Abstract
Background
Parenteral glutamine (Gln) supplementation can enhance the immune function of patients with colorectal cancer(CRC), regulate inflammatory response, nitrogen balance and protein synthesis, reduce the morbidity of postoperative complications and shorten the length of hospitalization. However, some guidelines and clinical studies have questioned the rationality of using glutamine-containing immunonutrition support in the perioperative period of CRC. Therefore, we conducted a meta-analysis of the effects of perioperative glutamine-enriched parenteral nutrition on short-term postoperative clinical outcomes in patients with CRC.
Methods
A comprehensive search of all relevant literature from the default date to June 2025 was performed using the following databases: PubMed, Embase, Web of Science, Cochrane Library, China Biology Medicine Database (CBM), China National Knowledge Infrastructure (CNKI), VIP Medical Information System (VIP), and Wanfang electronic database. RevMan 5.3 software was used for the meta-analysis. We calculated the outcomes using random- and fixed-effects models.
Results
Seventeen single-center randomized controlled trials (RCTs) involving 950 patients with CRC were included. The control group consisted of 470 patients who received traditional parenteral nutrition therapy and the experimental group consisted of 480 patients who received parenteral nutrition with Gln. The analyses showed that perioperative Gln-enhanced parenteral nutrition reduced the morbidity of infectious complications (Relative Risk [RR]=0.36,95% Confidence Interval [CI]:0.23–0.58) and non-infectious complications (RR=0.27, 95% CI: 0.13–0.55). The length of hospitalization was reduced by 2.18 days (mean difference [MD] = -2.18, 95% CI: -2.59–-1.78).
Conclusion
Parenteral Gln supplementation may potentially reduced the morbidity of postoperative complications, shortened postoperative hospitalization, improved some aspects of nutritional status, immune and inflammation function in patients with CRC, based on current evidence of low-to-moderate certainty.
Introduction
According to the latest global cancer statistics for 2020, 1 there were 560,000 new cases of colorectal cancer (CRC) and 290,000 deaths in China, ranking it 2nd in the cancer incidence spectrum and 5th in the cause of death spectrum. Currently, surgery-based comprehensive treatment is the main treatment for resectable CRC, 2 However, due to the high catabolic state of patients with tumors, ischemia-reperfusion injury caused by surgery and postoperative intestinal dysfunction can easily lead to impaired nutrient intake and impaired absorption. Previous studies have reported that the incidence of malnutrition in patients with CRC is as high as 45% to 60%. 3
Malnutrition often leads to decreased immune function, altered inflammatory response, delayed or failed wound healing, prolonged postoperative hospitalization, 4 and increased medical costs. 5 Therefore, perioperative nutritional support is crucial for patients with gastrointestinal cancer, particularly those undergoing surgery. 6 Glutamine (Gln) has immunomodulatory functions and plays an important role in nutritional metabolism and mammalian host health. 7 In highly catabolic scenarios, such as trauma, surgery, or infection, the body’s requirements are increased, while its production capacity is impaired, and the dietary intake of Gln and endogenously synthesized Gln are insufficient to meet the body’s requirements; in such cases, Gln supplementation may be required.8,9 Although enteral feeding is the preferred method of nutritional support, the provision of parenteral nutritional supplements can be clinically beneficial. Some patients are unable to receive enteral feeding because of tumor obstruction and delayed postoperative intestinal recovery; it is clinically beneficial to provide them with non-enteric nutritional supplements. Numerous studies have shown that parenteral supplementation with Gln increases the intestinal lymphocyte count, thereby modulating the immune function in the mucosa of the small intestine and reducing intestinal permeability and bacterial translocation.10-12 In addition, it also regulates the inflammatory response, nitrogen balance and protein synthesis, and ultimately reduces the morbidity of infection.13,14
However, the 2021 European Society for Clinical Nutrition and Metabolism stated that the degree to which immunonutritional support with parenteral supplementation of Gln positively affects patients undergoing colorectal surgery is unclear. 15 In addition, some reports have raised doubts regarding the reasonableness of routine use of immune nutrition before colon cancer surgery. For instance, a randomized controlled trial (RCT) by Lee et al. 16 which included 176 patients with colon cancer, administered an oral immunonutrition supplement (400 mL/day) to the intervention group (n = 88) for 7 days prior to surgery. The results indicated no significant differences between the immunonutrition group and the control group in terms of morbidity of infectious complications (17.7% vs. 15.9%, P = 0.751) or overall morbidity of complications (31.6% vs. 29.3%, P = 0.743). The length of hospital stay was also comparable between the two groups (7.6 ± 2.5 days vs. 7.4 ± 2.3 days, P = 0.635). Furthermore, a network meta-analysis by Ye et al. 17 which incorporated 34 RCTs involving 2,841 participants, found that Gln supplementation did not yield significant benefits in maintaining serum albumin level, body weight, or body mass index in patients undergoing CRC surgery or chemotherapy. These findings contradict the conclusions of some previous studies, thereby raising questions about the rationale for employing immunonutrition, particularly Gln, in CRC. To further investigate the efficacy of immunonutrition, represented by Gln, in patients undergoing CRC surgery, we conducted a meta-analysis focusing on the short-term postoperative clinical outcomes in these patients who received parenteral nutrition support during the perioperative period. This study aims to provide an evidence-based evaluation of the role of parenteral Gln supplementation as an immunonutrition strategy for CRC patients.
Materials and Methods
The reporting of this systematic review and meta-analysis was designed and conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement. 18 As a meta-analysis utilizing only data extracted from previously published studies, this research did not involve direct interaction with human subjects. Therefore, in accordance with standard ethical guidelines for secondary research, it was classified as exempt from ethical approval processes, including the need for informed consent.
Literature Search
Two researchers independently searched the following databases: PubMed, Embase, Web of Science, Cochrane Library, Chinese Biomedical Database (CBM), China National Knowledge Infrastructure (CNKI), VIP Medical Information System (VIP), and the Wanfang Electronic Database. The search period was from the default time to June 2025 and the search strategy was conducted according to the patient/population, intervention, comparison, and outcomes (PICOS) principles, which were based on the terms “Rectal Neoplasms” and “Colorectal Neoplasms”, “Colonic Neoplasms”, “Glutamine” and “randomized controlled trial” as subject terms combined with their corresponding free words in all possible combinations. Other articles were searched manually in the reference lists of articles selected for the full-text search. For overlapping authors, institutions, or patients, the most recent or highest-quality article was selected. Finally, the two researchers compared their respective search results and discussed the inconsistent results to reach a consensus; otherwise, a third researcher was involved in conducting a discussion to decide whether to retain or discard the results.
Inclusion and Exclusion Criteria
Inclusion criteria: (1) Population (P): patients diagnosed with CRC, undergoing elective radical R0 resection, without distant metastasis; no age, gender, race and ethnicity, or location restrictions; (2) Intervention (I): perioperative parenteral nutritional support enhanced with Gln, no restriction of the dose or maintenance time; (3) Comparison(C): blank control or isonitrogenous and isocaloric conventional nutritional support in the control group, and parenteral administration of Gln in addition to the control group conditions in the experimental group; (4) Outcome (O): included studies were required to include at least one of the following outcome indicators, infectious complications, non-infectious complications, postoperative hospitalization, nutritional status indicators [albumin, prealbumin, peripheral blood lymphocyte count], nitrogen balance, immune function indicators (IgG, IgM, CD3+, CD4+, CD8+, CD4+/CD8+), inflammation indicators [tumor necrosis factor α (TNF-α)]; (5) Study design (S): only studies of RCTs were included, whether single-center or multi-center. Exclusion criteria: (1)Studies with co-morbidities (e.g.intestinal obstruction, etc.): or combined with other diseases; (2) Animal experiments; (3) Intervention modalities that involved radiation therapy, chemotherapy, or combinations of other immunonutrients (e.g., probiotics, arginine, nucleotides, and omega-3 polyunsaturated fatty acids etc.); (4) Studies of perioperative enteral supplementation of Gln; (5) Narrative literature review, meta-analysis, and inability to download the complete full text; (6) non-RCTs; (7) retrospective studies; and (8) inconsistent reported outcome indicators; those who met any of the above were excluded.
Literature Screening and Data Extraction
Two researchers, respectively, evaluated all the literature retrieved according to the search strategy. First, the retrieved literature was imported into Notepress software to automatically remove duplicates. Second, after removing duplicates the literature was reviewed for titles and abstracts, and literature such as reviews, meta-analyses, animal experiments, and incompatible studies were deleted. Third, the literature was downloaded in full text. The literature was read in full to confirm eligibility. When the two researchers had different opinions on the included literature, a third researcher was involved in the negotiation to reach a consensus. The extracted data including the study name, year, tumor type, sample size, age, sex, Gln dose, timing, maintenance time, and outcome indicators were entered into a pre-established Excel spreadsheet.
Literature Quality Evaluation and Risk of Bias Assessment
The included literature was assessed independently by two researchers according to the Cochrane Risk of Bias assessment tool for literature quality, 19 which included: (i) the method of randomization, (ii) allocation concealment, (iii) whether blinding was used, (iv) the completeness of outcome data, (v) the selective reporting of results, and (vi) other sources of bias (e.g., early trial termination, inconsistent baseline). Colors (green, red, yellow) and symbols (“+”, “-”, “?”) are used to indicate “low risk of bias,” “high risk of bias”, and “unclear”, respectively. In cases of disagreement, the two researchers discussed and reached a consensus; otherwise, a third researcher’s opinion was sought to reach a final agreement.
Outcome Measures
The main measures were the morbidity of infectious and non-infectious complications and the length of postoperative hospitalization. The secondary outcomes were nutritional status (albumin, prealbumin, peripheral blood lymphocyte count), nitrogen balance, immune function (IgG, IgM, CD3+, CD4+, CD8+, CD4+/CD8+), and inflammatory level (TNF-α).
Data Synthesis and Statistical Analysis
The meta-analysis was based on the principles of the Cochrane Handbook for Systematic Reviews of Interventions. The meta-analysis was performed using RevMan 5.3 statistical software and the corresponding forest plots were generated to visualize the combined results. For continuous variables (postoperative length of hospitalization, albumin, prealbumin, peripheral blood lymphocyte count, nitrogen balance, IgG, IgM, CD3+, CD4+, CD8+, CD4+/CD8+ and TNF-α), pooled results were expressed as mean difference (MD) and 95% confidence intervals (95% CI), if included studies were analyzed at different measurement scales, the pooled results were then expressed using standardized mean difference (SMD). For dichotomous variables (complications), the pooled results were expressed as the relative risk (RR) and 95% CI. Heterogeneity was examined using Cochran’s Q test and quantified by the I2 statistic (low: I2 < 50%; medium: 50% ≤ I2 ≤ 75%; high: I2 > 75%). Meta-analysis was performed if I2 < 50% at the baseline level, otherwise rounded. For endpoint levels, meta-analysis was performed using the Mantel-Haenszel fixed-effects model if I2 ≤ 50% and the DerSimonian-Laird random-effects model if I2 > 50%. P<0.05 was considered statistically significant. For highly heterogeneous studies (I2 >75%), to explore the source of heterogeneity, we conducted sensitivity analysis or subgroup analysis by excluding studies individually to explore possible statistical or clinical heterogeneity. Finally, we used visual inspection of funnel plots to investigate the publication bias when 10 or more studies were included in the meta-analysis.
Results
Literature Screening Results
A total of 540 articles were obtained in the initial inspection, 211 were automatically removed as duplicates, 192 were excluded after reading the titles and abstracts, 120 were excluded after reading the full text of the remaining 137 potentially included articles (9 were combined with co-morbidities, 14 were combined with other immunonutrients, 47 were related to radiation or chemotherapy in the interventions, 18 were combined with other diseases, 5 did not match the study design, 3 did not match outcome indicators, 7 had inaccessible full text, 1 had duplicated literature, 6 had irrelevant content, and 10 had enteral nutrition). Finally, 17 articles were included. Among them, 2 written in English and 15 written in Chinese (Figure 1). A PRISMA flow diagram is presented to illustrate the screening of studies
Basic Characteristics of the Included Literature
The Basic Characteristics of the Literature

Note. CRC: colorectal cancer; RC: rectal cancer; CC: colon cancer; POD: postoperative; Pre-op: preoperative; NR: not mentioned; ①Albumin ②Prealbumin ③Peripheral blood lymphocyte count ④Nitrogen balance ⑤CD3+ ⑥CD4+ ⑦CD8+ ⑧CD4+/CD8+ ⑨IgG ⑩IgM ⑪TNF-α ⑫Infectious complications ⑬Non-infectious complications ⑭Length of postoperative hospital stay.
Quality Evaluation Results
With regard to selection bias in the 17 studies, three studies were randomized by the random number table method, one was randomized according to the order of surgery, and one was randomized according to the different protocols of perioperative medication. The remaining 12 studies only stated “randomization” without describing the specific method, which was rated as “low risk”. None of the studies described whether the allocation scheme was hidden and were rated as “unclear”. One study was blinded to the investigator and subjects by having the infusion solution prepared by a clinical pharmacist who was not involved in the study and was rated as “low risk”. The remaining studies were not blinded and were rated as “high risk”. The risk of bias from incomplete outcome data or selective reporting of findings was low because the majority of the included RCTs adequately described all outcomes of interest, which was “low risk”. No other bias was found in the remaining studies, which were “low risk” (Figure 2). (A) Summary plot of risk of bias and (B) plot of risk of bias for each study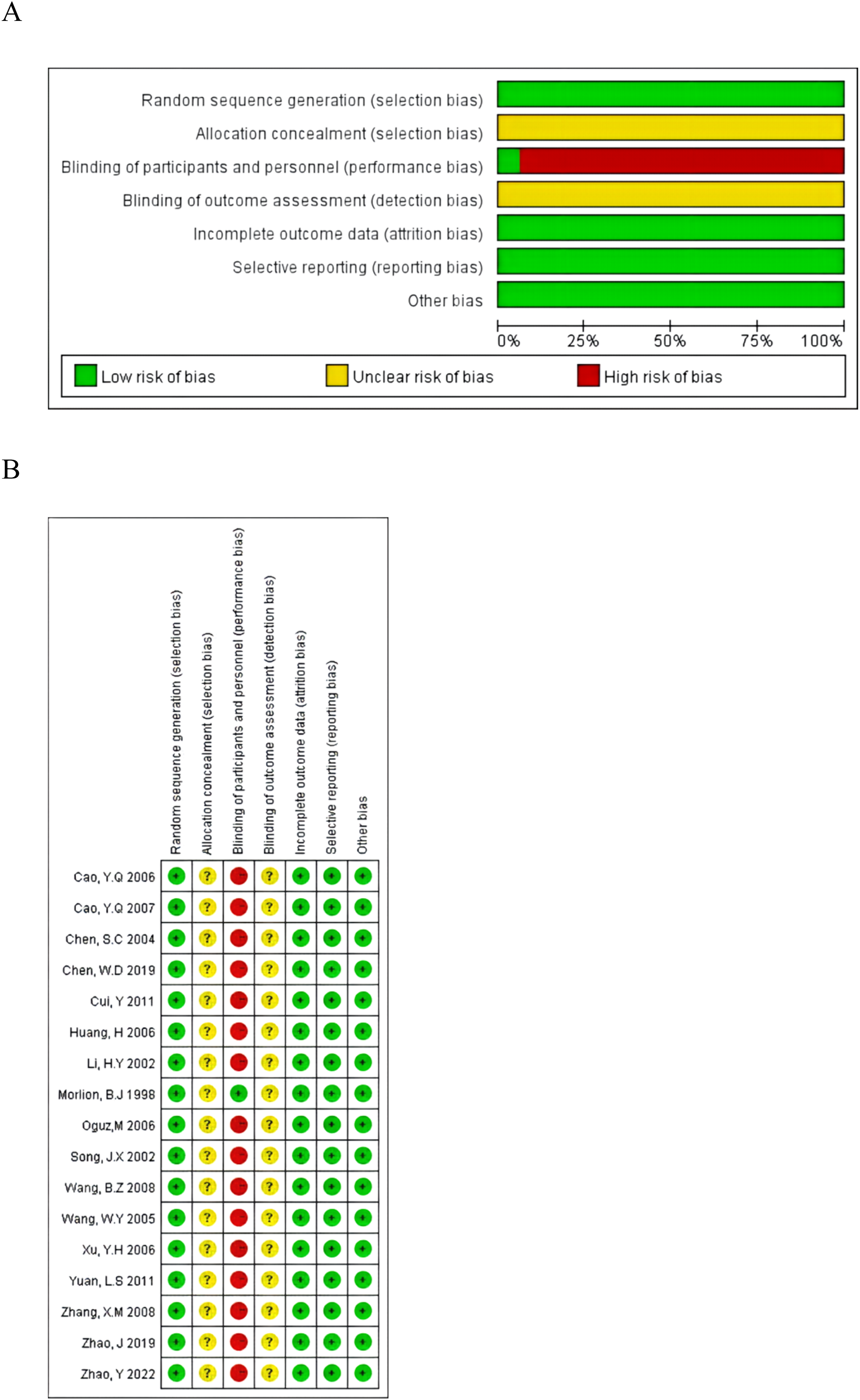
Meta-Analysis Results
Infectious Complications
Nine studies20-28 reported the effect of parenteral Gln supplementation on the morbidity of postoperative infectious complications in patients with CRC, with an morbidity of infectious complications of 7.05% in the experimental group and 19.31% in the control group. Fixed-effects model analysis showed that Gln supplementation initiated before surgery reduced postoperative infectious complications (RR =0.30, 95% CI: 0.17-0.55, Z=4.00, P<0.0001). However, there was no significant difference in the morbidity of postoperative complications in which Gln supplementation was initiated after surgery (RR =0.50, 95%CI: 0.23-1.12, Z=1.67, P=0.09). The overall analysis showed that Gln reduced the morbidity of postoperative infectious complications (RR=0.36, 95%CI:0.23-0.58, Z=4.27, P<0.0001) (Figure 3). Forest plots of the morbidity of infectious complications.The area of each gray square represents the weight of the study, and horizontal lines represent the 95% CI, and the solid vertical line represents null effect. Black diamonds represent The overall effect estimate. Using the fixed-effects models
Non-Infectious Complications
Seven studies20,21,24,27-30 reported the postoperative non-infectious complications. The morbidity of non-infectious complications was 3.03% in the experimental group and 13.22% in the control group. The fixed-effects model analysis showed that the difference was statistically significant between the two groups. Gln supplementation reduced the morbidity of postoperative non-infectious complications (RR =0.27, 95%CI: 0.13-0.55, Z = 3.56, P=0.0004) (Figure 4). Forest plots of the morbidity of non-infectious complications. The area of each gray square represents the weight of the study, and horizontal lines represent the 95% CI, and the solid vertical line represents null effect. Black diamonds represent the overall effect estimate. Using the fixed-effects models
Postoperative Hospitalization
Five studies23,24,26,28,31 reported postoperative hospitalization. Random effects model analysis showed that Gln supplementation shortened postoperative hospitalization by 2.64 days (MD =-2.64, 95%CI: -4.26 to -1.01, Z=3.18, P=0.001), but the heterogeneity was high (I2=85%). The study by Morlion et al. was found to have a greater impact on the results by the one-by-one exclusion method, and after excluding it, the heterogeneity decreased to I2=0%. Fixed-effects model analysis showed that patients supplemented with Gln had postoperative hospitalization shortened by 2.18 days and the result still indicated a statistically significant reduction in hospital stay (MD = -2.18, 95% CI: -2.59 to -1.78, Z=10.67, P<0.00001) (Figure 5). This suggests a consistent overall trend, while the initial high heterogeneity was primarily driven by this single trial. Forest plots of length of hospital stay (A) and forest plots of length of hospital stay after excluding the study by Morlion et al (B). The area of each gray square represents the weight of the study, and horizontal lines represent the 95% CI, and the solid vertical line represents null effect. Black diamonds represent the overall effect estimate. Using the random-effects and fixed-effects models
Nutritional Status Indicators
Regarding the effect of parenteral Gln supplementation on the postoperative nutritional status of patients with CRC, Twelve studies20,21,23-27,29,30,32-34 reported the albumin level and five studies20,21,25,27,30 reported the prealbumin level. Four studies24,29,30,32 reported the peripheral blood lymphocyte count. The random effects model analysis showed that the difference in the postoperative albumin level between the two groups was statistically significant if parenteral Gln supplementation was initiated in the preoperative period (MD = 4.39 g/L, 95% CI: 2.80-5.99, Z=5.40, P<0.00001). In contrast, there was no statistically significant difference in postoperative parenteral addition of Gln alone (MD = 0.95 g/L, 95% CI: -0.39-2.29, Z=1.39, P=0.16). The overall analysis showed that the difference between the two groups was statistically significant (MD = 2.32 g/L, 95%CI: 1.06-3.58, Z=3.61, P=0.0003) (Figure 6A). In the funnel plots of the twelve included studies, visual inspection was essentially symmetrical, suggesting no significant publication bias (Figure 7). In addition, the prealbumin levels (MD = 12.34 mg/L, 95%CI: 8.33-16.35, Z=6.03, P<0.00001) and the peripheral blood lymphocyte count (MD = 0.21× 109/L, 95%CI: 0.07-0.35, Z=2.94, P=0.003) Compared, after stratified analysis based on the timing of Gln supplementation, similar results were obtained, and the differences were all statistically significant (Figure 6B and 6C). Forest plots of albumin (A), prealbumin (B), peripheral blood lymphocyte count (C). The area of each gray square represents the weight of the study, and horizontal lines represent the 95% CI, and the solid vertical line represents null effect. Black diamonds represent the overall effect estimate. Using the random-effects and fixed-effects models Funnel plot of albumin

Cellular Immune Function Indicators
Regarding cellular immune function, four studies29,30,32,35 reported the CD3+ level, seven studies21,27,29,30,32,33,35 reported the CD4+ level, Six studies21,27,30,32,33,35 reported the CD8+ level and three studies21,30,35 reported the CD4+/CD8+ ratio. The results showed that Gln supplementation enhanced partial cellular immune function, this was demonstrated by statistically significant differences in the postoperative CD4+ level (MD = 5.14%, 95% CI: 3.73-6.55, Z=7.13, P<0.0001) and the CD4+/CD8+ ratio (MD = 0.30, 95% CI: 0.02-0.58, Z=2.10, P=0.04) in two groups. However, there was no difference in the CD3+ level (MD = 2.41%, 95%CI: -1.23-6.04, Z=1.30, P=0.19). For the CD8+ level, preoperative initiation of Gln supplementation was statistically significantly different between the two groups (MD = -5.73%, 95% CI: -9.39 ∼ -2.07, Z=3.07, P=0.002), and postoperative initiation yielded effect (MD = -1.14%, 95% CI: -7.58 - 5.30, Z=0.35, P=0.73) and the total effect (MD = -3.31%, 95% CI: -7.57 to -0.95, Z=1.52, P=0.13) were not statistically significant different between two groups (Figure 8). Forest plots of CD3+ level (A), CD4+ level (B), CD8+ level (C), CD4+/CD8+ ratio (D). The area of each gray square represents the weight of the study, and horizontal lines represent the 95% CI, and the solid vertical line represents null effect. Black diamonds represent the overall effect estimate. Using the random-effects and fixed-effects models
Humoral Immune Indicators
Regarding humoral immune function, four studies25,32,33,35 reported IgG and IgM levels. Pooled analysis showed that after parenteral Gln supplementation, the difference was statistically significant between two groups, with the experimental group having higher IgG (SMD = 0.77, 95% CI:0.23 -1.31, Z=2.79, P=0.005) and IgM levels (MD = 0.19 g/L, 95% CI: 0.10-0.29, Z=4.06, P<0.0001) compared to the control group (Figure 9). Forest plots of IgG level (A), IgM level (B). The area of each gray square represents the weight of the study, and horizontal lines represent the 95% CI, and the solid vertical line represents null effect. Black diamonds represent the overall effect estimate. Using the random-effects and fixed-effects models
TNF-α, Nitrogen Balance
In terms of regulating the inflammation level and negative nitrogen balance, two studies22,36 reported the TNF-α level, and three studies25,26,29 reported nitrogen balance. The pooled analysis showed that the level of TNF-α(SMD = -1.49, 95%CI: -2.45 to -0.53, Z=3.05, P=0.002) and negative nitrogen balance (MD =1.38g/d, 95%CI: 0.58-2.17, Z=3.40, P=0.0007) was lower in experimental group (Figure 10). Forest plots of TNF-α level (A), nitrogen balance (B). The area of each gray square represents the weight of the study, and horizontal lines represent the 95% CI, and the solid vertical line represents null effect. Black diamonds represent The overall effect estimate. Using the random-effects and fixed-effects models
Sensitivity Analyses
We performed sensitivity analysis using the removing one by one method for pooled outcomes with marked heterogeneity (I2 > 75%) to evaluate the robustness of the combined effect estimates. Results indicated that for indicators including albumin, peripheral blood lymphocyte count, CD3+, and CD8+, omitting any single study did not substantially alter the pooled effect size or its confidence interval, suggesting good robustness of these findings (Figure 11). In contrast, sensitivity analysis for TNF-α showed that the pooled estimate was sensitive to the exclusion of individual studies, indicating limited robustness of the conclusion. This is likely attributable to the small number of studies included for this outcome, which makes the result susceptible to influence from individual trials. Therefore, the current pooled result for TNF-α should be interpreted with caution, and further validation with more studies is warranted in the future. Sensitivity analyses was performed on the pooled analysis results with high heterogeneity (I2 >75%) to verify the robustness of the pooled analysis for albumin level (A), peripheral blood lymphocyte count (B), CD3+ level (C), CD8+ level (D), TNF-α level (E)
Discussion
Gln is the most abundant free amino acid in the human body and is mainly stored in skeletal muscle. In tumors with high metabolism, the continuous release of Gln by skeletal muscle leads to the depletion of Gln systemically,37,38 which negatively affects immune function, intestinal barrier integrity, and intestinal flora. There have been several clinical studies on perioperative Gln supplementation in patients with CRC; however, the vast majority were single-center, small-sample studies, and the results varied, failing to reach a consistent conclusion. Therefore, this study comprehensively searched the domestic and international literature on parenteral Gln supplementation in patients with CRC and included 17 RCTs for meta-analysis. Our study showed that parenteral Gln supplementation reduced the morbidity of postoperative complications, shortened the length of postoperative hospitalization, improved nutritional status, enhanced immune function, reduced postoperative inflammation, and corrected negative nitrogen balance, in patients with CRC.
The initial use of Gln in clinical practice has mostly been in critically patients. A meta-analysis by Stehle et al. 39 involving 16 RCTs with 842 critically patients showed that parenteral Gln supplementation, based on balanced nutrition and clinical guidelines, significantly reduced hospital mortality, morbidity of infectious complications, and length of postoperative hospitalization. Another meta-analysis that included 40 RCTs also showed that parenteral Gln supplementation in critically patients significantly reduced infectious complications and shortened the length of postoperative hospitalization by 2.35 days but failed to reduce mortality. 40 In recent years, it has been found that most patients with tumors have an impaired nutritional status. Malnutrition is an independent risk factor for complications after gastrointestinal surgery41,42 and inpatient admission with a risk of malnutrition prolongs postoperative hospitalization.43,44 With the widespread application of immunonutritional therapy during the perioperative period for gastrointestinal malignancies, many studies have explored the clinical efficacy of Gln. A recent retrospective study of patients who had 1,004 CRC surgeries (of whom 660 received intravenous Gln supplementation) found that the morbidity of postoperative complications in the Gln and control groups was 14.9% and 36.8%, respectively. The morbidity of postoperative infection in the Gln group was significantly lower than that in the control group (10.5% vs. 28.9%), and postoperative hospitalization was significantly shorter (P<0.001). A study by Yang et al., 44 which included 31 studies with a total of 2,201 postoperative patients with CRC undergoing radical surgery, showed that the surgical site infection (RR=0.48,95%CI:0.30 - 0.75, P=0.001), anastomotic leakage (RR=0.23,95% CI: 0.09 - 0.61, P=0.003), and postoperative hospitalization (SMD=-1.13,95% CI: -1.68 ∼ -0.58, P=0.000) were significantly lower in Gln group.
These findings are consistent with our analysis showing that parenteral Gln supplementation reduced the morbidity of postoperative infectious and non-infectious complications and shortened postoperative hospitalization in patients with CRC. For infectious complications, which are of greatest concern in clinical practice, our subgroup analysis found that Gln supplementation, initiated preoperatively, reduced its postoperative occurrence, but not if it was initiated only from the postoperative period. It is hypothesized that preoperative administration can achieve sufficient plasma immunonutrient concentration in the early postoperative period to produce a positive effect. Therefore, we suggest that Gln supplementation is initiated before surgery. Another point that should not be overlooked is that four of the studies in the meta-analysis by Yang et al. were supplemented with Gln parenterally, and some patients received Gln in combination with other immunonutrients, such as arginine and probiotics, which may differ from the effects of parenteral application of Gln alone. Finally, the results for shortened postoperative hospitalization after parenteral addition of Gln should also be analyzed with caution. First, the initial pooled analysis showed significant statistical heterogeneity (I2 = 85%). This heterogeneity was largely attributable to the strong effect observed in a single trial. Second, discharge criteria and the application of ERAS (enhanced recovery after surgery) protocols varied considerably and were not uniformly reported across the included trials. According to Greco et al., an effective ERAS protocol could reduce postoperative hospitalization by almost 2 days. 45 Therefore, the observed effect may not be solely attributable to Gln supplementation but could reflect its synergistic contribution within an ERAS framework or the influence of other unmeasured confounding factors. Future trials should more rigorously control and report core ERAS elements to better isolate the independent effect of Gin.
Perioperative Gln immunonutritional support had beneficial effects on nitrogen metabolism, inflammatory factors, and immunological parameters. In animal studies, Lin et al. 46 found that Gln-rich parenteral nutrition can reduce the expression of leukocyte integrins after gastrectomy and trigger a faster immune response to injury. Wang et al. 47 found that Gln parenteral nutrition supplementation improved innate immunity and resistance to bacterial mucosal invasion, in mice, demonstrating the potential importance of parenteral nutrition in protecting the intestinal mucosa. In a clinical study, Lu et al. 48 found that patients with a gastrointestinal tumor who received Gln parenteral supplementation seven days after surgery had a higher serum prealbumin level (P=0.039) and nitrogen balance (P=0.048), and the IL-6 level (P=0.01) and serum C-reactive protein level (P=0.013) were significantly lower than those in the standard group. Cheng et al. 49 treated 87 elderly patients with gastrointestinal tumors with parenteral nutrition combined with Gln 0.6 g/(Kg·d) for eight days and the results showed that albumin, prealbumin, and transferrin decreased significantly on postoperative day four (P<0.05) and recovered significantly on postoperative day eight (P<0.05). Nitrogen balance was poor in the early postoperative period but recovered significantly on postoperative day eight (P<0.05).
A recent meta-analysis of 26 RCTs involving 1,678 CRC patients showed that Gln significantly increased postoperative plasma protein marker levels (albumin [SMD= 0.79,95% CI: 0.55 ∼ 1.03], proalbumin [SMD= 0.94, 95%CI: 0.69 ∼ 1.20], nitrogen balance [SMD= 1.11, 95%CI: 0.46 ∼ 1.75]. Conversely, TNF-α [SMD= -1.86, 95%CI: -2.21 ∼ -1.59] and C-reactive protein [SMD= -1.94,95% CI: -2.41 ∼ -1.48] decreased significantly. 50 The above study is consistent with our meta-analysis showing the positive effects of Gln in improving postoperative nutritional status, immune function, decreasing the inflammation level, and improving negative nitrogen balance in patients with CRC. The difference is that our results showed that parenteral Gln supplementation did not improve CD3+ and CD8+ levels, possibly due to the small number of studies included in the correlation analysis.
This study has some limitations. First, we included 17 single-center RCTs, most of them were insufficiently reported in terms of random sequence generation, allocation concealment and blinding implementation, which suggests methodological limitations. Second, the majority of the studies included in this meta-analysis were conducted in China. It is well recognized that significant differences exist across countries and healthcare systems regarding perioperative care standards, nutritional support protocols, and the specific implementation details of ERAS protocols. Consequently, the findings of this study primarily reflect the intervention effects within the specific clinical context of China. High caution should be exercised when applying these results to clinical decision-making for non-Chinese populations or within different healthcare settings. Future high-quality studies conducted across broader geographical regions and more diverse clinical practices are warranted to validate the generalizability of these findings. Third, in most studies, the patients were not correctly stratified according to their nutritional status, and it is unclear whether the benefits reported in this meta-analysis can be generalized to other diseases. Finally, Although the RCTs we included underwent rigorous literature screening and exclusion screening and there was still homogeneity, some existing reports did not allow us to clearly determine the extent to which the enrolled patients received treatment, and the nutritional interventions used in the control groups were not identical. Therefore, it is particularly necessary to conduct multi-center clinical studies with larger sample sizes in different regions or countries in the future.
Conclusion
Current low-to-moderate certainty evidence indicates that parenteral Gln supplementation may be associated with improved short-term outcomes in CRC patients, such as reduced morbidity of complications, shorter hospitalization, and better nutritional and immune-inflammatory parameters. It should be noted that the observed benefits were not uniform across all immunological parameters (e.g., CD3+, CD8+), and the strength of evidence varies across outcomes due to the limited number of studies for some endpoints. Therefore, our findings suggest a potential benefit rather than a definitive clinical effect, and they should be interpreted with caution pending further high-quality, large-scale RCTs. In the future, the specific mechanism of action, time, optimal dose, and efficacy of Gln in adjuvant therapy, such as radiotherapy and chemotherapy, for patients with CRC, deserves further study.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China, No. 81960105; and Medical Research Union Fund for High-quality Health Development of Guizhou Province, China, No. 2024GZYXKYJJXM0155.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
