Abstract
Objective
Inavolisib, a novel highly selective PI3Kα inhibitor, is approved for treating PIK3CA-mutated, hormone receptor-positive/human epidermal growth factor receptor 2-negative advanced breast cancer (PIK3CA-mutated, HR+/HER2− ABC). Given its significant efficacy, as confirmed by the updated INAVO120 III trial, but also its high price, a comprehensive evaluation of its value is warranted. The purpose of this study was to investigate whether inavolisib plus palbociclib–fulvestrant is cost-effective for patients with PIK3CA-mutated, HR+/HER2− ABC from the U.S. healthcare system perspective.
Methods
A Markov model comprising three health states was developed to simulate the progression of PIK3CA-mutated, HR+/HER2− ABC. Parametric survival models, fitted to and extrapolated from survival data, were used to estimate long-term clinical outcomes. Lifetime costs and health outcomes were calculated. Willingness-to-pay (WTP) thresholds were set at $100,000, $150,000, and $200,000 per quality-adjusted life year QALY to reflect general and more affluent regional standards. One-way and probabilistic sensitivity analyses were conducted to assess model robustness, and subgroup analyses explored the variation in benefits across patient clinical characteristics.
Results
Compared with placebo, inavolisib provided an additional 0.6 QALYs while increasing costs by $160,490.07, resulting in an incremental cost-effectiveness ratio (ICER) of $268,457.82 per QALY. Sensitivity analysis identified the utility of the progression-free state as the most sensitive factor in the model. Within the WTP threshold range of $100,000 to $200,000 per QALY, the inavolisib regimen was not considered cost-effective. To achieve cost-effectiveness at WTP thresholds of $100,000, $150,000, and $200,000 per QALY, the per-cycle price of inavolisib would need to be reduced to 59.5%, 72.0%, and 86.5% of its current price, respectively.
Conclusion
For patients with PIK3CA-mutated HR+/HER2− ABC, the inavolisib regimen is not cost-effective in the U.S. healthcare setting. Negotiating price reductions and adjusting decision thresholds based on patient characteristics may be viable strategies to meet the extensive treatment demand in the U.S.
Introduction
In the United States, breast cancer is the second leading cause of cancer death among women, with over 280,000 new cases diagnosed each year. 1 Although modern therapies can effectively prevent the recurrence of metastatic disease, about 15% of patients are eventually diagnosed with distant metastatic recurrence. 2 Hormone receptor-positive/human epidermal growth factor receptor 2-negative breast cancer represents the most frequent subtype, comprising 68% of all breast cancer cases. 3 For early-stage patients with this type of cancer, endocrine therapy is the standard treatment. While around 30% of early-stage patients initially respond to endocrine therapy, resistance inevitably emerges over time; notably, approximately 50% of patients with advanced or metastatic disease exhibit resistance to endocrine therapy. 4
Genetic alterations underlying this resistance include activating mutations in the PIK3CA gene, which are present in approximately 40% of HR+/HER2− breast cancers.4,5 Activating mutations in the PIK3CA gene not only lead to abnormal activation of the PI3Kα enzyme, resulting in continuous PI3K (phosphoinositide 3-kinase)/AKT (serine/threonine kinase)/mTOR (mechanistic target of rapamycin) pathway signaling and uncontrolled tumor growth, but also serve as a key driver of endocrine therapy resistance.4,6 Preclinical studies have demonstrated that concurrently targeting the PI3K pathway, Cyclin-Dependent Kinase 4 and 6 (CDK4/6), and estrogen receptor can enhance treatment response and delay the emergence of resistance in PIK3CA-mutated breast cancer models.7,8 This rationale has prompted the clinical development of targeted agents, including inavolisib—a potent, orally bioavailable PI3Kα inhibitor with a dual mechanism of action. On one hand, it shuts down the downstream PI3K/AKT/mTOR signaling pathway through enzyme inhibition, thereby suppressing tumor cell proliferation. 9 On the other hand, it directly degrades the mutant p110α protein (the catalytic subunit of the PI3Kα enzyme encoded by the PIK3CA gene), blocking signal transmission at its source and thereby reducing the risk of drug resistance. 10 However, the clinical development of PI3Kα inhibitors has faced challenges, as exemplified by previous combination regimens of alpelisib with CDK4/6 inhibitors, which were limited by adverse events including grade ≥3 hyperglycemia in 32.7% of patients. 11 Biochemical assays demonstrated that inavolisib exhibits over 300-fold greater selectivity for p110α (the alpha isoform of PI3K) compared to the p110β, δ, and γ isoforms. 10 This high selectivity profile translates to a widened therapeutic window, enabling enhanced efficacy when combined with standard treatment strategies, as demonstrated in the GO39374 trial. 12
It is noteworthy that the INAVO120 III clinical trial (NCT04191499), at the recent data cutoff (July10, 2025), was the first to compare the efficacy and safety of inavolisib plus palbociclib–fulvestrant versus placebo plus palbociclib–fulvestrant in patients with PIK3CA-mutated, HR+/HER2− locally advanced or metastatic breast cancer following prior endocrine therapy. 13 In this global Phase III trial, the inavolisib regimen demonstrated significant efficacy compared with placebo, with a statistically significant and clinically meaningful improvement in overall survival (OS) (34.0 vs 27.0 months, hazard ratio [HR] 0.67; 95% CI, 0.48 to 0.94). However, this clinical gain was accompanied by a higher incidence of grade 3 or 4 adverse events compared to the placebo arm (90.7% vs. 84.7%). In the United States, value assessment frameworks such as that developed by the Institute for Clinical and Economic Review (ICER) have been used to guide coverage decisions and clinical practice. 14 ICER’s framework integrates cost-effectiveness analysis with budget impact assessment, typically using WTP thresholds ranging from $100,000 to $150,000 per QALY to evaluate long-term value for money. 15 Within this framework, the clinical promise of PI3Kα inhibition must be carefully weighed against its economic implications, as interventions with incremental cost-effectiveness ratios (ICERs) exceeding $150,000 per QALY are often considered not cost-effective from a U.S. payer perspective. For instance, Alpelisib, the first approved PI3Kα inhibitor, when combined with fulvestrant in patients with PIK3CA-mutated, HR+/HER2− advanced breast cancer, yielded an incremental cost-effectiveness ratio (ICER) of $340,153.30 per QALY. 16 From a U.S. payer perspective, this regimen was not deemed cost-effective at current prices, despite a clinically meaningful 7.9-month median survival extension. This prior evidence raises a critical concern: whether inavolisib, despite its substantial clinical efficacy, may face a similar cost-effectiveness challenge in the same patient population, particularly given its associated high costs and modestly increased toxicity burden. To date, economic evidence specific to inavolisib in this setting remains lacking.
To address this evidence gap, the present study evaluates the cost-effectiveness of the inavolisib-based regimen versus placebo from a U.S. healthcare payer perspective among patients with PIK3CA-mutated HR+/HER2− advanced breast cancer following endocrine therapy. By integrating clinical efficacy, safety, and economic data, this analysis provides essential economic evidence to inform clinical decision-making and support the value-based management of metastatic breast cancer.
Methods
Patients and Regimens
In this trial, 325 patients with HR–positive, HER2-negative locally advanced or metastatic breast cancer were randomly assigned to receive inavolisib plus palbociclib–fulvestrant (inavolisib group) or placebo plus palbociclib fulvestrant (placebo group). Genetic testing revealed PIK3CA mutations in all patients, and the tumor PIK3CA mutation status in the majority of patients was assessed primarily by circulating tumor DNA (ctDNA) testing. Patients eligible for enrollment had experienced disease recurrence or progression within 12 months of completing adjuvant endocrine therapy. Additional inclusion criteria included a fasting blood glucose level below 126 mg/dL (7.0 mmol/L), an HbA1c (glycated hemoglobin) level below 6.0%, and measurable disease as defined by Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. 17 In inavolisib group, patients received inavolisib (at a dose of 9 mg, administered orally, once daily on days 1 to 28 of each 28-day cycle), each given in combination with palbociclib (at a dose of 125 mg, administered orally, once daily on days 1 to 21 of each 28-day cycle) and fulvestrant (at a dose of 500 mg, administered intramuscularly, on days 1 and 15 of cycle 1, and approximately every 4 weeks thereafter) until the occurrence of disease progression, unacceptable toxic effects, withdrawal of consent, or death. Patients in the placebo group received a once-daily placebo, with the dosing schedules for the placebo itself as well as the other two agents mirroring those of the inavolisib group. Following disease progression, 74.8% of patients in the inavolisib group and 75.7% in the control group received post-progression therapies, while the remaining individuals received palliative care. The second-line treatments included chemotherapy, antibody–drug conjugates, PI3K inhibitors, mTOR kinase inhibitors, and CDK4/6 inhibitors. The detailed distribution of patients receiving each regimen is provided in the appendix of the INAVO120 trial. 13
In this economic assessment, no individual patient-level data were used for analysis. As it does not meet the criteria for human research, review or exemption from an institutional review board is not required, nor is approval from an ethics committee necessary.
Analytic Model
A Markov model was constructed in TreeAge Pro 2024 utilizing data from the INAVO120 trial to simulate disease progression. The model consisted of three mutually exclusive health states: progression-free survival (PFS), progressed survival (PS), and death. All patients entered the model in the PFS state and were randomized to either the treatment or control arm. The model adopted a unidirectional transition structure, allowing patients to transition only from the PFS state to either the PS state or death over each Markov cycle (Supplemental Material Figure S1). In alignment with the trial’s dosing regimens, a 28-day cycle length was adopted for the Markov model, and transitions were permitted each cycle. To ensure that 99% of the cohort transitions to the death state, we set the time horizon to 30 years. This study evaluated total costs, QALYs, and the ICERs as primary outcomes, employing semicircular correlation analysis to estimate costs and survival rates. Costs were estimated from the U.S. payer’s perspective, incorporating only direct medical expenses. A 3% discount rate per year was applied for both cost and effectiveness in the U.S. 18 Consistent with empirical evidence, we established WTP thresholds at $100,000, $150,000 and $200,000 in the current analysis, the highest recommended threshold by Neumann et al. 19
Log-Logistic Parameters

θ: scale; κ: shape; ECF/ECX: docetaxel, oxaliplatin, leucovorin, fluorouracil; FLOT: epirubicin, cisplatin, fluorouracil or capecitabine.
Utility and Cost
For each subject, expected QALYs were estimated by multiplying the cumulative duration in each health state by the specific utility value assigned to that state. The following utility values were used: 0.85 (0.64-1.00) for the PFS state and 0.52 (0.39-0.65) for the PS state. Utility values were assumed to be identical for all patients in the same health state, regardless of treatment arm. To account for the impact of treatment-related Grade III/IV adverse events (AEs), the utility value of the progression-free state was adjusted by subtracting the disutility associated with AEs. Disutility parameters were derived from published literature, and the corresponding AE incidence rates were obtained from the results reported in the INAVO120 trial.16,20 Consistent with clinical practice and expert suggestions, the model incorporated grade III/IV AEs with an incidence rate of at least 5%, specifically hyperglycemia, stomatitis, neutropenia, thrombocytopenia, and anemia. The per-cycle utility decrement per patient was calculated as the sum of each AE’s incidence rate multiplied by its corresponding disutility value. Each AE was assumed to incur a one-time utility loss only in the cycle in which it occurred, thereby avoiding double counting. Throughout the model’s time horizon, QALYs were discounted at an annual rate of 3%.
Basic Parameters Input to the Model and the Ranges for Sensitivity Analyses

aAll costs were inflated to 2025 U.S. dollars using the Medical Care Consumer Price Index (Medical CPI). The adjustment was performed using the following formula: Adjusted Cost = Original Cost (Year Y) × (CPI_2025 / CPI_Year Y) where CPI_2025 is the annual average Medical CPI for 2025, and CPI_Year Y is the annual average Medical CPI for the original cost year. 23
Sensitivity Analysis
The robustness of our model was tested through one-way sensitivity analyses. A tornado diagram illustrates how variations in individual parameters influence the results. Details regarding the parameters’ median values, distributions, and the ranges used for these analyses are provided in Table 2. For parameters without such data, we applied a hypothetical range of ±20% around the base-case value. A probabilistic sensitivity analysis (PSA) was conducted to assess parameter uncertainty of the model through 10,000 Monte Carlo simulations. The results are presented in the form of cost-effectiveness acceptability curves (CEACs) to predict the probability of cost-effectiveness of treating according to the study protocol at WTP thresholds of $100,000–$200,000 per QALY. A treatment strategy was considered cost-effective if it either: (1) yielded more QALYs with cost savings, or (2) increased QALYs at a higher cost but with an ICER below the predefined WTP threshold.
Subgroup Analysis
In the subgroup analysis, all patient subgroups reported in the INAVO120 trial were included. It was assumed that all subgroups shared the same baseline survival function for both OS and PFS, with the only differentiating factor being the application of subgroup-specific hazard ratios (HRs). Specifically, the survival function for a given subgroup was derived by applying the corresponding HRS to the overall population survival function, as follows: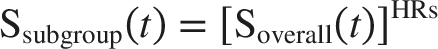
Result
Base-Case Results
Summary of Cost (US Dollars) and Outcome Results in the Base-Case Analysis

One-Way Sensitivity Analysis
The outcomes of the one-way sensitivity analysis were presented in a tornado diagram (Figure 1). The five most influential parameters were the utility of progression-free state, the cost of inavolisib, utilities of post progression state, discount rate, and the cost of Alpelisib. The utility of progression-free state was the most sensitive parameter affecting the ICER, causing the ICER to range from $206,664/QALY to $461,748/QALY. The analyses suggested that all parameters in defined ranges failed to make ICER lower than the $200,000/QALY WTP of threshold selected for affluent regions, and none of the parameters resulted in an ICER reaching the $100,000/QALY WTP threshold established for general regions. Tornado diagram for univariable sensitivity analyses. ICER incremental cost-effectiveness ratio.
Probabilistic Sensitivity Analysis
Considering the uncertainty of the model parameters, we conducted 10,000 iterations by varying all parameters simultaneously in a Monte Carlo simulation for probabilistic sensitivity analysis (PSA). Figure 2 shows the PSA results via a CEAC. The probability of inavolisib plus palbociclib-fulvestrant being cost-effective was 0% at a WTP threshold of $100,000/QALY, 3.3% at $150,000/QALY, and only 22.23% even when the threshold increased to $200,000 per QALY. The probability of inavolisib regimen being cost-effective exceeds 50% only when the WTP threshold surpasses $252,000/QALY. The inavolisib regimen reached cost-effectiveness in most cases (about 90%), and became cost-effective in virtually all cases at a WTP threshold above $573,000/QALY. Cost-effectiveness acceptability curves of inavolisib versus placebo group in patients with PIK3CA-mutated, HR+/HER2− locally advanced or metastatic breast cancer following prior endocrine therapy.
Scenario Analysis
We conducted a scenario analysis modeling progressive price reductions for inavolisib to explore its potential to achieve cost-effectiveness thresholds. In our probabilistic sensitivity analysis, we used WTP thresholds of $100,000, $150,000, and $200,000 per QALY, assessing the impact of drug price on the cost-effectiveness probability of the inavolisib regimen at these different values (Figure 3). When the price of inavolisib per cycle was reduced to 59.5 % and 42.8%, the proportions of simulations with cost-effectiveness for inavolisib regimen were 50% and 90%, respectively, at the WTP of $100,000/QALY. To achieve a 50% and 90% probability of cost-effectiveness, the per-cycle price of inavolisib must be reduced to 72% and 48% of its current price at the $150,000/QALY WTP threshold, and to 86.5% and 54% at the $200,000/QALY threshold, respectively. This scenario analysis demonstrates that to meet standard US cost-effectiveness benchmarks, a substantial reduction in the drug’s acquisition cost is essential. Probability of cost-effectiveness of inavolisib versus placebo group at different costs of inavolisib’price. QALY, quality-adjusted life-year; WTP, willingness-to-pay.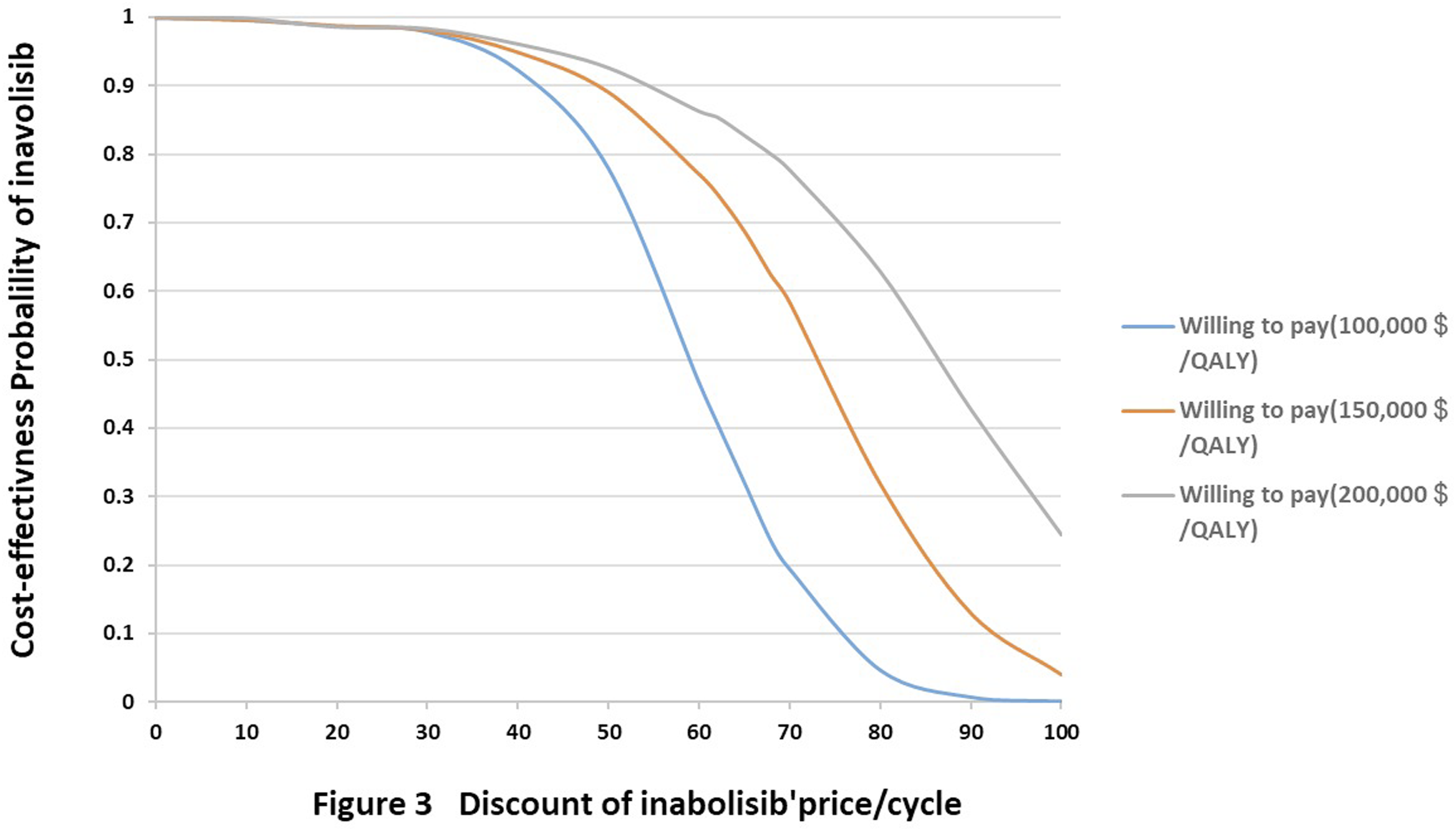
Subgroup Analysis
At the WTP threshold of $150,000/QALY, adding inavolisib was most cost-effective among the following subgroups, in descending order: patients with 2 organ metastases at enrollment (39.2%), patients outside of Asia, North America, or Western Europe (32.21%), HR-positive patients (14.71%). When the threshold was increased to $200,000/QALY, the list of potentially cost-effective subgroups expanded to include patients with two organ metastases at enrollment (81.91%), patients from regions outside Asia, North America, or Western Europe (77.41%),HR-positive patients (57.62%), patients aged ≤65 years (39.01%). The results of the subgroup analyses are presented in Supplemental Material Table S3.
Discussion
This is the first study to evaluate the cost-effectiveness of inavolisib plus palbociclib–fulvestrant versus placebo plus palbociclib–fulvestrant in patients with PIK3CA-mutated, HR+/HER2-advanced breast cancer following prior endocrine therapy by developing an economic model. In this population, the inavolisib treatment group demonstrated superior modeled efficacy outcome (i.e.,QALYs) compared to the control group, with an ICER of approximately $268,457.82/QALY gained for inavolisib. However, from the perspective of the U.S. healthcare system, inavolisib may not offer a cost-effective advantage under the current circumstances.
One-way sensitivity analysis identified the utility value for the PFS state as the most influential parameter in the model. As the PFS utility increased from 0.64 to 1.00, the ICER decreased from $461,748 to $206,664 per QALY, demonstrating a strong inverse relationship between the ICER and this utility parameter. Because the inavolisib group spent substantially more time in the PFS state than the placebo group (17.3 months vs. 7.3 months), improvements in the PFS utility value led to a disproportionately greater QALY gain for the treatment regimen, while the gain for the control egimen was relatively limited. If patients maintain a good quality of life during the progression-free period, the economic value of the inavolisib group would be substantially enhanced. Conversely, if the actual quality of life during PFS is lower (e.g., approaching the lower bound of the 95% confidence interval at 0.64), potentially due to treatment-related toxicity or other factors, the ICER would increase markedly, rendering the regimen not cost-effective. Given that the PFS utility value was identified as the most sensitive parameter, future research should prioritize the use of standardized, preference-based instruments such as the EQ-5D or SF-6D to systematically collect real-world health-related quality of life data from patients receiving inavolisib. 32 Such efforts would enable economic evaluations to be updated in line with real-world clinical outcomes, thereby providing more robust evidence to inform pricing and reimbursement decisions.
This study also focuses on the impact of price fluctuations of inavolisib, because its cost is the second factor affecting the results of sensitivity analysis, and the drug is still in the early stage of market supply, so its price may have potential room for price adjustment over time. Sensitivity analysis indicated that as the WTP threshold increases, the likelihood of inavolisib being cost-effective significantly rises. This finding is consistent with the earlier research on alpelisib by Wenhua Wu et al. 16 Given the heavy disease burden and steadily rising healthcare expenditures, we followed the recommendation of several economists and organizations by setting a maximum WTP threshold of US $200,000/QALY.2,19 Scenario analyses using different WTP thresholds demonstrated that the inavolisib regimen is still unlikely to be considered cost-effective under the current U.S. WTP when compared to the placebo regimen, based on results of long-term health benefits and cost differences. The findings demonstrate that at WTP thresholds of $100,000, $150,000, and $200,000 per QALY, the inavolisib group becomes cost-effective when its price is reduced to 59.5%, 72.0%, and 86.5% of the current level, respectively, with a cost-effectiveness probability exceeding 50%. Therefore, appropriately adjusting the price of inavolisib could be an effective approach to enhancing the economic value of this treatment regimen and could thereby secure a more favorable formulary position.
Moreover, we applied suitable HRs to determine whether the cost-effectiveness of inavolisib group varies in subgroups. The subgroup analysis demonstrated that the inavolisib regimen was most likely to be cost-effective in patients with no more than two organ metastases at enrollment. At WTP thresholds of $150,000/QALY and $200,000/QALY, the probabilities of cost-effectiveness reached 39.2% and 81.91%, respectively. Other potential subgroups that could achieve cost-effectiveness include patients outside of Asia, North America, or Western Europe and those with an ER-positive and PR-positive status. Their probabilities of cost-effectiveness each exceeded 50% at a WTP threshold of $200,000/QALY. Based on these findings, a benefit analysis could help identify which patients are better candidates for the treatment. From an economic perspective, this precision screening based on patient characteristics provides a basis for reimbursement decisions to better match the benefits of specific patient groups. Notably, several health technology assessment agencies have recognized the use of thresholds based on prevalence adjustment or disease severity, rather than applying a uniform threshold across all indications, which is consistent with our findings. 15
Although CDK4/6 inhibitors (i.e., palbociclib) combined with endocrine therapy have been the first-line treatment for PIK3CA-mutated, HR+/HER2- ABC, drug resistance remains a significant challenge. 33 Given the multiple resistance mechanisms, such as alterations in the PI3K/AKT/mTOR signaling pathway and ESR1 mutations, we should perform precise molecular testing upon disease progression, focus on mutations within these pathways, and provide targeted treatment. 34 Compared to capivasertib, which targets the midstream of the pathway, and everolimus, which acts downstream, inavolisib offers a novel upstream intervention with potentially stronger efficacy.35,36 In contrast, alpelisib, also a PI3K inhibitor, is associated with more prominent toxicities such as hyperglycemia, diarrhea, and rash, and has received only a Category 2A recommendation in the NCCN guidelines, whereas inavolisib has achieved a Category 1 recommendation in the latest guidelines. 37 It should be noted that the patient populations in the INAVO120 and SOLAR-1 trials differed. A head-to-head phase III trial, INAVO121, is currently underway to directly compare the efficacy and safety of inavolisib versus alpelisib in patients who have progressed on prior CDK4/6 inhibitor therapy.
We acknowledge several limitations in this study. First, although validated extrapolation techniques were employed, uncertainty remains regarding outcomes beyond the follow-up period of the INAVO120 trial. Second, the utility values used in this model were derived from published literature and may not fully capture the health-related quality of life specific to the patient population under investigation. Third, treatment pathways in the trial setting were protocol-driven and may not fully capture the heterogeneity of real-world clinical practice in the United States. As such, uncertainty inherent in the treatment sequencing assumptions remains and may influence long-term cost and outcome estimates. In addition, the impact of adverse drug events was not fully captured, as less severe adverse events were excluded. The sensitivity analysis confirmed the robustness of the findings, showing that AEs exerted only a modest influence across the two treatment strategies. Finally, as a newly approved drug in the United States, the market price of inavolisib has not yet stabilized, and evolving U.S. healthcare policy represents an important source of uncertainty in its pharmacoeconomic evaluation. Furthermore, the head-to-head phase III trial comparing inavolisib with the similar agent alpelisib—the INAVO121 study—is still ongoing. Therefore, ongoing monitoring of relevant healthcare policies and timely updates to model inputs are warranted.
Conclusions
To our knowledge, from the perspective of US payers, evaluating the use of inavolisib combined with palbociclib-fulvestrant in patients with PIK3CA-mutated, HR+/HER2- ABC is not cost-effective compared with the placebo receiving palbociclib-fulvestrant. Although inavolisib has shown significant benefits in clinical trials, its cost needs to be reduced to provide more favorable economic benefits and improve patient accessibility.
Supplemental Material
Supplemental Material - Economic Evaluation of Inavolisib Combined With Palbociclib–Fulvestrant for PIK3CA-Mutated, HR+/HER2− Advanced Breast Cancer in USA
Supplemental Material for Economic Evaluation of Inavolisib Combined With Palbociclib–Fulvestrant for PIK3CA-Mutated, HR+/HER2− Advanced Breast Cancer in USA by Hanqing Zeng, Li-Ying Song, Ren Guo, Dan Ding, Xiaohui Zeng and Qiao Liu in Technology in Cancer Research & Treatment
Footnotes
Ethical Considerations
This study does not involve human participants or animal subjects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Provincial Natural Science Foundation [Grant numbers 2021JJ80080].
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
No additional data available.
Provenance and Peer Review
Not commissioned; externally peer reviewed.
Trial Registration Number
NCT04191449.
Supplemental Material
Supplemental material for this article is available online.
