Abstract
Introduction
Breast cancer remains a leading cause of cancer-related morbidity and mortality in sub-Saharan Africa, with Uganda experiencing a reported 5% annual increase in cases. Alarmingly, 87% of women in Uganda present with advanced-stage disease that is less responsive to treatment, contributing to the region's disproportionately low survival rate. Early breast cancer detection will be the fundamental intervention to reverse the mortality resulting from breast cancer in Uganda. This study aims to enhance breast cancer screening and early detection in Ugandan women who are at risk through innovative use of quantitative MRI to differentiate between benign and malignant breast lesions for women at risk.
Methods
The study prospectively recruited women at risk of breast cancer who underwent breast ultrafast DCE-MRI from July 2023 to April 2024. A 3.0-T MRI system with a16 channel breast dedicated coil was used with scan durations of up to 10 min. The T1 weighted pre-contrast, T1 weighted post-contrast, T1 weighted dynamic subtracted and Maximum Intensity projection (MIP) sequences were acquired and the histology blinded pharmacokinetic analysis for the breast lesion was done. The initial area under the curve in 30 s after contrast injection (
Results
A total of 52 women were recruited and imaged and 36 lesions were found. Unlike the
Conclusions
Abbreviated DCE-MRI protocols with quantitative analysis can effectively differentiate malignant from benign breast lesions with improved compliance and can be adopted as a model of breast cancer screening and early detection for women at risk.
Introduction
Breast cancer (BC) is a major public health problem, accounting for 2.3 million new cases and 685 000 deaths recorded globally in 2020 alone.1,2 In sub-Saharan Africa, BC is the leading cause of cancer; and in Uganda, a 5% annual rise in BC incidence has been reported.3,4 GLOBOCAN (2020) estimated that 2640 new cases of BC and 1370 deaths occurred in Uganda. 5 This high mortality is because most cases present with non-treatable disease due to late detection. This study aims to assess the feasibility and diagnostic utility of magnetic resonance imaging (MRI) techniques for early BC detection in high-risk women in Uganda. It also aims to inform the development of practical and scalable early detection approaches in low-resource settings that will improve BC outcomes. We hypothesized that the application of innovative MRI techniques that include Abbreviated MRI (AB-MRI), Dynamic Contrast-Enhanced MRI (DCE-MRI), Ultrafast MRI (UF-MRI), and Quantitative (qMRI) will significantly enhance the early detection of BC compared to conventional approaches. This will reduced mortality and improve BC survival. BC survival in Uganda remains disproportionately low with the 5-year overall survival among women of 51.8%. 6 The impact of lack of modern BC screening and early detection methods like MRI is exacerbated by relatively high incidence of BRCA1 and BRCA2 mutations in Uganda and other countries in Sub-Saharan Africa. 7 The low survival rate has been attributed to the scarcity of early detection programs, modern technologies like MRI, lack of adequate diagnosis and treatment facilities, stigma, providers and health system delays, as well as costs associated with transport and treatment along the BC care pathways.1,7–11 To date, there are no well-developed, innovative approaches like use of MRI for BC screening and early detection programs in Uganda.12,13 Strengthening BC early detection programs has been demonstrated to reduce BC mortality by at least 20% in several developed countries.12,14 However, in Uganda and the sub-Sahara Africa, there are no effective BC screening and early detection methods. Currently, there is no data about the utility of MRI in early detection of BC in the region. Therefore, reliable, fast, inexpensive MRI methods for BC early detection can have a huge impact in Uganda and also in surrounding countries. This study was designed to improve early detection through the use of modern and appropriate imaging technologies, including AB-MRI, DCE-MRI, UF-MRI and qMRI. The rationale of these techniques was to achieve significantly shortened scan with AB-MRI, to leverage the angiogenesis of the tumor by DCE-MRI, to capture contrast inflow in the lesion with UF-MRI and to study the pharmacokinetics with qMRI. We therefore addressed the critical need for early BC detection by integrating with modern technologies. The breast AB-MRI is a shortened version of a full MRI protocol or standard breast MRI exam, currently performed for screening breast cancer. The concept of the abbreviated protocol is to reduce the length of the acquisition and interpretation time of a screening breast MRI examination from more than 45 min to a minimum. For this study, we used breast AB-MRI protocol with scan time of less than 10 min, with patient handling times varying between 5 to 10 min. In 2014, Kuhl and colleagues were the first to report the feasibility of the simplest form of abbreviated MRI. 13 Their version of abbreviated protocol consisted of an unenhanced T1-weighted sequence and the first contrast-enhanced T1-weighted sequence, subtraction images with a single maximum intensity projection (MIP). 13 These are UF-MRI protocols. UF-MRI is developed to capture the inflow of contrast in breast lesions and hence enable dynamic analysis of contrast wash-in rather than contrast wash-out. The technique builds largely on earlier attempts to analyze the pharmacokinetic characteristics of breast cancer vasculature, which was shown to allow good separation between benign and malignant breast lesions.15,16 Furthermore, the kinetic assessment of the ultrafast DCE-MRI was shown to be comparable to the standard kinetic assessment obtained from the combination of early and delayed phases of standard DCE-MRI for distinguishing between benign and malignant lesions.17,18 In this study, we pioneered the application of AB-MRI, DCE-MRI, UF-MRI, and qMRI for BC screening in a sub-Saharan African setting. By evaluating diagnostic performance and feasibility of this approach, we aimed to lay the groundwork for an effective and accessible BC screening and early detection model in Uganda and similar low-resource environments.
Material and Methods Study
Design and Site
This was a prospective pilot cohort study targeting 52 women at a high risk for BC in Uganda. This study was conducted at one of the leading cancer centers in Uganda that is mandated to, among others, undertake and coordinate the management of cancer and cancer-related diseases in the East African region. This study was approved by the Uganda cancer institute ethics committee (Reference number 2023-74: Date 01/10/2023) located at upper Mulago hill road Kampala and the Uganda national council of science and technology (Reference number HS 3386ES: Date 06/08/2024) located plot 6 Kimera road Ntinda Kampala. This study was conducted in accordance with Good Clinical Practice and the Declaration of Helsinki. All participants provided written informed consent for undergoing MRI, biopsy of the breast lesions and use the clinical data for research and publication purposes. This study has been conducted following the relevant equator guide line and the reporting conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE). 19
Patient Population
Through the community outreach, as indicated in Table 1, we screened women in the Central Region of Uganda and recruited a total of 52 women with the risk of BC, as indicated in Figure 1. These were women with dense breasts. Women with a family history of breast cancer, meaning those with a first degree relative with a history of breast cancer. These relatives include sisters, mothers, and daughters. Women with suspicious clinical findings that included those with hormonal factors like early menstruation, late menstruation, hormone replacement therapy, and other risks like obesity, not breastfeeding mothers and those who gave birth late. These further included women who had abnormal findings on mammography and ultrasound that were scored BI-RADS 3 to BI-RADS 5 or with suspicious breast lesions on clinical evaluation. Our inclusion criteria were women aged between 19 and 80 years, women who had willingness to provide informed consent, and women who had a strong family history of BC. This included being a first-degree relative to a person with or had BC. The inclusion criteria were also women with abnormal Breast Mammogram or Ultrasound within 1 year since the last imaging. Women with any prosthesis and active breast cancer (confirmed BC on histology and within 5 years of diagnosis) were excluded. However, for this as a pilot study, we aimed at evaluating the diagnostic performance of imaging parameters and identifying optimal cut-off points for BC early detection, therefore there was no formal sample size calculation conducted. The sample size for this study was based on feasibility and available resources, with the intention of identifying parameters with acceptable diagnostic accuracy to inform future larger studies.
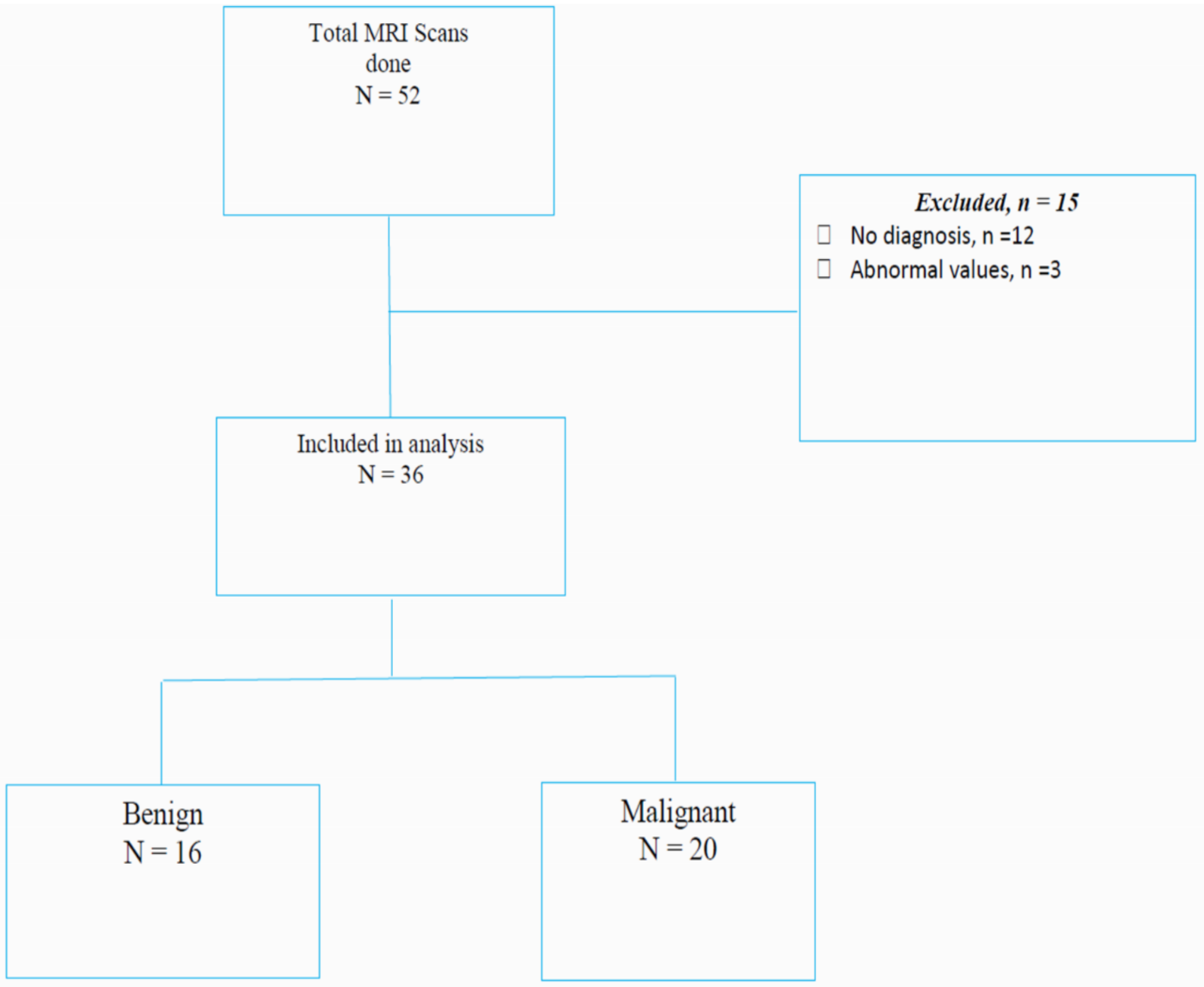
Consort Diagram Demonstrating the Participant Flow for This Study.
Characteristics of the study population

Data are presented as mean (SD) or median (IQR) for continuous measures, and n (%) for categorical measures
SD - Standard deviation; IQR - Interquartile range (Q1 - Q3), Min - Minimum, Max - Maximum
Imaging Technique
Women who met the criteria underwent bilateral breast MRI scan (Germany made Siemens 3.0 Tesla with syngo MR EII software version) equipped with a specialized 16-channel breast coil. We performed ultrafast AB-MRI with T1 weighted pre-contrast, T1 weighted post-contrast T1 weighted dynamic subtracted and Maximum Intensity projection (MIP) that could take up to 10 min. The women received a contrast media injection of gadolinium (Omniscan 0.5 mmol/mL) at a dose of 0.2 mL/kg. This was injected manually, approximately up to 5 s maximum, and was followed by a bolus of 10 mL of (0.9%) saline flush. The T1-weighted sequences that were acquired before the injection were with TR = 5.4 ms, TE = 2.5 ms, slice thickness = 4 mm, gap = 10%, FOV = 280 mm, matrix = 320 × 182 and flip angle = 73°. The DCE sequences consist of DCE T1-weighted with TR = 3.7 ms, TE = 1.1 ms, slice thickness = 4 mm, gap = 10% mm, matrix = 320 × 182, flip angle = 73⁰. The acquired images were reconstructed to the requirements of the clinical protocol and processed for viewing as DICOM data. This was mainly targeted for the entire breasts, breast parenchyma, and the tumor's solid components or solid nodules. All these acquired images were exported for quantitative analysis. However, all the image analyses were performed with observers blinded to the histopathologic outcomes. The analysis was done through the analysis pipeline indicated in Figure 2.

The Pipeline of Quantitative Analysis to Demonstrate the Sequential Step by Step Analysis of the MRI Images to Come up with the Kinetic Parameter.
Quantitative Analysis
Data analysis was performed with an in-house MATLAB platform and 3D Slicer. The image analysis was done in a number of steps as indicated in Figure 2. Our first step in the image analysis was to convert all the DICOM images to MATLAB variables for each participant. A non-rigid registration was used to minimize participants’ motion. 20 The motion corrected images were manually segmented, and then they were then subjected to pharmacokinetic (PK) analysis.
Segmentation
In this step, the images were subjected to the anatomic boundaries delineation by computer-assisted contouring. This was the step of image segmentation. Anatomic segmentation was to allow analysis of what is of interest to the breast images (the breast tissue, immediate chest wall, and axilla) while discarding that which is not clinically useful (surrounding air, thoracic cavity, and abdomen). This anatomical segmentation was performed using a 3D slicer 5.4.0. 21 Breast MRI segmentation was divided into three consecutive tasks: 1) the delineation of breast–chest wall and breast–air. 2) The separation of breast parenchyma tissue from fat. 3) Distinguishing abnormal enhancement from normal background parenchymal enhancement (BPE). To identify parenchyma, seed pixels were manually selected in a slice where the maximum amount of parenchyma was visible. Then the ‘level Tracing’ editing tool in 3D Slicer automatically generated a border of region by searching for voxels with a similar intensity value in the same slice. Volumetric parenchymal ROIs were capture using the ‘Grow from seeds’ editing tool by growing the ‘seed’ area into a 3D volume. The same segmentation step was used for segmenting tumor but unlike for the breast boundary and for the breast parenchyma segmentations where the study used T1-weighted pre-contrast sequences, for the breast lesion segmentations the study used dynamic contrast-enhanced subtracted sequences. The segmentation of tumor was confirmed by a Radiologist with the experience in breast imaging. The validation to reduced inter-observer variability was done by inspecting the segmented images and comparing them original MRI images to verify the accuracy of the segmentation. Comparison with the previous imaging that included mammography and ultrasound was done. This was to ensure that accurate segmentation of the breast MRI images with manual use of the 3D slicer was achieved.
Mathematical Quantitative Analysis
Signal enhancement ratio over time per voxel within the lesion and normal parenchyma, 
Where
Then, the secondary parameter maximum uptake slope:
The other secondary parameter area under the curve for initial 30 s after contrast uptake: iAUC30 was also calculated, which is the integration of Eq. 1, ie:
In addition to EMM, Tofts model was used to obtain quantitative pharmacokinetic parameters.
22
Here we used the Tofts two-compartment model, which describes the exchange of contrast agent between the blood plasma and extracellular extravascular space (EES) in tissue. Contrast agent concentration curve (
Where

The MRI Image Analysis of a 28 years old who had a Painful Right Breast Mass Note: (A) Kinetic map of the First Breast Mass, (B) ROI of the First Tumor and the BPE, (C) Kinetic map of the Second Breast Mass, (D) ROI of the Second Breast Mass, (E) Plotting of the First and Second ROI (F) the Kinetic Parameters.

A 60-Year-Old who had a Painful Right Breast Mass. Kinetic map, (B) ROI of the tumor and the BPE (C) plotting of the ROI and BPE (D) the kinetic parameters.
Histology
Histopathology was used as the standard of reference in all lesions by using image-guided needle biopsy and surgery. An experienced pathologists read all cases. In malignant tumors, the Nottingham histology score, reflecting tumor cell differentiation based on tubule formation, nuclear grade, and mitotic rate, was used for grading as Grade 1 (well differentiated), Grade 2 (moderately differentiated), or Grade 3 (poorly differentiated). However, the tumors were generally discriminated as malignant or benign. These results were validated in the pathology laboratory by use of quality control measures, where the known positive and known negative tissues are used as controls to ensure the reliability of the results. Also, the pathology laboratory has histology books that include a histology atlas that provides detailed description of histology images. These were used as the standard references in histology.
Statistical Analysis
The statistical analysis aimed to evaluate the diagnostic performance of MRI kinetic parameters that included
Optimal cut off is 10.355 however, there were significant differences to the sensitivity, specificity, positive predictive and negative predictive values. As indicated in Figure 5, the area under the ROC curve (

The ROC Curve for MRI iAUC30 Values Indicating the Ability of qMRI Ability to Discriminate Benign and Malignant Breast Lesion Using iAUC30.
Interpretation and Discussion
To the best of our knowledge, this study is the first exploration of this novel approach in this region and demonstrates promising techniques with the potential to discriminate benign and malignant breast lesions. Table 2 indicates that the parameters
Optimal cutoff, Sensitivity, Specificity and ROC iAUC30 of MRI Parameters

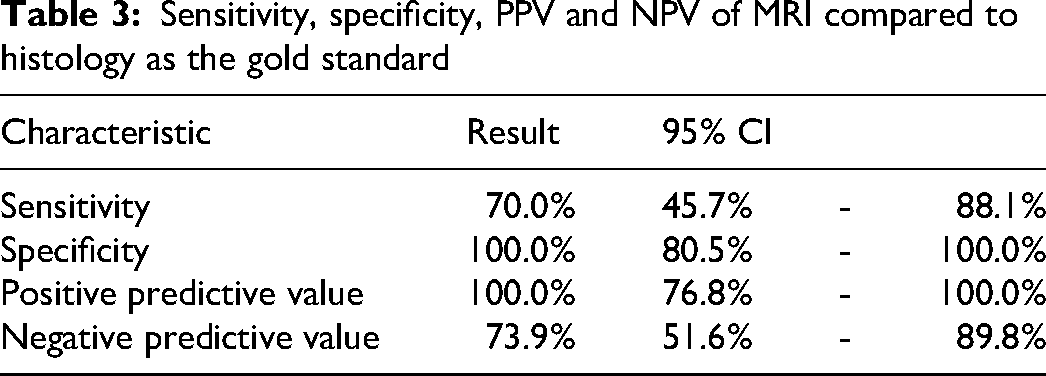
Sensitivity, specificity, PPV and NPV of MRI compared to histology as the gold standard
In this study these protocols have been leveraged on angiogenesis as an essential step in the pathophysiology of soft-tissue tumor growth.24,25 The vessels of normal tissue are intact, and the capillary bed tends to be mature. However, stimulation from the microenvironment of soft-tissue tumor, such as hypoxia and weak acid, could increase the level of vascular endothelial growth factor.
26
With vascular endothelial growth factor stimulation, a soft tissue tumor tends to generate a large number of tumor vessels. The tumor vascular structure is morphologically incomplete and lacks intact muscular and basal layers.
27
Owing to a high level of permeability, the reactivity and feeding capacity of tumor vessels are functionally low.
27
Therefore, microcirculation in the newly formed tumor is disorganized, and the fragile vessels are highly permeable. These were the basis of this study, where the kinetics of the contrast agent transit depends heavily on tissue perfusion, vessel permeability, and volume of the extravascular extracellular space.
28
Biological behaviors and microcirculation characteristics vary in benign and malignant soft-tissue tumors, which result in differences in the pharmacokinetic distributions of the contrast agent. DCE-MRI aids in the differentiation of these pharmacokinetic distributions and can enable conversion of the distributions into objective parameters.
29
This is the basis for offering an improved diagnostic accuracy of early BC detection as an outcome of this study. The DCEMRI images can be analyzed qualitatively, semi quantitatively, and quantitatively.28–30 This study explicitly did not perform a qualitative analysis that is widely used. This is because qualitative analysis is the basic analysis of DCE-MRI that is based on the subjective evaluation of the time signal intensity curve to determine the inflow rate and clearance time of the contrast agent into the lesion.
31
It can also help determine the degree of enhancement and blood supply characteristics of the lesion.
31
However, this approach is intuitive and prone to errors due to technical expertise requirement and bias introduction, therefore, the results of a qualitative analysis are subjective.28,29,32 In addition, this type of analysis does not provide quantifiable parameters (eg, the rate of tracer uptake or washout) or measurements of tissue perfusion and permeability.
32
Though this can be regard as a limitation, in order to eliminate errors associated with subjectivity, semi quantitative and quantitative analysis were performed in this study. We therefore performed the calculation of the pharmacokinetic parameters that included

A Box Plot Comparing iAUC30 Among the Benign with Malignant Group Indicating the Variation of the iAUC30 Values of the Benign and Malignant Breast Lesions.
However, with the sensitivity of 70% for
A 2 × 2 table of histology results and MRI results (sensitivity analysis)

Overall, our findings demonstrate the superiority in diagnostic accuracy of innovative MRI protocols, reduced scan time, patient compliance, reduced operational costs, the increased throughput and that unnecessary biopsies can be avoided. We also acknowledge the infrastructural and economic constraints that currently limit widespread implementation of MRI in Uganda for BC early detection on full-scale population is not immediately feasible due to the high costs of equipment, maintenance, and the need for skilled personnel. However, this study is grounded in a translational research framework, aiming to inform a phased and targeted deployment strategy that is leveraged on the existing referral system for women at risk of BC. We envision MRI to be integrated initially into tertiary care centers or regional referral hospitals for high-risk or symptomatic patients, where its diagnostic advantage is most impactful. Compared to mammography and ultrasound, MRI, when used strategically in high-risk groups, can overall significantly reduce the downstream costs associated with delayed diagnosis and advanced stage disease management. We also envision integration with telemedicine, public-private partnerships, and capacity-building initiatives for sustainability. This strategic model will serve as a pilot framework for scaling the use of MRI in low-resource contexts. To further enhance our approach, the study emphasized the aspect of communication, community outreach and education about BC.
Limitations of the Study
The study had limitations, including a small and unevenly distributed sample, predominantly comprising Invasive Ductal Carcinomas (IDCs), highlighting the need for a larger cohort in future research. The modest sample size also limited us to conduct of reliable subgroup analyses that would be statistically meaningful. The manual injection of contrast medium, though skillfully done, may have had inconsistent flow of contrast medium and could have introduced human errors. This technical variability could have affected the results. To be consistent, future studies should use the automated contrast media injectors. The analysis did not consider the impact of high temporal resolution on diagnostic performance, which could provide valuable insights into differentiating between benign and malignant breast lesions. The manual segmentation and delineation of regions of interest (ROIs) may have introduced variability in parameters. Additionally, the study focused solely on ultrafast DCEMRI for distinguishing between benign and malignant breast lesions, without comparing it to conventional DCE-MRI, an area that could be explored in future studies. Also, given the lack of delayed post-contrast sequences in most abbreviated MR imaging protocols, slow-enhancing malignancies may be missed because some neoplasm can present as non-mass enhancement. The other limitation is that genetic testing was not performed during the study to aid identification of women at risk.
Conclusion
These preliminary findings provide supporting evidence that the innovations in MRI enhance diagnostic accuracy for early BC diagnosis by accurately differentiating between benign and malignant masses and delineating normal tissue. This approach could reduce unnecessary biopsies and serve as an effective early detection tool for BC for women at risk. The developed MRI protocols are not only cost-effective and rapid but also improve patient compliance, management and hence offer improved outcome. The study suggests that larger clinical trials could contribute to better breast cancer screening and early detection strategies.
Recommendations
Given the study's limited sample size and demographic scope, these findings should be validated in larger cohorts, and further research should focus on optimizing sensitivity without compromising specificity. Clinicians should use the MRI
Footnotes
Abbreviations
Acknowledgements
We acknowledge and thank all participants who contributed to the completion of this study including the staff of the breast clinic and pathology unit at the Uganda Cancer Institute.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Kiphart center. The funders had no role in the design of the study; the collection, analysis, and interpretation of the data; the writing of the manuscript; and the decision to submit the manuscript for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors also declared that this work is original and has not been published or submitted elsewhere and that the information provided is accurate and reliable.
Data Availability Statement
The data presented in this study are available upon reasonable request from the corresponding author.
