Abstract
Introduction
Despite technological advances in radiotherapy, trismus persists as a treatment-limiting toxicity in T3-T4 nasopharyngeal carcinoma (NPC) survivors, with current guidelines lacking evidence-based dose constraints for masticatory muscle sparing. This study aimed to establish dose-response relationships and propose clinically actionable thresholds for trismus prevention using volumetric modulated arc therapy (VMAT) with collapsed cone convolution (CCC) dose calculation.
Methods
In this retrospective cohort study, 60 T3-T4 NPC patients treated with VMAT (CCC algorithm) were stratified by trismus severity. Dosimetric parameters (Dmean/Dmax) of jaw-opening (digastric, mylohyoid, geniohyoid) and closing muscles (masseter, pterygoids) were analyzed. Univariate (t-tests) and multivariable logistic regression identified predictors; ROC curves determined optimal dose constraints.
Results
Univariate analysis: digastric Dmean showed the strongest association with trismus (p = .0049), followed by mylohyoid Dmean (p = .088) and masseter Dmean (p = .063). Multivariable analysis: digastric Dmean retained marginal significance (β=−1.58 × 10−3, p = .092), with each 1 Gy increase reducing trismus odds. ROC analysis: digastric Dmean (AUC = 0.70) and mylohyoid Dmean (AUC = 0.60) yielded optimal thresholds of ≤26 Gy and ≤28 Gy, respectively (Youden's index).
Conclusion
Proactive sparing of jaw-opening muscles—particularly the digastric (Dmean ≤26 Gy) and mylohyoid (Dmean ≤28 Gy)—during VMAT planning may mitigate trismus risk in advanced NPC. These preliminary constraints, derived from CCC-optimized dosimetry and based on optimal classification cutoffs (Youden's index), help address a critical gap in current OAR protocols.
Introduction
Radiotherapy (RT) is a cornerstone treatment for nasopharyngeal carcinoma (NPC), achieving 10-year local and regional control rates of 84% and 94%, respectively. 1 However, patients often experience treatment-related toxicities, with trismus (restricted mouth opening) being a critical late effect that significantly compromises quality of life. 2 Although advancements like volumetric modulated arc therapy (VMAT) and improved dose-calculation algorithms (collapsed cone convolution) have reduced radiation exposure to organs at risk (OARs), clinically observed sequelae—including xerostomia, dysphagia, and trismus—persist.
The reported incidence of trismus in head and NPC patients ranges from 5% to 50%. 3 Impaired mouth opening or pain can limit oral intake, impair speech, and exacerbate poor oral hygiene. 4 Jaw movement is governed by paired masticatory muscles: the closing group (masseter, medial and lateral pterygoids) and the opening group (digastric, mylohyoid, and geniohyoid muscles). Proactive measures, such as guided exercises and dose constraints for these muscles during treatment planning, may mitigate trismus risk. 5 Despite recognition of its clinical impact, the pathophysiology of RT-induced trismus and optimal dosimetric thresholds remain poorly defined. 6
Both irradiated site and tissue volume influence trismus development. For example, patients with laryngeal, hypopharyngeal, or nasopharyngeal carcinomas frequently exhibit high-dose exposure to masticatory structures due to their proximity to target volumes. The spatial overlap between these muscles and beam paths often results in unavoidable low-to-intermediate dose deposition. However, the importance of constraining doses to these structures is frequently overlooked.
Current NPC guidelines specify dose limits for the temporomandibular joint (TMJ) but omit constraints for masticatory muscles. 7 Moreover, as elective nodal irradiation is standard for T3–T4 NPC, this study expands the anatomical definition of jaw-related muscles 8 to include the complete functional unit for jaw movement (digastric, mylohyoid, and geniohyoid). Our aims are to: Establish dose- response relationships between RT and trismus in NPC patients; Propose evidence-based dose thresholds for masticatory/suprahyoid muscles to reduce trismus risk.
Methods
Patient Characteristics
This retrospective study included 60 patients diagnosed with nasopharyngeal carcinoma (NPC) between 2023 and 2024, with a follow-up period of 1 year. The reporting of this study conforms to STROBE guidelines. 9 The study was approved by the Institutional Review Board of the First Affiliated Hospital of Fujian Medical University (Approval No. MTCA,ECFAH of FMU [2015]084-3) on third, April, 2025 . As a retrospective analysis using anonymized data, written informed consent was waived under ethical approval, and all patient information was securely protected in the hospital database. The study was conducted in accordance with the Declaration of Helsinki to ensure patient confidentiality. Flow diagram can be seen in Figure 1.

Flow Diagram.
Inclusion Criteria, Eligible patients met the following criteria:
Histologically confirmed T3 or T4 NPC (staged according to the eighth edition AJCC staging system). Treatment-naïve patients with no prior therapy at other centers. Uniform treatment protocol: All patients received volumetric modulated arc therapy (VMAT) with the collapsed cone convolution (CCC) algorithm, and target dose coverage met standardized criteria. No severe systemic comorbidities that could interfere with treatment or follow-up. Voluntary participation in follow-up with documented informed consent.
Patient characteristics are summarized in Table 1.
Patient Characteristics and Dosimetric Parameters.

At the 1-year follow-up, trismus (defined as the ability to fit 1-2 fingers between the incisors) was observed in 31 out of 60 patients (51.7%).
Target Delineation and Definition Criteria
All patients received definitive 6-MV x-ray volumetric modulated arc therapy (VMAT) delivered by a linear accelerator (Trilogy, Varian Medical Systems, USA). Target volumes were delineated on treatment planning CT scans co-registered with diagnostic magnetic resonance imaging (MRI) .Target volumes were defined in accordance with the International Commission on Radiation Units and Measurements (ICRU) Reports 50 and 62. Clinical target volume (CTV) delineation followed international consensus guidelines for nasopharyngeal carcinoma. Dose distributions were optimized to meet organ-at-risk (OAR) constraints specified in Radiation Therapy Oncology Group (RTOG) protocols 0615 and 0225, ensuring protocol-compliant plan quality (Figure 2).

Representative Dose Distribution for a T3-T4 Nasopharyngeal Carcinoma Patient Treated With VMAT. The Delineated Target Volumes are as Follows: Gross Tumor Volume of the Nasopharynx (GTVnx, Solid red); Gross Tumor Volume of Metastatic Lymph Nodes (GTVnd, Green); High-Risk Clinical Target Volume (CTV1, blue); low-Risk Clinical Target Volume (CTV2, Cyan). The Corresponding Planning Target Volumes (PTVs), Generated by a 3-mm Expansion From Their Respective CTVs, are: PTVnx (Lavender); PTVnd (Orange); PTV1 (Magenta); PTV2 (Yellow). The Plan Demonstrates Conformal Dose Coverage of the Target Volumes.
Target volume definitions:
Gross tumor volume (GTV) included: Primary tumor (GTVnx), prescribed 66-76 Gy in 30-33 fractions Metastatic cervical lymph nodes (GTVnd), prescribed 66-70.6 Gy in 30-33 fractions Clinical target volume (CTV) encompassed: High-risk regions (CTV1), prescribed 60-62 Gy in 30-33 fractions Low-risk regions (CTV2), prescribed 50-56 Gy in 30-33 fractions
All patients were treated with one fraction daily, five times per week, over 6-7 weeks.
Delineation of Masticatory Muscles
The jaw-opening (digastric, mylohyoid, geniohyoid) and closing (masseter, medial and lateral pterygoids) muscles were contoured as new organs at risk on planning CT scans using bony landmarks for guidance, co-registered with MRI for soft-tissue reference. For the jaw-opening muscles: the digastric muscle was contoured in its entirety, including both anterior and posterior bellies and the intermediate tendon at the hyoid bone; the mylohyoid was contoured as the muscular sling forming the floor of the mouth; and the geniohyoid was defined on the midline superior to the mylohyoid. For the jaw-closing muscles: the masseter was outlined from its origin on the zygomatic arch to its insertion on the mandibular ramus; the medial pterygoid was contoured deep to the mandibular ramus, extending from the pterygoid plates to the medial surface of the mandibular angle; the lateral pterygoid was delineated within the infratemporal fossa, with its superior head attaching to the infratemporal surface of the sphenoid bone and its inferior head to the lateral pterygoid plate (Figure 3). All contours were performed by a single experienced radiation oncologist and verified by a senior colleague to ensure consistency. The mean volumes of the contoured muscles were listed in Patient characteristics (Table 1).
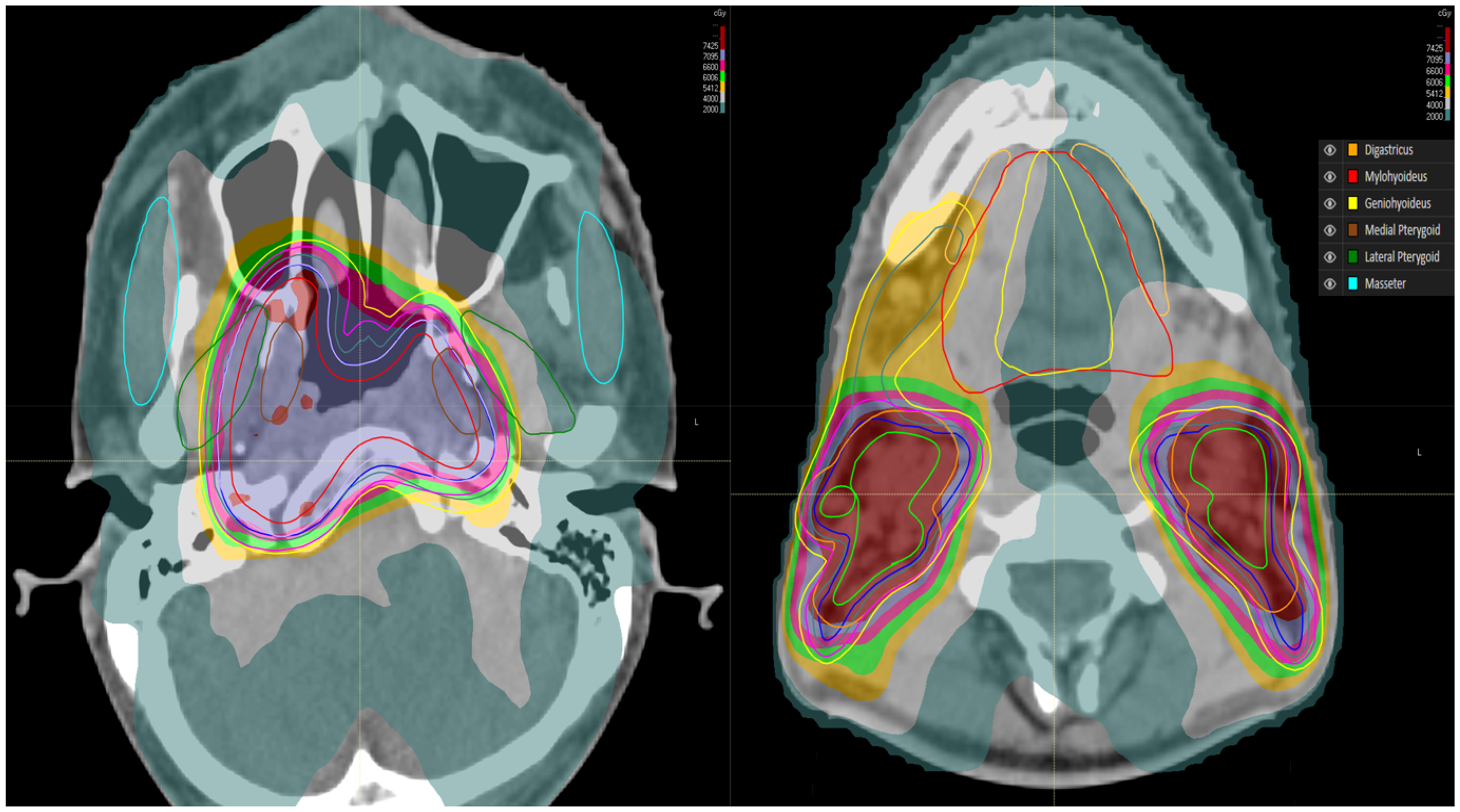
Anatomical Location and Delineation of the Defined Masticatory and Suprahyoid Organs at Risk (OARs) on Axial CT Imaging. The jaw-Opening and jaw-Closing Muscles Were Contoured on Planning CT Scans a Color Scheme was Adopted: Digastric (Orange), Mylohyoid (red), Geniohyoid (Yellow), Masseter (Brown), Medial Pterygoid (Green), and Lateral Pterygoid (Cyan).”
Evaluation Criteria for Trismus
Patients were evaluated during a follow-up survey conducted 1 year after treatment. Participants were contacted via multiple communication methods, including email and telephone. The survey focused exclusively on assessing the presence and severity of trismus.
Trismus was self-reported by participants based on the maximum number of vertically stacked fingers that could be placed between their central incisors or gums, with reference to the LENT-SOMA scale.
10
The evaluation consisted of two questions:
"How wide can you open your mouth?” "How many fingers can you place vertically between your upper and lower teeth or gums?”
Responses were categorized as follows:
Three or more fingers: No trismus At least two fingers: Moderate trismus At least one finger: Severe trismus Fewer than one finger (unable to place a single finger between teeth/gums): Very severe trismus
Dose Acquisition Using the CCC Algorithm in RayStation Treatment Planning System
All target volumes and organs at risk (OARs) in this study were calculated using the Collapsed Cone Convolution (CCC) algorithm within the RayStation treatment planning system (TPS). Clinical treatment plans were required to meet our institutional standards of 100% coverage for CTV and ≥95% coverage for PTV.
Under these clinical constraints, dose calculations and extractions were performed for the relevant masticatory and suprahyoid muscle groups (opening/closing muscles). The corresponding dose distribution is illustrated. Dose statistics were acquired and analyzed using the integrated dose evaluation system within the TPS platform.
In this study, dose calculations were performed using RayStation's collapsed cone convolution algorithm to ensure accurate dose modeling. Strict institutional coverage criteria were applied, requiring 100% coverage for the clinical target volume (CTV) and ≥95% for the planning target volume (PTV). Dose analysis was focused on functionally relevant jaw musculature, utilizing the built-in treatment planning system (TPS) tools for comprehensive dose evaluation.
This systematic approach ensured consistent and reproducible dose assessment across all study cases while maintaining clinical treatment standards. The integrated DOES system provided standardized dose- volume histogram parameters for subsequent statistical analysis. The dose display of muscle groups is shown in Figure 4.

Dose Distribution Patterns in Masticatory and Suprahyoid Muscle Groups. Faceted Boxplots of Dmax and Dmean across Masticatory Suprahyoid Muscles in T3-T4 NPC Patients Treated with VMAT. DIG=Digastric; MYL =Mylohyoid; MAS=Masseter; MP/LP=Medial/Lateral Pterygoids; GEN=Geniohyoid.
Statistical Analysis
Statistical analyses were performed using SPSS (IBM SPSS Statistics for Windows, Version 26.0) and R (version 4.2.1). Patients were stratified into two cohorts based on mouth opening capacity: the limited opening group (y = 1), defined by the ability to insert 1–2 fingers vertically between the incisors, indicating clinically significant trismus; and the adequate opening group (y = 0), comprising those able to accommodate 3–4 fingers, representing preserved mandibular function. This dichotomization enabled comparative analysis of dosimetric parameters between these functionally distinct groups.
To identify potential biomarkers associated with the outcome variable (y), we first performed univariate statistical comparisons between the two groups (y = 1 vs y = 0). Continuous variables were assessed using independent two-sample t-tests under the assumption of equal variance. The resulting p-values were used to rank the features based on their discriminatory power between groups. Features with p-values < .1 were considered potentially relevant for further analysis, while those with p-values > .1 were excluded from subsequent modeling to reduce dimensionality and avoid overfitting.
A multivariable logistic regression model was fitted to assess the independent associations between dosimetric parameters and the binary outcome (y), while adjusting for potential confounding effects among variables. The model incorporated all available features simultaneously, with maximum likelihood estimation used to compute regression coefficients. The Wald test was employed to evaluate the statistical significance of each predictor. Features with p-values exceeding 0.1 were considered non-significant in the multivariable context and were excluded from subsequent interpretation. To identify a dose threshold with optimal discriminatory power between patient groups, the cutoff value was selected as the point on the receiver operating characteristic (ROC) curve that maximized Youden's index (J). It is important to note that this approach identifies a cutoff for classification accuracy rather than a clinical risk threshold.”
In addition to the ROC analysis, a risk-based analysis was performed to quantify the absolute risk of trismus associated with specific dose levels. Univariate logistic regression was used to model the probability of trismus as a function of the mean dose to the most significant predictor from the univariate analysis. The resulting dose-response equation was derived from the model coefficients. The predicted probabilities of trismus across a range of clinically relevant dose levels were calculated using the formula: P(Trismus) = 1 / (1 + e^-(β0 + β1 * Dose)). A dose-response curve was plotted to visualize the relationship between the dose and the predicted probability of trismus.
Result
At the 1-year follow-up, trismus (defined as the ability to fit 1-2 fingers between the incisors) was observed in 31 out of 60 patients (51.7%). The univariate analysis revealed significant differences in several features between the two groups. The top-ranking features based on p-values were: Dmean Digastric (p = .0049, t = -2.93), indicating a significantly lower mean value in the y = 1 group compared to y = 0. Dmean Masseter (p = .063, t = 1.90), suggesting a trend toward higher mean values in the y = 1 group. Dmean Mylohyoid (p = .088, t = -1.74), showing a marginal but notable decrease in the y = 1 group.
Additionally, Dmax Geniohyoid (p = .099, t = 1.68) exhibited a borderline significant difference. However, none of the other features reached the pre-specified significance threshold (p < .1) and were thus excluded from further consideration. The statistical results are shown in Table 2.
Univariate Analysis of Dosimetric Parameters Associated with Trismus.

The multivariable analysis revealed distinct feature importance patterns compared to the univariate analysis,Model fit was assessed using deviance statistics (Null deviance: 83.11; Residual deviance: 64.95 on 47 degrees of freedom). Dmean Digastric emerged as the most significant predictor (β1 = -1.58 × 10−³ /cGy, p = .092), maintaining its marginal significance from the univariate analysis, with each unit increase associated with decreased log-odds of y = 1. Dmax Digastric showed suggestive evidence of association (β = -9.40 × 10−⁴, p = .178), though below the pre-specified significance threshold. No other features reached statistical significance (all p > .1) in the multivariable setting, including previously notable univariate candidates Dmean Masseter (p = .248) and Dmean Mylohyoid (p = .310) (Table 3).
Multivariable Logistic Regression Analysis of Dosimetric Predictors for Trismus.

The receiver operating characteristic (ROC) curve analysis demonstrated area under the curve (AUC) values of 0.7 for Dmean Digastric, 0.6 for Dmean Mylohyoid, and 0.38 for Dmean Masseter (Figure 5). Based on the maximum Youden's index, cutoff values were identified at Dmean Digastric ≤26 Gy and Dmean Mylohyoid ≤28 Gy. These cutoffs represent doses that best differentiate between patients with and without trismus in our cohort; however, their associated clinical risk profiles require further validation.
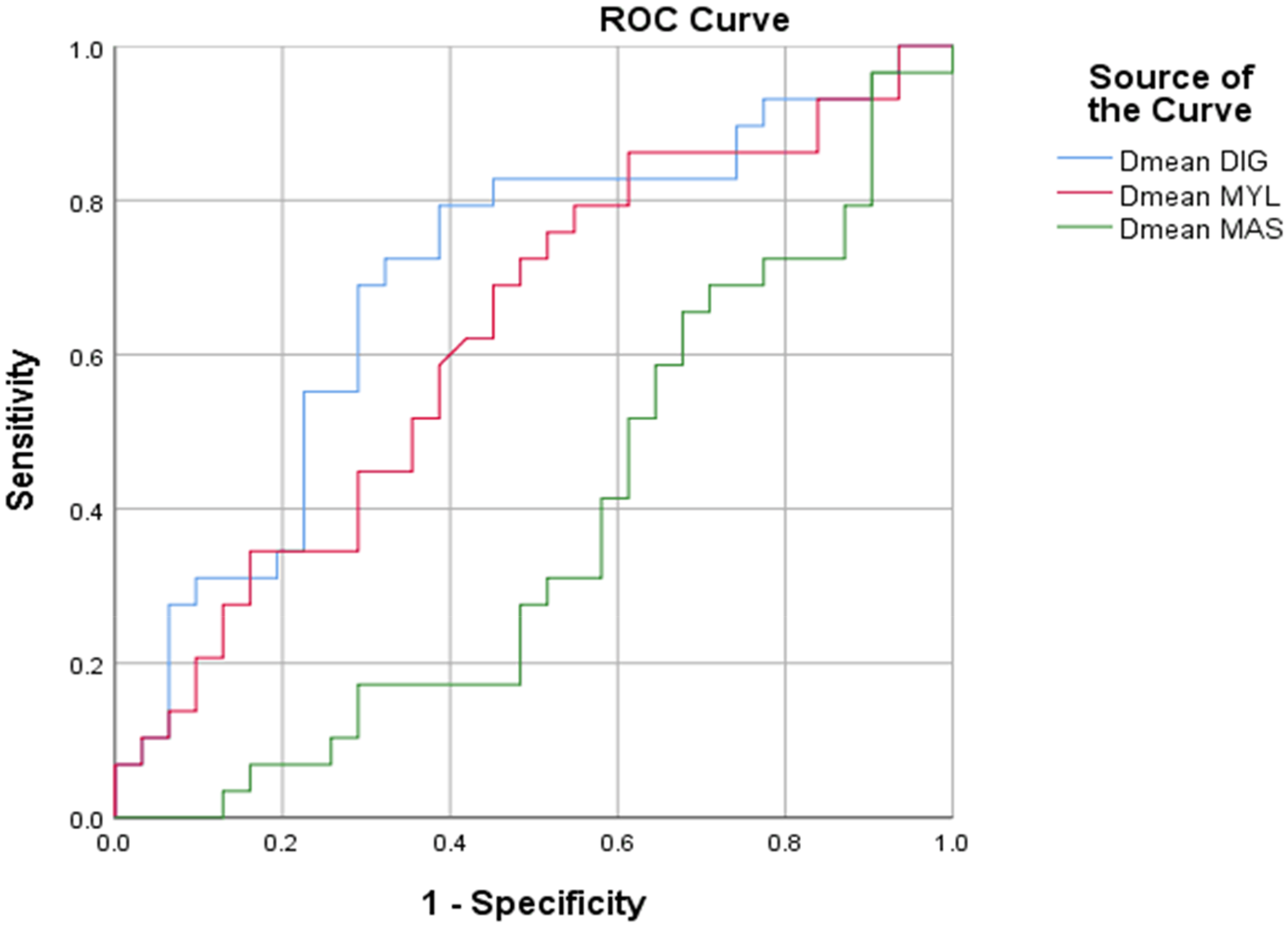
Receiver Operating Characteristic (ROC) Curves for Dosimetric Predictors of Trismus. ROC Curves for Dmean of Digastric (DIG), Mylohyoid (MYL), and Masseter (MAS) Muscles Predicting Trismus in T3-T4 NPC Patients. Dashed Diagonal Line Represents Random Chance (AUC=0.5). Optimal Cutoffs Were Selected by Maximizing Youden's Index.
The dose-response relationship was derived from the logistic regression model defined in the Methods section. Using the estimated coefficients (β₀ = 3.296, β₁ = -1.58 × 10−3 /cGy), the equation is: Log(Odds of Trismus) = 3.296 - (1.58 × 10−3) * (Dmean_Digastric).The corresponding dose-response curve is presented in Figure 6. This model indicates a high baseline risk of trismus in this cohort of advanced-stage (T3-T4) NPC patients. The predicted probability of trismus exceeded 90% across most of the observed dose range.

Dose-Response Curve for Trismus Risk Based on Digastric Muscle Mean Dose. Linear Dose-Response Relationship Between the Mean Dose to the Digastric Muscle and the Predicted Risk of Trismus. The Filled Circles Represent the Individual Observed Data Points for Each of the 60 Patients in the Cohort.
Discussion
All nasopharyngeal carcinoma (NCC) patients treated at our center underwent volumetric modulated arc therapy (VMAT) with the collapsed cone convolution (CCC) algorithm to address prolonged treatment durations and potential adverse events during radiotherapy. Studies have demonstrated that VMAT significantly reduces radiation doses to the parotid glands, thereby decreasing the incidence of post-treatment xerostomia. Additionally, VMAT optimizes dose distribution to non-target normal tissues, 11 exhibiting superior physical and technical advantages compared to intensity-modulated radiation therapy (IMRT).
This study exclusively enrolled patients receiving VMAT, with consistent dose constraints for both target volumes and organs at risk (OARs), thereby minimizing systematic errors and confounding factors to ensure result homogeneity. Dosimetric evaluations were performed using the RayStation treatment planning system with the CCC algorithm. Comparative analyses have shown no significant dosimetric differences from other algorithms under standard dose calculation conditions, confirming its reliability for clinical applications at our institution.12,13
This retrospective study evaluated patients with ≥1 year of follow-up and revealed that trismus persists even with contemporary radiotherapy advancements. Multiple etiological factors contribute to trismus in nasopharyngeal carcinoma (NPC) patients post-radiotherapy: (1) direct tumor invasion of masticatory muscles or temporomandibular joints, (2) neural infiltration affecting pharyngeal/oral motor function, and (3) radiation-induced fibrosis of articular structures and masticatory musculature, leading to restricted mouth opening and reduced mobility. 14
Prior studies have established a strong association between trismus and the medial/lateral pterygoid and masseter muscles, 15 with most research focusing on jaw-closing muscle groups.16,17 However, our study incorporated key jaw-opening muscles (digastric, mylohyoid, and geniohyoid), revealing previously unreported dosimetric deviations. This discrepancy may stem from our cohort exclusively comprising clinically confirmed T3+ NPC patients, where extended irradiation of lower neck nodal regions resulted in higher radiation exposure to inferomedial muscle groups.
In clinical practice, treatment planning prioritizes target coverage, yet this study demonstrates that sparing adjacent normal structures—particularly jaw-opening/closing musculature—should not be overlooked when technically feasible. Implementing dose constraints to these functional muscles while maintaining target dosimetry may significantly reduce post-radiotherapy trismus risk, representing a clinically actionable strategy to preserve patients’ quality of life.
The most effective approach to safeguard quality of life in NPC survivors lies in preventing trismus onset and progression. Consequently, dose limitation to non-target masticatory structures should inform RT planning decisions. Prior studies have explored dose-response relationships for trismus in NPC patients.18,19 For instance, van der Molen et al identified Dmean, Dmax, V20, V40, of mastication muscles (masseter, temporalis, pterygoids) as key predictors of trismus at 10 weeks post-RT. 20
While existing research frequently incorporates volumetric parameters, our study focused on Dmean and Dmax based on both clinical and methodological considerations. The primary rationale for this selection is clinical pragmatism: Dmean and Dmax constraints are straightforward to implement during treatment planning and are readily translatable to routine clinical practice, streamlining workflow without compromising plan quality. Furthermore, for small, functionally critical muscles like those involved in mastication, maximum dose points may have clinical implications for functional outcomes.
We selected Dmax as it provides a direct measure of peak dose exposure, which may be particularly relevant in serial structures where functional integrity can be compromised by focal high-dose irradiation. Although near-maximum dose (D2%) is also commonly used and provides a more stable estimate of high-dose regions, we chose Dmax for its immediate interpretability and established use in clinical practice for toxicity assessment. We acknowledge that both metrics have value and incorporate D2% for complementary analysis in the next step of future research
For clinicians, our Dmean and Dmax thresholds offer actionable guidance compatible with ICRU-91's emphasis on simplified constraint paradigms. This approach balances dosimetric rigor with practical feasibility in resource-variable settings.
Based on the initial discriminatory ability in our cohort, we identified preliminary dose cutoffs (digastric Dmean ≤26 Gy; mylohyoid Dmean ≤28 Gy) using Youden's index. It is critical to emphasize that these cutoffs represent the values that best separate patients with and without trismus for classification purposes within our specific cohort, and they should not be misinterpreted as established safe dose thresholds.
The subsequent risk-based analysis revealed a high predicted probability of trismus (>90%) across the entire observed dose range for the digastric muscle. This indicates that our study population, consisting exclusively of advanced T3-T4 NPC patients, had a very high baseline risk of trismus due to extensive disease and treatment volumes. This high baseline risk likely created a “ceiling effect,” limiting our ability to detect a positive dose-response relationship and potentially leading to the counterintuitive negative coefficient observed in the regression analysis. In such a context, where the outcome is almost ubiquitous, conventional dose-response modeling has limited discriminative power.
Therefore, rather than proposing a single dose constraint, we provide a dose-response curve (Figure 6) to illustrate the absolute trismus risk across dose levels in this high-risk population. This allows clinicians to understand the baseline risk context and make informed planning decisions based on their institutional risk tolerance levels. The utility of these dose-risk relationships for predicting absolute trismus risk in populations with lower baseline risk remains to be validated in future, larger prospective studies.
A counterintuitive yet notable finding from our analysis was the negative association between digastric Dmean and trismus. The following considerations help interpret this paradox. First, the anatomical proximity of the suprahyoid muscles to the primary nasopharyngeal tumor and upper nodal stations in advanced T3-T4 disease means that these structures frequently lie within or immediately adjacent to the high-dose clinical target volumes. the digastric muscle was commonly situated within or immediately adjacent to the high-dose clinical target volumes (CTVs), particularly in cases requiring comprehensive coverage of the inferior tumor extension and nodal regions. This creates an inherent confounding scenario, particularly in T4 tumors with extensive local invasion. This creates an inherent confounding scenario: patients with more extensive disease inevitably receive higher doses to these muscles and have a higher intrinsic risk of trismus due to the tumor's direct effects, making it difficult to isolate the independent effect of radiation dose. Second, while heterogeneous dose distributions could theoretically influence outcomes, the consistent negative trend for both Dmean and Dmax metrics makes it unlikely that alternative dose-volume parameters would fundamentally alter this association in the present cohort.
Therefore, the observed negative association is most parsimoniously explained by confounding by indication, where the primary driver of the outcome (advanced tumor stage) is also the primary determinant of the exposure (higher radiation dose to adjacent muscles). The uniformly high baseline risk of trismus in this cohort further limited the statistical power to detect a positive dose-response relationship. These findings underscore that in advanced T3-T4 NPC, tumor-related factors may dominate trismus risk, and future studies should aim to incorporate quantitative measures of tumor invasion to better disentangle these effects.
A notable finding is that trismus shows a stronger association with the radiation dose to jaw-opening muscles (digastric/mylohyoid) than to jaw-closing muscles (masseter/pterygoids), which may be ascribed to the following factors. First, anatomically, for advanced T3-T4 nasopharyngeal carcinoma (NPC), the inferior extension of the tumor and elective nodal irradiation often require low-neck irradiation. This directly overlaps with the suprahyoid and infrahyoid jaw-opening musculature, resulting in higher and more consistent doses to these muscles than to the jaw-closing muscles. The latter are located more superiorly, with some parts potentially spared from high-dose regions. Second, jaw-opening muscles typically have smaller volumes and less functional redundancy. A moderate dose may be well-tolerated by large muscles such as the masseter, whereas the same dose to smaller but critical opening muscles (eg, digastric) can induce significant fibrosis and functional impairment. Finally, radiologically, jaw-closing muscles are often partially included in the primary tumor volume in advanced cases, making their exposure to high doses inevitable and thus reducing the discriminative power for dose-response analysis. In contrast, the interpatient variation in dose to jaw-opening muscles is greater, providing stronger statistical power to detect a dose-response relationship with trismus.
While providing clinically actionable insights, some limitations should be considered. Firstly, as a single-institution investigation with a cohort of 60 T3-T4 NPC patients, our findings require validation in larger, multi-center studies to enhance generalizability and account for potential variations in clinical practice. We are actively expanding our recruitment to over 200 cases to address this. Secondly, we acknowledge the limitation of our trismus assessment methodology. The patient-reported finger-width measurement was adopted as a pragmatic tool to maximize long-term follow-up rates and directly capture the patient's perspective on functional limitation, which is strongly correlated with quality of life. However, this method is inherently subjective. Future prospective studies would greatly benefit from incorporating objective measures, such as caliper-based interincisal distance, to complement the patient-reported outcomes and provide a more comprehensive assessment of trismus. The dose-response here represent a significant step forward in trismus prevention; however, the ultimate test of their clinical utility will be their successful application and validation in independent patient cohorts and prospective clinical trials. We encourage other institutions to evaluate these constraints in their own practices.”
Conclusions
This study demonstrates that the mean dose to the jaw-opening muscles, particularly the digastric, possesses discriminative value for trismus in patients with advanced T3-T4 nasopharyngeal carcinoma treated with VMAT and the CCC algorithm. The findings highlight the potential role of the suprahyoid musculature in radiation-induced trismus, suggesting the need for a more holistic appraisal of functional muscular units during treatment planning. The characteristically high baseline risk in this advanced cohort underscores the challenge of defining dose-response relationships. These observations provide a foundation for future validation in broader patient populations.
Glossary of Abbreviations

Footnotes
Acknowledgements
Thank you to everyone who participated in the research, the research was conceived and designed by RFC and FBG. The manuscript was written by RFC. XYL and JC were in charge of implementation of the trial. JZ, LLS contributed the Patient management and patient enrollment. YG and LZL were in charge of radiation treatment programs. The final version for submission was approved by all of the authors.
Ethics Statement
Ethical approval for this study was obtained from the Ethics Committee for Medical Research and Clinical Technology Application of the First Affiliated Hospital of Fujian Medical University (Approval No. MTCA,ECFAH of FMU [2015]084-3). The committee reviewed and approved the secondary use of inpatients’ medical information and biological specimens in accordance with the Declaration of Helsinki, China's Good Clinical Practice (GCP), and the Measures for Ethical Review of Biomedical Research Involving Humans. This approval is valid from April 3, 2025, to April 2, 2028, following a full committee meeting review (8/8 unanimous approval) on April 29, 2025. The requirement for informed consent was waived as the study utilized de-identified retrospective data and archived specimens.
Author Contribution Statements
Runfan Chen, Conceptualization; Investigation; Writing original Draft
Jun Chen, Projection administration; Resources
Xuanyu Liu, Data curation; Formal Analysis
Linlong Shao, Resources
Jiang Zeng, Resources
Yi Guo, Supervision
Linzhen Lan, Writing editing
Feibao Guo, Supervision; Validation
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Startup Fund for Scientific Research of Fujian Medical University (2024QH1112) and Leading Project Foundation of Science and Technology, Fujian Province of China (2023Y0017)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
