG029815). Dr Weintraub's and Dr Rademaker's roles were supported in part by Alzheimer's Disease Core Center Grant (AG13854) to Northwestern University (PI: Mesulam).
