Abstract
Intrafraction organ deformation may be accounted for by inclusion of temporal information in dose calculation models. In this article, we demonstrate a quasi-4-dimensional method for improved risk estimation. Conventional 3-dimensional and quasi-4-dimensional calculations employing dose warping for dose accumulation were undertaken for patients with liver metastases planned for 42 Gy in 6 fractions of stereotactic body radiotherapy. Normal tissue complication probabilities and stochastic risks for radiation-induced carcinogenesis and cardiac complications were evaluated for healthy peripheral structures. Hypothetical assessments of other commonly employed dose/fractionation schedules on normal tissue complication probability estimates were explored. Conventional 3-dimensional dose computation may result in significant under- or overestimation of doses to organ at risk. For instance, doses differ (on average) by 17% (σ = 14%) for the left kidney, by 14% (σ = 7%) for the right kidney, by 7% (σ = 9%) for the large bowel, and by 10% (σ = 14%) for the duodenum. Discrepancies in the excess relative risk range up to about 30%. The 3-dimensional approach was shown to result in cardiac complication risks underestimated by >20%. For liver stereotactic body radiotherapy, we have shown that conventional 3-dimensional dose calculation may significantly over-/underestimate dose to organ at risk (90%-120% of the 4-dimensional estimate for the mean dose and 20%-150% for
Keywords
Introduction
It is vital that the doses actually delivered to tumors and normal tissues in stereotactic body radiotherapy (SBRT) of the liver are accurately reported 1,2 so that the correlation between dose and clinical outcome may be better known. The SBRT of the liver can offer excellent local control 3,4 but is subject to the difficulties introduced by respiratory motion and associated organ deformation. 2
Such complexities have driven the development of 4-dimensional (4D) treatment planning in radiotherapy. 5 One approach for the calculation of accumulated dose in different phases of the breathing cycle is via “dose warping.” 6 –11 In previous work, we have demonstrated that this may be performed accurately 12 using the DEFGEL dosimetry system. 13 This deformable image registration-based approach revealed that conventional 3-dimensional (3D) planning approaches can significantly underestimate the dose to the planning target volume (PTV) in liver SBRT. 14
The purpose of this study is to investigate the impact of deformation on untargeted healthy organs and evaluate corresponding risk estimates in liver SBRT. Specifically, we demonstrate a methodology for assessing the risk of detriment using conventional 3D and 4D dose-warping approaches in terms of normal tissue complication probabilities 15,16 and latent risks of radiocarcinogenesis and cardiac complications. 17 Whether for prospective estimation of patient risks or retrospective analysis of plan efficacy, we demonstrate that the quasi-4D dosimetry methodology is important for accurate estimation of doses and corresponding risks.
Methods
Dose Calculation: 3D and 4D
To illustrate the proposed methodology, 4 consecutive patients with 5 liver metastases were simulated with 4D computed tomography (CT). A personalized vacuum mold (BlueBAG; Elekta, Stockholm, Sweden) was used for immobilization, while a respiratory signal was monitored via pneumatic bellows (fixed to the abdomen). Patients were imaged using a Brilliance Big Bore (Phillips Healthcare, Cleveland, Ohio) helical CT. The 4D CT data sets were comprised of 10 phases (0%-90%) per patient, acquired at regular time intervals throughout the respiratory cycle (ie, equal-time weighting). This inherently accounts for organ/tumor position in such a way that does not require further weighting. The 0% and 50% phases were observed to correspond to end inspiration and end expiration, respectively. The reference phase was chosen to be the end-exhalation phase, as one would expect smaller tumor motion and thus minimized artifacts. 18 For the 3D planning, internal tumor volumes (ITVs) were delineated using maximum intensity projection (MIP) data. Dose calculation and normal tissue delineation are performed using the 4DCT and its derivatives (MIP and average); the gross tumor volume (GTV) underlying the ITV is based on a contrast CT (triphasic/triple phase scan) performed only in the region of the target with the patient in breath hold. A 5-mm margin was isotropically added to the ITV to define the PTV. The liver and other anatomy (the left and right kidney, spinal cord, heart, esophagus, bowel, stomach, and duodenum) were contoured at the reference phase in the 4D case and on the average intensity projection (AIP) in the 3D case. All radiotherapy techniques are prone to potential uncertainties arising from discrepancies between imaging and (intrafraction) treatment. Despite such complexities, the 4D methodology is expected to provide more a realistic representation than 3D approaches. The original plan was calculated on the AIP. The SBRT treatments were planned on Eclipse v8.9 for treatment on a Varian 21-EX with mini-multileaf collimator (Varian Medical Systems, Palo Alto, California), operated at 6 MV and 600 MU/min using 7 to 9 coplanar fields with 1 to 3 noncoplanar fields. Doses were calculated using the analytical anisotropic algorithm 19 –22 with a grid size of 2.5 mm. Although the number of patients studied is few, our primary intent is to highlight the methodology and considerations that ought be given within the context of deformation in liver SBRT.
Deformable image registration was performed using an optical flow algorithm
23
we have determined to be the most accurate
12
of a range of commonly available algorithms for mass and density conserving deformations. Calculations were undertaken such that the reference image (end-exhalation phase) was the “target” image, to which the “source” images, that is, the other respiration phases were mathematically morphed to match. All calculated image sets were compared and validated against the original CT data. Associated deformation vector fields (DVFs) were used to implement warping of the dose distributions. Dose distributions (3D plan) were generated for each individual phase. Nine DVFs were then applied to the corresponding dose distributions, such that the dose was accumulated in the reference phase. A similar method has been recently employed by Jung
Calculation of Normal Tissue Complication Probability for Deterministic Effects
Calculation of the normal tissue complication probability (NTCP) in this work follows the Niemierko approach
25
and first involves determination of the isoeffective biologically equivalent dose (

The equivalent uniform dose (EUD) may then be evaluated
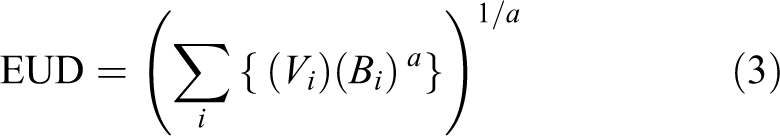

Organ-specific parameters associated with calculation of the NTCP.

Abbreviations: NTCP, normal tissue complication probability; TD50, tolerance dose corresponding to a 50% likelihood of a complication.
Accurate estimation of normal tissue complication probabilities relies on minimal uncertainties in model parameters. Due to the limited clinical data available in model parameters for hypofractionated treatments, such certainty does not exist for the organs at risk (OARs) considered in liver SBRT. In the case of normal liver, Xu
Risk Calculations for Latent Effects
Risk estimates were undertaken based on the methodologies recommended by the biological effects of ionizing radiation VII (BEIR-VII) report
17
for low-dose cancer induction. Estimated risks are presented as excess relative risks (ERRs), defined as the relative risk (RR) compared to background cancer incidence, minus 1; that is,

In other words, an ERR of zero implies zero risk above normal background cancer incidence.
For solid cancer induction, the following approach was taken to estimate the ERR in terms of the dose, 
For the liver, βmale = 0.32 (95% confidence interval [CI95%] 0.16-0.64), βfemale = 0.32 (CI95% 0.10-1.0), γ = −0.3, and η = −1.4; for the stomach, βmale = 0.32 (CI95% 0.11-0.40), βfemale = 1.40 (CI95% 0.31-0.73), γ = −0.30, and η = −1.4; and for other sites, βmale = 0.27 (CI95% 0.15-0.50), βfemale = 0.45 (CI95% 0.27-0.75), γ = −0.30, and η = −2.8. The approach is based on Life Span Study (LSS) cohort data, using analyses of cancer incidence from cases diagnosed in the period 1958 to 1998, and is essentially similar to those used in recent reports by Radiation Effects Research Foundation Researchers (RERFR, Japan/United States). There are also links between radiation exposure and noncancer mortality; this is discussed in detail by, for instance, Shimizu
Results and Discussion
The foremost objective in planning is to achieve coverage of the primary tumor. From a clinical perspective, however, and in accordance with the
This highlights the utility of 4D path-integrated dose accumulation (to supplement conventional 3D-planned dose calculations) for better estimation of the dose received by both targeted and untargeted volumes in liver SBRT. Motion compensation strategies such as the ITV concept serve to ensure GTV coverage, but the TPS calculation of the dose distribution will not account for the movement of healthy tissue

A and B, Dose volume histogram (DVH) of the spinal cord of the first and second patients, respectively (inset: ratio of 3D to 4D calculated dose). This illustrates that the 3D estimate of the dose to the healthy spine may be either (A) poor or (B) good. C, Organ doses calculated using 3D method, that is, not taking into account organ motion. The mean dose, near-maximum dose
Differences between contoured volumes in the 4D reference phase and the 3D AIP are generally small, for example, 7% for the liver and 6% for the kidney. The inclusion of this region contributes further uncertainty to the risk estimates derived from a 3D estimate of the dose distribution. We have contoured organs on the reference phase for the dosimetric comparison, which is consistent with the practices of other clinics undertaking similar work. 24 However, at our clinic, contouring is frequently undertaken on the AIP. We have quantitatively assessed both approaches and discovered that (on average) contouring on the reference phase provides a more accurate estimate of the dose (ie, reduced disparity between 3D and 4D methods). The percentage of increase in disparity in EUD when organs are contoured on the average is liver 3.3%, left kidney 1.7%, right kidney 21%, esophagus 0%, spinal cord 1.7%, heart 1.1%, bowel 36%, and stomach 0.7%.
It is generally accepted that the expected toxicity associated with radiotherapy cannot be defined by the prescription dose alone, 47 and it is for this reason that physical models and simple dosimetric descriptors are essential in plan evaluation.
One such means of assessing the impact on healthy tissues is via normal tissue complication probabilities. We have computed these, using the Niemierko approach, for the healthy liver volume, kidneys, esophagus, spinal cord, heart, bowel, stomach, and duodenum. The EUD (see Equation 3) are shown in Figure 2. The NTCP for all OARs were found to be very small (<0.2%) for the fractionation scheme used (data not shown). Repeating the calculations for normal liver using the LKB approach
44
and model estimates based on Xu

The mean ratio of equivalent uniform dose (EUD; 3D/4D) for various healthy organs; the error bars indicate maxima and minima for the patient cohort studied. 3D indicates 3-dimensional; 4D, 4-dimensional.
The NTCP (%) of the Healthy Liver Calculated Using Alternative Fractionation Schemes.a

Abbreviations: NTCP, normal tissue complication probability; 3D, 3-dimensional; 4D, 4-dimensional.
aComparison of the Niemierko-based (2 Gy per fraction) and Xu-based (4.6 Gy per fraction) estimates. Some cases would have been rejected or replanned but are shown here for completeness.
Differences in absolute values of NTCP were expected based on the data and methods used to derive model parameters (including differences in fractionation schemes) and also differences in toxicity end points.
48,49
Although the absolute values for the NTCPs were different (0.5%-2.0% using the parameters of Xu
In this case, 42 Gy was delivered in 6 fractions of 7 Gy each. However, exploration of alternative fractionation schemes does reveal significant differences, as shown in Table 2. This shows NTCP data for the (healthy) liver for several of the most common 50 liver SBRT fractionation approaches. The results in Table 2 demonstrate that significant uncertainties exist in the estimation of NTCP and that substantial differences in these values may exist across the different fractionation schemes. The NTCP calculated using the Xu data is in general higher but exhibit similar differences between 3D and 4D. These model parameters are derived from clinical observations and estimates of the doses delivered. Although the estimates of NTCP for the OAR appear to be low, considerable uncertainty exists in the modeling parameters. Furthermore, it is likely that this uncertainty increases when the dose fractionation scheme differs notably from that employed when the parameters were originally derived. As clinical data emerge from this rapidly growing treatment modality, we believe that realistic estimates of the dose actually delivered to the liver (eg, through the quasi-4D method presented here) will lead to greater confidence in the model estimates. Such information is vital if treatment prescriptions are to be informed by NTCP estimates. Clearly, the more accurately we can estimate the true delivered dose to the patient, the better we can define model parameters to predict clinical outcomes in the era of hypofractionated treatments. 1
Although the deterministic effects appear to be low for the fractionation scheme ultimately employed, one cannot ignore the notable differences in calculated healthy organs dose for the 2 methods. Out-of-field doses and potential stochastic detriment (eg, radiocarcinogenesis) are becoming increasingly of interest within the context of stereotactic radiotherapy, 2 particularly since the difference in out-of-field dose between small and large fields is not as significant as one may intuitively expect. 51 –53 Of particular note, centres with SBRT employ it to treat over half of liver cases, 50 and survival rates are increasing. 54 Although the mean age for such patients is around 60, patients as young as 30 may present. 55 As survival improves, radiocarcinogenesis presents an increasingly important adverse potential outcome. This highlights the importance of accurate dosimetry, both for prospective risk estimation and retrospective plan analysis. As such, we have computed the risks of radiation-induced cancer using methods recommended by the BEIR-VII 17 report, and the results are presented in Figure 3. Although extrapolation of NTCP approaches to the SBRT regime is sometimes considered contentious, one could suggest that in the case of stochastic effects the opposite is true—that single/few high-dose fractions better represent the cohort forming the basis of the risk estimates, that is, the atomic bomb cohort. We have assumed the age-of-exposure model of >30 years (see Equation 6) and an attained age of 60. It is clear from Figure 3 that the ERR is greater than zero for all organs studied.

A, Based on the conventional 3D dose estimates, the excess relative risk (ERR) for different healthy organs calculated using biological effects of ionizing radiation VII (BEIR-VII) recommended methods
18
for various healthy organs based on the total organ dose and on maximum dose received by at least 2% of the volume (
For the purpose of comparison, we have also calculated the equivalent risk estimates calculated using the oft-employed International Commission on Radiological Protection 103 (ICRP-103) approach
56
(see Figure 4A). The ratio of results from the 3D to 4D methods is the same for both BEIR and ICRP methods as a result of linearity. It is also possible to determine the latent risk of heart disease using data from the Japanese LSS cohort. Preston
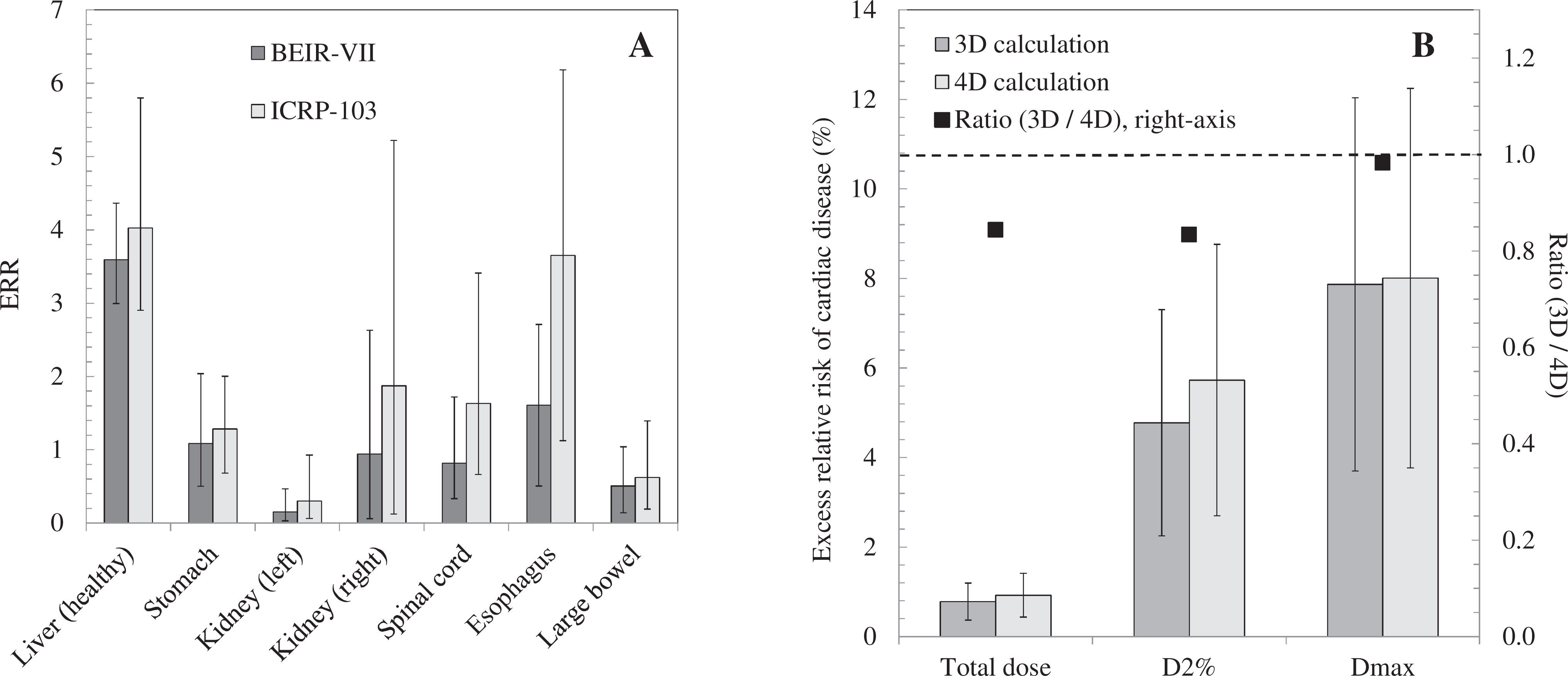
A, A comparison of excess relative risks calculated using International Commission on Radiological Protection 103 (ICRP-103) data57 against the biological effects of ionizing radiation VII (BEIR-VII) phase 2 estimates
17
employed in this work. The error bars reflect the spread in patient doses about the mean, not uncertainties in the risk estimates. B, The calculated radiation-induced ERR for cardiac disease is shown for an example patient. Calculations are shown for integral dose, maximum dose received by at least 2% of the volume (
The expectation of a correlation between the magnitude of motion and/or deformation and the magnitude of errors quantifiable by the 4D approach suggests the possibility for patient selection rather than routine 4D calculations. For instance, a typical clinical workflow might involve:
Patients exhibiting large tumor excursion to be flagged for additional analysis. Cross-check to be undertaken using quasi-4D methodology. Undertaken by physics/RT using optical flow algorithm. Dose and risk data presented to complement 3D information. If large differences in dose calculations are noted, we recommend that the treating clinician be notified and the most conservative approach be taken in estimating clinical risk to critical organs.
Conclusion
Accurate dosimetry is needed for accurate DVH-based risk estimation and appropriate reporting of doses associated with observed end points. We have demonstrated that the use of conventional 3D dose estimates (that do not take into account temporal changes) may be insufficiently accurate for this purpose and that a quasi-4D dose-warping-based dose-accumulation method may yield more suitable data.
For our illustrative cohort of liver SBRT patients
Estimation of the NTCP risks, while low for the fractionation scheme employed, typically vary by about 30% between the 3D and 4D methods. Excess RRs for radiocarcinogenesis range up to ERR > 4 and the prediction thereof depends greatly on the use of either 3D or 4D methods (by tens of percent).
In general:
The doses and risks to healthy organs calculated by the path-integrating quasi-4D methodology differ significantly from those computed with the conventional 3D approach.
Footnotes
Acknowledgments
The authors would like to thank Prof Martin Ebert for his assistance with the LKB NTCP calculations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Health and Medical Research Council (NHMRC), 555420.
