Abstract
Low temperature plasma (LTP), as an emerging cancer treatment technology, has shown significant therapeutic potential due to its unique physical and chemical properties and biological effects. This article reviews the basic characteristics of LTP and its multiple mechanisms of application in tumor treatment. LTP can induce various cell death modes, including apoptosis, pyroptosis, and autophagy, through its unique chemical and physical properties. Additionally, studies have demonstrated that the combination of LTP with traditional chemotherapy drugs (such as cisplatin and paclitaxel) can enhance the anti-tumor efficacy of the drugs while reducing drug resistance. The combined application of LTP and nanomaterials also shows promising prospects. However, LTP still faces some challenges and limitations in medical applications. Future research needs to further explore the specific applications of LTP in different tumor types, optimize treatment plans, and develop more portable and efficient LTP devices to promote its application in clinical treatment.
Introduction
Cancer is one of the major diseases threatening global human health. The difficulty of treatment is due to the high heterogeneity, dynamic adaptability and complex microenvironment regulation mechanism of tumor cells. Although modern medicine has developed a variety of treatments such as surgery, chemotherapy, radiation therapy and immunotherapy. The treatment options we have are often associated with more serious side effects. First, the dose-limiting side effects such as bone marrow suppression and neurotoxicity induced by traditional treatment significantly affect the quality of life of patients. Second, acquired drug resistance caused by genomic instability is widespread. Third, there is a lack of effective and precise intervention policies for metastatic lesions. These thorny issues are still the boulders that stand in the way of cancer patients’ pursuit of health.
As the fourth state of matter, plasma is a quasi-neutral particle system composed of ionized gas, including electrons, ions, excited species and molecules. 1 Excited state matter refers to the non-ground state species formed by the transition of electrons in atoms, molecules or ions to higher-level orbitals after energy absorption. The energy of the excited state is significantly higher than that of the ground state. This high energy state gives atoms stronger chemical reaction activity, and is one of the key active components in plasmas. The unique non-equilibrium characteristics of plasma make it show significant advantages in energy transport and chemical reaction regulation. In recent years, Low Temperature Plasma (LTP) technology can achieve the targeted generation of active species while ensuring the temperature safety of biological tissues by precisely controlling the decouping effect between the electron temperature (≈1-10 eV) and the gas temperature. New modes of action of energy are provided that are thermodynamically compatible. Especially in the field of tumor therapy, LTP produces multi-modal effects such as Reactive Oxygen and Nitrogen Species (RONS), electromagnetic fields and ultraviolet rays, showing significant tumor selective killing properties.
In the medical field, plasma technology is widely studied and used in different areas. It mainly includes wound healing, 2 cancer treatment, 3 dental treatment,4,5 skin disease treatment and other fields.6,7 Among them, tumor therapy is one of the most revolutionary and challenging fields in the application of plasma medicine. Medical studies have shown that plasma can inhibit cell migration and invasion by inducing tumor cell apoptosis, iron death and other programmed death pathways, while maintaining normal tissue activity.8–16 While maintaining normal tissue activity, this characteristic of plasma provides a new idea for tumor treatment. In addition, the combination of plasma and other tumor therapies also performed well. It can not only improve the efficacy of the original single treatment, but also effectively reduce the toxic side effects of treatment.
This article systematically reviews the multi-dimensional mechanisms and synergistic treatment strategies of LTP in the field of tumor treatment, providing an integrated perspective for understanding its multiple effects of “physical-chemical-biological”. The core pathways of cell death induced by LTP (such as apoptosis, Ferroptosis, autophagy, etc) and their synergistic potential with traditional chemotherapy and new nanotherapeutics were summarized. At the same time, the limitations of the current study, such as the lack of standardization of device parameters and the lack of long-term safety verification in vivo, were discussed in depth, which provided a key framework for future research directions. By integrating interdisciplinary research results, this review aims to promote the translation of LTP from basic research to clinical practice, and lay a theoretical foundation for the development of safe and precise new strategies for tumor treatment.
Overview of LTP
LTP Concept
Plasma, as the fourth state of matter different from solid, liquid and gas, can be divided into Thermal plasma and LTP according to thermodynamic equilibrium characteristics. In thermal equilibrium plasma, electrons, ions and neutral particles reach thermodynamic equilibrium through full collision, and the system temperature can reach the order of 10³-10⁴ Kelvin. Its strong thermal radiation characteristics are widely used in industrial cutting and material processing. In sharp contrast, LTP usually excites the gas with the help of applied electric field or electromagnetic radiation, so that the electrons obtain sufficient kinetic energy, resulting in gas ionization. 17 And its temperature is close to room temperature through the uncoupling of electron temperature and gas temperature. The common generation methods include DC arc discharge, RF discharge and microwave discharge. The uncoupling of the electron temperature to the gas temperature is important during these discharges. During the discharge process, the electron temperature can reach thousands of Kelvin, while the ion and neutral particle temperature is kept near room temperature. Since the mass of electrons is much smaller than that of ions and neutral particles, although the temperature of electrons is higher, the temperature of the latter rises more slowly. This non-equilibrium system results in a LTP capable of conducting high-energy electron-driven chemical reactions without significantly increasing the overall temperature. Because of this, LTP can effectively kill tumor cells without causing excessive heat damage to surrounding healthy tissues. This characteristic gives LTP the advantages of high precision and low thermal damage in medical applications. 18
Active Species in LTP
The active species in LTP is a general term for free radicals, charged particles and excited substances produced during the discharge of gas/liquid media. Excited state matter refers to the non-ground state species formed by the transition of electrons in atoms, molecules or ions to higher-level orbitals after energy absorption. The energy of the excited state is significantly higher than that of the ground state. This high energy state gives atoms stronger chemical reaction activity, and is one of the key active components in plasmas. The generation of reactive species in LTP involves gas-phase electron collision dissociation, excited state particle reaction, liquid phase dissolution of free radicals, and subsequent chain reactions. The synergistic action of these species is the central mechanism of the biological effects of plasma. For example, when a high-energy electron e − collides with an O2 molecule, it can cause dissociation of O2 to form an oxygen atom O, which then reacts with H2O to form OH•. Upon discharge in a liquid medium, H2O2 reacts with NO2 − to form the strong oxidant ONOO −. Detailed chemical reactions between the active species can be seen in Table 1.
Chemical Reactions Between the Active Species.
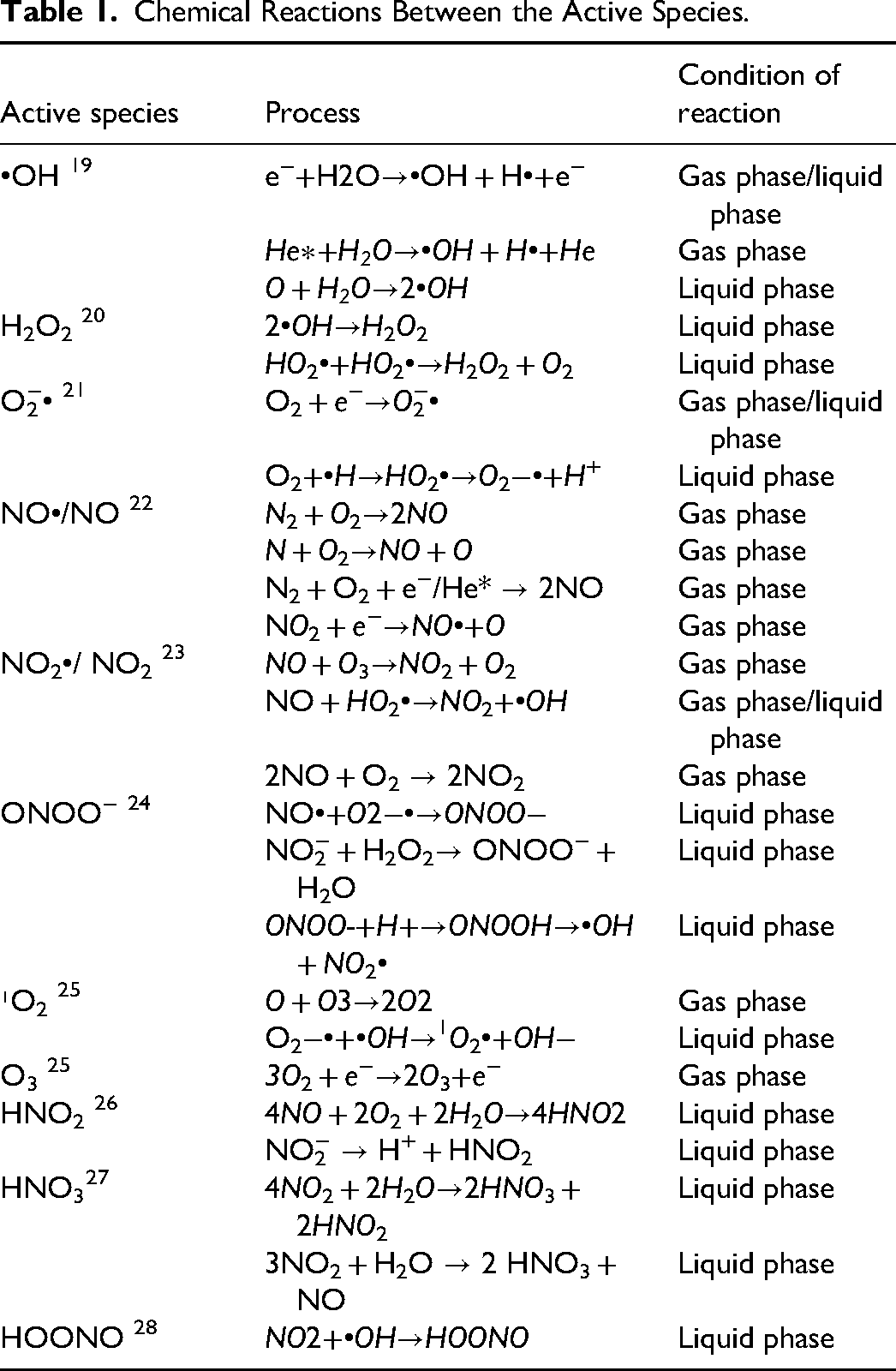
The stability and reactivity of an active species are key factors in determining its effectiveness in LTP-related reactions. According to the characteristics of reaction kinetics, it can be divided into short-lived species and long-lived species. Long lived active substances (τ > 10²s) were mainly stable molecules such as hydrogen peroxide (H2O2), nitrite (NO2−), and ozone (O3), and their persistence was due to the low reaction rate constant.29–31 Short-lived active species (τ = 10−⁹-10−⁶s) include hydroxyl radical(•OH), superoxide anion(O2−•), etc They are typically high-energy and highly reactive, capable of rapidly triggering oxidative stress reactions within the plasma-acting region (gas-liquid/gas-tissue interface). The whole inactivation process is usually in the range of nanoseconds to microseconds, and they decay and lose their anti-tumor activity. Besides changes in temperature, humidity and chemical environment can also cause the loss of activity of short-lived active substances. Nevertheless, these slightly fragile nitrogen-oxid-rich living species have important applications in fields such as medicine, environmental governance, and materials science. The temporal-spatial synergistic effect of short-lived species which destroy tumor cell structure through rapid oxidation reaction and long-lived species which affect cell function through continuous chemical action is an important mechanism of plasma therapy selectivity.
LTP Device
As the application of LTP in cancer treatment is in an active research stage, the design and development of its generating devices have also become a research hotspot. Currently, there are several LTP devices under research and development and application, all of which have the distinct advantage of precisely suppressing cancer while minimizing damage to adjacent tissues. Table 2 shows the structure diagram of the three LTP devices and their comparison.
Shows the Structure Diagram of the Three LTP Devices and Their Comparison.
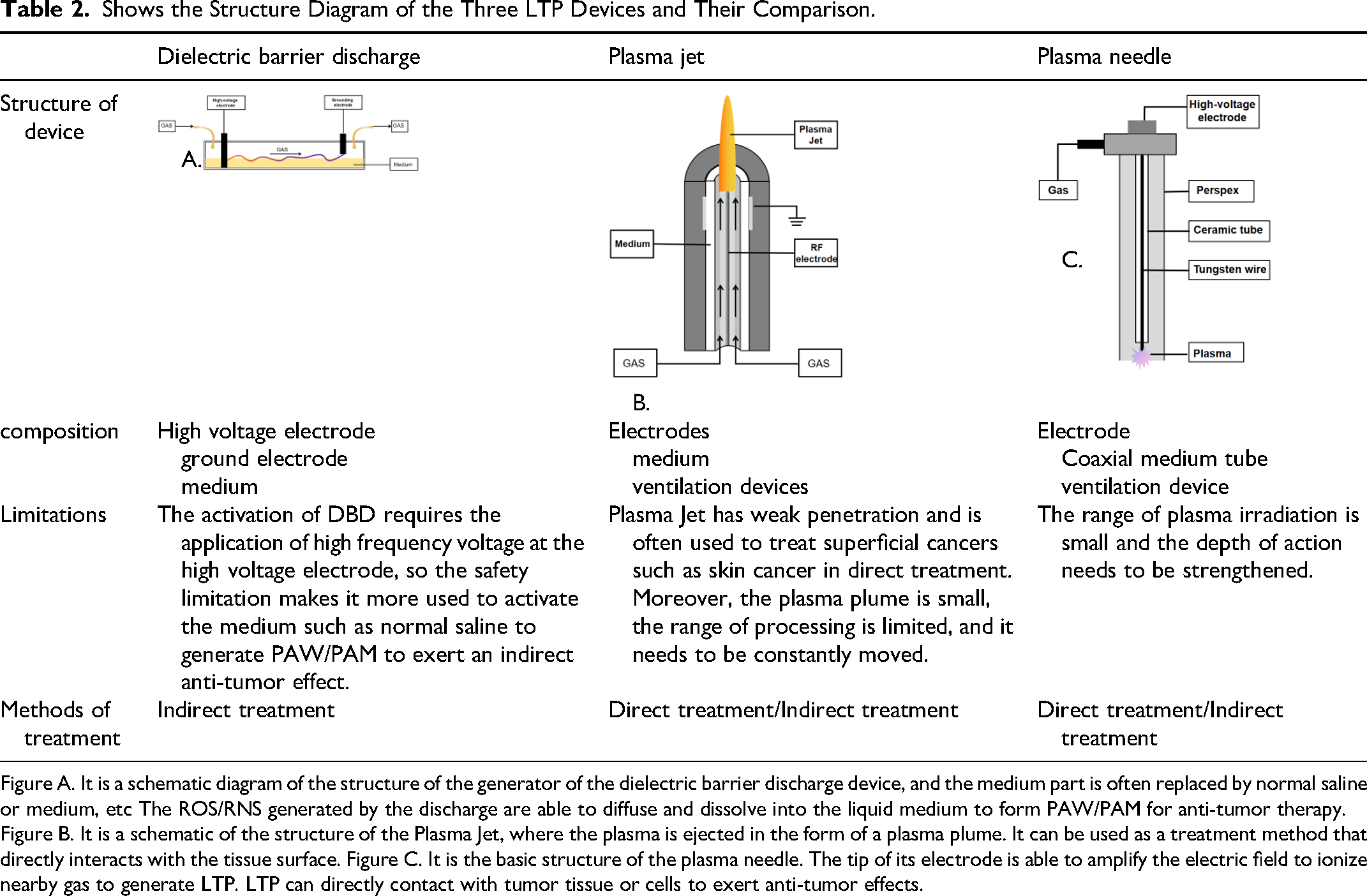
Figure A. It is a schematic diagram of the structure of the generator of the dielectric barrier discharge device, and the medium part is often replaced by normal saline or medium, etc The ROS/RNS generated by the discharge are able to diffuse and dissolve into the liquid medium to form PAW/PAM for anti-tumor therapy. Figure B. It is a schematic of the structure of the Plasma Jet, where the plasma is ejected in the form of a plasma plume. It can be used as a treatment method that directly interacts with the tissue surface. Figure C. It is the basic structure of the plasma needle. The tip of its electrode is able to amplify the electric field to ionize nearby gas to generate LTP. LTP can directly contact with tumor tissue or cells to exert anti-tumor effects.
The dielectric barrier discharge (DBD) plasma reactor is one of the most common plasma generating devices. Its basic structure consists of two electrodes and an insulating medium (such as glass or ceramic) in between. When a high-frequency/high-voltage alternating electric field is applied, the gas between the electrodes ionizes to form plasma, and the entire discharge process usually occurs within microseconds or nanoseconds. The insulating medium has a dual regulatory function: it limits the current to prevent large-scale arc discharge, facilitating the accumulation of the electric field and stabilizing the entire discharge process. Secondly, it promotes the generation of microdischarges. 32 The advantage of DBD lies in ensuring the uniformity and stability of the discharge, which is a crucial factor for generating LTP at atmospheric pressure. DBD is typically applied in the indirect stimulation and inhibition of tumors. Because the medium between the electrodes can be replaced with different liquids, such as cell culture medium, physiological saline, or Ringer's solution, the unique active species of LTP can quickly dissolve in these liquids to form plasma-activated water (PAW), thereby exerting its biological effects indirectly through PAW. For instance, in tumor treatment, PAW containing a large amount of active nitrogen oxides can be directly applied to the surface of tumor cells to achieve indirect inhibition of tumor cells by LTP. 33
The plasma jet device is another device that generates LTP through electrical discharge. However, unlike the above principle, it produces a directional plasma jet through the combined effect of the electric field and gas flow (such as argon or air). 34 When the gas passes through the jet channel, ions are accelerated by the electric field and ejected at high speed, forming a plasma plume. The stability of the jet is mainly determined by the matching of gas flow rate and electric field strength. The electric field provides sufficient energy to maintain the ionization of the jet. The gas flow rate determines the transmission efficiency of the plasma and the shape of the jet. Therefore, by adjusting the voltage and gas flow rate, the length and density of the jet acting on the surface of tumor cells can be precisely controlled to achieve precise treatment of tumor tissues. Similarly, the plasma jet device can also achieve indirect tumor treatment effects through the activation of PAW/PAM. 35 kINPen, which is currently clinically approved in Europe, has been clinically approved. Its micro-plasma nozzle is suitable for local treatment in a small area, and may achieve synergistic therapeutic effect when combined with other treatment regimens. 36
Plasma Needle is a convenient LTP Device, which can stimulate non-equilibrium glow discharge by radiofrequency electric field under normal pressure. The generation of reactive oxygen species (ROS) and nitrogen species (RNS) can be regulated by adjusting gas composition (such as helium-air mixing ratio) and power parameters. The main structure consists of a sharpened stainless steel needle electrode and a grounded metal cylinder surgery. Due to the very small radius of curvature of the tip of the electrode (about 0.1 mm), a local strong electric field region was formed. Compared with DBD, plasma needle can directly achieve stable discharge through electrode geometry design and gas flow field control without dielectric barrier layer. It has the unique advantages of high spatial resolution (submillimeter range) and low energy input, which provides a new physical tool for local tumor treatment.37–40
Anti-Tumor Mechanisms of LTP
Apoptosis
Apoptosis is a highly conserved and tightly regulated form of programmed cell death in a cell organism. It plays a vital role in body growth, development, tissue homeostasis maintenance and disease defense. 41 In physiological states, apoptosis dynamically balances cell numbers, removes dysfunctional cells, and shapes organ architecture. And when the cell receives too much external stimulation and is unable to compensate or repair, the apoptosis process is initiated. During this process, cells first undergo morphological changes such as volume reduction and nuclear chromatin aggregation. The nucleus and cytoplasm then break down into small vesicular apoptotic bodies. Apoptotic bodies are rapidly engulfed and degraded by surrounding phagocytes. The initiation and execution of apoptosis depends on a series of complex signal transduction pathways. These pathways involve not only signals inside and outside the cell, but also precise regulation of a series of specific proteins and enzymes.42–46 LTP activates apoptosis by causing cell damage and disrupting the normal physiological function of cells. Next, we will explore the pathway and molecular mechanism of LTP induced apoptosis of tumor cells in detail.
Cell Membrane Injury
The cell membrane is the first line of defense for cell survival and physiological function. Cell membrane regulates the entry and exit of substances through selectivity and permeability, protects the stability of the cellular environment and isolates potentially harmful substances from the outside world. The cell membrane is mainly composed of phospholipid molecules that form a hydrophobic internal barrier. The heads (hydrophilic) of the phospholipid molecules are oriented laterally (extracellular fluid and cytosol) and the tails (hydrophobic fatty acid chains) are aligned inwards. In addition, receptor proteins on the surface of the cell membrane recognize and respond to signaling molecules to maintain the physiological functions of the cell. The bilayer structure of the cell membrane makes it easier for fat-soluble substances to penetrate the cell membrane, while the corresponding hydrophilic substances, ROS, are difficult to penetrate the cell membrane. Moreover, there are few protein carriers involved in the transmembrane transport of ROS on the cell membrane. However, a large number of reactive nitrogen oxides (RNS and ROS), which are unique to LTP, will oxidize lipids and proteins in the cell membrane upon contact with tumor cells, thereby destroying the structural stability of the cell membrane. This oxidative reaction weakens the skeleton structure of the membrane, leading to increased membrane permeability and disordered intracellular environment, which in turn triggers the apoptotic mechanism. Some scholars have simulated the transmembrane transport process of active substances under oxidation conditions and the effect of electric field. The results showed that hydrophilic ROS itself could not directly pass through the lipid layer of the cell membrane, but its strong oxidation could cause the oxidation of the phospholipid bilayer, weaken the integrity of the membrane, and provide the basis for ROS transmembrane. In addition, oxidative damage also reduces the electric field threshold required for the formation of pores in the membrane, providing a new channel for other active substances to penetrate the membrane, leading to the collapse of the membrane and inducing apoptosis. In vitro experiments, changes in G-/ F-actin ratios, activation of apoptosis-related enzymes caspase-3 and caspase-7, and increased levels of DNA degradation in osteosarcoma cytoskeleton were observed after co-incubation of LTP-activated substrates with osteosarcoma cells. These experimental results further confirmed the close relationship between plasma damage to tumor cell membrane and cell apoptosis. 47
Activation of Endogenous Apoptotic Pathways
Apoptosis is a complex biological process regulated by multiple signal pathways. The mitochondrial apoptosis pathway plays a very important role. When cells are subjected to endogenous damage or exogenous signals, pro-apoptotic BH3-only proteins (such as Bim, Bid, Puma, Noxa) are activated, and the activated BH3-only proteins can directly bind to and antagonthe anti-apoptotic protein bcl-2. This activates the pro-apoptotic effector proteins bax and bak, which undergo conformational changes and oligomerization to form pores in the mitochondrial membrane, namely mitochondrial outer membrane permeability. MOMP causes the loss of mitochondrial membrane potential (ΔΨm), mitochondrial swelling, and the release of a variety of pro-apoptotic substances such as Cytochrome c, Smac/DIABLO, and AIF (apoptosis inducing factor) into the cytoplasm. The most important of these, cytochrome c, binds to Apaf-1, which recruits pro-Caspase-9 through the CARD domain to form apoptotic bodies. pro-Caspase-9 can self-cleave into active Caspase-9, which activates effector caspases such as Caspase-3 and Caspase-7, and then clefts multiple structures involving the nucleus, cell membrane, cytoskeleton and other structures, leading to the characteristic changes of apoptosis.48,49 This pathway dynamically regulates mitochondrial outer membrane permeability (MOMP) through the Bcl-2 protein family, that is, the balance between pro-apoptotic members (such as Bax, Bak) and anti-apoptotic members (such as Bcl-2, Bcl-xL) determines the apoptotic process. 50 Studies have found that LTP stimulation can significantly reduce the Bcl-2/Bax ratio of tumor cells, promote the expression of pro-apoptotic proteins while inhibiting anti-apoptotic proteins, and finally activate the caspase cascade to induce apoptosis. 51 At the same time, in vivo experiments showed that LTP could significantly increase intracellular ROS, trigger the decrease of mitochondrial membrane potential and induce cell apoptosis in a dose-dependent manner. 52
The regulation of apoptosis is a sophisticated and complex network. In the process of mitochondrial apoptosis pathway as the main line, it also involves the cross influence of other pathways, among which JNK pathway is also a key. JNK (c-Jun N-terminal kinase) is an important member of the MAPK (mitogen-activated protein kinase) family, which mainly responds to cellular stress signals (such as oxidative stress, DNA damage, inflammatory factors, etc). It participates in cell apoptosis, proliferation and differentiation by regulating the phosphorylation of downstream target proteins. In apoptosis, activation of the JNK pathway promotes programmed cell death through a variety of molecular mechanisms. The phosphorylation of JNK leads to the phosphorylation of Bcl-2 family member Bim and transcription factor c-Jun, which in turn promotes the activation of apoptotic protein caspase-3/7. In a study to investigate the role and mechanism of LTP in inducing mitochondrial-mediated apoptosis in human cervical cancer cells (HeLa), researchers showed that ROS accumulation caused increased Cyto C release and JNK and p38 phosphorylation. It was further concluded that LTP activated the mitochondrial apoptotic pathway through the Cyto C/JNK/p38 signaling pathway. This conclusion was also confirmed by in vitro experiments. 53
Above all, ROS can also co-promote apoptosis through the p53 pathway: LTP stimulates the activation of ATM/ATR kinase and inhibits MDM2-mediated p53 ubiquitination degradation through phosphorylation. After nuclear translocation of stable p53 protein, on the one hand, upregulation of p21 causes cell cycle arrest, and on the other hand, activation of mitochondrial apoptosis pathway through increasing Bax/Bcl-2 ratio. 54 The PI3K/AKT pathway maintains cell survival by inhibiting apoptosis-related proteins and activating pro-survival factors. PI3K catalyzes the generation of the second messenger PIP3, which activates Akt protein. Akt prevents the release of cytochrome C from mitochondria by phosphorylating the pro-apoptotic protein Bad, which binds to the anti-apoptotic members of Bcl-2 family (such as Bcl-2 and Bcl-xL) and thus prevents the occurrence of apoptosis. At the same time, Akt can phosphorylate Caspase-9 (inhibiting its activity) and Caspase-3 precursor, blocking the execution phase of apoptosis. Studies have shown that LTP can cause the down-regulation of p-PI3K and p-Akt in cells, and inhibit the anti-apoptotic function of PI3K/AKT pathway. In the study of exploring the anti-tumor effect of LTP on lung cancer cells, researchers found that LTP inhibited the PI3K/AKT pathway, and its ability to inhibit the activity of GSK-3β was weakened due to the decreased expression of activated AKT, which enhanced the degradation of β-catenin by GSK-3β, resulting in the synergistic inhibition of PI3K/AKT-Wnt pathway.55–58
Death Receptor Pathway
The death receptor pathway is another important mechanism of apoptosis. This pathway activates apoptosis signals through the binding of cell surface death receptors to their ligands, the core of which lies in the activation of caspase-8. Taking the Fas/FasL system as an example, when Fas ligand (FasL) binds to the Fas receptor, it can induce the trimerization of the death receptor, and then recruit the FAS-associated protein with death domain (FADD). 59 FADD binds to caspase-8 precursors through a death effect domain (DED) to form a death inducing signaling complex (DISC), thereby activating caspase-8. Activated caspase-8 can directly activate downstream effector caspases (such as caspase-3 and caspase-7), ultimately leading to apoptosis.
Studies have shown that LTP can induce apoptosis of tumor cells by activating Fas pathway. 60 ROS produced by LTP play a key role in this process. LTP treatment can up-regulate the expression of Fas and FasL in tumor cells, which may be related to the change of cellular oxidative stress. Oxidative stress not only promotes the cell surface expression of Fas receptors, but also enhances FasL synthesis, thereby increasing the efficiency of FAS-FASL interactions. And activated caspase-8 can also generate tBid by cutting the Bid protein, thus connecting the exogenous apoptosis pathway mediated by death receptors and the endogenous apoptosis pathway mediated by mitochondria. 61
Dysfunctional DNA Damage and Repair Mechanisms
The mechanism of LTP-induced apoptosis of tumor cells involves multi-level complex regulation. Among them, DNA damage causing apoptosis is also a hot topic. When the human body receives endogenous or exogenous stimuli to cause DNA damage, it will initiate self-protection mechanisms of repair and apoptosis. When DNA is damaged, ATM/ATR can recognize and activate. ATM/ATR further phosphorylates p53 (eg, Ser15, Ser20), prevents its ubiquitination and degradation by MDM2, and stabilizes p53 and enters the nucleus. Stable p53 can up-regulate the expression of apoptotic proteins and inhibit the expression of anti-apoptotic proteins, which leads to the increase of the ratio of BAX/Bcl-2 and induces cell apoptosis. On the other hand, p53 activates the cell cycle checkpoint kinase CHK1 to arrest the cell cycle and buy time for DNA repair. When DNA damage is too severe to repair, it will cause cell cycle arrest and apoptosis. In a study on the anticancer effect of LTP on oral squamous cell carcinoma (OSCC) and its molecular mechanism, researchers found that LTP could induce DNA damage and activate the ATM/p53 pathway. The expressions of phosphorylated ATM (Ser1981), p53 (Ser15/20/46), p21 and γH2AX (DNA damage markers) were up-regulated in LTP treated OSCC, and ATM knockdown could reverse the expression of these molecules. The LTP human colon cancer cell study also suggested that ROS could directly cause DNA damage and induce subsequent apoptosis including P53 pathway and mitochondrial pathway62–67. However, there is heterogeneity in the response of different tumor cells to LTP: Although LTP treatment generally induces up regular expression of the DNA damage marker γ-H2AX, its effect is directly related to the apoptosis process, rather than simply reflecting the physical damage of DNA. 68 Moreover, antioxidant experiments show that although ROS clearance can inhibit cytotoxicity and gamma-H2AX induction, it cannot completely block DNA damage, indicating that the mechanism of LTP not only depends on the direct destruction of ROS, but also may involve the direct intervention of physical factors (such as electric field effects) on DNA structure. Or through epigenetic modification, mitochondrial apoptosis pathway and other indirect regulation. In summary, the killing effect of LTP on tumor cells has multi-target characteristics: on the one hand, it directly destroys DNA and cell membrane integrity through ROS attack and physical effects. On the other hand, the DNA damage response (DDR) pathway is activated and cascaded with the apoptosis signaling network. This synergistic effect of multiple mechanisms not only provides a new idea for overcoming drug resistance in cancer therapy, but also suggests that future studies should further analyze the interaction mechanism of key signaling pathways and optimize clinical application strategies based on the characteristics of tumor subtypes.
Endoplasmic reticulum Stress and Unfolded Protein Response (UPR)
Endoplasmic reticulum (ER) stress is the core site of protein folding and post-translational modification, and its homeostasis can trigger ER stress. 69 Er stress manifests as abnormal accumulation of Unfolded or misfolded proteins in the ER lumen, which activates the Unfolded protein response (UPR) to restore ER function. Studies have found that LTP can directly activate the UPR signaling network through oxidative stress or membrane potential perturbation, and initiate three classical UPR branching pathways to coordinate stress adaptation or apoptotic decision-making. 70 Additionally persistent ER stress triggers disturbances in the endoplasmic reticular-mitochondrial Ca² + signal axis: release of the calcium bank of the endoplasmic reticulum leads to cytoplasmic Ca² + overload. In this process, up-regulation of the UPR end-effector molecule CHOP (C/EBP homologous protein) further inhibits the activity of anti-apoptotic protein BCL-2, promoting the transfer of apoptotic signals from the endoplasmic reticulum to the mitochondria. 71 This mechanism suggests that LTP-induced ER stress is not only involved in the dynamic balance regulation of the UPR pathway, but also closely related to the signal interaction between organelles and the integration of apoptosis signal networks.
Others
Since apoptosis is a complex and multi-factor regulated biological process, LTP can induce apoptosis of tumor cells through other ways besides the above-mentioned mechanisms. Studies have shown that LTP is able to oxidize and degrade histone mRNA during the early S phase of the cell cycle. In vitro experiments have shown that at least 16 histone mRNAs in LTP-treated breast cancer cells are oxidized and rapidly degraded to form 8-oxoguanine (8-oxoG). 72 8-oxoG can cause base substitution from G to T, thus triggering mutations. 8-oxoG. In addition, the expression of DNA damage response genes was significantly upregulated 12 h after LTP treatment, suggesting that 8-oxoG modification and histone mRNA degradation (rather than direct DNA damage) are the main mechanisms of LTP-induced cancer cell death independent of cancer cell subtypes.
In addition, LTP-stimulated tumor cells also exhibit features of immunogenic cell death (ICD), including calreticulin (CRT) exposure and ATP release. These changes induce an anti-cancer immune response by activating the innate immune system. 73 At the same time, LTP stimulation significantly increased the expression and secretion of high mobility group protein B1 (HMGB1) in tumor cells, and the aggregation of calreticulin (CRT) on the cell membrane was observed. These phenomena are typical markers of immunogenic cell death. 74
Ferroptosis
Ferroptosis is a form of cell death caused by disruption of the homeostasis of the cellular antioxidant system, the core mechanism of which involves abnormal iron metabolism and lipid peroxidation. First, dysregulation of iron metabolism is one of the key drivers of iron death. Normal cells maintain intracellular iron balance through ferritin and transferrin, but when cells are in pathological conditions, intracellular ferritin degradation increases to produce a large amount of divalent iron, which is the most important manifestation of ferroptosis. LTP stimulation can lead to an increase in the concentration of free iron ions in cells. These iron ions react with hydrogen peroxide (H2O2) to generate hydroxyl radical (•OH) through Fenton reaction, and then damage cell DNA. This DNA damage activates the DNA repair enzyme poly ADP-ribose polymerase-1 (PARP-1), and the Poly ADP-ribose produced during its repair induces mitochondrial release of apoptosis-inducing factor (AIF), thereby promoting apoptosis. 75 In addition, activation of PARP-1 consumes a lot of energy, leading to cell energy exhaustion and death. 76 Due to the high dependence on energy for the rapid proliferation of cancer cells, this energy depletion effect makes cancer cells more prone to death. It is important to note that the accumulation of poly (ADP-ribose) induced by PARP-1 activation can further activate ion channel TRPM2, resulting in a large amount of calcium ion inflow and accelerating the apoptosis process of cancer cells. 77
On the other hand, another hallmark of ferroptosis is the breakdown of the antioxidant system. Among them, glutathione peroxidase 4 (GPX4) is the only key enzyme that can reduce lipid peroxides, and its activity depends on reduced glutathione (GSH). When System Xc− function, which consists of SLC7A11 and SLC3A2, is inhibited, cysteine uptake is reduced and GSH synthesis is blocked, leading to GPX4 inactivation. Studies have shown that LTP stimulation can reduce the expression of ferroptosis suppressor protein 1 (FSP1) in lung cancer cells, thereby increasing the level of lipid peroxidation and promoting iron death in tumor cells. Animal experiments have shown that the proportion of lipid peroxidation markers 4-hydroxynonenal (4-HNE) and prostaglandin peroxidase synthase 2 (PTGS2) in tumor samples of mice treated with LTP increased significantly, but no significant damage to liver and kidney function was observed. 78 Similarly, in an anti-tumor study of LTP in oral cancer, LTP-stimulated cells showed increased expression of 4-HNE and 8-OHdG (markers of oxidative DNA damage) and decreased expression of GPx4 by immunohistochemistry. Iron chelators DFO and Fer-1 significantly reversed LTP cytotoxicity. 79
Autophagy
As an important physiological process of cell self-degradation, autophagy plays a key role in maintaining cell homeostasis and responding to stress stimuli (such as nutrient deficiency, hypoxia, drug stress, etc). This process realizes the recycling of cell components by degrading damaged organelles, abnormal protein aggregates and other macromolecular substances. 80 Existing studies have shown that LTP can induce autophagy of tumor cells by regulating LC3-II/LC3-I transformation, Beclin-1 expression, p62 degradation, PI3K/AKT/mTOR/p70S6K signaling pathway and other multiple mechanisms. 81
The mTOR signaling pathway occupies a core position in the autophagy regulatory network. Its functional complex mTORC1 inhibits the biological activity of ULK1 kinase and VPS34 complex through phosphorylation modification, thereby negatively regulating autophagy initiation and autophagosome membrane nucleation. 82 Furthermore LTP treatment can relieve the inhibition of mTOR on autophagy through two mechanisms: on the one hand, mTOR activity is reduced by inhibiting the phosphorylation cascade of PI3K/AKT pathway. On the other hand, it directly induces the inactivation of the mTORC1 complex and ultimately activates the autophagy program of tumor cells.83,84
As a key regulator of autophagosome formation, Beclin-1 catalyzes the generation of PI3P lipid signaling molecules by forming complexes with Class III PI3K (VPS34). This molecule promotes isolation membrane extension and autophagosome maturation by recruiting effector proteins such as DFCP1 and WIPI2. Under LTP-induced stress, BH3-only proteins such as pro-apoptotic proteins Bad and Bim can competitively bind to Bcl-2/Bcl-XL, release bound Beclin-1 molecules, and then activate autophagy cascade. 85
The lipidation modification process of LC3 protein is a classic biomarker for monitoring autophagy activity. Specifically, the cytoplasmic LC3 precursor is cleaved by Atg4 protease to produce LC3-I, which is subsequently covalently bound to phosphatidylethanolamine (PE) through Atg7 (E1 ligase) and Atg3 (E2 ligase) to form membrane-bound LC3-II. LTP treatment can significantly increase the LC3-II/LC3-I ratio in tumor cells, which indicates accelerated autophagosome membrane formation, indicating substantial enhancement of autophagy flux. 86
Pyroptosis
Pyroptosis is a form of dissolved programmed death that is accompanied by a violent inflammatory response. Its typical features include changes in membrane permeability, cytoplasmic swelling and release of intracellular proinflammatory factors. 87 The pathophysiological manifestations were bulbous protrusions and eventual dissolution of the cell membrane. 88 Functional activation of GSDME as a porogen precursor depends on proteolytic cleavage. in the resting state, GSDME remains inactive through self-inhibition of C-terminal domain. After being cut by proteases such as caspase-3, its n-terminal domain (Gasdermin-E-NT) can oligomerize to form a plasma membrane pore with a diameter of 10-15 nm, triggering osmotic pressure imbalance and driving the scorch death process. 89 The expression level of GSDME is closely related to the transformation of cell death patterns: in the high-expression model of GSDME, LTP treatment can significantly promote the pyrodeath phenotype, while the low-expression group mainly presents non-inflammatory apoptosis characteristics. 90 The mechanism study shows that the enhancement effect of coke death induced by LTP is closely related to the characteristics of oxidative stress. ROS produced by LTP activate the JNK-cytochrome c pathway, thereby activating the caspase-9/caspase-3 cascade. Activated caspase-3 not only cleaved GSDME to produce functional N-terminal fragments, but also cooperated with the granase system to amplify the pyrodeath signal, and finally realized the dynamic transformation of apoptosis-pyrodeath mode.
LTP Joint Application
As the need for cancer treatment continues to evolve, researchers are exploring the use of LTP in combination with other treatments to further improve treatment effectiveness and reduce side effects. Some researchers have found that chitosan colloids treated by LTP irradiation can produce chitooligosaccharides and reactive small molecules, which exhibit selective anti-tumor activity, enhanced antioxidant and mucosal adhesion, and excellent biocompatibility. This provides a new strategy for the development of low toxicity and high efficiency tumor therapeutic agents. 91 In addition, LTP also performed well in combination with other treatment regimens. Here are the main ways LTP technology can be used in combination with other treatments.
Combined Application of LTP and Chemotherapy
The synergistic anti-tumor effect of LTP and chemotherapy drugs has been verified in many studies, and its mechanism of action mainly involves pharmacokinetic changes and regulation of cellular stress pathways. In the cisplatin combination therapy system, LTP has a synergistic effect with platinum compounds by damaging DNA molecules. 92 This synergistic effect enables the equivalent anti-tumor effect to be maintained even when the dose of cisplatin is reduced, and systemic toxic reactions such as renal toxicity and myelosuppression can be significantly alleviated. In the doxorubicin (DOX) combination therapy regimen, LTP exhibits a unique “synergy-attenuating” dual mode response. In vitro experiments confirmed that LTP pretreatment of MCF-7 breast cancer cells can coordinate the effects of DOX by down-regulating anti-apoptotic protein Bcl-2 and up-regulating pro-apoptotic protein Bax to activate mitochondrial apoptosis pathway. Animal models further showed that the tumor volume was reduced more significantly in the combination treatment group than in the monotherapy group. 93 In addition, studies have confirmed that LTP can effectively improve the sensitivity of tumor cells to DOX, and reduce the cardiotoxicity of drugs on the basis of the same efficacy. The combination of LTP and paclitaxel can significantly increase the apoptosis rate and increase the frequency of DNA breakage. Studies have shown that LTP combined with paclitaxel can significantly increase the proportion of tumor dead cells, which initially confirms the potential of LTP in enhancing the efficacy of paclitaxel. 94 The combination of chloroquine and LTP exerts a synergistic effect by regulating autophagy. Treatment of tumor cells by chloroquine alone led to abnormal autophagosome formation and disruption of mitochondrial homeostasis to induce cell death. When LTP was combined with chloroquine, Drp1 mitochondrial fission protein was down-regulated, while PINK1 protein was up-regulated. This suggests that chloroquine may further promote cell death by increasing LTP-dependent mitochondrial instability. 95 In the experimental study of the combination of LTP and temozolomide in glioblastoma, LTP pretreatment can make tumor cells more sensitive to TMZ, which is related to the increase of cell membrane permeability and ROS level induced by LTP. 96 Studies on the combined treatment of LTP and metformin in glioma cells also showed the synergistic effect of LTP, which may be related to the activation of c-Fos pathway. More importantly, this conclusion further supports the potential of LTP in combination with metabolic inhibitors. 97 In the combination of LTP and vitamin C, a large number of reactive oxygen species (ROS) produced by LTP can further enhance the anticancer activity of vitamin C. Specifically, LTP stimulation upregulates the expression of aquaporin-3 (AQP3) in tumor cells. This increases the uptake of exogenous H2O2 by cancer cells, thus significantly elevating the level of intracellular ROS. In addition, ROS produced by LTP works with vitamin C to activate the JNK signaling pathway and induce cell apoptosis. This synergistic mechanism not only enhances the anti-tumor effect, but also shows the potential to improve the therapeutic effect while reducing the drug dose. 98
Combined Application of LTP and Nanotechnology
The collaborative treatment system of LTP and nanotechnology shows the potential of precision therapy, and its core advantage is to enhance tumor targeting through physico-chemical coupling effects. 99 Small size gold nanoparticles can enhance the effect of LTP-induced apoptosis by consuming intracellular glutathione (GSH) and regulating oxidative stress. It was found that the construction of p-FAK antibody-coupled gold nanoparticles could enter melanoma cells and induce apoptosis by binding p-FAK. In order to further improve the selective killing effect of tumor cells, the researchers introduced GNP stimulated by LTP into tumor cells. The results showed that the killing effect was significantly higher than that of gold nanoparticle – antibody complex alone. 43
In another study, low-dose PEG-coated gold nanoparticles combined with LTP effectively inhibited the proliferation of cancer cells by inhibiting the PI3K/Akt signaling pathway. In addition, combination therapy upregulates epithelial markers (such as e-cadherin) and downregulates interstitial markers (such as n-cadherin, Slug, and Zeb-1), further inhibiting tumor cell growth in solid tumors. 100
In addition, iron nanoparticles, another common class of nanomaterials, are commonly used as heating media in cancer treatments. Under the action of magnetic field, iron nanoparticles generate heat through photothermal effect, thus enhancing the killing effect on cancer cells. The combination of LTP and iron nanoparticles significantly aggravated the DNA damage of tumor cells and led to changes in the BAX/BCL-2 ratio, thus promoting apoptosis. 101
Notably, it has been shown that pretreatment of LTP is able to promote drug influx to enhance the efficacy of chemotherapeutic agents topotecan (TPT) and temozolomide (TMZ) against glioblastoma (GBM) and other cancers. Moreover, this technology can be adapted to a variety of drugs, and has the potential to be applied to the treatment of other tumors 102 .Recently, researchers have focused on the combined use of LTP and methylene blue-loaded nanoparticles (Micelle-MB). Through simulation algorithms, it is proposed that LTP can successfully optimize the structure of MB loaded micelles and verify their stability and drug localization. At the same time, the synergistic effect of LTP and Micelle-MB is significantly better than that of free MB in vitro experiments, which provides a new combination strategy for the treatment of glioblastoma. 103
The combination of LTP with chemotherapy drugs and nano-therapy is another innovation in the innovative concept of LTP anti-tumor therapy. The toxic side effects of traditional tumor treatment and the drug resistance that cause the poor prognosis of tumor patients are still problems that plague us. However, the combination of LTP with chemotherapy and nanotechnology can reduce the dosage of drugs and improve the sensitivity of tumor cells to drugs, which provides a new idea to overcome the above problems.
Conclusion
As an emerging biomedical technology, LTP provides an innovative intervention strategy for disease treatment due to its unique physicochemical properties. This review systematically analyzed the principle of LTP and the progress of its medical application, focusing on its molecular mechanism and transformation potential in tumor therapy. Studies have shown that LTP can precisely regulate the cell fate decision process by producing output molecules such as reactive oxygen species/nitrogen species (ROS/RNS). On the one hand, LTP selectively kills tumor cells by inducing programmed death (including apoptosis, autophagy and ferroptosis). On the other hand, it can exert synergistic anti-tumor effects by combining with other therapeutic methods. This dual regulatory property gives LTP a unique advantage in overcoming tumor therapy.
There is no doubt that LTP has the ability to kill tumor cells, but there are still many problems behind this powerful killing. Which is worthy of attention,different concentrations of ROS/RNS in PAW/PAM activated by LTP can even produce completely opposite biological effects. Previous studies have found that treatment of breast cancer cells with low concentrations of paw resulted in a significant decrease in cell viability due to cell toxicity, but higher dilutions of paw caused an increase in cell viability. This completely opposite effect suggests that the anti-tumor effect of LTP is concentration-dependent on ROS/RNS concentration. However, due to the lack of complete and unified use standards, more exploration is still needed in the dose management of further conversion therapy. 104 Moreover, LTP could weaken the anti-tumor effect of antioxidants when used together with them. 105 This proves that the clinical translation of LTP still faces many challenges.
The current difficulties in clinical translation of LTP can be summarized in the following two aspects. From the perspective of LTP, the current technological development of LTP equipment has crossed the basic preparation stage, but the systematic construction of its core parameters still faces significant challenges. Firstly, the diversity of device structure directly leads to the differentiation of key parameters. At the level of power supply design, the dielectric barrier discharge device and the plasma jet device adopt different types of alternating and pulsed power supply, respectively, and the resulting electric field strength, electron density and energy distribution show great differences. In terms of electrode configuration, due to the different electric field focusing characteristics of the needle structure and the coaxial structure, the local field strength of the output and the coverage of the active particles are essentially different. However, the surface discharge devices significantly affect the discharge uniformity and stability due to the differences in the physical characteristics of the dielectric materials. In addition, although the component regulation of working gas can directionally regulate the types of active substances, its proportion fluctuation will still lead to uncontrollable deviation of biological efficacy. In a study on the in vitro efficacy and cellular mechanism of cold piezoelectric plasma targeting cholangiocarcinoma cells, Piezo Brush PZ2 device with two discharge modes was used. Pz-DBD (Dielectric barrier discharge): a dielectric barrier discharge DBD that generates a uniform discharge through a dielectric barrier. And direct needle discharge corona discharge Pz-CD. The DBD had a lower power (13 ± 5 mW) while the needle-shaped corona had a higher power (74 ± 15 mW) and produced more reactive nitrogen species. 106 This fragmentation of the parameter system further exposes the lag of the standardization process. Although international organizations have begun to develop relevant medical device standards, there is no consensus on the measurement methods of core parameters. For example, the electrical and optical detection methods of power density still have systematic deviations in complex scenarios, and the definition of bioequivalent dose remains in macroscopic dimensions such as time and distance for a long time, which fails to effectively reflect the spatial heterogeneity of plasma plumes. The confusion of parameter system has formed multi-dimensional constraints on technology transformation. In the field of basic research, the same biological model using different equipment may reach contradictory conclusions, which seriously weakens the horizontal comparability of research data. At the industrialization level, the heterogeneity of device interfaces and consumables greatly increases the complexity of supply chain and indirectly drives up the cost of clinical translation. In the future, it is urgent to establish an interdisciplinary collaboration mechanism, systematically construct the correlation model between equipment parameters and biological effects, and form a universal dose system through large-scale clinical verification, so as to break through the core bottleneck of LTP technology from the laboratory to the widespread application.
This fragmentation of the parameter system further exposes the lag of the standardization process. Although international organizations have begun to develop relevant medical device standards, there is no consensus on the measurement methods of core parameters. For example, the electrical and optical detection methods of power density still have systematic deviations in complex scenarios, and the definition of bioequivalent dose remains in macroscopic dimensions such as time and distance for a long time, which fails to effectively reflect the spatial heterogeneity of plasma plumes. Although the emerging standards try to introduce microscopic indicators such as active substance flux, the verification of their applicability in complex anatomical structures is still insufficient. The confusion of parameter system has formed multi-dimensional constraints on technology transformation.
From the perspective of tumor treatment, first of all, the mechanism of LTP has not been fully elucidated. This is because the mechanism of cell death caused by LTP is not independent. Different LTP stimulation methods can cause different responses in cells, and sometimes different responses will intricately cross, promote or inhibit, which requires in-depth study of its specific action pathways at the molecular and cellular levels. Moreover, due to the limited experimental technology, only a few liquid phase active particles can be detected, so the key active substances and biological mechanisms of LTP anticancer effects have not been determined. Currently, there are phase I clinical trials of LTP in the treatment of skin cancer, and the clinical application scenarios include direct treatment of ulcerative tumors, ablation of intradermal lesions, and inactivation of residual cancer cells in the tumor bed after surgery. The results of this study provide preliminary evidence of its safety and efficacy. In the integration of existing therapies, cold plasma can replace some surgery, or be used as an adjunct to radiotherapy/targeted therapy to reduce the dose and side effects. But the study did not mention whether toxic side effects occurred. In the future, the safety and long-term side effects of LTP still need to be systematically reviewed through large-scale, randomized controlled clinical trials. 107
Compared with the systemic administration of traditional chemotherapy, LTP can act locally on tumor cells or tissues, reduce systemic side effects, and make the treatment more precise and effective. In addition, the combination of LTP with other tumor techniques, such as nanotechnology, as described above, could further improve the effectiveness of targeted precision therapy. However, LTP still does not solve the problems such as unnecessary side effects, dose management difficulties, and possible drug resistance mechanisms in malignant tumors. This is indeed the core challenge for the anti-tumor research of LTP to move towards clinical application.
Future research should focus on understanding the mechanism of LTP, developing individualized treatment regimens, optimizing treatment parameters, improving device design, and conducting large-scale clinical trials. At the same time, it is of great significance to standardize the application standards of LTP in the medical field to ensure its safety and effectiveness, so as to promote the clinical application of LTP technology. In addition, ai learning models should be developed to adjust plasma parameters in real time according to tumor type, location, and patient metabolic characteristics to achieve individualized treatment. The single-cell transcriptome was used to reveal the heterogeneous response induced by LTP, and the spatial and temporal distribution of ROS/RNS was tracked by metabolomics to draw the dynamic molecular network. A global database of LTP equipment parameters (such as ROS/RNS generation rate and penetration depth) was established to promote the development of international standards. In summary, the medical application of LTP is standing at a key node from “technical breakthrough” to “clinical revolution”. Only through interdisciplinary collaboration, technological innovation and multi-dimensional promotion of ethical norms, can its transformative potential in precision cancer treatment be fully realized.
All authors have read and agreed to the published version of the manuscript.
Footnotes
Acknowledgments
I would like to express my sincere gratitude to all those who contributed to the completion of this work. First and foremost, I extend my deepest appreciation to my co-authors for their invaluable expertise, intellectual input, and collaborative efforts throughout this research. This work was supported by theDalian University of Technology-Liaoning Cancer Hospital Medical Engineering Cross-Union Fund under Grant/Award Number LD202309. The financial assistance and academic resources provided were essential for the execution of this study. Finally, I acknowledge the institutional support from the patience of my family/friends throughout this research endeavor.
Ethical Considerations
This review does not address ethical issues.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dalian University of Technology-Liaoning Cancer Hospital Medical Engineering Cross-Union Fund. No. LD202309.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent
Human experiments are not involved in this review.
