Abstract
Aim:
To explore the relationship of clinicopathological features and the proteins of C-met expression in the prognosis of cholangiocarcinoma.
Methods:
Clinical data and the completed follow-up information of patients with cholangiocarcinoma who underwent cholangiocarcinoma operation from January 2004 to December 2010 were analyzed retrospectively. The relationship of clinicopathological features and C-met in the prognosis of the patients was analyzed.
Results:
Patients with high expression of C-met had significantly shorter overall survival than those with low expression of C-met, the difference being statistically significant (P = .003). Patients with high C-met expression had significantly shorter disease-free survival time than those with low expression of C-met, the difference being statistically significant (P = .009). By COX multivariate analysis, high C-met expression in tumor tissues was an independent risk factor in predicting overall survival and disease-free survival for patients with cholangiocarcinoma (P = .038, .048, relative risk = 1.390, 1.427).
Conclusion:
Patients with high C-met expression in cancer tissues had shorter disease-free survival and overall survival. High expression of C-met is an independent risk factor for overall survival and disease-free survival.
Introduction
Cholangiocarcinoma (CC) is one of the most common malignant tumors in China, diagnosis is often delayed, and prognosis is poor. Surgery is the only possible way of CC cure, but most patients with CC had inoperable tumor treatment, and due to the rapid development of CC with obvious symptoms, surgical patients also have a high replacement rate. 1 To fully understand the biological characteristics and important factors of CC that may affect treatment strategies, and to implement standardized and individualized treatment are the key to the treatment of CC. Recently, a new treatment strategy for CC has been proposed, in light of better understanding of the molecular mechanisms of carcinogenesis. It has been proposed that receptor tyrosine kinases (RTKs), such as epidermal growth factor receptor (EGFR), vascular epithelial growth factor (VEGF), and C-met, are promising targets for treatment of CC. 2,3 In a previous report, we have indicated that EGFR and VEGF could be promising molecules for targeted therapy of CC. 3,4 Sia and his colleagues 5 analyzed 119 cases of patients with CC on the gene expression profile and screened out 2 different sets of gene tags, that is, proliferation and inflammation classes. Proliferation class was related to some differences to the copy number of oncogenes, including KRAS (kirsten rat sarcoma viral oncogene homolog) and BRAF (B-Raf proto-oncogene, serine/threonine kinase) signaling pathways, and RAS (retrovirus associated sequence oncogene), MAPK (Mtiogen-Activated Protein Kinase), and MET (MET proto-oncogene, receptor tyrosine kinase) signal transduction pathway. RAS-RAF-MEK-ERK signal of the proteins encoded by these genes forms part of the conduction path. RAS-RAF-MEK-ERK signal transduction pathway can promote cell proliferation or activation of phosphatidylinositol-3-kinase-AKT-mammalian target of rapamycin (PI3K-AKT-mTOR) signaling pathway that promotes cell survival. When combined with hepatocyte growth factor (HGF) and C-met, HGF/C-met signaling pathway is activated, and first of all 4 phosphorylation sites of intracellular tyrosine residues autophosphorylate near C-met, which raise downstream Gab-1, Drb-2, Shc, and adapter proteins such as c-Cb1 (adaptor), followed by a series of phosphorylation reactions that activate PI3K, ERK1/2, phospholipase Cγ, STAT (signal transducer and activator of transcription), and focal adhesion kinase and other important signaling molecules and the corresponding signal, thereby regulating pathways of tumor cell proliferation, migration, and invasion.
C-met proto gene encoding has the tyrosine kinase activity of HGF receptor. C-met activates multiple downstream signal transduction pathways. C-met has a relation between a variety of regulator proteins and cancer gene products involved in cell signal transduction and cytoskeletal rearrangement and is an important factor in differentiation, movement, and proliferation. Part of the RAS-RAF-MEK-ERK conductive signal pathway is formed by these encoded gene proteins. RAS-RAF-MEK-ERK signaling pathway can promote cell proliferation or activation of PI3K-AKT-mTOR signaling pathway to promote cell survival. C-met with a variety of regulatory proteins and cancer-related gene products involved in cellular signal transduction and regulation of cytoskeletal rearrangement is an important factor of cell differentiation, movement, and proliferation. C-met plays an important role in tumor migration and metastasis. 6 –8 The aim of this study was to explore the relationship of C-met and clinicopathological features, the expression of C-met in the prognosis of tumor. Moreover, this study provides a certain reference value of clinical evidence for CC-targeted therapy, especially for C-met protein targets.
Materials and Methods
Tumor Tissues
A total of 291 resected specimens from patients who underwent operations for CC from January 2004 to December 2010 at the Department of General Surgery, General Hospital of Air Force, Fucheng Road, Haidian District, Beijing, China, were analyzed retrospectively. Clinical data (including age, gender, and tumor pathological features) were collected. The patients had a clear diagnosis of CC and had received neither radiotherapy and chemotherapy before surgery nor preoperative liver transplantation. Those with pathological diagnosis of sarcoma, mixed cell carcinoma, and hepatocellular carcinoma was excluded. After surgery, all tissue samples were fixed in 4% formalin for 24 hours and then embedded in paraffin for hematoxylin and eosin (H&E) and immunohistochemical (IHC) staining. Sections were cut at 4 µm. The H&E-stained samples were individually examined microscopically by 2 independent pathologists (L.R. and Z.-Y.M.). Clinicopathological characteristics of the tumors are shown in Table 1.
Clinical and Pathological Features of 291 Cases.

Immunohistochemical for C-met
C-met (clone: EP1454, dilution 1:200; Abcam, Cambridge, United Kingdom) was used and pre-heated using heat-induced epitope retrieval (pre-heated in pressure cooker for 5 minutes in citrate buffer, pH 6), which was employed prior to C-met staining. Primary antibodies were then added and slides incubated for 2 hours at 37°C. Slides were then processed on an immunostainer (LabVision Autostainer 360, Fujian, China). The primary antibody was replaced by phosphate-buffered saline as a negative control to assess the specificity of the antibodies. Hematoxylin-counterstained sections were mounted in aqueous mounting medium and observed under a light microscope.
The IHC staining (percentage of stained cells × staining intensity) for C-met was scored for each case after semiquantitative evaluation by 2 independent pathologists (L.R. and Z.-Y.M.). The percentage of stained cells observed in every 100 positive cells/100 × 100% was the percentage of positive cells. Staining intensity was negative (0), weak (light yellow; 1), moderate (yellow; 2), or strong (deep yellow; 3). The final score of the specimen area was percentage of positive cells × staining intensity × 100. The final score for each antibody IHC staining ranged from 0 to 300 points, with a median score as a borderline, and they were assigned to a high-expression group and a low-expression group, and the median value was assigned to the high-expression group.
Statistical Analysis
The SPSS 19.0 statistical software was used for statistical analysis. Chi-square (χ2) test was used to compare rates, Spearman test was used for correlation analysis, Kaplan-Meier survival curves were used to calculate survival, the Log-rank test was used for univariate analysis, and COX regression was adopted for multivariate survival analysis. P value <.05 was considered statistically significant.
Results
Expression of C-met in the Tumor Tissues and Adjacent Tissues
The expression of C-met in the tumor tissues and adjacent tissues is presented in Table 2. The C-met staining in membrane and perinuclear granules is shown in Figure 1. The high C-met expression rate in tumor tissues was 53.1% (135 of 254) and 22.3% in adjacent tissues (55 of 247). C-met expression in tumor tissues was higher than that in the adjacent tissues.
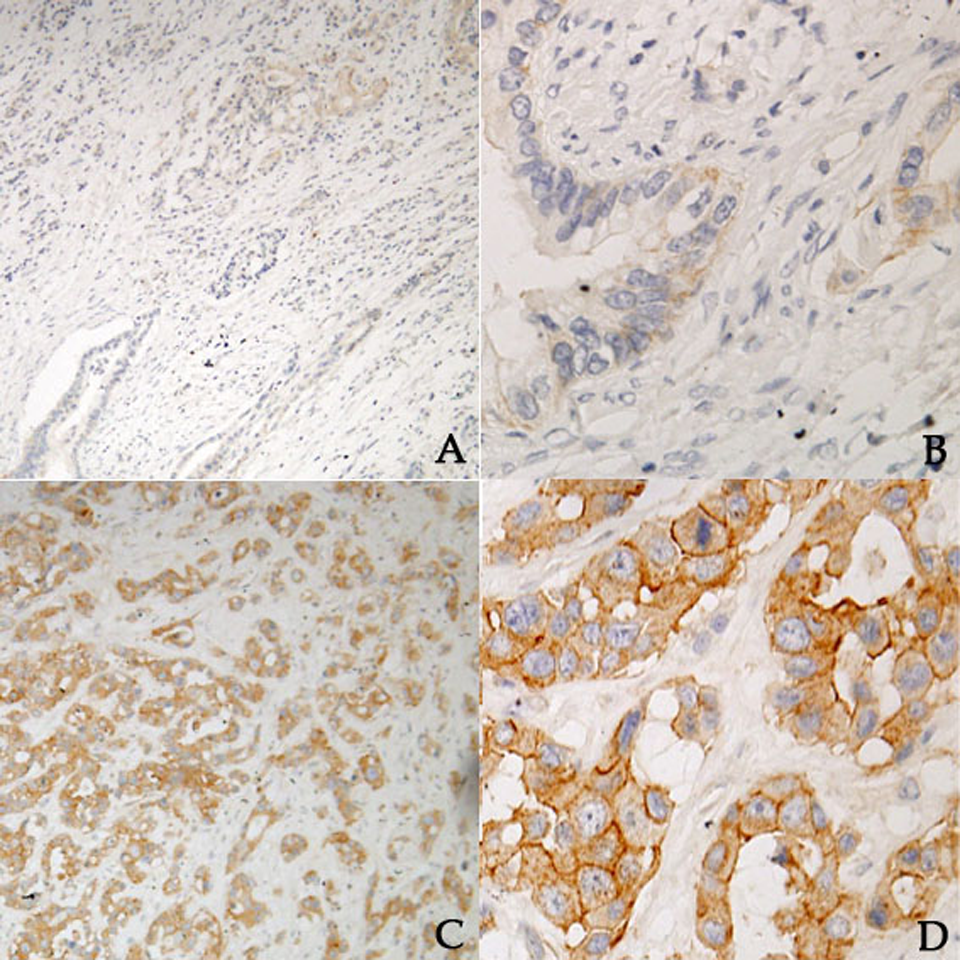
Immunophenotypes of the investigated antigens in tumor tissues. C-met in membrane staining: (A) C-met low expression, ×100, (B) C-met low expression, ×400, (C) C-met high expression, ×100, and (D) C-met high expression, ×400.
The Expression of C-met in the Tumor Tissues and Adjacent Tissues.

aDue to off-chip during the dyeing process or no tumor tissue, so the number of cases is different from the previous.
Correlation Between C-met Expression Levels and Clinicopathological Characteristics of Tumors
The correlation between C-met expression and clinicopathological features is shown in Table 3. C-met expression is correlated with Tumor Node Metastasis (TNM) stage, depth of invasion, and surgical methods. With the increase in the TNM staging, high rates of C-met expression are, respectively, 43.4%, 59.7%, 78.8%, and 53.8%, the differences mean significance in the science of statistics (P = .002), as shown in Figure 2. In cases of tumor confined to the partial and full-thickness infiltration of C-met expression is 45.9%, while the growth of the tumor to the surrounding invasive cases of C-met expression was 63.2%, the differences mean significance in the science of statistics (P = .007), as shown in Figure 3. The high expression of C-met in radical surgery in cases of CC is 54.7%, while in the case of palliative surgery it is 18.2%, the differences mean significance in the science of statistics (P = .017), as shown in Figure 4. There is no significant difference between the expression rates of C-met with other clinicopathological features. The positive expression rates of C-met is significantly related to the TNM stage, infiltration, and surgical approach (Figures 2 –4).
The Relation of Expression of C-met With Clinicopathological Characteristics in Tumor Tissue.


C-met expression in tumor tissues and TNM staging histogram.

C-met expression in tumor tissues and infiltration histogram.
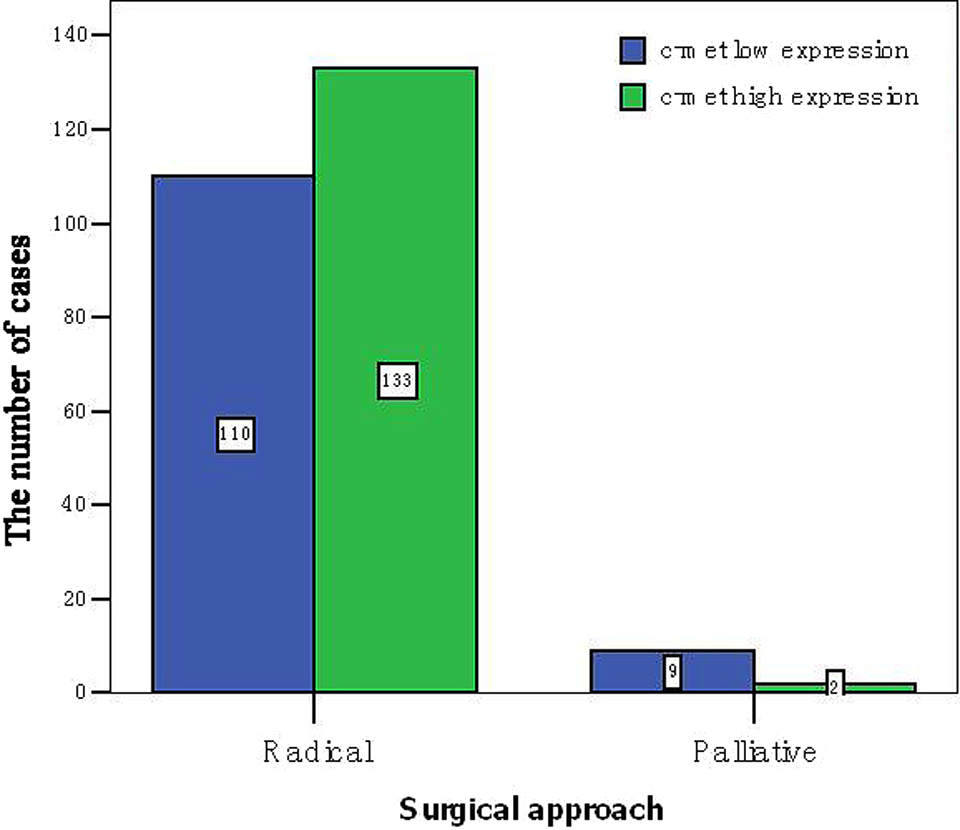
C-met expression in tumor tissues and surgical approach histogram.
Correlation Between C-met Expression in Tumor Tissues and Prognosis
Patients with high expression of C-met had significantly shorter overall survival (OS) than those with low expression, the difference being statistically significant (P = .003). Patients with high C-met expression had significantly shorter disease-free survival (DFS) than those with low C-met expression, the difference being statistically significant (P = .009). Based on COX multivariate analysis, the high C-met expression in cancer tissues was an independent risk factor in predicting OS and DFS for patients with CC (P = .038, .048, relative risk =1.390, 1.427).
Univariate analysis showed that patients with high C-met expression in tumor tissues had significantly shorter progression-free survival and OS than those with low expression, and the difference was statistically significant (mean OS: 27.47 vs 15.00 months, mean DFS: 30.80 vs 12.00 months; P = .003 and .009), as shown in Figures 5 and 6.

C-met expression in tumor tissues and disease-free survival (DFS), blue line: C-met low expression and green line: C-met high expression (Color version of the figure available online).

C-met expression in tumor tissues and overall survival (OS), blue line: C-met low expression and green line: C-met high expression [Color version of the figure available online].
Multivariate Analysis in C-met Expression and Prognosis of Patients With CC
The clinical and pathological features of patients with CC were analyzed with univariate analysis, and the statistically significant results obtained and C-met were then included in the COX multivariate analysis to observe the effects of the indicators on the prognosis. As shown in Table 4, the high C-met expression in tumor tissues is an independent risk factor for OS and DFS of patients with CC, and patients with high C-met expression in tumor tissues had 1.390 times higher risk of death than patients with low C-met expression. Patients with high C-met expression in tumor tissues had 1.427 times high risk of recurrence than patients with low C-met expression of 1.427 times.
C-met Expression and Clinicopathological Features of Multivariate Survival Analysis in Cancer Tissues.

Abbreviation: CI, confidence interval.
Discussion
Abnormal HGF/C-met signaling is closely related to cancer occurrence and development. 9 –20,21,22 Tumor cells can release a series of cytokines such as interleukin, and these cytokines can stimulate adjacent fibroblasts to exudate HGF. Abnormal expression of HGF sustained dependent activation of the HGF signaling pathway of C-met and raised on the cell surface expression of C-met protein, thereby enabling the tumor cells to exhibit invasion and metastasis activity. This invasion and metastasis is closely related to the activation of HGF/C-met signaling pathway. C-met is highly expressed or mutated in a variety of inflammation-related malignancies, such as 23,24 esophageal cancer, liver cancer, pancreatic cancer, and other tumors. Herrera et al took measure of protein and messenger RNA of C-met in normal esophageal tissue, Barrett esophagus, and esophageal adenocarcinoma by immunohistochemistry and reverse transcription polymerase chain reaction, the results showed that expression of C-met is low in the normal esophagus and well-developed Barrett esophagus, and expression of C-met is high in esophageal cancer and hypogenetic Barrett esophagus. Yu et al studied and found that 25 the expression of C-met in pancreatic cancer tissues was significantly higher than that of chronic pancreatitis and normal pancreatic tissue, but the study also showed that only 1 change in the expression of C-met itself does not induce benign to malignant transformation, that is, chronic pancreatitis tissue cells into pancreatic epithelial cell transformation. Currently, the industry considered that C-met relates closely to a variety of malignancies, that is, its development and metastasis; in the majority of tumors, there is a high-expression of C-met and amplification of the gene in the process of the development and metastasis of tumors. The results of our study showed that the expression of C-met in patients with bile duct cancer tissues were significantly higher than that of the expression in adjacent tissues, and the high expression of C-met is an independent risk factor for DFS and OS for the patients with CC. Miyamoto research shows 26 that there is poor OS and DFS in patients with intrahepatic CC where expression of C-met is high, and the high expression of C-met is an independent predictor of OS and DFS. Overexpression of C-met is linked with EGFR expression, and it is a poor prognosis factor in CC, which is consistent with the results of this study. But Miyamoto merely researched intrahepatic CC, and this study researched both kinds of CCs (including intrahepatic and extrahepatic CC). In this study, although the cases with extrahepatic CC are large in number after statistical analysis the expression of C-met is not affected by the tumor site. Therefore, this study fully describes the expression of C-met and the influence of CC prognosis.
Because tumor invasion and metastasis are the main causes of death in patients with cancer, interfering with C-met signaling appears to be a promising therapeutic approach. So C-met is a target for the treatment of CC. Tyrosine kinase receptor C-met 27 -32 as a target for anticancer drugs in cancer research has become very active in the field of antitumor therapy, which provides a new method. C-met activation is regarded as one of the molecular mechanisms involved in the acquisition of resistance to anti-EGFR therapy, as activation of the alternative RTK pathway would bypass the EGFR pathway. 33 Therefore, the application of EGFR inhibitors can inhibit the synthesis of C-met. 34 Many studies have confirmed this conclusion. 22,35 –41
Although there are only a few cases with TNM stage IV, the present study has already shown patients with high expression of C-met are more than patients with low expression of C-met. So if the sample size is increased, it can be believed that in cases with TNM stage IV, the expression of C-met will show further expansion. There is a statistical significance of expression of C-met in all cases of bile duct cancer, and we infer that cases with C-met mutations can be curbed using C-met target therapy. In summary, there are only a limited number of cases in our study, and the next step is to expand the sample size to study further. Therefore, C-met inhibitors as a treatment of CC new program need to be further confirmed, and this study provides a helpful theory of clinical treatment of CC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
