Abstract
Skeletal metastases are a common cause of severe morbidity, reduction in quality of life and often early mortality. Consequently, improvements in therapies are necessary. Electroporation uses electric energy to alter cancer cell membrane permeability and enhance the local uptake of chemotherapeutics, thus leading to local tumor control. The aim of this study was to investigate the feasibility and safety of delivering electric field protocols causing electroporation of healthy bone and structures of clinical relevance using small and large animal models. Reversible electroporation was used in the rabbit sciatic nerve by applying 2 series of 8 pulses 100ms long at 1000 V/cm. Irreversible electroporation was used in rabbit distal femur condyles and in sheep vertebral body by applying 120 pulses 100ms long at 1750 V/cm. Any effect on surrounding sensitive structures was investigated. Reversible electroporation of sciatic nerve was associated with transient foot functional deficit that completely recovered at 30 days. Irreversible electroporation removed cells from trabeculae in the femurs of rabbits and in the vertebral body of sheep. After irreversible protocol, histology and microtomography demonstrated that the trabecular structure was maintained, the presence of new bone marrow cells, osteoblasts, and mineral apposition characterized by new trabeculae thinner than controls (P = .005) and a significant reduction in the ablated areas (−225%, P = .0219). Spinal cord, vertebral pedicles and spinal nerves showed transient edema in the absence of functional or structural alterations. Collectively, these results show that electroporation can be safely applied to bone even in the proximity of neuronal structures.
Introduction
Malignancies in the bone can be of different origins. Cancer cells originate either from the bone tissue or the bone marrow itself or are metastasized from a tumor that originated elsewhere in the body. The special milieu of the bone microenvironment provides a fertile soil for many cancers to metastasize. The consequences of bone metastases are devastating. Severe bone pain, pathologic fractures, hypercalcemia, and spinal cord compression greatly impair patients’ quality of life. Treatment strategies are mostly palliative or aimed at the primary tumor. 1-2 There are a number of options to treat bone metastases (eg, chemotherapy, surgery, X-ray radiation, bisphosphonates, thermal ablation). However, level of severity of patient conditions, tumor pathology, and anatomical localization and extent of the metastases prevent standardization of treatment protocols. Traditionally, surgery is considered to be the most appropriate treatment for bone metastases. However, such an aggressive approach is not possible in many patients. In fact, surgery of bone metastases in the vertebral body is extremely complex and needs to preserve the anatomical structures proximal to the metastasis, spinal cord, nerves, and blood vessels. In other localizations, like to pelvis, surgery may result in permanent damage, or in long bones, it may require major surgery and joint replacement. Several ablative modalities have been introduced in order to locally control tumor without impairing sensitive structures or functions of healthy surrounding tissues and organs: radiation therapy, thermal ablation, or cryoablation. An ablation modality, which would ensure therapeutic effectiveness in eradicating metastases while preserving the functionality of sensitive structures, would allow new bone apposition in a short time and would consequently limit or prevent pathological fractures, offering an innovative useful therapeutic approach.
Electroporation (EP) is a physical method that uses microsecond length electric pulses to alter cell membranes temporarily without affecting cell viability: reversible electroporation (REP). Reversible electroporation is used to facilitate the intracellular diffusion of low permeant hydrophilic drugs (bleomycin or cisplatin) or large molecules (DNA and RNA). 3 Reversible electroporation of the cell membrane is used for gene delivery (electrogenetherapy) 4 and for cytotoxic drug delivery (electrochemotherapy [ECT]). 5 When a high number (80-120) of electrical pulses are used, it may induce cell death directly: irreversible electroporation (IRE). 6 Based on extensive preclinical studies that defined the parameters for effective tumor treatment and elucidated the mechanisms of action, ECT is by now in clinical use since 2006. 7 -9 Electrochemotherapy is a local treatment that uses REP of tumor cells to increase the uptake of cytotoxic drugs, such as bleomycin or cisplatin. 10 In the case of bleomycin, up to 1000-fold increase in cytotoxicity was observed. 10 -12 Currently, ECT is used in the treatment of cutaneous and subcutaneous tumors of different histological types with an objective response rate of 80% and long-lasting complete responses of 70%. 7,13,14 Moreover, the European Standard Operating Procedures of Electrochemotherapy (ESOPE) trial has shown that after a single short session of ECT, 74% of the treated cutaneous and subcutaneous tumor nodules had undergone a complete response and 11% a partial response. 7 No major negative side effects have been reported after ECT, and only minor acceptable side effects, such as pain, muscle contraction or ache, superficial or deep necrosis, hyperpigmentation, bleeding, and local infection, have been reported. 7,15 Isobe et al successfully used EP combined with methotrexate in mice implanted with osteosarcoma cells, 16 and consequently, ECT has been considered for the treatment of bone metastases. 17
An in vivo preclinical study was conducted to investigate whether the presence of the mineralized trabeculae would interfere with electric field distribution and the ability to ablate bone cells and cells among trabeculae. 18 Combinations of electric field amplitude and number of pulses were tested to achieve bone cell ablation without affecting the recovery of osteogenetic activity and bone biomechanical competence. Subsequently, ECT was used to treat experimental osteolytic bone metastases in rats successfully. 19 On the basis of these preclinical data, a phase I–II clinical trial was approved by the local institutional review board for the treatment of patients with bone metastases 20 ; however, the study excluded patients with metastases to the vertebrae for the proximity of sensitive structures, such as spinal cord and nerves.
We hypothesize that EP could be applied directly to bone and nerves without permanent damages to sensitive structures and clinically relevant organs and/or tissues, such as spinal nerves and spinal cord. Thus, specific EP protocols were adopted for each tissue, also selecting the more suitable animal model, in order to investigate the feasibility and safety of delivering electric field protocols on different tissues or organs that are often targeted by tumor or metastasis growth and have to be spared by any permanent toxic effects because of their vital functions.
Materials and Methods
Electroporation Apparatus
A custom-made Cliniporator (IGEA SpA, Carpi, Italy) was used for in vivo EP. Square-wave electrical pulses 100 µs long were delivered, and amplitude was set at 1000 V/cm for REP and at 1750 V/cm for IRE. Pulse frequency was set at 1 and 5000 Hz for IRE and REP, respectively. Pulse protocols used 2 series of 8 pulses for REP and 120 pulses for IRE. The electric field distribution in the bone tissue of sheep was calculated using computer software (COMSOL Multiphysics, Brescia Italy)
The rabbit and sheep studies were performed in compliance with Italian and European Laws on animal experimentation (Legislative decree of January 27, 1992, No. 116). The experimental protocols were approved by the Ethical Committee of the Rizzoli Orthopaedic Institute (Prot. 10434 and 10499 for rabbits and Prot. 8309 and 583 for sheep) and by the Italian Ministry of Health.
Rabbit Animal Model
Surgery
Seven New Zealand male rabbits (body weight 2.500 ± 0.250 kg, 14 weeks old) were housed under controlled conditions (room temperature: 22°C ± 0.5°C; relative humidity: 55% ± 5%) and were supplied with standard diets and water ad libitum. At the time of surgery, general anesthesia was induced with an intramuscular injection of 44 mg/kg ketamine (Farmaceutici Gellini SpA, Latina, Italy) and 3 mg/kg xylazine (Bayer AG, Leverkusen, Germany). Antibiotic therapy (cefazolin, 100 mg/kg) was administered preoperatively, immediately after surgery, and after 24 hours. Analgesics (metamizole chloride, 50 mg/kg) were prescribed in the immediate postoperative period. Animals were checked daily by an experienced veterinarian and clinically examined until scheduled experimental time to detect the onset of any sign of pain, stress, or side effects 21 and functional alterations in gait and peripheral sensibility to hindquarters.
Three animals were killed at 7 days and 4 at 30 days after EP by pharmacological euthanasia under general anesthesia with intravenous administration of Tanax (Hoechst, Frankfurt am Main, Germany).
Irreversible electroporation of bone
A 2-cm skin incision was made bilaterally on the lateral surface of the distal femur: 4 stainless steel cylindrical electrodes (0.7 mm in diameter) were inserted into the epiphyseal trabecular bone at 4 mm distance in a square configuration by a positioning mask and a drilling device. Electroporation was applied to the right condyles (1750 V/cm and 120 pulses), whereas the left condyles were used as sham controls (insertion of electrodes only without IRE). At the end of IRE treatment, the electrodes were cut at the bone surface and the inserted portion left inside the bone as markers for subsequent analysis. Soft tissues were closed in layers with Dexon 3-0 and silk 3-0. To label bone formation, oxytetracycline (30 mg/kg) was injected intramuscularly (im) on the second and third day after surgery in animals killed at day 7 and on the 27th and 28th day after surgery in animals killed at day 30.
Reversible electroporation of sciatic nerves
The animals underwent REP on the right sciatic nerve: 2 series of 8 pulses were delivered at 5 KHz using plate configuration electrodes. Briefly, 2 stainless steel plates 1 cm large and separated by 1 mm were used. The nerve to be pulsed was positioned between the plates, and REP protocol was delivered. In each animal, the left sciatic nerve was used as contralateral control, and it received no treatment.
Histology and histomorphometry
After euthanasia, the distal femoral condyles were excised and cut in the lateral and medial compartment (EXAKT Cutting and Grinding Systems, Germany). The lateral condyles were processed, undecalcified, and embedded in polymethyl methacrylate. Briefly, the samples were fixed in 4% paraformaldehyde for 48 hours, and then dehydrated in graded series of alcohols. After a 24-hour infiltration period in methyl methacrylate, they were finally embedded in polymethyl methacrylate (Merck Schuchardt, Hohenbrunn, Germany). The lateral femur blocks were sectioned along a sagittal plane using a diamond saw microtome (Leica 1600 SpA, Milan, Italy) for histology. Tetracycline emission by new bone formation of unstained sections was performed by manual measurements: 3 nonconsecutive sections were observed under fluorescence by a light microscope (λ = 410 nm; Olympus BX41, Melville), the region of interest (ROI; 1556 × 2084 pixel) among the 4 electrodes was identified, and images were captured at ×1.25 magnification. The ablated area was manually quantified by measuring the zone in the ROI in which tetracycline-labeled bone did not emit fluorescence (Leica Q-Win Imaging Systems, Cambridge, England). Measurements were performed blindly by 2 experienced investigators. Sections were subsequently thinned, polished, and stained with toluidine blue and fast green for cell morphology examination and histomorphometric measurements of the trabecular bone thickness (TbTh, μm) calculated according to the nomenclature and the methodology approved by the American Society of Bone and Mineral Research. 22
The medial femoral condyles were fixed in 10% neutral-buffered formalin for 24 hours, decalcified in a formic/nitric acid solution for 36 to 40 hours, dehydrated, and processed into wax by an automated tissue processor. Sagittal sections of 4 μm were routinely stained with hematoxilin–eosin. Immunohistochemical determination of cell apoptosis by B-cell lymphoma 2 antibody (Bcl-2; Dako Italia SpA, Milan) was performed. Briefly, the antigen retrieval was carried out in buffered solution at pH 8 in water bath at 95°C for 20 minutes. After cooling at room temperature, sections were incubated for 20 minutes with prediluted primary antibody, washed with phosphatase buffer pH 7.5, and subsequently incubated with biotinilated secondary antibody and with alkaline phosphatase/red rabbit (mouse Dako real detection system, cod. K5005; Dako Italia SpA.) on an automated autostainer antibody. The immunoreaction was visualized by the addition of a red chromogen. Negative controls and known positive specimens were included to check proper specificity and performance of the applied reagents.
Sciatic nerve histology
Sciatic nerves were fixed in 4% buffered paraformaldehyde (phosphate buffer 0.13 mol/L, pH 7.2) for 2 hours, postfixed in 1% osmium tetroxide (phosphate buffer 0.13 mol/L, pH 7.2) for 2 hours, dehydrated in increasing graded ethanol, and embedded in epoxy resin (Durcupan ACM, Sigma-Aldrich, St Louis (MO), USA). Afterward, they were transversally and longitudinally sectioned in different points (proximal to the electroporated area, in the electroporated area, and distally to the electroporated area) with a diamond knife (Ultracut-Reichert Microtome, Wien, Austria) to obtain thin sections. Transversal sections were collected on a glass slide, stained with a toluidine blue solution for a few seconds on a warm plate, rinsed in water, dried, and mounted. Longitudinal sections were routinely stained with hematoxilin–eosin staining. Specimens were observed and photographed under a Zeiss Axiophot microscope, Carl Zeiss Italia, Arese (MI), Italy equipped with a Zeiss AxioCam ERc 5s digital camera, using a ×40 magnification objective.
Sheep Animal Model
Surgery
Three adult female sheep crossbred (merinos–sarda) weighing 65 ± 5 kg, (Pancaldi, Budrio, Bologna, Italy) were used. The animals were housed in individual boxes with a grid floor at room temperature and controlled humidity: 22°C ± 1°C and 55% ± 10% relative humidity, with ventilation of 10 air/changes per hour. The animals were fed a standard maintenance diet (Mucedola, Settimo Milanese), clover, and water ad libitum. After a premedication with intramuscular administration of ketamine (10 mg/kg, Imalgene 1000; Merial, Italia spa) and xylazine (0.3 mg/kg, Rompun; Bayer, Milano, Italy) and subcutaneous atropine sulfate 0.125 mg/kg to avoid bradycardia, general anesthesia was induced by intravenous bolus infusion of thiopental sodium in 2.5% (Thiopental, Inresa, Freiburg) solution at a dose of 6 mg/kg. Mechanical ventilation (Servo Ventilator, 900D; Siemens Italia, Milan, Italy) was maintained by endotracheal tube with the administration of O2/N2O 60%/40% (7 L/min) mixture with isoflurane 2% (Aerrane, Baxter SpA, Roma). Under general anesthesia and in a sterile environment, a posterior midline incision was made, and the lumbar spine was exposed, and using a specifically prepared mask, 2 holes were drilled through the vertebral bone in the L3 and 2 in the L4 vertebrae (1.8 mm diameter). Irreversible electroporation protocol (1750 V/cm and 120 pulses) was applied. The 4 electrodes were connected in consecutive couples, and the same number of pulses was applied to each electrode couple in all the 6 possible combinations: 4 sides and 2 diagonals. Following the same procedure, 2 electrodes were inserted into the L2 vertebra, but no pulses were applied to investigate the mechanical effects of drilling on bone trabeculae.
During the postoperative period, antibiotics (cephalosporin 1 g/d for 5 days; Cefazolin Cefamezin Pfizer Italia Srl, Latina) and analgesics (metamizole sodium 50 mg/kg/day for 3 days; Farmolisina Ceva Vetem, SpA, Agrate Brianza) were administered, and the sheep were housed in single boxes under the same environmental conditions. To label bone formation, oxytetracycline 30 mg/kg (Terramicina 100; Pfizer Italia Srl, Latina, Italy) was injected on the third day after surgery. At 7 days from surgery, 2 animals were pharmacologically euthanized with the intravenous administration of Tanax (Hoechst, Frankfurt am Main, Germany) under general anesthesia. Vertebral bodies and surrounding sensitive structures, spinal cord, vertebral pedicles, and spinal nerves were harvested and processed for histological and microtomographical investigations. Animals were checked daily by an experienced veterinarian and was clinically followed for 30 days after EP to detect the onset of any sign of pain, stress, or side effects. 21
Histology and histomorphometry
The vertebral bodies were processed, undecalcified, and embedded in polymethyl methacrylate as described previously. The embedded blocks were cut through the transversal plane using EXAKT cutting and grinding systems (EXAKT Cutting and Grinding Systems, Germany). Tetracycline emission by new bone formation of the unstained section was observed under fluorescence by a light microscope (λ = 410 nm; Olympus BX51). Three sections were subsequently automatically thinned to 20 ± 5 μm, polished, and stained with toluidine blue and fast green for cell morphology examination. After qualitative analysis, a grid consisting of 40 ROIs with dimension of 0.5 × 0.5 cm was superimposed on each section of the vertebral bodies. The number of osteoblasts and osteoclasts present in the ROI was assessed by 2 blinded investigators using computerized image analysis (Axio Vision 4.6; Carl Zeiss, Arese (MI), Italy) at resolution ×20.
The spinal cord, the vertebral pedicle, and the spinal nerve were fixed in 10% neutral-buffered formalin for 24 hours. Then, the vertebral pedicles were decalcified in 4% HCl, 5% formic acid, and after decalcification, dehydrated and then processed for paraffin embedding.
Microtomography of electroporated bone
Microtomographic assessment was carried out on the vertebral bodies using computed microtomographic system Skyscan 1172 (Kontich, Belgium). The scans were performed with 100 kV of voltage source and 100 μA of current source. Images were acquired with a pixel size of 12 μm, an aluminium filter 0.5 mm, a sample rotation of 180°, and 0.4° of rotation step. The scan images were later reconstructed to obtain microtomographic sections. Reconstruction was performed using NRecon software (v1.6.2.0): a specific alignment was used for each sample, and a medium intensity ring artifact correction was applied. Microtomographic images of both control and EP-treated samples were acquired and reconstructed with the same setting of scan and reconstruction parameters and then qualitatively evaluated.
Histological analysis of surrounding tissues
Spinal cord, vertebral pedicles, and spinal nerves were fixed in 10% neutral-buffered formalin for 24 hours. Then, the vertebral pedicles were decalcified in 4% HCl, 5% formic acid, and after decalcification, dehydrated and then processed for paraffin embedding, whereas the spinal cord and spinal nerve were immediately dehydrated and processed for paraffin embedding. Sections of 4 μm thick were cut (Microm HM340E; Microm International GmbH, Heidelberg, Germany) and stained with hematoxilin–eosin.
Statistical Analysis
The statistical analysis was performed using the IBM SPSS Statistics 21 software. Data are reported as mean ± standard deviation (SD) at a significant level of P < .05. Ablated area data in the EP-treated or control samples were analyzed statistically by applying Student t test. For TbTh parameter, the general linear model with Sidak adjusted multiple comparison test with “group” (control and EP treated) and “experimental time” (7 and 30 days) as fixed effects was performed to assess the influence of EP treatment on bone thickness. Precision of the reported coefficients was assessed by creating 1000 bootstrap samples from the entire data set and replicating the estimation process. Standard deviations and significances were obtained using this bootstrap method of the corresponding sampling distributions.
Results
Rabbit Animal Model
Histology and histomorphometry
Animals survived the procedure without any complication related to EP. Figure 1A shows the tetracycline labeling in epiphyseal bone 7 days after electrode insertion in the control femur, and new bone apposition was evident in the whole trabecular bone around and within the electrodes. Histology showed normal osteoblasts lining trabeculae and osteocytes entrapped within bone lacunae (Figure 1B).

Control samples at 7 days after surgery (electrodes inserted with no electric field applied). A, Fluorescence imaging of new bone apposition showing a tetracycline labeling maintained also in proximity of the electrode surface: surgical trauma does not affect the new bone deposit (unstained section, ×2 magnification). B, Histological image of normal osteoblasts lining trabeculae and osteocytes within lacunae (toluidine blue and fast green staining, ×10 magnification). Asterisk indicates the area occupied by the electrode.
The electrode position is well visible and allows the identification of the area subjected to IRE, and 120 pulses prevented tetracycline labeling among the electrodes (Figure 2A). Histology shows the ablation of osteoblasts from the trabeculae, and the osteocyte lacunae are occupied by pyknotic nuclei (Figure 2B). Metaphyseal cartilage was also affected by EP showing signs of alteration, cellular enlargement, and pyknotic nuclear damage (Figure 3A and B).

Electroporated samples at 7 days after surgery (irreversible electroporation [IRE] protocol: 1750 V/cm and 120 pulses). A, Ablation of fluorescence within the electrodes (unstained section, ×1.25 magnification). B, Histological image of nude trabeculae without osteoblasts or pyknotic osteocytes (toluidine blue and fast green staining, ×10 magnification). Asterisk indicates the area occupied by the electrode.

Electroporated samples 7 days after surgery (1750 V/cm at 120 pulses). A, Histological image of the area of mataphyseal cartilage involved by ablation showing signs of cell damage (hematoxilin–eosin [H&E] staining, ×2 magnification). B, Altered nuclear appearance at higher magnification (alcian blue staining, ×50 magnification). Asterisks indicate the area occupied by the electrode.
In animals killed at day 30, tetracycline labeling showed the preexisting bone stock with an important new bone deposition among the electrodes. No osteogenetic activity was present in the proximity of the electrode surface (Figure 4A) but was appreciable in the area inside the electrodes, moving from the peripheral trabecula toward the central area defined by the electrodes. Furthermore, histological analysis confirmed a large number of mesenchymal cells arising from bone marrow, driving osteogenetic activity and endochondral ossification (Figure 4B) and suggesting a significant remodeling activity. Osteocytes in the lacunae of old trabeculae appeared apoptotic (Figure 4C). Cartilage did not stain, and no repair activity was observed (Figure 4D).

Electroporated samples 30 days after surgery (1750 V/cm at 120 pulses). A, Fluorescence image of new bone deposit among the electrodes but not in proximity of the electrode surface where irreversible electroporation occurred (unstained section, ×2.5 magnification). B, Histological image highlighting the presence of mesenchymal stromal cells arising from bone marrow compartment, presence of osteoblasts lining cells, and endochondral ossification (toluidine blue and fast green staining, ×20 magnification). C, Immunohistochemical determination of osteocyte apoptosis in old trabeculae (Bcl-2 antibody, ×10 magnification). D, Histological image of the degenerated metaphyseal cartilage (toluidine blue and fast green staining, ×10 magnification).
Histomorphometric data shown in Figure 5 revealed that at 7 days, the bone tissue femurs had TbTh values not affected by IRE protocol with no statistically significant difference in comparison with controls. At 30 days in comparison with the 7 days, the results revealed a significant decrease in TbTh values of IRE samples (P = .019). Moreover, the bone tissue of IRE-treated samples showed significantly thinner trabeculae than those of controls (P = .005). No ablated bone area was present in the control samples either at 7 or at 30 days, thus confirming that the surgical procedure did not affect cell viability. Irreversible electroporation induced a peak bone ablation area at 7 days (2.6 ± 1.2 mm2). At 30 days, the extent of the ablated area was significantly reduced by up about to one-fourth of the initial extent (0.8 ± 0.4 mm2, P = .0219) consistent with a repair process and new bone apposition.
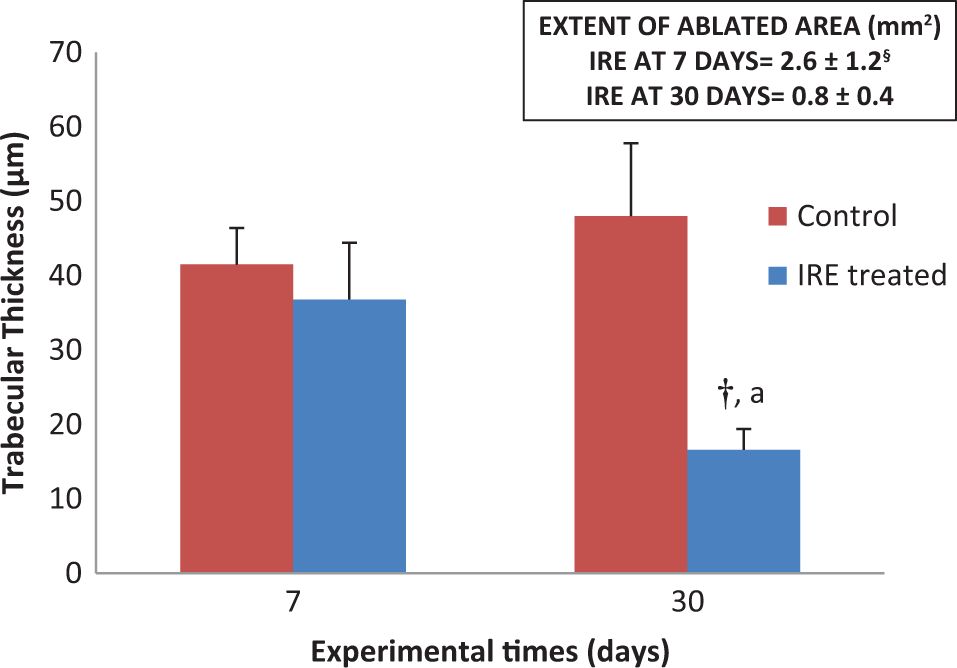
Histomorphometric 2-dimensional (2D) measurements of trabecular thickness in the irreversible electroporation (IRE)-treated bone and controls at 7 and 30 days. In the insert, measurements of ablated area. General linear model (GLM) with adjusted Sidak multiple comparison test: †30 days: IRE treated versus control, P = .005; aIRE treated: 30 days versus 7 days, P = .019. Student t test: §P = .0219.
Sciatic nerve histology
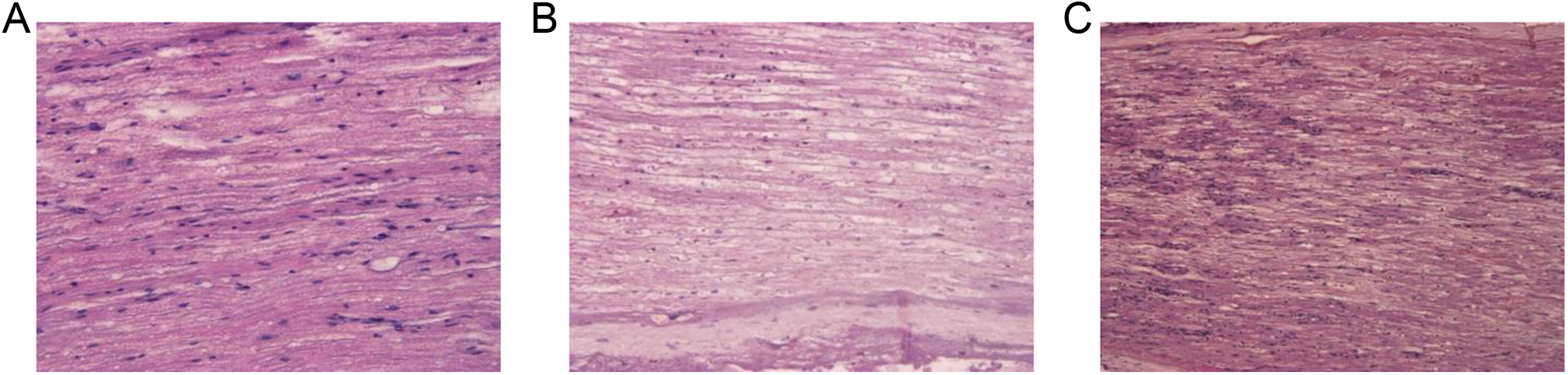
Figures 6A and 7A show the aspect of the control nerve: axons was regularly myelinated and the connective tissue around them was normally arranged. On the contrary, sciatic nerve 7 days after REP showed signs of thinning and delamination of the myelin sheath with a slightly edematous connective tissue in the area where plates were positioned (Figure 6B). The adjacent areas (Figure 6C) appeared normal, as in control sections. Thirty days after treatment, the REP area (Figure 6D) showed residues of altered myelin sheaths. Adjacent areas were similar in appearance to the contralateral nerve, especially in the distal part.

Control and electroporated nerves (reversible electroporation [REP] protocol: 1000 V/cm and 2 series of 8 pulses; ×40 magnification, toluidine blue staining). A, Control nerve showing regularly myelinated axons. B, Electroporated area 7 days after treatment. Notice the thinning and delamination of the myelin sheath and the edematous connective tissue. C, Proximal section of a nerve removed 7 days after electroporation. D, Electroporated area 30 days after treatment. Remains of altered myelinated sheaths are present.
Control and electroporated nerves (reversible electroporation [REP] protocol: 1000 V/cm and 2 series of 8 pulses; ×10 magnification, longitudinal sections, hematoxylin–eosin staining). A, Control nerve. B, Electroporated section at 7 days after treatment. Notice the edematous connective tissue. C, Proximal section of a nerve removed 7 days after electroporation
Functional deficit involved the motor component of the sciatic nerve with hypotonia and hypomotility and was detected in 2 animal legs within the first 7 days after REP. Animals completely recovered motor abilities before scheduled killing at 30 days.
Sheep Animal Model
Clinical follow-up
Figure 8A to D shows the electric field distribution graph (Figure 8A) and images of the surgical procedure steps that allowed proper electrode positioning in L3 and L4 by specifically prepared masks (Figure 8B and C) and the radiological aspect of the explanted vertebral body, in which electrodes were cut and left inserted as markers for subsequent analyses.

Irreversible electroporation (IRE) protocol applied to sheep vertebral body of L3 and L4 (1750 V/cm, 120 pulses). A, The electric field distribution calculated using computer software COMSOL Multiphysics. B and C, The steps of the surgical procedure: mask positioning and electrode insertion. D, Radiological image of the explanted vertebral body in which the electrodes were cut after IRE and left inserted as markers for subsequent analysis.
The animals survived the procedure at the spine without any acute complication related to IRE or surgery. During the 30-day follow-up period, the animal submitted to IRE and scheduled for the evaluation of possible clinical complications showed no changes in physiological habits and no alterations in gait, peripheral sensibility to hindquarters, or pain.
Bone histology
Vertebral body
Seven days after electrode insertion, tetracycline labeling in the L2 vertebral body showed the apposition of newly formed bone in the whole trabecular bone around the electrodes, thus highlighting that the insertion procedure does not influence bone tissue viability (Figure 9A). In the L3 and L4 vertebral bodies, tetracycline labeling among the electrodes was absent (Figure 9B), following IRE. Figure 10A shows the histological analysis of the L2 vertebral body with preexisting trabeculae among the electrodes completely covered by osteoblasts with evident evenly spread osteocytes, oriented with their longest axis in the direction of the lamellae contained in the bone lacunae. However, an absence of osteoblast and osteocyte nuclei within bone lacunae was observed in the L3 and L4 vertebrae following IRE (Figure 10B).

A, Doughty fluorescence of bone tissue around the electrodes in the untreated vertebra (L2) 7 days after electrode insertion (unstained section, ×20 magnification). B, Absence of fluorescence in the L3 vertebral body 7 days after electroporation (irreversible electroporation [IRE] protocol: 1750 V/cm and 120 pulses; unstained section, ×20 magnification).

Histological sections of the vertebrae (toluidine blue, fast green staining). A, Untreated L2 vertebral body: toluidine blue staining of the trabeculae showing osteoblasts aligned on the surface with trabecular lamellar bone (×20 magnification). In the insert, evident evenly spread osteocytes, oriented with the longest axis in the direction of the lamellae contained in the bone lacunae (resolution ×40). B, Irreversible electroporation (IRE)-treated L4 vertebral body: absence of osteoblasts on the trabeculae surface and pyknotic osteocytes (×20 magnification). B indicates bone; OB, osteoblast; IT, inflammatory tissue; ET, hematopoietic tissue; AT, adipose tissue; OS, osteocyte.
Histomorphometry
The histomorphometric evaluation on the L2 vertebral body showed that the number of osteoblasts, in each quadrant of the superimposed grid, around the electrodes was 450 ± 50, whereas in the L3 and L4 vertebral bodies, the number of osteoblasts was reduced to 25 ± 5 and 15 ± 5, respectively, confirming the ablation of bone cells. Osteoclasts were not detected in any of the examined samples.
Microtomography
To study the morphologic changes of the vertebral body, we also generated a 2-dimensional (2D) image obtained using microcomputed tomography (CT). At 7 days, 2D micro-CT images showed intact structure of cortical and trabecular bone in both untreated and EP-treated vertebral bodies (Figure 11A and B).

Two-dimensional (2D) micro-computed tomographic (CT) images. No structural differences are observable in cortical and trabecular bone of (A) untreated and (B) EP-treated vertebral bodies.
Histology of surrounding tissues
Vertebral pedicle
Histological analysis of vertebral pedicles showed the presence of inflammatory tissue, rich in granulocytes and macrophages, in both the untreated (Figure 12A) and the IRE-treated spinal pedicles (Figure 12B and C) probably due to the insertion of electrodes through the tissue. Figure 12A also shows the presence of bone trabeculae in the untreated group completely covered by numerous osteoblasts, mainly cylindrical in shape; in addition, there are numerous osteocytes regularly distributed in lenticular cavities (bone lacunae). In the IRE group, bone trabeculae without osteoblasts and with numerous empty bone lacunae were present (Figure 12B and C).

Histological sections of vertebral pedicle (×20 magnification; hematoxylin–eosin staining). A, Untreated L2 vertebral pedicle: bone trabeculae completely covered by numerous osteoblasts and with osteocytes regularly distributed in bone lacunae. B, Irreversible electroporation (IRE)-treated L3 vertebral pedicle. C, Irreversible electroporation-treated L4 vertebral pedicle: bone trabeculae without osteoblasts and with numerous empty bone lacunae. B indicates bone; OB, osteoblast; IT, inflammatory tissue; OS, osteocyte.
Spinal cord
Figure 13A shows the untreated spinal cord, with recognizable white matter, characterized by a myelin sheath of cellular processes and numerous blood vessels, and gray matter, characterized by cells, fibers, and some capillaries. The spinal cord, adjacent to the IRE-treated bone, has the same morphology and structure as the untreated spinal cord, but the axons were bound by a myelin sheath with increased vacuolation (Figure 13B).

Histological sections of spinal cord (×10 magnification; hematoxylin–eosin staining). A, Untreated spinal cord. B, electroporation (EP)-treated spinal cord: myelin sheath with increased vacuolation.
Spinal nerve
Compared to untreated spinal nerves, the fascicular architecture of nerve adjacent to the IRE-treated bone showed signs of disorganization with an enlargement of the spaces between groups of nerve fibers, due to interstitial edema. However, axons were closely packed and the myelin sheath structures were similar to those of controls and normal structures (Figure 14A and B). Regarding the perineurium lining nerve fibre bundles, there were no morphological or structural differences between IRE-treated and untreated spinal nerve.

Histological sections of spinal nerve (×40 magnification; hematoxylin–eosin staining. A, Untreated spinal nerve. B, Electroporation (EP)-treated spinal nerve: disorganization of nerve fibers. A large number of myelin sheaths have lost their granular structure. P indicates perineurium; NF, nerve fibers.
Discussion
Electroporation is an ablative modality based on electric field delivery to tissues that alters, at cellular level, the cell membrane potential, causing the formation of pores. Depending on the electric field amplitude and the number and duration of delivered pulses, EP can be reversible (REP), leading to cell survival, or irreversible (IRE), meaning that cells can’t maintain the intracellular homeostasis and thus undergo apoptosis.
Irreversible electroporation has been cleared by the FDA as a nonthermal soft tissue ablation modality, however, its use in cancer treatment is limited to clinical trials. 23 -25 Reversible electroporation is applied to the tumor nodules in order to locally enhance intracellular diffusion of low permeant drugs (ECT), thus leading to local tumor control. 7
Nowadays, ECT is used in clinical trials as palliative treatment of cutaneous and subcutaneous tumor nodules of different histological types with response rate of 80% and long-lasting complete responses of 70% or when other therapeutic strategies have failed or are insufficient. Patients with metastases to bone can benefit from local treatments, which include surgery, radiation therapy, thermal ablation and selective arterial embolization, and thermal ablation. The choice of local treatment is based on number, location, and size of lesions, patient’s general performance status, age, comorbidities, and symptoms caused by the metastasis. Surgery is indicated to prevent or treat pathologic fractures, to release spinal cord compression, or to resect small or isolated lesions. 26 Treatments are aimed at pain relief, preservation of function, and quick mobilization of the patient. 27 Radiation therapy in fractionated doses is indicated in bone reconstruction 27 and metastases: advantages over surgery are no need for anesthesia, lower morbidity, good immediate tolerance, and ambulatory treatment, but when used in appendicular skeleton, it may increase bone fragility and the risk of fracture. Radiofrequency thermal ablation is associated with surgery or cement to protect the treated bone from fracture. Embolization is used for large vascularized metastases. Systemic chemotherapy is used for the treatment of multiple metastases to prevent bone reabsorption. 28 All ablative strategies share the major problem consisting in ablating tumor and margins precisely, which often is not possible owing to the presence of sensitive structures. Electroporation, being a nonthermal process, maintains the extracellular matrix structure. 18,29 Thus, the aim of this study was to evaluate the in vivo feasibility and safety of EP in bone tissue and in the neighboring clinically relevant structures by using small and large animal models, considering the importance of determining the effect of electric pulses on neuronal and other sensitive structures, such as nerves or spinal cord, while avoiding serious consequences.
In the rabbit sciatic nerve, REP highlighted a thinning and delamination of the myelin sheath 7 days after the procedure, whereas the adjacent, both distally and proximally, areas did not show any structural or morphological damage. At a longer experimental time, 30 days after treatment, the EP area showed histological residues of altered myelin sheaths, but the functional limb insufficiency, observed early after REP, resulted transient and recovered within 30 days. Functional damage without nerve degeneration might indicate a reversible neuropraxia. 30 Moreover, the adopted REP pulse protocol delivered a number of pulses at least twice that were used in clinical practice, 16 versus 8 pulses, to overstress the tissues.
In the rabbit femoral condyles, the ablation induced by IRE was complete and detectable 7 days after surgery, and histological analyses showed the detachment of osteoblasts from trabeculae, the transitory inhibition of bone apposition, pyknotic osteocytes, and empty lacunae, as well as degenerative changes in the growth cartilage. On the contrary, electrodes insertion without application of electric field did not affect bone tissue viability and mineral apposition. Thirty days after IRE treatments, animals demonstrated an intense osteogenetic activity in the area ablated among the electrodes, as determined by tetracycline labeling and new bone apposition characterized by new dense and thin trabeculae and the presence of vessels.
The reparative activity was not present immediately around the electrodes (at a distance of about 120 μm from the electrodes), and actually this area experienced the highest electric field values.
Regarding vertebral body of sheep, 7 days after IRE, our results revealed no tetracycline fluorescence and complete ablation of osteoblasts from the trabeculae. Additionally, the heterogeneous structure of the bone in the vertebral body and in the vertebral peduncle did not prevent the complete EP of cells in the space between the electrodes, whereas the structural integrity of bone and extracellular matrix is maintained. Finally, transient interstitial edema and vacuolization at early experimental times in sheep spinal nerves and spinal cord were noted, respectively, but there were no permanent functional deficits attributable to the IRE, thus showing the feasibility of EP in the spine.
Promising results were obtained from this study, however, some limitations of the study should be considered. First, the small animal sample size, particularly for the sheep model. Second, all morphometric parameters were measured by 2D image analysis and in a structure as complex as bone, whereas 3-dimensional analysis would have given a more complete vision of the organization and structure of the sample.
The choice of the rabbit animal model strengthened and extended the previous work on trabecular bone of rabbit femurs 18 by analyzing in detail the effects of IRE on bone cells and metaphyseal growth cartilage by histology and static and dynamic histomorphometry. Then, to confirm results obtained in rabbits, we selected to apply IRE to the spine of sheep, a widely used model in the literature for studies on the spine and therefore well characterized with regard to surgical access, dimensions, and anatomy. 31 This model ensures at the same time adequate anatomical dimensions suitable for electrode positioning and the presence of near clinically relevant structures.
In our study, we decided to assess the effects of EP on the healthy tissues of 2 distinct animal models, to gain safety information from 2 sequential models rather than from only 1. Moreover, this methodology, by adopting models of increasing complexities, will increase the effectiveness and predictability of the preclinical data.
To date, to our knowledge, the effect and safety of EP protocols were investigated in few studies. Rabussay et al 32 evaluated ECT on major arteries and nerves in a preclinical model. Results revealed that healthy tissue close to major arteries and nerves, as well as the vessels and nerves themselves, were not significantly affected by treating the tissue surrounding these structures. Moreover, our previous study evaluated the feasibility and safety of REP applied to the pancreas and ECT in vitro on highly drug-resistant pancreatic cancer cells, concluding that REP is a safe procedure because it does not affect normal parenchyma and it has a potentiating effect on cytotoxicity of bleomycin in pancreatic tumor cells. 33 Regarding IRE, efficacy and safety were investigated with promising results on tumors of liver, pancreas, pelvis, kidney and lung 34,35 and on vessel patency and healthy tissues. 36,37 However, no data are available on the safety of different EP pulse protocols on spinal nerves and clinically relevant structures lining spine.
In summary, our preclinical investigation shows that EP can be feasible and safely applied on rabbit healthy bone tissue and sciatic nerve, thus indicating that these structures are well preserved after EP. Most importantly and of greater interest in the clinical setting, no functional or permanent deficits at the level of spinal cord and spinal nerve structures were observed when EP was performed in the L3 to L4 vertebrae of sheep. This study provides attractive preclinical results, increasing the understanding of the effects of EP on healthy tissues, and showing the safety of 2 different pulse protocols on very sensitive structures, such as spinal cord and nerves.
These results, together with further evidence of ECT safety and efficacy in in vivo models of tumors, should support translation of this methodology into clinical practice, providing the rationale for the treatment of tumor nodules also in the presence of critical anatomical structures, such as in bone metastases and in head and neck cancers.
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank the late Dr Marco Alberghini for his unequalled help and precious suggestions in histology interpretation of samples and Dr Annapaola Parrilli for microtomographic acquisition and reconstruction of samples.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mattia Ronchetti is a full-time employee of IGEA SpA, manufacturer of Cliniporator for the in vivo study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by the Operational Programme ERDF 2007-2013 in the region Emilia-Romagna: Activity The 1.1 “Creation of technology centers for Industrial research and technological transfer” and by Project. n. A20/1837/00/X06, ‘Studio e sviluppo di un’innovativa Piattaforma Tecnologica per Elettropoazione (PTE) per la terapia dei tumori profondi’ of Ministero dello Sviluppo Economico.
