Abstract
A proof-of-concept for an advanced-level computerized training tool for cryosurgery is demonstrated, based on three-dimensional cryosurgery simulations and a variable insertion depth strategy for cryoprobes. The objective for system development is two-fold: to identify a cryoprobe layout in order to best match a planning isotherm with the target region shape and to verify that cryoprobe placement does not violate accepted geometric constraints. System validation has been performed by collecting training data from 17 surgical residents having no prior experience or advanced knowledge of cryosurgery. This advanced-level study includes an improved training session design in order to enhance knowledge dissemination and elevate participant motivation to excel. In terms of match between a planning isotherm and the target region shape, results of this demonstrate trainee performance improvement from 4.4% in a pretest to 44.4% in a posttest over a course of 50 minutes of training. In terms of combined performance, including the above-mentioned geometrical match and constraints on cryoprobe placement, this study demonstrates trainee performance improvement from 2.2% in the pretest to 31.1% in the posttest. Given the relatively short training session and the lack of prior knowledge, these improvements are significant and encouraging. These results are of particular significance, as they have been obtained from a surgical resident population which are exposed to the typical stress and constraints in advanced surgical education.
Introduction
Cryosurgery is a surgical procedure that destroys cancerous tissues by freezing. Minimally invasive cryosurgery may require the strategic placement of multiple cryoprobes within a target region, with the objective of maximizing injury within the target region, while preserving the surrounding tissues. In prostate cryosurgery, for example, the target region may be the entire prostate gland or a portion of it. 1 For a given cryosurgery system, successful procedure outcome mandates the optimal selection of three key parameters: the number of cryoprobes to be used, their layout, and their thermal protocol. Optimal selection of these parameters results with an ideal match between a planning isotherm and the contour of the target region.
The planning isotherm is a clinically relevant temperature selected by the cryosurgeon based on common practices. This planning isotherm most frequently represents the onset of freezing as visualized via medical imaging, although the so-called lethal temperature—a temperature threshold below which the maximum tissue destruction is reached—should also be considered. 2 Unfortunately, the relationship between the planning isotherm and the lethal temperature is not explicitly known during cryosurgery.
With the above-mentioned key parameters in mind, cryosurgery planning is largely a subjective process, where the surgeon relies on personal experience and common practices. Suboptimal cryosurgery planning may leave viable tissue within the target region to be destroyed, lead to destruction of healthy surrounding tissues, require an excessive number of cryoprobes, result in an elongated procedure, and trigger postoperative complications. 3 It follows that optimal cryosurgery planning may increase the quality of the medical treatment while decreasing its cost. 3
Current methods used in minimally invasive cryosurgery training are limited. Cryosurgery training follows the apprenticeship model, wherein novice physicians observe a surgical procedure performed by an experienced physician after reviewing a set of guidelines believed to yield surgical success. Often, the observed procedure may integrate software provided by cryosurgery hardware manufacturers to assist in the selection of optimal cryosurgery parameters. Following the apprenticeship approach and relying on available clinical cases may be constrained by opportunity, availability of patients, and instruction time and may even conflict with clinical operations. 4
The quality of cryosurgery instruction is limited by the cases available during the specific training period in the training facility, the cryosurgery hardware at hand, and the personal experience and knowledge of the instructing cryosurgeon. Furthermore, the lack of standardization in current cryosurgery practices is likely to yield surgical techniques and supporting concepts that vary significantly among instructing cryosurgeons. These variations may give rise to some sense of uncertainty under the current training paradigm, although the fundamental principles of modern cryosurgery have been investigated for more than half a century 5 and the underlying science of cryobiology is well understood. 2
Computer-based training can circumvent many limitations imposed by traditional education methods, such as increased opportunities for demonstrations and practice and systematic means to assess knowledge and competences. 6 In turn, computerized training offers mitigation of ethical issues associated with training on patients and increased cost effectiveness of training. 7 At present, computer-based training is widely accepted in medical education and has been successfully integrated into numerous medical specialties. Computer-based training systems are most frequently found in the following forms: (1) part-task trainers comprised part- or full-body mannequins capable of replicating pathophysiological vital signs, (2) virtual reality simulators often accompanied by haptic systems that replicate visual, tactile, and kinesthetic experience of performing a specific task, and (3) multimedia applications that often provide case-based computer-assisted instruction to facilitate decision making and diagnostic skills. 8 –12 A review of available computerized training systems has been presented recently. 6
Based upon a recently developed proof-of-concept, 6 the current study presents an advanced-level simulation-based cryosurgery training system. This study uses prostate cryosurgery as a development model and optimal match between the shapes of the frozen region and the organ as a training target. Key advances in the current study are associated with the migration from two- to three-dimensional planning, where a single insertion depth was constrained in the previous study, whereas a variable insertion depth strategy is the focus of the current study. Furthermore, the current study aims at improved tutor–trainee interaction and training session design.
Methods
Conceptual Design of the Trainer
The overarching goal of the Trainer is to create a cryosurgery education environment by means of virtual case practices. The current study focuses on 3D cryoprobe layout design based on two key features: common geometric constraints for cryoprobe placement and the match between the shapes of the target region and a planning isotherm. The mismatch between those shapes is defined as a
Figure 1 displays the four main components of the Trainer: cryosurgery simulator, cryoprobe layout planner, tutor, and database. The simulator uses an efficient numerical technique to simulate the bioheat transfer process for a given cryoprobe layout. 15 The simulator’s output includes the resulting 3D temperature field at the end of the simulated procedure, the defect distribution, and its overall value. The cryoprobe layout planner generates an optimized cryoprobe layout for a specific target geometry based on the bubble packing method. 14,16 The tutor encompasses means to present the trainee with a planning case and the corresponding trainee layout evaluation. The database contains all case-related information, including organ geometries, previously solved cases, and trainee track record.

Cryosurgery Trainer architecture, including the cryosurgery computational tools (CCT) developed previously. 6
Trainer Design Modifications
The first Trainer prototype has been presented recently 6 where design modifications are highlighted subsequently. Beyond advancing the planning scheme from 2D to 3D, system design expands the aim of this study at encouraging the trainee to follow preferred cryosurgery practices while improving trainer–trainee interaction. Figures 2 and 3 display the graphical user interface (GUI) of the modified trainer prototype where design changes from the previous version of the system 6 are marked with “*.”
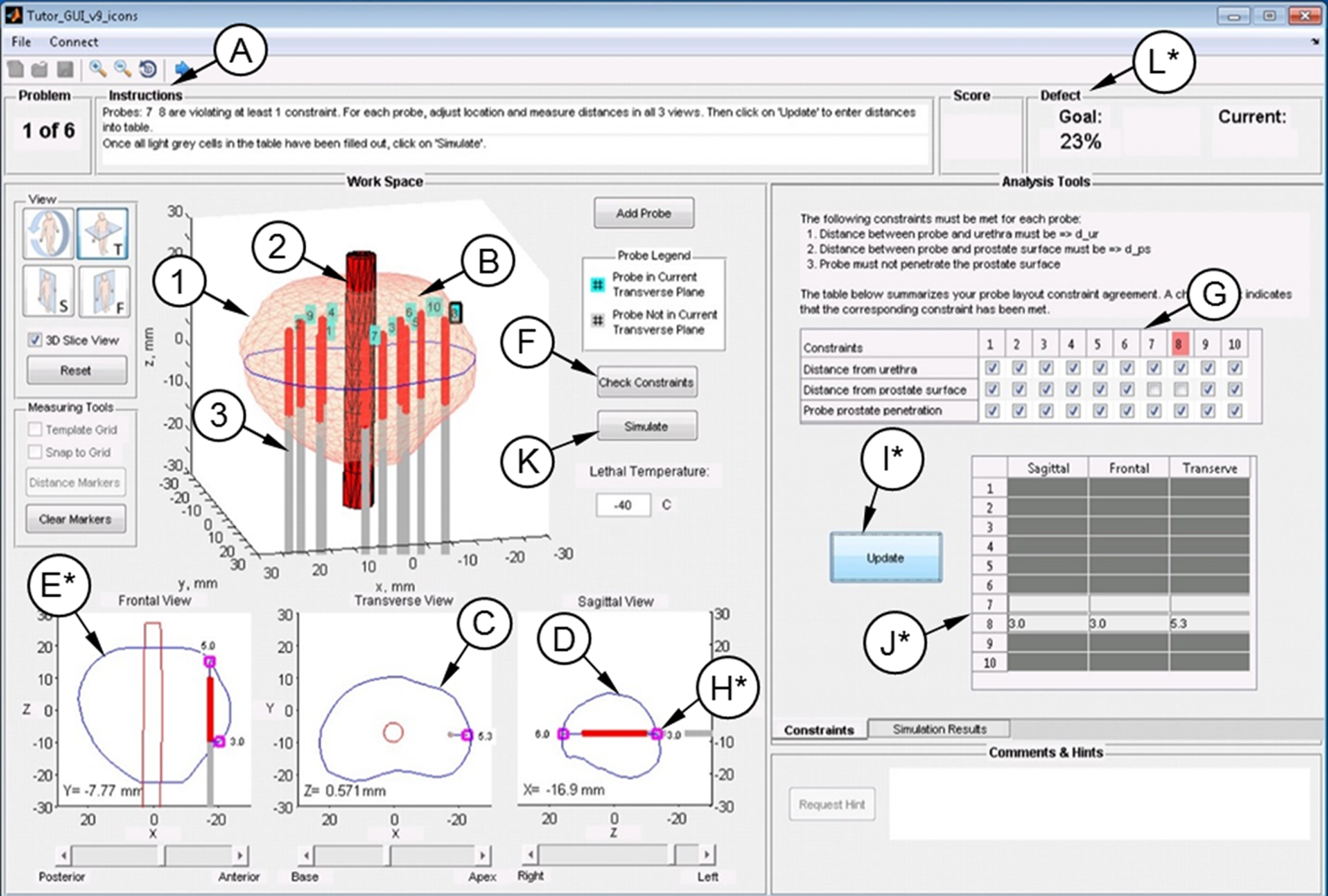
A representative screen of the graphical user interface (GUI) of the cryosurgery Trainer: (A) instructions panel; (B) geometrical presentation of the prostate to be treated, where (1) is the prostate capsule, (2) is the urethral warmer, and (3) is a cryoprobe; (C) a transverse cross section of the prostate and the urethra, with a sliding bar for sagittal location; (D) a sagittal cross section of the prostate, with adjustable transverse location with the sliding bar below; (E) a frontal cross section of the prostate, with adjustable sagittal location with the sliding bar below; (F) constraints violation check button; (G) table summarizing all probe violations; (H) markers for distance measurements; (I) probe distance update button for constraint violating probes; (J) table with measured distance values for violating probes; (K) a bioheat simulation button; and (L) panel displaying the total defect percentage. Key modifications in the current study are marked with “*.”

A representative screen of the graphical user interface (GUI) of the cryosurgery Trainer: (A) temperature map presented at the end of a cryosurgery simulation, where pink represents the prostate contour, black represents defect region, and the red-blue spectrum is correlated with temperature according to color scale (B); (C) video panel for automated display of the simulated temperature map; (D) goal defect, calculated based on a computer-generated cryoprobe layout; (E) actual defect of the most recent trainee planning; and (F) combined training score based on constraint violations and defect size. Key modifications in the current study are marked with “*.”
Variable insertion depth
Depending on the target geometry, a surgeon may choose to insert all cryoprobes at the same depth or in a staggered formation. The variable insertion depth planning is more complex due to the additional degree of freedom (z-coordinate), as reflected by the three orthogonal views in Figure 2-C to E. It should be noted that only transverse (Figure 2-C) and sagittal (Figure 2-D) transrectal ultrasound scan images are available in clinical practice. It was found favorable to move cryoprobes on the
Trainer feedback
Two features were integrated into the Trainer in the previous study to identify constraint violations—a check table (Figure 2-G) and a distance visualization tool. 6 These features were found to be equally effective, and thus, the latter was eliminated in the current study in favor of an improved GUI design. Although the summary of constraint violations is readily available in the check table, the strategy on how to make use of this information has been changed in the current study. Similar to ultrasound imaging analysis, the Trainer includes a distance measurement tool which helps identify and eliminate cryoprobe placement violations but requires voluntary use. Many trainees have not elected to make use of this tool in the previous study, 6 and since distance measurements are deemed critical for planning success, the training strategy has been modified in the current study as displayed in Figure 4. In particular, the phases of layout planning (phase 1), constraints verification (phase 2), and cryosurgery simulation (phase 4) were simplified, while a probe distance measurement phase (phase 3) was added. The trainee could potentially skip distance measurements and not be penalized if no constraint violation has occurred. However, when a violation did occur, the trainee must measure distances from the cryoprobe to the relevant surfaces in each of the cross-sectional views (Figure 2-C,D,E), which automatically updates the content of the distance table (Figure 2-J). Only after all violating probes have been measured and corrected, can the trainee move onto the simulation phase 4 of the specific case.

Schematic illustration of the sequence of operations during trainee–tutor interaction. Compared with the previous case structure, 6 phases 1, 2, and 4 have been simplified and phase 3 has been added, requiring distance measurement for all cryoprobes violating at least 1 constraint.
Automated simulation display
It was found in the previous study 6 that the level of trainee interest in the simulation of temperature field plots (Figure 3-A) has declined over time during the training session. This decreased level of interest may be attributed to (1) the manual scrolling method through the 3D temperature field and (2) underappreciation of the need in such a review in order to develop a correlation between the defect distribution and the cryoprobe layout. For this purpose, an automated movie presentation of the temperature field at the end of simulation has been integrated into the modified Trainer, which cannot be bypassed but can be replayed and paused at will (Figure 3-C).
Assessment metrics
Although the overall defect represents an easy-to-follow output indicator of the Trainer GUI, it represents only one aspect of training where constraint violations may be equally important. Furthermore, the absolute defect value in itself (i.e., defect volume) is less informative than its relative value to the volume of the target region, which is presented in percentage in the modified Trainer (Figure 3-E). The goal defect (Figure 3-D) represents the outcome of the computer-generated, automated planning. 14 With these two observations in mind, a combined score is integrated into the modified Trainer as follows (Figure 3-F):

where
Study Design
The objective of the current study is to evaluate the effectiveness of the modified cryosurgery trainer prototype by measuring trainee learning gains. Trainer evaluation was conducted in two-hour sessions of up to 6 trainees following the time breakdown in Table 1. Each session started with a brief introduction to cryosurgery, combined with the long-term goals of this line of research. The introduction was followed by an interactive demonstration of the training software prototype where trainees followed along with the presenter on their own machines, each working through one planning exercise. The interactive demonstration reviewed the Trainer tools, with emphasis on verification of cryoprobe constraint criteria and interpretation of temperature field results.
Training Session Outline in the Current Study.

Prior to training, a pretest was administered where each trainee had only one attempt to solve three selected cases. During the pretest, the trainee had access to the distance measuring tool, but no additional means of feedback were made available. In each case, the trainee was presented with a unique prostate model (including the urethra) and required to plan a layout using 10 cryoprobes. It is noted that the selection of the ten cryoprobes for the current proof-of-concept demonstration is a choice of practice. Although current clinical practices are based on a dozen or more cryoprobes, frequently using a pullback strategy, 17 variable insertion depth—the focus of the current study—is yet to be reduced in practice. Variable insertion depth represents a higher level of freedom in layout planning using fewer cryoprobes. Although ten cryoprobes were found appropriate for the trainee’s first layout in the context of the current investigation, future studies will attempt to optimize the number of cryoprobes in this new strategy. Prostate models were generated by geometrically deforming base prostate shapes, reconstructed from preclinical ultrasound scanning, as described previously. 18 During the training session, trainees had 50 minutes to complete as many cases as possible (up to 6). A case was successfully completed when a planning score of 100% was achieved, which means that both defect and geometric constraint criteria have been met or acceded. Trainees were allowed an unlimited number of attempts to reach a score of 100%. The study session concluded with a posttest in which the trainee solved the same 3 cases as the pretest.
This study was performed on a total 9 prostate geometric models, all having a volume of 35 mL, which is an average volume for cryosurgery candidates. These models were created from 5 preclinical ultrasound data sets of the prostate by means of geometrical deformations.
18
With reference to Table 1, three models were used for pretest, 6 models were used for training, and the same three models from the pretest were used again for the posttest. Note that planning solutions were not given at the end of the pretest on the same cases. The applied thermal history was selected to simulate the cooling capabilities of Joule-Thomson-based cryoprobes, as discussed in detail in the study by Lung
Geometric constraints are obtained from the accepted prostate cryosurgery practices as follows: (1) all probes should be placed at a given safety margin from the prostate capsule, (2) all probes should be placed at a given safety margin from the urethra, and (3) the active cooling surface of each cryoprobe must be encompassed within the target region. Although the actual value for the above-mantioned safety margin may vary among clinicians, a 3 mm distance was deemed appropriate for the purpose of the current demonstration without affecting its generality. A planning isotherm of −22°C was selected for the current study. Defect is calculated as any volume external to the target region with temperatures below the planning isotherm and, conversely, any volume internal to the target region with temperatures above it. It is important to note that the presented criteria do not lead to a unique solution nor do they necessitate a specific planning strategy—it is possible to generate a class of cryoprobe layouts that meet the above-mentioned criteria. In the current study, the reference (computer generated) solution represents a local minimum on the defect in an optimization process and not simply the first-found, computer-generated solution. The reference solution is considered as an expert solution in the current study.
The human subject study was approved by the Allegheny Singer Research Institute, Allegheny Health Network (AHN), Pittsburgh, Pennsylvania (institutional review board protocol #14-006). This study was performed at the Simulation, Teaching and Academic Research (STAR) Center of AHN. Eighteen participants were recruited from the AHN surgical residency programs. Of the 17 residents, 12 (71%) were males and 5 (29%) were females, not by choice but due to the current demographics of the surgical residency program at AHN. All bioheat transfer simulations and defect calculations were performed with a recently developed graphics processing unit-based code, with a typical runtime of a few seconds on personal workstations located at the Biothermal Technology Laboratory (BTTL) at Carnegie Mellon University. 19 In order to make use of superior computation capabilities at the BTTL while benefiting from the unique educational environment at the STAR Center, human subject studies were performed by means of remote networking between the AHN and the BTTL, using a framework that further signifies the potential for internet-based training.
All statistical analyses were performed using Statistical Package for the Social Sciences (SPSS; IBM Co, Armonk, NY). Comparison between the pretest and posttest results was performed using the Wilcoxon signed rank sum test, and correlation between two variables was determined using Spearman ρ correlation coefficient.
Results
Figure 5 displays the mean number of planning attempts made by all trainees for the respective cases during the training session. Of the 17 trainees, case 1 data for 4 trainees were omitted—3 trainees’ data were lost due to technical problems and 1 trainee did not comprehend that probes could also be moved in the z-direction (i.e., axial direction). These problems were corrected in the subsequent cases solved. Overall, the required number of attempts decreases with increasing number of cases solved. Within the allotted time of 50 minutes, all trainees completed at least two cases, while four trainees advanced fast enough to complete the six available cases.
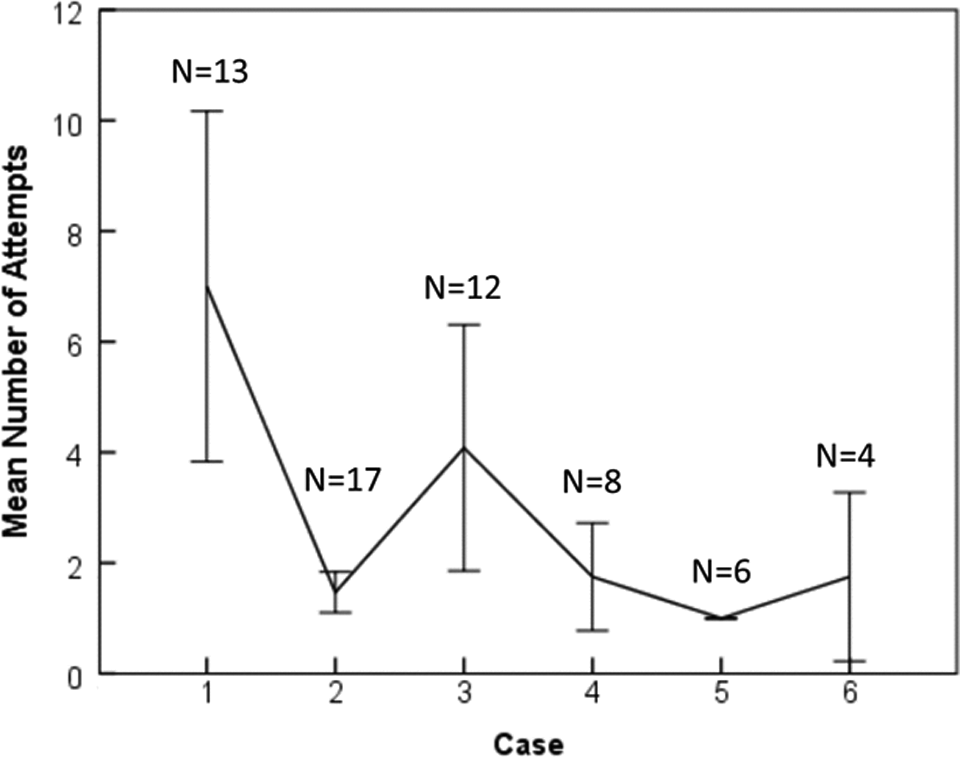
Mean attempts per case until a 100% overall training score has been obtained. Error bars represents standard error at 95% confidence interval. N represents the number of trainees who completed the respective training case.
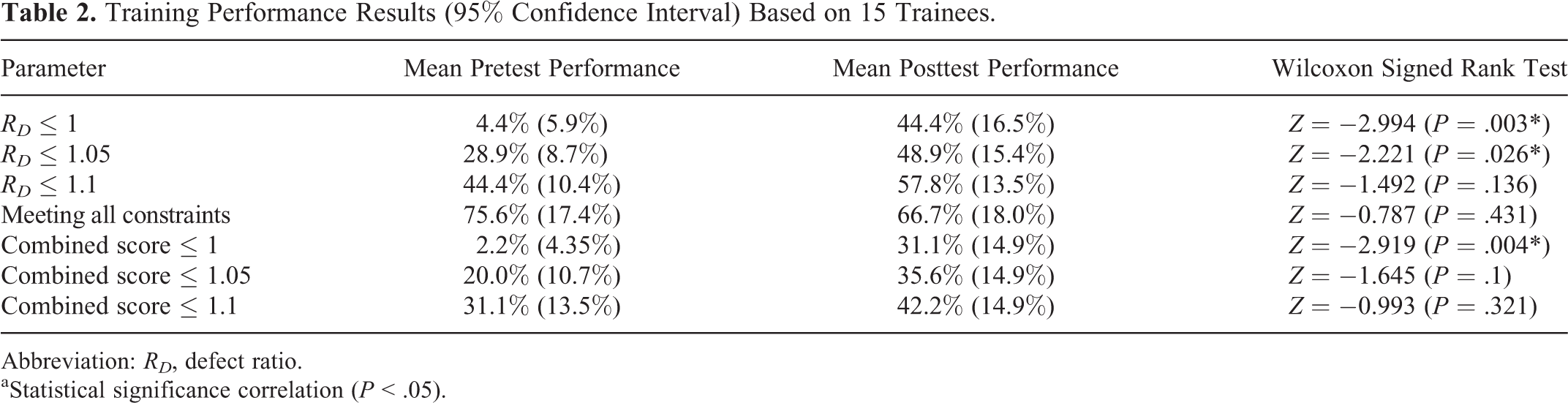
Table 2 lists a summary of pretest and posttest performance, where the defect ratio is defined as:

Training Performance Results (95% Confidence Interval) Based on 15 Trainees.
Abbreviation:
aStatistical significance correlation (
where
Table 2 lists evaluation results for three
In order to assess the effectiveness of the trainer, it is not only important to apply the pass/fail criteria detailed previously but also to measure the incremental performance improvements between the pretest and the posttest. Figure 6 displays the posttest

Comparison of pretest versus posttest results for 15 participants based on defect ratio calculations. Green symbols represent an improvement in terms of constraint violations from pretest to posttest, whereas red symbols represent no improvement. Actual symbols correspond to improvement categories, whereas “n” indicates that any number of probe constraint violations have been recorded, ranging between 1 and 10 in the current study. The area under the dashed horizontal line represent posttest goal, whereas any symbol below the diagonal line represents an improvement between the pretest and the posttest in terms of defect calculations.
Cases of no improvement in constraint violations are marked red in Figure 6, that is, the number of probe constraint violations has remained the same or became worse. About 80% of trainees in case 1 and 67% of trainees in cases 2 and 3 demonstrated improvement in terms of constraint violations (recall, cases 1, 2, and 3 correspond to prostate models 1, 2, and 3, respectively). Although the Wilcoxon signed rank test revealed no statistically significant difference between the pretest and posttest with respect to meeting the constraint criteria (evaluated using binary pass–fail scheme, where layouts with no constraint violations are scored as 100% and 0 otherwise), the actual percentage of trainees who were able to demonstrate an improvement over the course of their training is quite significant. Note that Figure 6 summarizes only 15 trainees, as 2 trainees did not practice adjustment of the cryoprobe insertion depth during their pretest.
The dotted horizontal line in Figure 6 represents the posttest goal defect for all cases. The proportional line in Figure 6 signifies changes from pretest to posttest, where all data points below this line correspond to a decreased defect ratio for a specific trainee. With reference to the posttest alone, 9, 8, and 3 trainees met the defect criteria in cases 1 through 3, respectively. However, of the 15 trainees, 13, 9, and 12 trainees reduced their pretest defect ratio in cases 1 through 3, respectively. Additionally, the number of trainees who met both constraints and defect criteria in the posttest is 7, 5, and 2 trainees in cases 1 through 3, respectively. The variation in the number of trainees who were able to meet the planning criteria between the 3 posttest cases may be associated with the inherent geometric complexity of each case, with case 3 appearing to be the most difficult.
Discussion
It can be seen from Table 2 that perfect trainee performance in terms of the defect ratio (
It is noted that all training sessions in this study were held at 8 AM at the middle of the week, where the trainee population (surgical residents) represents a collection of individual participants to a varying level of stress. Some trainees benefited from a good night sleep and free time on their hand, some rushed in from earlier professional commitments in a different hospital across town, whereas others joined the session quite tired after an overnight shift. These prior activities may well have an effect on the individual’s performance and the general ability to follow recommended practices. Nevertheless, there is no practical way to isolate such stress-inducing activities, although the current trainee population potentially represents the group who would most benefit from computerized training as a whole. Hence, this study is perceived to simulate the realistic conditions for medical training, which further signifies the reported training performance.
Although minimizing defect is a complicated task requiring a multitude of concepts and skills, planning a layout free of constraint violations is essentially a procedural task. With unlimited time and access to adequate measuring tools, creating a layout that meets the constraint criteria may be perceived as a simple task, with no apparent technical barrier to reach a perfect score. Based on recorded keystroke data and regardless of specific case scores, cryoprobe distance measurements were taken in only 20 of 45 cases during the pretest and reduced to 7 of 45 cases during the posttest. These numbers may reflect an incorrect perception of trainee ability, over self-confidence in the ability to eyeball distances, lack of motivation, or tangential reasons not related to the Trainer activity.
Some of these observations are consistent with a previous study, 6 which led to modifications in the Trainer’s structure and the training session design, as described in the Methods section. For example, the current Trainer is redesigned to force cryoprobe organ surface distance measurement (a necessary task, which is consistent with current clinical practice), but the trainee behavior remained uncorrected. It may be that additional measures must be taken to correct the trainee behavior, such as imposing a greater penalty for unmeasured probe violations during training. As another example, the introductory portion of the training session, including the interactive demonstration (Table 1), has been redesigned to stress the need for distance measurements, but the practice of knowledge dissemination can be improved.
The current study enables a variable insertion depth in contrast to the previous study 6 which utilized a uniform insertion depth for all cryoprobes. Although a variable insertion depth is quite uncommon in clinical practice, an intermediate approach is frequently applied, known as the pullback procedure. 17 The great majority of trainees were able to satisfy planning criteria in the current study using variable insertion-depth strategy. From an engineering optimization perspective, 3 the quality of the variable insertion depth planning is potentially higher, as a better match may be found between the planning isotherm and the target region shape.
The previous study required 6 training cases, 6 whereas the current study requires 50 minutes of training for each trainee. Only 4 of the 15 trainees were able to complete the 6 available training cases in the current study (Figure 5). With the improved trainee performance in the current study, it may be concluded that the variable insertion depth is more intuitive to learn and possibly that the training exposure time is more important than the number of cases practiced. Future studies should include specific tasks to test those hypotheses.
Although training outcomes in the current study are comparable with the previous study, 6 it should be noted that the training methodologies are quite different, with an intelligent tutoring system (ITS) as a main theme in the previous study. Following that concept, only the suboptimal portion of the trainee layout was required to be replanned in the consecutive iteration, whereas the remaining portion of the layout became locked. By identifying and focusing on suboptimal areas, the ITS training session offered intermediate information, while going faster through training sessions and covering more training cases within a specific time slot. Nevertheless, the ITS does not necessarily offer more overall training opportunities. Furthermore, the ITS may decrease the intellectual challenge associated with replanning, potentially adversely affecting trainee motivation to excel. Although no ITS concepts have been utilized in the current study, the average trainee performance in the current study has improved. This observation does not necessarily suggest that the ITS concept is ineffective but that significant modifications to it may be necessary.
Summary
This study presents an advanced-level simulation-based cryosurgery training system. This study uses prostate cryosurgery as a development model and optimal match between the shapes of the frozen region and the organ as a training target. Key advances in the current study are associated with 3D planning, improved tutor–trainee interaction, and training session organization. These advances have demonstrated improved training outcomes.
In terms of geometrical match between the planning isotherm and the target region shape, this study demonstrates trainee performance improvement from 4.4% in the pretest to 44.4% in the posttest over a course of 50 minutes of training. In terms of combined performance, including geometrical match and constraints of cryoprobe placement, this study demonstrates trainee performance improvement from 2.2% in the pretest to 31.1% in the posttest. Given the relative short training session and the lack of experience by the trainees, these improvements are significant and encouraging. These results are of particular significance, as they have been obtained on a surgical resident population subject to the typical stress of medical education.
In general, trainees did not use all available means in order to satisfy the required planning criteria. Further development of training methodologies must seek ways to increase motivation to use all available means in an effort to improve planning outcomes.
Footnotes
Abbreviations
Acknowledgments
Special thanks to the residents of the General Surgery Residency Program of the Allegheny Health Network for volunteering time and effort to support this study.
Authors’ Note
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute or the National Institutes of Health.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by Award Number R01CA134261 from the National Cancer Institute. This study has also been supported in part by the Simulation, Teaching and Academic Research (STAR) Center of the Allegheny Health Network, Pittsburgh, Pennsylvania.
