Abstract
Activation of the phosphoinositide 3-kinase (PI3K)/Akt signaling pathway is a novel poor prognostic indicator of neuroblastoma (NB), and the positive effects of chemotherapy on NB have been confirmed. In this study, we investigated the effect of small molecule PI3K inhibitor PI103 on chemosensitivity. The PI3K inhibitor cooperates with doxorubicin to synergistically induce apoptosis and to reduce tumor growth of NB in in vitro and in vivo models. Human NB cells, SH-SY5Y and SK-N-BE(2), were treated with PI103 combined doxorubicin-enhanced Bid cleavage, activated Bax, and caspase 3. Activation of caspase 3 was also observed in xenografts of NB in nude mice upon combination of doxorubicin with the specific PI3K inhibitor PI103. Cell viability was assessed with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assays. Both PI103 and doxorubicin inhibited growth of NB in vitro and PI103 induced a G1 arrest of NB cells. PI103 combined doxorubicin significantly inhibits the growth of established NB tumors, induced apoptosis of tumor cells, and improved the survival of mice in vivo. Taken together, our findings suggest that PI3K inhibition seems to be a promising option to sensitize tumor cells for chemotherapy in NB, which may be effective in the treatment of NBs.
Introduction
Neuroblastoma (NB) is the most common solid extracranial tumor in early childhood, and the neoplasm is most frequently diagnosed during infancy. 1 The hallmark of NB is heterogeneity, varying from spontaneous regression or differentiation to relentless progression. 2,3 However, the majority of NBs are malignant with metastatic spread that is difficult to cure with current treatment modalities. The advanced stages of this malignancy are difficult to treat, and despite intense therapeutic intervention, the cure rates for high-grade NB have only improved marginally over the recent years. 4 The prognosis of children older than 1 year with stage 4 disease is still very poor with long-term survival rates of only 40%, despite intensive and multimodal therapies. 5 Therefore, novel strategies are required for the treatment of advanced stage NB.
The phosphoinositide 3-kinase (PI3K)/Akt/mammalian target of rapamycin (mTOR) pathway plays an important role in mediating prosurvival signals. Abnormal activation of the PI3K/Akt signaling pathway is a common event in human cancers and has been validated through epidemiological and experimental studies as being essential for several human tumors, including NB. 6 -8 Once activated, Akt controls various biological responses including cell survival, cell cycle progression, cell metabolism, migration, angiogenesis, and resistance to drug-induced apoptosis, indicating that this pathway represents a clinically relevant target for therapeutic intervention. 6 Much effort has been put into developing agents that target this pathway. This has resulted in the development of several small-molecule inhibitors targeting key molecules in the PI3K/Akt pathway. 9 PI103 is a dual PI3K/mTOR inhibitor, and it has been shown to block proliferation 10,11 and enhance both radiosensitivity 12 and chemosensitivity 13 in glioma as well as priming NB cells for chemotherapy. 14 Monotherapy consisting of PI3K/mTOR inhibition alone is ineffective in NB that does not harbor MYCN amplification. 15
Tumors with favorable biologic features (very low risk, low risk, and intermediate risk) are less aggressive and nonmetastatic. These can be treated with surgical resection and shorter courses of chemotherapy. 16 The frontline chemotherapeutic drugs (eg, etoposide, doxorubicin [Dox], and cisplatin) for treating NB are all DNA-damaging agents, which induce DNA damage to exert their antitumor activities. 17 However, most high-risk NB shows an initial response to therapy but ultimately relapse, and it is reasonable to conclude that acquired drug resistance or selection of therapy-resistant cells prevents current treatment from becoming a cure. Inhibition of PI3K/Akt-mediated signaling strongly amplifies cell death induced by a wide range of chemotherapeutics. 14 Given the fact that elimination of tumor cells requires induction of cell death pathways, both PI3K/Akt inhibitors and chemotherapeutic drugs can induce the apoptosis of tumor cells. In this study, we evaluated the combined effects of PI3K/Akt inhibitor PI103 and chemotherapeutic drug Dox on the apoptosis of tumor in vitro and in vivo.
Materials and Methods
Cell Culture
SH-SY5Y and SK-N-BE(2) cell lines were cultured in Roswell Park Memorial Institute (RPMI) 1640 (Gibco Life Technologies [Sundbyberg, Sweden]). Media were supplemented with 10% fetal bovine serum (Hyclone Labs [Logan, UT]) and 1% penicillin/streptomycin. All cell lines were obtained from American Type Culture Collection (ATCC).
Antibodies and Reagents
Rabbit anti-β-actin (#4967), rabbit anti-Bax (#2772), rabbit anti-Bid (#2003), and rabbit anticaspase 3 (#9662) antibodies were purchased from Cell Signaling Technology (Beverly, MA). PI103 and Dox were purchased from Sigma-Aldrich (St. Louis, MO). PI103 and Dox were dissolved in dimethyl sulfoxide (Sigma-Aldrich). Stock solutions were further diluted in RPMI 1640 to desired in vitro concentrations. For in vivo studies, the stock was diluted in 0.5% methylcellulose (w/v; Sigma-Aldrich) and 0.1% Tween 80 (v/v; Sigma-Aldrich) in sterile water.
Western Blotting Assay
Soluble proteins were extracted from the SH-SY5Y and SK-N-BE cells untreated or treated with PI103 or/and DOX for 24 or 48 hours and subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis. Separated proteins were electrophoretically transferred onto polyvinylidene difluoride membranes (Thermo Fisher Inc [Braunschweig, Germany]) and immunoblotted with anti-β-actin, anti-Bax, anti-Bid, and anticaspase 3. Immunoreactive proteins were visualized using the Odyssey Infrared Imaging System (LiCor, Lincoln, NE) as described by the manufacturer.
Determination of Bax Activation
Bax activation was determined by immunoprecipitation. Briefly, cells were lysed in CHAPS lysis buffer (10 mmol/L HEPES [2-[4-(2-Hydroxyethyl)-1-piperazinyl]ethanesulfonicacid], pH 7.4; 150 mmol/L NaCl; 1% CHAPS [3-[(3-Cholamidopropyl)dimethyl-amino]-1-propanesulfonate]). Protein (1 mg) was incubated with 2 μg mouse anti-Bax antibody (6A7; Sigma) overnight at 4°C followed by addition of 10 μL Dynabeads Pan Mouse immunoglobulin G (Dako [Glostrup, Denmark]), incubated for 2 hours at 4°C, washed with CHAPS lysis buffer, and were analyzed by Western blotting using rabbit anti-Bax (Cell Signaling) antibody.
Viability Assay and Colony Formation
Cell viability was assessed by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay according to the manufacturer’s instructions (Roche Diagnostics [Mannheim, Germany]). For colony assay, cells were plated in 0.3% agarose/RPMI 1640 media containing the indicated drugs with indicated concentration on top of a 0.5% agarose/RPMI 1640 layer. After 14 days, colonies were stained with MTT and counted.
Flow Cytometric Analysis of Cell Cycle
SH-SY5Y and SK-N-BE cells treated with PI103 or/and Dox for 48 hours. Cells were harvested and fixed with ice-cold 70% (v/v) ethanol for 24 hours. After centrifugation at 200g for 5 minutes, the cell pellets were washed and stained with propidium iodide (50 μg/mL), Triton X-100 (0.1%, v/v), and DNase-free RNase (1 μg/mL). DNA contents were determined by flow cytometry (FACS Calibur [Becton Dickinson, San Jose, CA]), and cell cycles were performed by FlowJo Software (Tree Star Inc., Ashland, OR, USA) according to the manufacturer’s instructions.
In Vivo Therapeutic Experiments
Female Naval Medical Research Institute (NMRI) nu/nu mice were xenografted at 6 weeks. The mice were anesthetized and 0.1 mL SK-N-BE(2) cells (2 × 107 cells) were implanted subcutaneously in the right hind leg of the animal. At a tumor volume of ≥200 mm3, the animals were randomized to receive PI103 (10 mg/kg intraperitoneal [ip], n = 10) daily and/or Dox (1.2 mg/kg ip, n = 10) every other day for 10 consecutive days or no treatment (controls, n = 10). Tumors were measured daily with a digital caliper, and tumor volume was evaluated as tumor volume index (TVI), calculated by the starting volume divided by the tumor volume at each day of treatment. The animals were weighed daily and assessed for signs of toxicity. At day 10, the animals were killed, the tumors were excised and tumor weight recorded. Tumor materials was fixed in 4% paraformaldehyde and frozen in liquid nitrogen and stored at 4°C or −80°C, respectively.
Statistical Analysis
Survival was plotted as Kaplan-Meier survival curves, and the statistical significance was determined by the log-rank test. The statistical significance of tumor size was determined by the Student t test for 2 groups or by the Kruskal-Wallis test followed by Dunn posttest for more than 2 groups. Differences for which P was <.05 were considered statistically significant. All statistical computations were performed using GraphPad Prism 5 (GraphPad Software, Inc., CA).
Results
Inhibitory Effects of Dox or PI103 on NB Cells Viability
We first examined the inhibition effects of Dox or/and PI103 on SK-N-BE(2) cells and SH-SY5Y cells. The growth inhibitory effects of Dox or PI103 were investigated in vitro in NB cells over 24, 48, or 72 hours. The cell lines were chosen to comprise both MYCN-amplified [SK-N-BE(2)] and non-MYCN-amplified (SH-SY5Y) NB cells. Both Dox and PI103 induced time- and concentration-dependent inhibition on NB cell growth (Figure 1A–D, respectively). Neuroblastoma cells containing MYCN amplification, SK-N-BE(2), was significantly more sensitive to treatment with Dox or PI103 compared with no MYCN amplification cell line SH-SY5Y, 48 hours drug incubation: SK-N-BE(2) versus SH-SY5Y Dox: P < .01; PI103: P < .01. To investigate the combined effect of Dox and PI103 on long-term clonogenic survival, we performed colony assays. Both Dox and PI103 alone reduced the colony formation of NB cells. Importantly, addition of PI103 significantly enhanced Dox-mediated reduction in colony formation compared with cells treated with Dox alone (Figure 1E and F). These data show that inhibition of PI3K by PI103 sensitizes NB cells to Dox-induced inhibition.
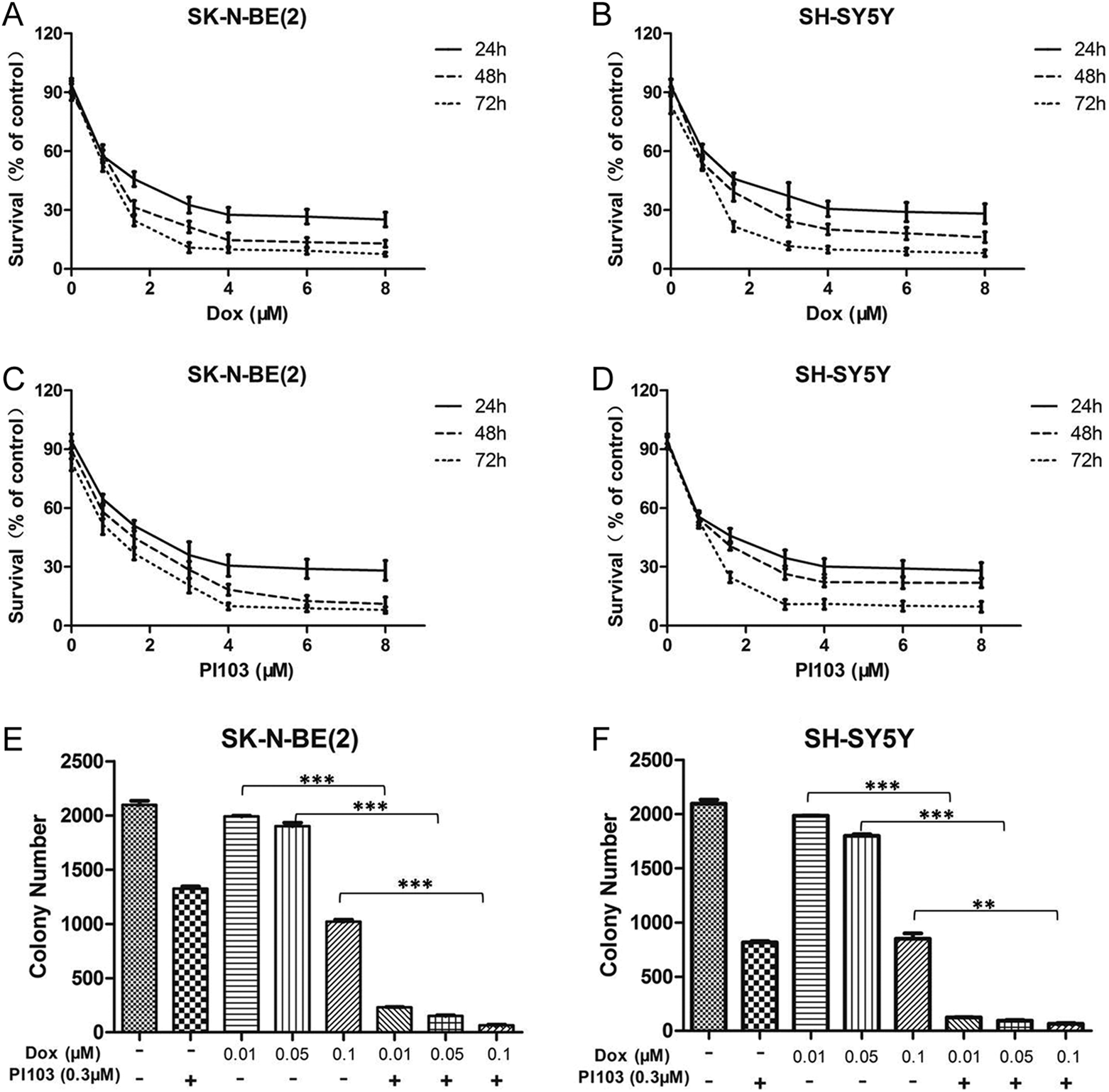
Dox and PI103 inhibit neuroblastoma growth in vitro. The MYCN-amplified, SK-N-BE(2), and nonamplified (SH-SY5Y) cell lines were incubated with increasing concentrations of Dox (A and B) or PI103 (C and D), and cell growth was measured using MTT assays after 24, 48, and 72 hours. The mean survival of 6 replicas is shown and the experiments were repeated twice. (E and F) The SK-N-BE(2) and SH-SY5Y cells were plated in 0.3% agarose/RPMI 1640 media containing the indicated drugs with indicated concentration on top of a 0.5% agarose/RPMI 1640 layer. After 14 days, colonies were stained with MTT and counted. These experiments were performed in 6 duplicate and reported as means with standard deviations (SDs). *P < .05, **P < .01, and ***P < .001 (Student t test, 2 tailed). Dox indicates doxorubicin; MTT, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; RPMI, Roswell Park Memorial Institute.
Dox or PI103 Exert Different Growth Inhibitory Effects on NB Cells
To study the growth inhibitory mechanisms of these small-molecule inhibitors on NB, we evaluated the effect of Dox and PI103 on cell cycle progression. Cell cycle analysis revealed a transient G1 cell cycle arrest after 24 hours treatment with Dox or/and PI103 in SH-SY5Y cell line (Table 1), but we observed a difference in SK-N-BE(2) cell line, treatment of SK-N-BE(2) cell line with PI103 alone or combined with Dox for 24 hours resulted in a G1 cell cycle arrest, while treatment of SK-N-BE(2) cell line with Dox alone for 24 hours resulted in a G2 cell cycle arrest(Table 1).
Cell Cycle Analysis of Neuroblastoma Cells Treated With PI103 or/and Dox.

Abbreviation: Dox, doxorubicin.
Sensitization of NB Cells to Dox-Induced Apoptosis by the PI3K Inhibitor PI103
To examine the effects of drugs on NB cell apoptosis, the effect of Dox or/and PI103 on apoptosis was investigated in the NB cell line SH-SY5Y. As shown in Figure 2A, treatment of the cells with 1 µmol/L Dox resulted in a modest apoptosis increase by 8%, whereas treatment of cells with PI103 (1 µmol/L) did not induce obvious apoptosis. However, Dox-induced apoptosis was significantly enhanced by PI103. The enhancement by PI103 was extremely significant (P < .001). In addition, PI103 was able to enhance Dox-mediated induction of caspase 3, Bid, and Bax activation (Figure 2B–D). Together, these data demonstrate that the combined effect of PI103 plus Dox involves caspase 3, Bid, and Bax activation. PI103 synergistically enhanced the effect of Dox-induced apoptosis on NB cells in vitro.
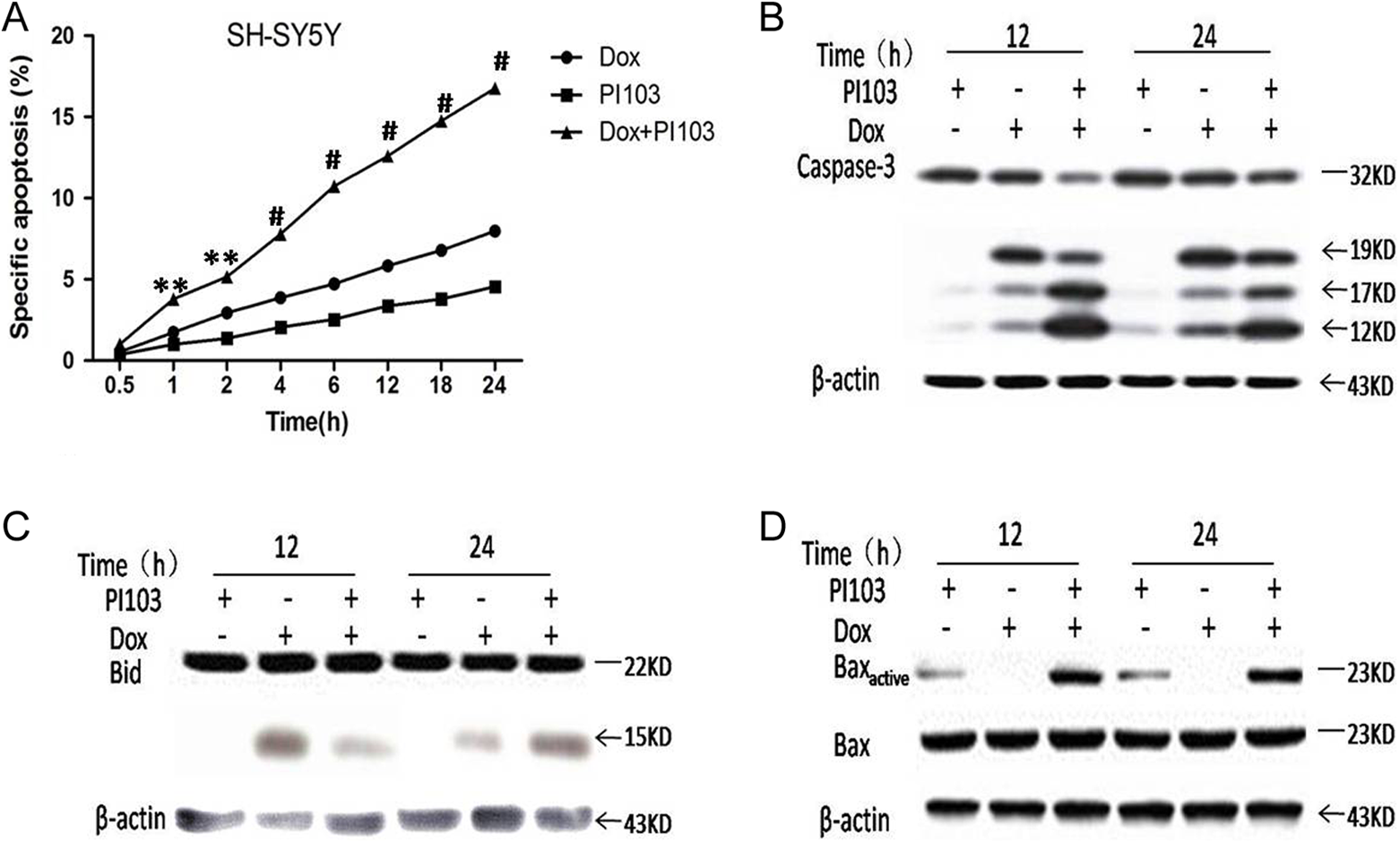
The PI103 sensitizes neuroblastoma cells to doxorubicin-induced apoptosis. A, The SH-SY5Y cells were treated with doxorubicin or/and PI103 for indicated times. Apoptosis was determined by fluorescence-activated cell sorting (FACS) analysis of DNA fragmentation of propidium iodide-stained nuclei and percentage of apoptosis is shown. Data represent mean with standard error of the mean of 3 independent experiments carried out in triplicate. For statistical analysis, Student t test was carried out comparing treatment with PI103 versus treatment without PI103. **P < .01 and #P < .001. B and C, The SH-SY5Y cells were treated with doxorubicin or/and PI103 for 12 or 24 hours. Protein extracts were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and immunoblotted with antibodies against caspase 3, Bid to detect the apoptotic cells. D, The Bax activation was analyzed by immunoprecipitation of protein lysates using the active conformation-specific anti-Bax antibody 6A7 and Bax expression was analyzed by Western blotting.
PI3K/mTOR Inhibitor and Dox Significantly Inhibit the Growth of Established MYCN-Amplified NB Tumors in Nude Mice
To investigate the therapeutic effects of Dox and PI103 in vivo, nude mice carrying established SK-N-BE(2) xenografts were randomized to treatment with Dox (1.2 mg/kg ip, n = 10) or/and PI103 (10 mg/kg ip, n = 10). Compared with untreated controls (n = 10), treatment with either Dox or PI103 resulted in a statistically significant reduction in the TVI (PI103 vs control: P < .05; Dox vs control: P < .05; Figure 3A), whereas the combination treatment had significantly reduced the tumor growth compared to monotherapy group (PI103 + Dox vs Dox: P < .01; PI103 + Dox vs PI103: P < .001; Figure 3A). Tumor weights were measured at killing, and treatment with Dox or/and PI103 significantly reduced the tumor weights compared to the control group (Dox vs control: P < .01; PI103 vs control: P < .05; Dox + PI103 vs control: P < .001; Figure 3C). The inhibitors also prolonged the survival of tumor-bearing mice, and mice of combination therapy had the longest survival time (PI103 + Dox vs Dox: P < .01; PI103 + Dox vs PI103: P < .001; Figure 3B). In tumors of nude mice, the combination of drugs increased the percentage of cells positive for active caspase 3 when compared to application of either Dox or PI103 alone (Figure 3D). These results suggest a significant contribution of PI103 to the inhibition effect of Dox on NB xenografts growth in vivo.

The Dox and PI103 significantly inhibit the growth of established neuroblastoma xenografts in nude mice. The NMRI nude mice engrafted with 2 × 107 SK-N-BE(2) cells subcutaneous (sc) were randomized at tumor take (200 mm3) to receive either PI103 (10 mg/kg ip, n = 10) daily and/or Dox (1.2 mg/kg ip, n = 10) every other day for 10 consecutive days or no treatment (controls, n = 10). Treatment effects are presented as tumor volume index (TVI; A). Treatment also reduced the tumor weight (C) and prolonged the survival time (B) of tumor-bearing mice. The tumor weight value is given as the mean with standard deviations (SDs). (D) Caspase 3 activity in tumors of nude mice isolated after 10 days of treatment with Dox or/and PI103 or solvent was detected by Western blotting. Dox indicates doxorubicin; ip, intraperitoneal.
Discussion
Neuroblastoma is the most common malignant extracranial solid tumor presenting in childhood and accounts for 8% to 10% of all childhood tumors. 18 Doxorubicin is a potent anticancer drug employed in the therapy of several solid tumors including NB. Its mode of action is not completely understood but interferes with apoptotic process and mediates the release of cell-damaging radicals. 19 Despite being one of the drugs of choice for NB treatment, the effect of Dox alone is limited by insufficient drug delivery to tumor and has much severe adverse effects. 16,20 Concomitant targeting of different cellular pathways is considered as a promising strategy in tumor treatment. In the present study, we examined the effects of PI3K/mTOR inhibitor PI103 to enhance the chemosensitivity of NB cells.
In this study, we provide convincing evidence that synthetic inhibition of PI3K activity greatly enhances Dox-induced cytotoxicity in NB cells both in vitro and in vivo. Our MTT assays in NB cell lines demonstrated highly synergistic antitumor activity when treated with Dox and PI103. Cell cycle and apoptosis analyses by flow cytometry and Western blotting revealed enhanced cell death accompanied by cleavage of caspase 3, Bax, and Bid when Dox was combined with PI103, demonstrating that PI103 enhanced the standard chemotherapy drug-induced apoptosis. Doxorubicin is frequently classified as a cell cycle-dependent topoisomerase IIα poison, 21,22 and our data suggest that Dox mainly causes G1 cell cycle arrest on SH-SY5Y cell line, while most cells show a G2 cell cycle arrest on SK-N-BE(2) cell line. This is in line with previous findings indicating that Dox treatment reduced the fraction of MYCN-amplified cells in S-phase and enriched the fraction of G2/M phase. 23 Based on the results in Table 1, it appears that PI103 is not able to further increase Dox-induced G2 cell cycle arrest on SK-N-BE(2) cell line. In the combined group, most cells arrested in G1 cell cycle similar to the PI103 group.
The main effects of Dox on tumors are DNA damage and cell apoptosis. The use of PI103 in combination with Dox may have some controversy, as Dox has been shown to be activator of PI3K/Akt signaling in a wide variety of malignancies 24 such as myelogenous leukemia 25 and breast cancer. 26 Interestingly, our results show that PI103 greatly sensitizes NB cells to Dox-induced apoptosis and inhibits the cell growth when administered as cotreatment (Figures 1E and f and 2), suggesting a more complex relationship between chemotherapeutic agent and PI3K inhibitor. We also evaluated the antitumor effects of these drugs in a nude mouse model. Injection of Dox at a dose of 1.2 mg/kg had a similar effect on tumor inhibition and life span prolongation compared to injection of 10 mg/kg PI103 (Figure 3A and B). However, when treated with Dox in combination with PI103, the tumors were significantly inhibited and the survival of mice was also greatly improved (Figure 3A and B). We tested the apoptosis of tumors and found that combination therapy significantly enhanced the caspase 3 cleavage in tumors. These results show that the level of cell apoptosis may be considered as an indicator of the efficacy of combination therapy.
In conclusion, our study demonstrates that PI103 synergistically enhanced the apoptotic effects of chemotherapeutic drug Dox on high-risk NB cells and inhibited the tumor growth in in vivo model. The results reported herein suggest that this treatment regime warrants further preclinical studies for use against NB. Further studies will be needed to investigate how the therapeutic efficacy of Dox can be further enhanced by PI103 and determine the most appropriate combination of these agents for treatment of NB. Nevertheless, combination therapy with Dox and PI103 is a promising novel strategy in the treatment of high-risk NB.
Footnotes
Authors’ Note
Hongshun Xing designed the experiment and analyze the results data and LingLing Xie provided technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
