Abstract
Background:
The treatment options for intractable metastatic colorectal cancer include regorafenib, trifluridine/tipiracil, and fruquintinib. In this study, we aimed to conduct a network meta-analysis for comparing the efficacy of these agents.
Methods:
We searched the PubMed, EMBASE, Cochrane Central Register of Controlled Trials, and ClinicalTrials databases for relevant literature, up to February 2020. The data were collected from randomized controlled trials on regorafenib, trifluridine/tipiracil, or fruquintinib, administered to patients with metastatic colorectal cancer who failed on treatment with oxaliplatin, irinotecan, or fluoropyrimidine. The primary end points, namely, the overall survival and progression-free survival, were analyzed for subsequent network analysis using the Review Manager and Aggregate Data Drug Information System software for performing direct and indirect comparisons.
Results:
A total of 7 trials were analyzed in this study. Trifluridine/tipiracil and regorafenib proved to be superior to the placebo, with respect to the overall survival (odds ratio: 0.38, 95% confidence interval: 0.27-0.52 for trifluridine/tipiracil; odds ratio: 0.47, 95% confidence interval: 0.26-0.84 for regorafenib) and progression-free survival (odds ratio: 0.18, 95% confidence interval: 0.05-0.67 for trifluridine/tipiracil; odds ratio: 0.06, 95% confidence interval: 0.04-0.09 for regorafenib). Regorafenib (80 mg) was superior to the placebo in terms of the overall survival and progression-free survival and inferior to trifluridine/tipiracil and fruquintinib. Network analysis revealed that the efficacy of trifluridine/tipiracil and fruquintinib was fundamentally similar, and both the agents were superior to regorafenib.
Conclusion:
Regorafenib (80 mg) was superior to the placebo, but inferior to 160 mg regorafenib, trifluridine/tipiracil, and fruquintinib. This study further revealed that the efficiency of trifluridine/tipiracil and fruquintinib is identical, but their toxicity profiles are different.
Introduction
Colorectal cancer (CRC) is one of most common malignant tumors. Morbidity and mortality due to CRC have gradually declined in the western countries, but the annual incidence of CRC is still increasing in China.
1
About 25% of patients with colon cancer have metastases at the time of diagnosis. Patients with inoperable metastatic colorectal cancer (mCRC) are mainly administered systemic chemotherapy, and the standard first-line and second-line treatments are usually a combination of oxaliplatin, irinotecan, and fluorouracil, administered orally or intravenously.
2
-5
For patients with
Regorafenib (160 mg),
7
trifluridine/tipiracil (TAS-102),
8
and fruquintinib
9
are approved for the treatment of refractory mCRC following disease progression after second-line chemotherapy. Regorafenib, a multitargeted tyrosine kinase inhibitor, which inhibits the processes involved in tumor angiogenesis (via vascular endothelial growth factor
We therefore aimed to evaluate the efficacy and adverse effects of regorafenib, TAS-102, and fruquintinib by performing a literature search and network meta-analysis.
Methods
The systematic review was reported to be consistent by the preferred reporting items for systematic reviews and meta-analysis.
Literature Search
We searched the PubMed, EMBASE, and Cochrane Central Register of Controlled Trials databases up to February 2020, using the following search strings: “metastatic colorectal cancer,” “TAS-102,” “fruquintinib,” and “regorafenib” for screening articles on prospective, placebo-controlled, randomized clinical trials (RCTs). We also searched the ClinicalTrials database for screening relevant articles. Systematic reviews and meta-analyses were also analyzed for checking whether any articles had been omitted in our search results. The specific search strategies are enlisted in Supplemental Table S1. Two reviewers (GZZ and FP) independently reviewed the full-text articles by screening the list of titles and abstracts, on the basis of the inclusion criteria. The disagreements between the 2 reviewers were resolved by consensus.
Inclusion Criteria and Outcome Measures
Studies on phase II/III, accessible, prospective, placebo-controlled RCTs on regorafenib, TAS-102, or fruquintinib in patients with mCRC were included in our study. Uncontrolled or single-arm studies, review articles, letters, meta-analysis data, case reports, commentaries, or publications that were not in the English language were excluded. The study participants had been assigned to receive one of the following agents: regorafenib, TAS-102, or fruquintinib, and the results were compared with those of the placebo or best supportive care groups. The studies reported the outcomes of progression-free survival (PFS), OS, and adverse events (AEs) following treatment with the aforementioned agents.
Data Extraction
In this study, OS and PFS were selected as the primary outcomes. Two reviewers (GZZ and LSX) extracted and tabulated the basic data from the articles, including the corresponding author, year of publication, name of the study, country, regimen, follow-up, AEs, and treatment arms. The inconsistencies were resolved by a third reviewer (FP).
Risk of Bias and Quality Assessment
Two reviewers (FP and LSX) individually assessed the quality of the studies included herein, on the basis of Cochrane risk of bias tool, and subsequently reviewed the results using Review Manager software, version 5.4. 11 The main points of quality assessment included the procedure of allocation, concealment of allocation, participant blinding, blinding of outcome assessment, incomplete outcome data, and selective reporting.
Statistical Analyses
We collected the data regarding the hazard ratio (HR) for survival time and the AEs observed in the different studies. All the data were processed by the Aggregate Data Drug Information System (ADDIS) software, version 1.8, 12 for performing direct and indirect comparisons. The results were subsequently verified with STATA software, version 14.2. 13
Results
Selected Studies
We screened a total of 639 studies, of which 39 were eligible for the assessment. Seven randomized controlled trials were included for the meta-analysis, as depicted in Figure 1. The 7 clinical trials selected herein included a total of 2755 patients. Three trials compared TAS-102 with placebo, 2 trials compared 160 mg regorafenib with placebo, 1 trial compared 160 mg regorafenib with 80 mg regorafenib, while 1 study compared fruquintinib with placebo. The common characteristics of the trials are enlisted in Tables 1 and 2, and in Figure 2.
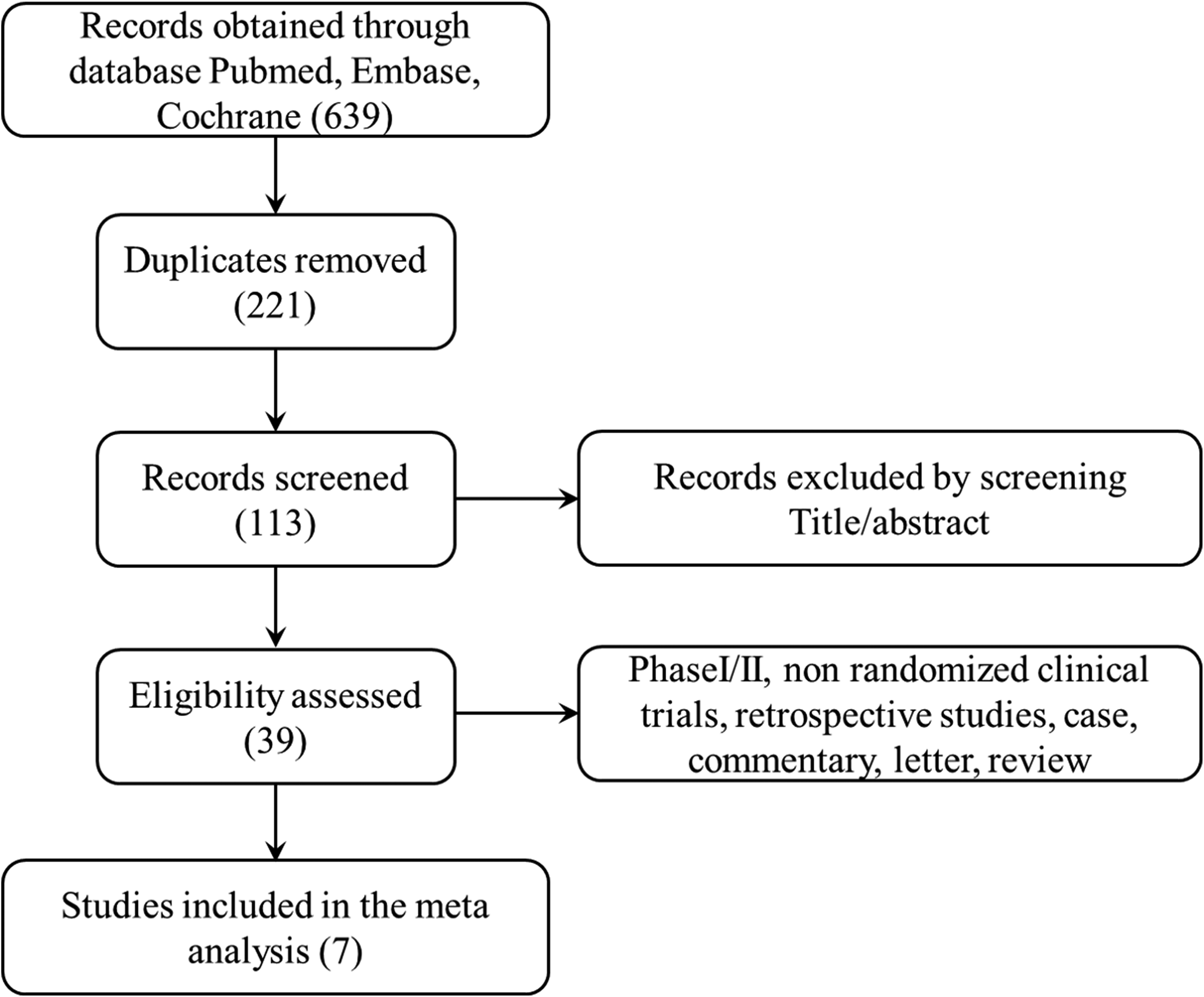
Flowchart of the screening process.

Network of the various interventions collected for the network meta-analysis. The numbers indicate the numbers of the trials. Regorafenib: Regorafenib 160 mg, Regorafenib80: Regorafenib 80 mg.
Characteristics of Studies Included in the Analysis.

Characteristics of Patients in the Studies at Baseline.
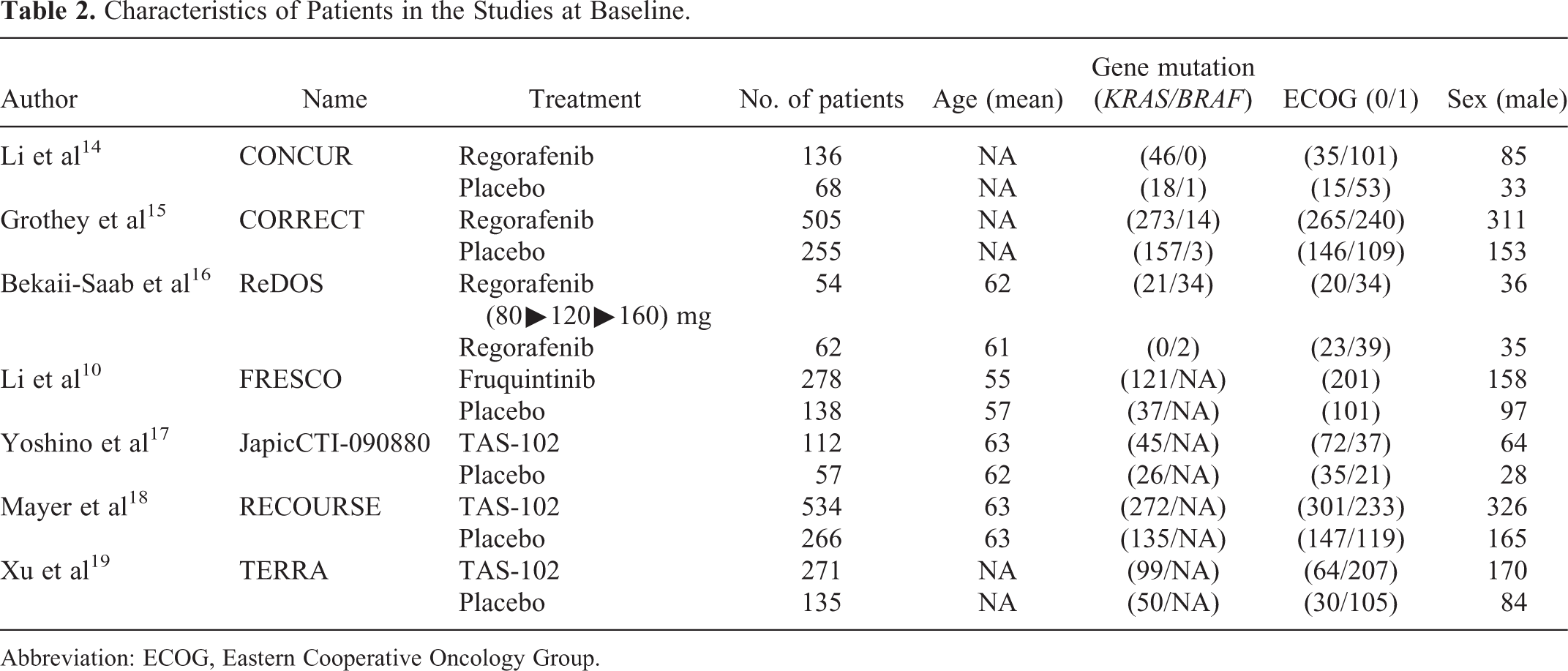
Abbreviation: ECOG, Eastern Cooperative Oncology Group.
Direct Meta-Analysis
Regorafenib versus placebo
The efficacy of 160 mg regorafenib was compared to that of the placebo in 2 trials, CONCUR 14 and CORRECT. 15 CONCUR was conducted in Asia, while CORRECT was conducted worldwide, in multiple centers. The results of the 2 trials were consistent. They demonstrated that regorafenib prolonged the OS (odds ratio [OR]: 0.47, 95% CI: 0.26-0.84) and PFS (OR: 0.06, 95% CI: 0.04-0.09) in patients with mCRC (Table 3; Figure 3).
Overall Survival and Progression-Free Survival.

Abbreviations: HR, hazard ratio; mOS, median overall survival; mPFS, median progression-free survival.

Direct analysis of the trials included in the study. A, Analysis of PFS; B, Analysis of OS. OS indicates overall survival; PFS, progression-free survival.
Trifluridine/tipiracil versus placebo
The efficacy of TAS-102 was compared with that of the placebo in 3 trials, namely, JapicCTI-090880, 17 RECOURSE, 18 and TERRA, 19 of which JapicCTI-090880 was a phase II study, conducted in Japan. We pooled and analyzed the results of these 3 clinical studies, and it was found that TAS-102 prolonged the OS (OR: 0.38, 95% CI: 0.27-0.52) and PFS (OR: 0.18, 95% CI: 0.05-0.67) of patients with mCRC (Table 3; Figure 3).
Indirect Network Meta-Analysis
A total of 7 RCT trials were included and analyzed in the network meta-analysis (Tables 1 and 2) that was performed with ADDIS, version 1.8, and subsequently verified by STATA. The results demonstrated that the efficacies of fruquintinib and TAS-102 were similar, with respect to the PFS (HR: 0.76, 95% CI: 0.16-3.77) and OS (HR: 1.30, 95% CI: 0.06-27.40) and agreed with the results of a previous study. 20 Intriguingly, both TAS-102 and fruquintinib were superior to regorafenib with respect to the PFS and OS (Table 4), and both were superior to 80 mg regorafenib with respect to the PFS and OS. All the results were subsequently verified by STATA (Supple mental Figure S1).
Summary of Adverse Effects Associated With TAS-102.

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; NA, not available; TAS-102, trifluridine/tipiracil.
Adverse Events
Both regorafenib and fruquintinib are multitarget agents, but differ from TAS-102. We therefore collected the descriptive data regarding these agents in this study (Tables 5 and 6). TAS-102 is a combination of 2 active pharmaceutical agents, trifluridine and tipiracil, of which tipiracil prevents the degradation of trifluridine, and consequently increases the activity of trifluridine. We therefore identified that the common AE of TAS-102 was the inhibition of bone marrow function. The AEs of regorafenib and fruquintinib were hypertension, hand-foot syndrome, and fatigue. The incidences of grade 3 and grade 4 AEs following regorafenib and fruquintinib therapy were found to be similar.
Collection of Adverse Effects Associated With Regorafenib and Fruquintinib.

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; NA, not available; TSH, thyroid stimulating hormone.
Network Analysis of the Studies.
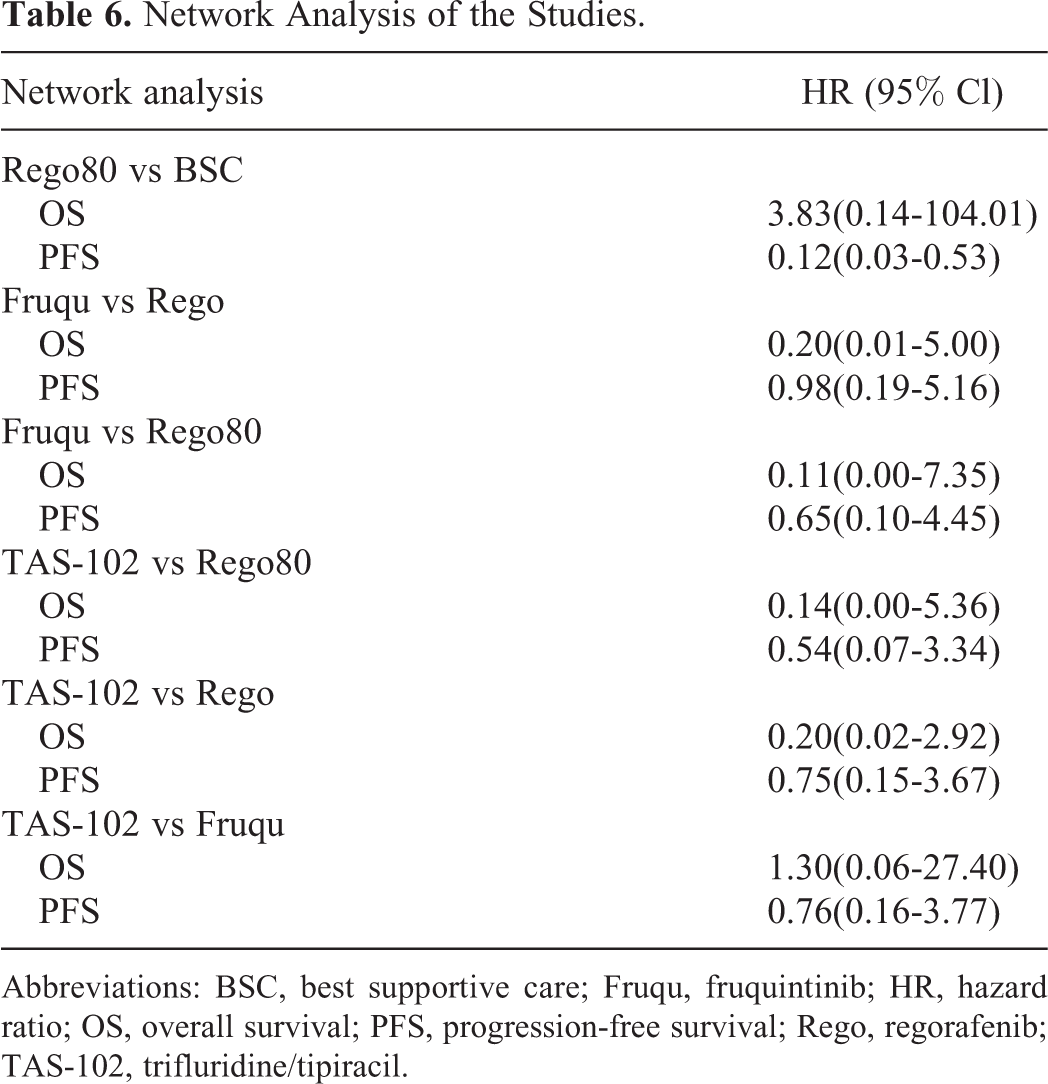
Abbreviations: BSC, best supportive care; Fruqu, fruquintinib; HR, hazard ratio; OS, overall survival; PFS, progression-free survival; Rego, regorafenib; TAS-102, trifluridine/tipiracil.
Quality Assessment and Risk of Bias
The qualitative assessment was conducted using the aforementioned tools 21 for calculating the risk of bias (Figure 4), in combination with the GRADE approach, 22,23 that comprises 4 steps for quality assessment. Overall, the trials had a low risk of bias. However, their quality was low for indirect network analysis as no closed-loop study had been performed for achieving the quality necessary for estimating the effects of the treatments.

Risk of bias: (A) summary; (B) graphical representation of the risk of bias.
Discussion
Decisions regarding subsequent systemic therapy for mCRC, following disease progression beyond second-line treatments, primarily depend on the agents that were previously used.
24
-26
TAS-102 is an antimetabolic agent that can prolong the OS of heavily treated patients with mCRC by 2 months.
18
On the other hand, regorafenib and fruquintinib are small molecule inhibitors that block multiple kinases, including
The sequence of treatments in refractory mCRC is a crucial factor in prolonging the OS of patients, as an optimal treatment sequence can maximize the benefits of chemotherapy. 29 To date, there are no RCTs on the outcomes of different treatment sequences. The results of the RECOURSE study demonstrated that TAS-102 is superior to the placebo in patients who previously received regorafenib, whereas the results of the FRESCO study 10 revealed that fruquintinib is superior to the placebo in patients who previously received anti-VEGF therapy. It is worth mentioning that regorafenib turned out to be inferior to fruquintinib and TAS-102, with respect to the OS, but not with respect to the PFS, and we identified that the primary cause for this observation was the results of the CORRECT study. The CORRECT study comprised 760 patients (regorafenib [n = 505] and placebo [n = 255]); of which, 445 patients expired within 1 year (1-year OS), which was a rare phenomenon in the other studies. This may narrow the difference between the 2 groups, and prospective RCTs are urgently necessary for identifying the best treatment sequence in this setting. Taken together, the results of our study demonstrated that various factors should be taken into account when selecting the appropriate treatment option from among regorafenib, fruquintinib, and TAS-102, including the economy, toxicity, and patient selection.
Chen
30
The present study has several limitations with regard to the individual studies and indirect analysis performed herein. In this study, we performed a fewer number of direct comparisons, and therefore, the assessment was primarily based on indirect analysis, which is a major source of bias. Additionally, the data were obtained by reviewing published data instead of the raw data published with the articles. These 2 factors reduced the reliability of our study.
To summarize, the OS of regorafenib, TAS-102, and fruquintinib was found to be superior to that of the placebo in patients with mCRC. Regorafenib (80 mg) was found to be superior to the placebo, but inferior to 160 mg regorafenib, TAS-102, and fruquintinib. It was observed that the efficacy of 160 mg regorafenib, TAS-102, and fruquintinib in treating mCRC was identical, but their toxicity profiles were different. Therefore, any of the aforementioned agents can be used in the clinics for the treatment of mCRC.
Conclusion
Regorafenib (80 mg) was found to be superior to the placebo, but inferior to 160 mg regorafenib, TAS-102, and fruquintinib. It was further observed that the efficacies of TAS-102 and fruquintinib are similar, however, their toxicity profiles are different.
Footnotes
Authors’ Note
Gao was responsible for the design. Chen and Fan provided the administrative support. Cao and Bao were responsible for the collection and assembly of data. Fu and Gao were responsible for manuscript writing. All the authors approved the final manuscript. Our study did not require an ethical board approval because it did not contain human or animal trials.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
