Abstract
Purpose:
To measure the scattered dose to ovary from radiotherapy for neuroblastoma in female children and to evaluate the relevant risks for radiation-induced ovarian damage.
Material and Methods:
Radiotherapy for child neuroblastoma was simulated on the water phantom. The scattered dose to ovary is measured by ionization chamber on the linear accelerator with 3-dimensional conformal radiation therapy and intensity-modulated radiation therapy treatment producing 6MV and 10MV X-rays. The treatment planning procedure was carried out on a computer system (TPS, Oncentra). Optimization of the number and orientation of beams were performed in order to minimize the ovarian dose.
Results:
For the target dose of 21.6 Gy, the scattered dose to ovary was ranged from 1.3 to 46.8 cGy depending on the treatment method and the energy of the beams. The ovarian dose of intensity-modulated radiation therapy is 1.32 to 1.64 times higher than that of 3-dimensional conformal radiation therapy. The ovarian dose of 6MV beam’s energy is 1.52 to 1.64 times higher than that of 10MV beam’s energy. For the radiotherapy, the scattered dose of ovaries on phantom by ionization chamber was 1.40 to 2.32 times higher than that on TPS calculated.
Conclusion:
The dosimetric data suggest that pediatric radiotherapy is not associated with a risk for permanent damage to the ovaries in female children. Through choosing the beams’ energy and treatment plan’s method, the scattered dose of ovaries can be reduced. The risk for development of hereditary disorders in offspring conceived after exposure is low.
Introduction
Neuroblastoma (NB) is one of the most common extracranial solid cancer in childhood and the most common cancer in infancy, with an incidence of about 150 cases per year in our department. Radiotherapy is used as a curative-intent treatment of NB, as well as for palliation. Recently, improvements in childhood NB treatment have been achieved, which resulted in longer survival. 1,2 External irradiation with mega-voltage beams is usually considered as a choice of the management of many pediatric malignancies. 3 The organs of children are particularly radiosensitive with low-dose tolerance comparing to the adult. The ovaries are regarded as the most important organs determining the risks of both gonadal failure and development of hereditary effects in future generations. Ovarian dose of the treatment is often calculated by treatment-planning systems when the gonads are included in the treatment volume or they are lying within a distance of 5 cm from the field edge. 4 The scattered dose of ovaries is associated with the treatment site and the distance separating the beam fields from the ovaries.
This study aims to investigate the scattered dose received by ovaries of female children who underwent radiotherapy, to assess the TPS’s dose by ionization chamber, and explore the associated risks for ovarian damage from the radiotherapy.
Material and Methods
Ionization Chamber
The ionization chamber was employed for the measurement of scattered dose in ovaries. The participants.
Simulation of the Treatment Planning
This study was based on treatment planning data for 5 female patients (median age, 4 years; range, 2-6 years) who underwent radiotherapy for truncal NB at our hospital from July 2017 to October 2018. According to the International Neuroblastoma Staging System, 5 children were classified as class IV. The ionization chambers were placed on the water phantoms simulating the radiotherapy treatment. The distance of ovaries from the edge of fields was measured by the images from simulated computed tomography images and the refused magnetic resonance imaging images on the software (MIM, version 6.7). The depth of the measurement points corresponding to ovaries was 7 to 8.5 cm from the anterior surface depending on the patient’s age. To measure the ovarian dose, the 2 points of measurements were located 3 cm from the median plane of the water phantom. Since the measurement’s point was out of the field, the scattered dose of ovaries is difficult to get. To measure the scattered dose accurately, the number of monitor units (MUs) in the measurement was 10 times of actual number. The data of the ovarian scattered dose were recorded. Irradiation with 21.6 Gy was administrated by accelerator (Elekta Synergy, Stockholm, Sweden) 3-dimensional conformal radiation therapy (3DCRT) and intensity-modulated radiation therapy (IMRT) treatment plan.
Treatment Planning
The treatment planning procedure was carried out on a computer system (TPS, Oncentra, Netherlands) and therapy simulator (Brilliance CT Big Bore; Philips Health Care, Cleveland, Ohio). For TPS data, we used the collapsed cone convolution calculation algorithm. For radiotherapy of the child NB, multileaf collimators were used to shield the normal tissues located inside the irradiated area. 3 The ovary is outside of the field, and all treatments were performed isocentrically. Regarding to different age of child, the treatment planning of 6MV or 10MV X-rays was performed. The parameters of the treatment planning and the distances separating the ovaries from the field edge are presented in Table 1. For organs far from the treatment field, TPS data were supplemented using analytical model based on other measurement results from a water phantom and ionization chamber.
Disease, Method of Treatment, Energy of Beams, Prescribed Tumor Dose, Field Size, and the Distance of Ovaries From the Edge of the Field.

Abbreviations: IMRT, intensity-modulated radiation therapy; 3DCRT, 3-dimensional conformal radiation therapy.
Phantom Irradiations
The chest and the abdomen are the primary tumor sites of the NB. The ovaries are the most proximal and important one to treatment fields. Scattered dose of ovaries measurements was performed. The water phantom was constructed from Perspex materials and filled with water. The solid phantom, constructed from materials (Perspex, Darwen, Lancashire, UK), was placed in the treatment field simulating the body of patient. Perspex is nonconductive, which has a dose absorption similar to that of soft tissue.
Risk of Ovarian Dose
To estimate the risk of NB treatment planning, total dose received by the ovaries was multiplied by the appropriate risk coefficient. The International Commission on Radiological Protection has recommended a sex-averaged risk factor of 1.3 × 10−2 per Gy for the whole population. 5
Results
The scattered dose to ovaries from radiotherapy for NB with 6MV and 10MV photon beam is shown in Table 2 and Figures 1 to 2. The scattered dose to ovaries ranged from 1.3 to 46.8 cGy depending on the irradiation site of the patient, energy of beams, and method of treatment. The difference about the scattered dose depending upon the energy of beam was shown in Table 3. The 10MV X-ray beams resulted in an ovarian dose of 1.52 to 1.64 times lower than that of using 6MV X-ray beams, depending on the patient’s age at the time of treatment and treatment site.
Ovarian Dose Measurements.
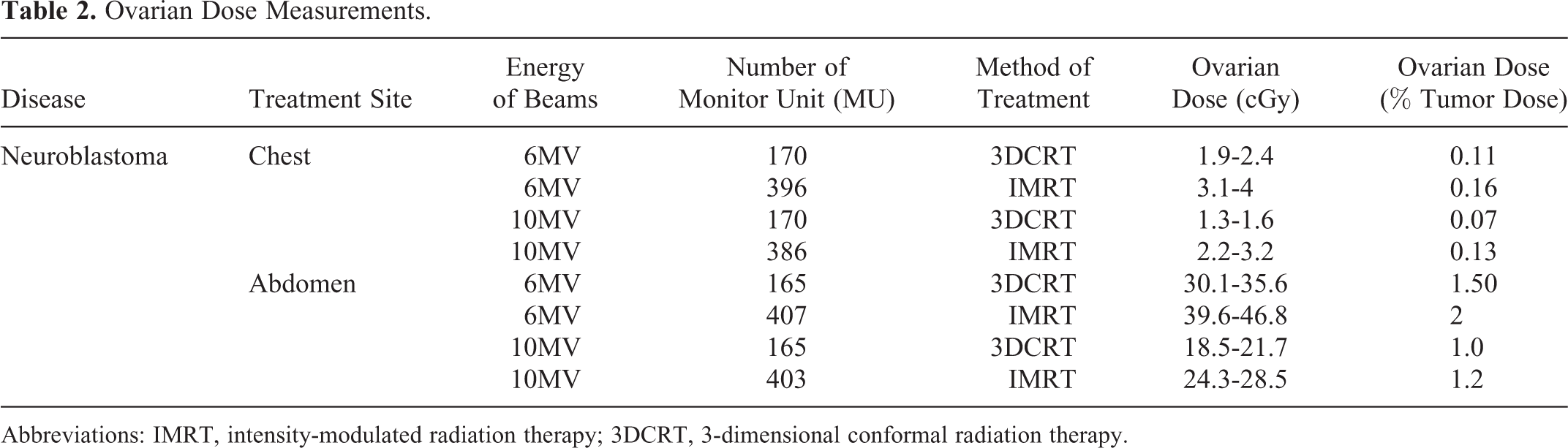
Abbreviations: IMRT, intensity-modulated radiation therapy; 3DCRT, 3-dimensional conformal radiation therapy.

The ovarian dose in patient—chest.

The ovarian dose in patient—abdomen.
Comparison of Ovarian Dose Measured With a 6MV Beam (D6) to That Measured With 10MV Beam (D10).

Abbreviations: IMRT, intensity-modulated radiation therapy; 3DCRT, 3-dimensional conformal radiation therapy.
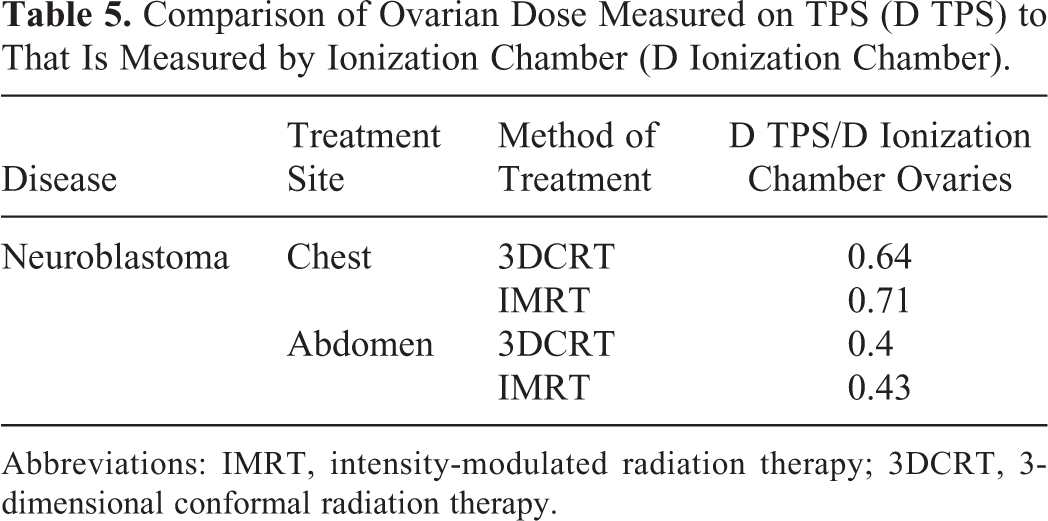
The IMRT treatment plan resulted in ovarian dose of 1.32 to 1.64 times higher than that of 3DCRT treatment plan. The result was shown in Table 4. The calculated scattered dose from TPS was compared to that from ionization chamber, and the result is shown in Table 5. For the radiotherapy, the scattered dose of ovaries on phantom by ionization chamber was 1.40 to 2.32 times higher than that on TPS calculated.
Comparison of Ovarian Dose Measured With a 3DCRT Treatment Plan (D3DCRT) to That Measured With IMRT Treatment Plan (DIMRT).

Abbreviations: IMRT, intensity-modulated radiation therapy; 3DCRT, 3-dimensional conformal radiation therapy.
Comparison of Ovarian Dose Measured on TPS (D TPS) to That Is Measured by Ionization Chamber (D Ionization Chamber).
Abbreviations: IMRT, intensity-modulated radiation therapy; 3DCRT, 3-dimensional conformal radiation therapy.
Discussion
Previous studies analyzing scattered dose of ovaries were mostly performed on phantoms using open fields of square dimensions in adult patients. Previous publications on the dose outside the geometric borders of the treatment fields report on measurements in water tanks or in water-equivalent solid phantoms using open fields of square dimensions. 4,6 –9 However, the influence of scattered dose of ovaries to female children is much more important than to adult. In the present study, we assessed the scattered dose comprehensively in female children who underwent radiation for NB through different method and different treatment plan.
TPS is commonly used to estimate the scattered dose to the organs. This study demonstrated that the scattered dose could be measured with ionization chamber, as there was no significant difference between dose measurements on ionization chamber and TPS-based estimation of doses in the field and at edge of the field. However, at remote sites, such as the measurement spot at long distance to the edge of the fields, TPS underestimated the scattered dose as compared to measurement by ionization chamber. We founded the scattered dose on TPS is lower than that measured by ionization chamber. To assess the safety of ovaries, ionization chamber is more accurate.
Compared with 3DCRT treatment plan, IMRT results in more scattered dose to the ovaries. Though the study, the number of MU decide the scattered dose out of the treatment field. However, IMRT treatment plan has more number of MUs than 3DCRT treatment plan. Taking into account for the scattered dose to the ovaries, 3DCRT treatment plan is better than IMRT for the radiotherapy of child NB. If we focus on the target dose or organ at risks dose, we should choose the IMRT plan which has a better dose distribution. The IMRT plan’s scattered dose of ovaries is in the limited range.
Regarding to the different thickness of body at the different age of child, this study used different energy of photon beams (6MV and 10MV). Compared with 10MV photon beam and 6MV photon beam, the higher dose energy can reduce the ovarian scattered doses up to 1.5 to 1.6 times. If the depth of 10MV photon beams is enough to the young child to treat the NB, we suggest to use 10MV energy or higher energy of photon beams in clinical so as to reduce the scattered dose. Wallace et al 10 have shown that the lethal dose required to kill the 50% of oocytes is <400 cGy. Ovarian doses <400 cGy won’t cause permanent ovarian dysfunctions. 11,12 Destruction of the fixed pool of oocytes and subsequent ovarian failure in women <40 years can occur after doses of 2000 cGy. 13,14 Young adult patients receiving >1500 cGy to ovaries develop hormonal failure. 15 The estimated risk values associated with pediatric radiotherapy should be regarded as low taking into account that the incidence of serious birth defects in human population ascribed as background radiation is 6%. 16
Conclusion
This study provides an analytic set of ovarian scattered dose measurement associated with radiotherapy of NB appearing during childhood. The dosimetric results in this study show that the treatment with mega-voltage photon beams does not carry any risk for permanent damage to ovaries located outside the irradiation field whether is 6MV photon beams or 10MV photon beams. The results in this study also show that method of treatment plan (3DCRT or IMRT) does not bring the risk of damage to ovaries. Through choosing the beams’ energy and treatment plan’s method, the scattered dose of ovaries can be reduced.
Footnotes
Authors’ Note
(a) The material has not been published in whole or in part elsewhere; (b) the paper is not currently being considered for publication elsewhere; (c) all authors have been personally and actively involved in substantive work leading to the report and will hold themselves jointly and individually responsible for its content; (d) our study did not require an ethical board approval because it did not contain human or animal trials. The author (Yichen Shen) testifies to the accuracy of the above on behalf of all the authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
