Abstract
Introduction
New treatment options and combination therapies have improved the outcome of prostate cancer (PCa) patients with an overall 5-year relative survival rate of > 98%. Along with long-term cancer survivorship comes an increased interest in reducing toxicity and improving quality of life after the different treatments.
When radiotherapy is applied, one strives to increase the dose to the tumor area while sparing organs at risk to balance between better oncological outcome and acceptable toxicity. To do so, there has been a shift from conformal radiotherapy techniques toward intensity-modulated radiation therapies. The latter creates a sharp dose fall-off which allows dose intensification to the areas of interest and simultaneously decreases the incidental dose delivered to the immediate vicinity of the target. 1
In the setting of PCa, modern techniques have reduced toxicity, 2 but there might be a reverse of the medal when reducing the dose to the immediate surroundings of the prostate.
The role of prophylactic irradiation of the lymph nodes (LNs) has been debated in many pathologies, including genitourinary tumors.3,4 Both the NRG/RTOG 9413 and GETUG-01 trial did not find an improvement in outcome when whole pelvic radiotherapy (WPRT) was applied compared to prostate-only radiotherapy (PORT) for high-risk PCa.5,6
Studies arguing against pelvic irradiation merely used conformal radiotherapy techniques so the question remains whether the absence of improved outcome is due to the nonneglectable incidental dose to the pelvic LN regions delivered with these techniques.
While intensity-modulated radiation techniques allows for intensifying the dose to the target, it also results in a spread out of the low and intermediate dose areas to the surroundings and a higher total body exposure. 1 Increasing the target volume from PORT to WPRT further increases the areas of normal tissue that are exposed to low to intermediate doses resulting in an increase in acute gastrointestinal toxicity when compared to PORT.7,8 Less is known about the impact of irradiating large volumes of pelvic bones, which is also the case for WPRT. The pelvic bone marrow accounts for more than half of the adult whole-body bone marrow and the active bone marrow with hematopoietic function accounts for 44%.9,10 As bone marrow is highly sensitive to radiation, WPRT can reduce bone marrow activity significantly. 10
Efforts to spare bone marrow might be beneficial and worthwhile to be evaluated prospectively.11,12
We performed a prospective randomized phase 3 trial in node-positive (pN1) PCa patients to evaluate whether WPRT improved clinical relapse-free survival compared to PORT. All patients were treated with volumetric-modulated arc therapy (VMAT).
This manuscript describes the incidental dose on the pelvic LN areas in the setting of PORT and compares differences in dose delivered to the organs at risk, including bone marrow, between PORT and WPRT when VMAT is applied. It also evaluates the impact of treatment volume on hematological parameters.
Materials and Methods
Patient Cohort
From May 2016 to June 2021, we performed a multicentric prospective phase 3 randomized trial, comparing PORT (ARM A) and WPRT (ARM B) combined with 18 to 24 months of androgen deprivation therapy (ADT) in pN1 PCa patients, after approval by the ethics committee of Ghent University Hospital on March 24, 2016 (EC/2016/0246) and registration at ClinicalTrials.gov (NCT02745587). A written informed consent from the patient was required for inclusion in the trial. The reporting of this study conforms to the CONSORT statement. 13
All patients had an adenocarcinoma of the prostate, without distant metastases (M0) on conventional imaging and received a local therapy to the prostate: either radical prostatectomy (RP) or external beam radiotherapy (EBRT). All patients, including patients treated with primary EBRT, received a diagnostic pelvic LN dissection as a staging modality. Time of referral of patients after RP was at the discretion of the treating physician so both adjuvant and salvage EBRT was allowed. All patients started with ADT (LHRH-(ant)agonist or anti-androgens) for a period of 18 to 24 months.
Treatment Planning
Pretreatment imaging consisted of a computed tomography (CT) scan and magnetic resonance imaging (MRI). The MRI was used for image fusion and planning. The clinical target volume (CTV) in ARM A consisted of the prostate (bed). For patients randomized to ARM B, also the pelvic LNs along the common, internal, and external iliac artery, obturator fossa, and the presacral nodes were irradiated. To evaluate the dose received by the nodal regions, each region was delineated separately. The delineation was based on the RTOG genitourinary radiation oncology specialists consensus statement on pelvic LN volumes (Supplemental Table S1). 14 For planning purposes, the different LN areas were summed to LN and expanded with a 2 mm margin to create the CTV_LNs.
The CTV was expanded by 5 mm to create a planning target volume (PTV).
Similar, a PTV_LNs was created by applying an isotropic expansion of 5 mm around the CTV_LNs.
The rectum, small intestine, sigmoid colon, femoral heads, and bone marrow were contoured as organs at risk. Bone marrow delineation was based on the publication of Mell et al., including the iliac, lumbosacral, and lower pelvic bone marrow areas. 15
Details on dose prescription and objectives are summarized in Supplemental Table S2. For bone marrow, the following dose objectives were used: volume of bone marrow receiving 7.5 Gy (V7.5) < 90% and a maximum of 18.9 Gy at 50% of the volume. These bone marrow constraints were applied in daily practice at our insitution at time of study design and were therefore adopted in the current trial.
All patients were treated with VMAT. Patients’ preparation and positioning were as per institute protocol.
An example of a treatment plan for PORT and WPRT is presented in Supplemental Figure 1.
Follow-up
Patients were followed weekly during therapy, 1 month after therapy, and 3-months thereafter until 1 year. Then, patients were followed every 6 months till 5 years and yearly thereafter.
A blood analysis to evaluate red blood cell (RBC), white blood cell (WBC), and platelet count was done before start of EBRT (pre-EBRT) and at the end of EBRT (post-EBRT). Additional blood analyses were requested at 3, 12, 24, 36, 48, and 60 months after EBRT.
For WBC and platelet count, hematological toxicity was scored according to CTCAE version 4.03. 16
Statistical Considerations
The study was initially designed to include 330 patients within an 8 years study period comprising a 5-year accrual period and 3-year follow-up period. Due to poor accrual, the study was closed prematurely after including 69 patients from whom dosimetric data and blood analyses were available in 64 patients. 17
The dosimetric parameters that were analyzed for the targets and nodal areas are median dose and dose received by 98% (D98) and 2% (D02) of the volume as a surrogate for minimal dose and maximal dose, respectively. For the rectum and sigmoid colon, we report on D02 and volume of the organ receiving a biological equivalent dose in 2 Gy fractions of 40, 50, 60, and 65 Gy. For small intestine and bladder, we looked at D02. For bone marrow, we evaluated the median dose and volume receiving a dose of 7.5 Gy (V7.5). Statistically significant differences between both groups were calculated using Welch t-tests.
To evaluate differences between PORT (ARM A) and WPRT in the evolution of RBC, WBC, and platelet count over time, three linear mixed models with a random intercept for the patient was fit with model terms randomization group (ARM A or B), study time point (pre, 0, 3, 12, 24, 36, 48, and 60), and the interaction between both categorical predictors. Model assumptions were checked visually. Based on this assessment, a natural log transformation was applied to WBC.
SPSS Statistics version 28 for Windows (IBM Corp., 2017.) was used, as well as R version 4.1.3 (R Core Team, 2022).
A P value < .05 was considered significant for the group differences.
For the mixed models, the Holm procedure was applied through adjustment of the significance level to correct for multiple testing (correcting for 7 comparisons).
Results
Dosimetric data and blood analyses were available for 64 patients. Thirty-three patients were treated in ARM A and 31 patients in ARM B. Most patients received postoperative EBRT (N = 26 in ARM A and N = 25 in ARM B).
Median follow-up for all patients was 36 months (3-60 months), 30 months (3-60 months), and 36 months (3-60 months) for ARM A and ARM B, respectively.
Dosimetric Differences Between PORT and WPRT
The planning objectives and dose constraints for the organs at risk were met in all patients except for a maximum dose of 18.9 Gy to 50% of the volume of bone marrow, which was not reached in 24 patients (all patients were treated in Arm B).
The dose received by the CTV_LNs, PTV_LNs, LN areas separately, and organs at risk are presented in Table 1.
Difference in Dose to the Target and Organs at Risk for Both Groups. Mean Doses are Presented in Gy ± Standard Deviation in Gy.
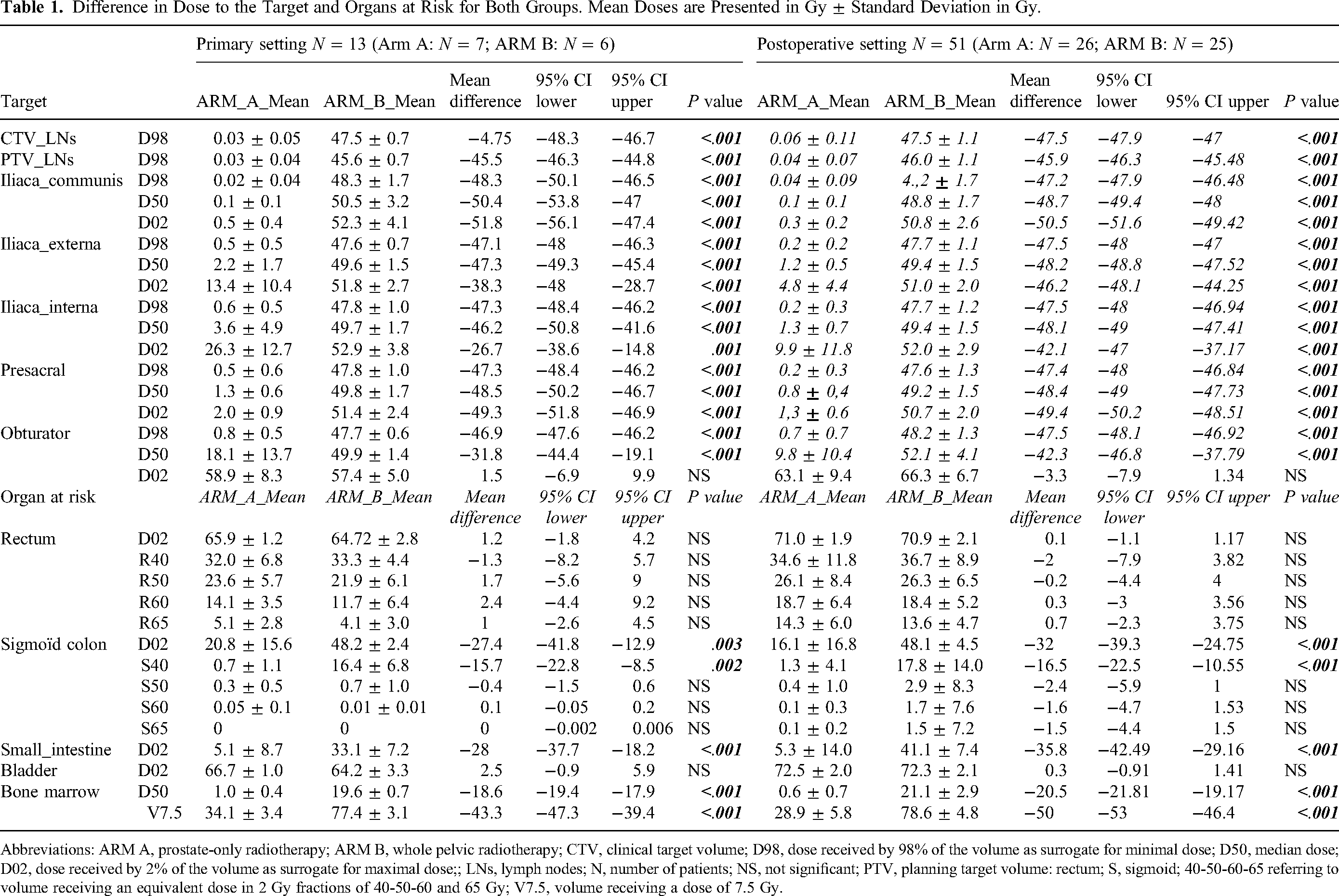
Abbreviations: ARM A, prostate-only radiotherapy; ARM B, whole pelvic radiotherapy; CTV, clinical target volume; D98, dose received by 98% of the volume as surrogate for minimal dose; D50, median dose; D02, dose received by 2% of the volume as surrogate for maximal dose;; LNs, lymph nodes; N, number of patients; NS, not significant; PTV, planning target volume: rectum; S, sigmoid; 40-50-60-65 referring to volume receiving an equivalent dose in 2 Gy fractions of 40-50-60 and 65 Gy; V7.5, volume receiving a dose of 7.5 Gy.
PORT significantly lowered the calculated D98, median, and D02 to the LNs. This was the case for all LN regions separately, except for the obturator region, where there was no significant difference in D02 between PORT and WPRT.
WPRT was associated with a significant increase in dose delivered to the sigmoid colon (D02 and S40), small intestine (D02), and bone marrow (D50 and V7.5).
Similar findings were observed whether patients were treated with primary radiotherapy or postoperative radiotherapy.
Hematological Differences Between PORT and WPRT
Only 1 patient in ARM B developed grade 1 leukopenia (defined as WBC < 3.0 × 10e9/L).
Supplemental Figure 2 gives an overview of the percentages of available blood analyses per time point and per group for RBC, WBC, and platelet count.
RBC
There were no significant differences in RBC count pre-EBRT between both arms. In contrast, the change in RBC count from time point pre-EBRT to 3 months was significantly different with ARM B showing a larger decrease (P = .0034; statistical significant difference after correction for multiple testing) (Figure 1).

Overview of changes over time in red blood cell (RBC) count for both groups separately. RBC is presented in 10^6/μL. A is referring to Arm A = prostate only radiotherapy. B is referring to Arm B = whole pelvic radiotherapy.
WBC
There were no significant differences in WBC count pre-EBRT between both arms. The difference in WBC count between time point pre-EBRT and month 3 (P = .0010) and month 12 (P = .0013) was significantly different, after correction for multiple testing, between both arms with a larger decrease observed again in ARM B (Figure 2).
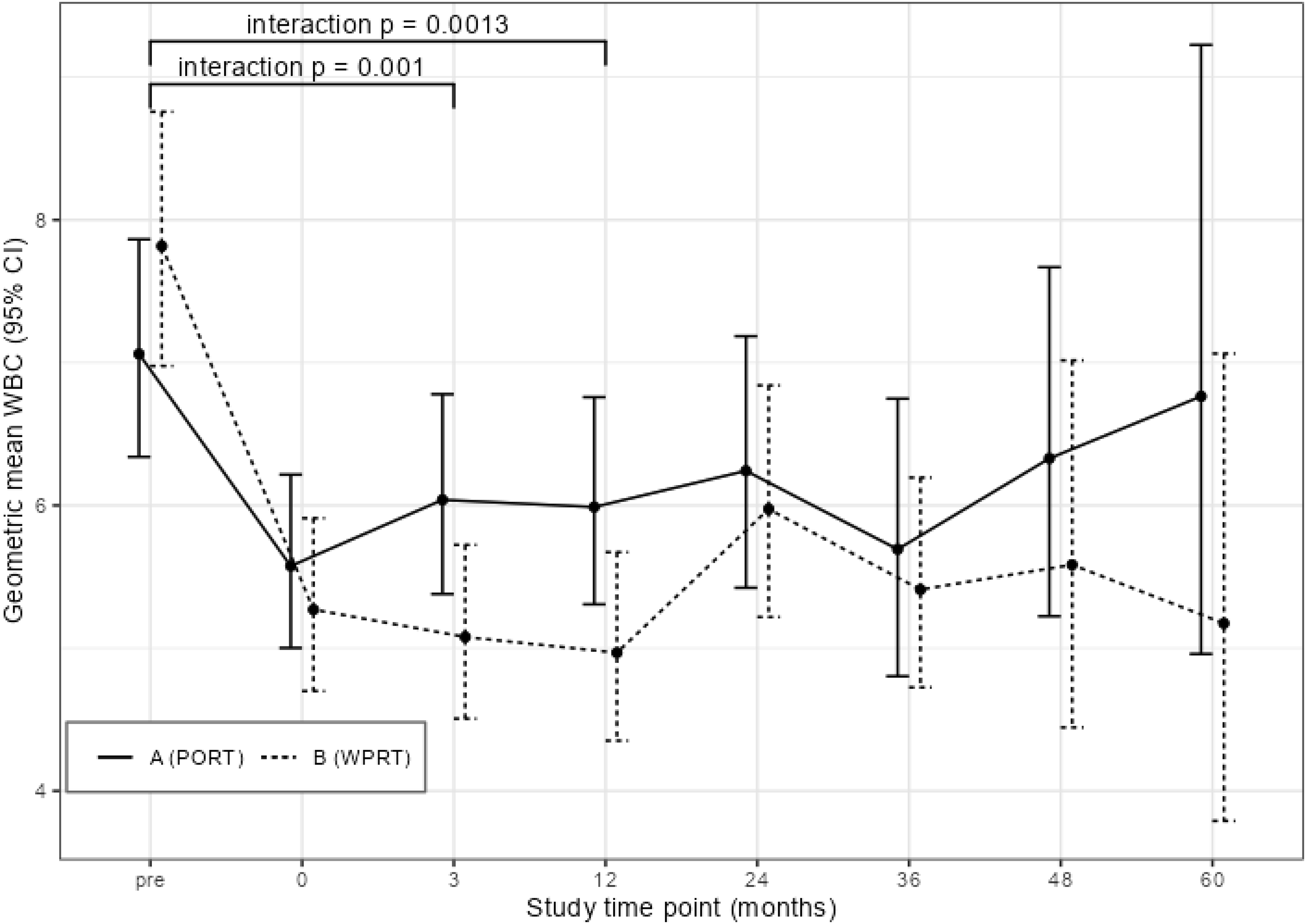
Overview of changes over time in white blood cell (WBC) count for both groups separately. WBC is presented in 10^3/μL. A is referring to Arm A = prostate only radiotherapy. B is referring to Arm B = whole pelvic radiotherapy.
Platelets
No significant differences between both arms were found in platelet count pre-EBRT. Changes in platelet count between time point pre-EBRT and other time points were not found to be significantly different between both arms (Figure 3).

Overview of changes over time in platelet count for both groups separately. Platelets are presented in 10^3/μL. A is referring to Arm A = prostate only radiotherapy. B is referring to Arm B = whole pelvic radiotherapy.
Discussion
As WPRT for PCa is associated with more acute gastrointestinal toxicity and late genitourinary toxicity, the decision to include the whole pelvis must be well-considered and reserved for the patients who truly benefit from it.
In the presented randomized phase 3 study, PORT was compared to WPRT in pN1 PCa patients to define whether including the pelvis is superior to PORT and improves clinical relapse-free survival in this specific population. Preliminary results showed that, after a median follow-up of 3 years, biochemical relapse-free survival was 79% and 92% for patients who received PORT compared to WPRT, and no pelvic recurrences were seen in the WPRT group. 17 The study confirmed that, even when modern techniques are applied, WPRT comes at the cost of more acute GI toxicity. 17
This manuscript focuses on incidental dose to the LN areas when PORT is delivered with modern radiotherapy techniques and looks at the impact on hematological parameters of irradiating a larger proportion of bone marrow in the setting of WPRT with VMAT.
The place of WPRT in high-risk N0 PCa has been a matter of debate for many years as randomized trials, performed with older radiation techniques, failed to show an advantage for WPRT over PORT.5,6 Especially when conformal radiotherapy techniques are used, a nonneglectable dose is administered to the pelvic LN areas in the immediate vicinity of the prostate. This unintended deliverance of dose has been associated with improved outcomes in PCa. Witte et al., for example, found that patients with high- and intermediate risk PCa without failure after 3D CRT to the prostate + /− seminal vesicles received a significantly higher average dose to the obturator regions and presacral regions. 18 Also, for PORT with image-guided intensity-modulated radiotherapy (IMRT), an association between treatment failure and incidental dose outside the prostate has been suggested for high-risk PCa patients. 19
Our study illustrates that the incidental dose to the surrounding LN areas is low when VMAT is used. In ARM A, a median dose of <4 Gy was found in all LN areas except for the obturator region where a median dose of 9 to 18 Gy was delivered. This is in line with the results reported in the study by Murthy et al., who also clearly showed that modern radiotherapy techniques reduce the incidental dose to the pelvic areas in the vicinity of the prostate due to steeper dose gradients. 20 The largest benefit was observed in the obturator area close to the prostate where the mean dose was lowered from 44 Gy to 29 Gy and 22 Gy when switching from 2D EBRT to 3D conformal radiotherapy and IMRT, respectively. 20
Recently, the benefit of applying WPRT in PCa patients with a high risk of pelvic LN involvement, defined as a risk of >20% based on the Roach formula, was shown in a randomized trial. 21 Another recent but retrospective study by Tilki et al., recommends adjuvant WPRT in the majority of pN1 PCa patients. 22 The first results of our prospective randomized trials also suggest an advantage for WPRT for pN1 PCa patients. 17
WPRT increases the volume of bone marrow that is irradiated with low-intermediate doses which can have hematological consequences. A meta-analysis based on 29 studies comparing bone marrow sparing with nonbone marrow-sparing radiotherapy in cervical cancer, supports the use of bone marrow protection therapy as this results in a significant reduction in hematological toxicity without increasing gastrointestinal or genitourinary toxicity. 11 Also, for rectal cancer, a bone marrow-sparing approach appears to be beneficial and results in lower incidence of acute hematological toxicity. 23 In 2016, Cozzarini et al., already reported leukopenia and thrombocytopenia after postprostatectomy WPRT. 24 Recently, available literature on hematological toxicity after prostate radiotherapy was resumed within a systematic review. 25 The authors concluded that the bone marrow should be regarded as an organ at risk and organ-sparing approaches are advised for patients undergoing WPRT. 24 A prospective study performed by Sini et al., found that the dose-volume parameter that was most correlated with the development of acute grade 3 and late grade 2 lymphopenia were medium-high doses of ≥40 Gy to the whole pelvic and iliac bone marrow, respectively. 26
The implementation of modern radiotherapy techniques allows for better bone marrow sparing with a positive effect on decrease in RBC, WBC, and platelets when compared to conformal radiotherapy in WPRT for prostate and bladder cancer. 27
Despite applying planning constraints for bone marrow and use of VMAT, we found a significant difference between both treatment arms in the evolution of RBC and WBC over time, with a greater decrease of both RBC and WBC count in the WPRT group. However, none of the patients developed severe hematological toxicity and only 1 patient developed grade 1 leukopenia.
Whether the observed drop in RBC and WBC count is clinically relevant is unclear although persistent fatigue is described after EBRT. Moreover, all patients in our study received concomitant ADT. ADT commonly leads to mild normochromic and normocytic anemia, merely without a significant clinical impact. However, in a minority of patients, this anemia can be more pronounced. For these patients, an additional decrease in RBC due to WPRT might amplify symptoms related to anemia.
The impact of radiation on the immune system is also of particular interest in the search for new biomarkers of response to radiation. Radiation may unintentionally cause lymphopenia which is associated with poor outcome across different tumor types. 28 Danckaert et al., looked at the impact of radiotherapy parameters on lymphopenia in urological tumors. 29 Since the bone marrow is the primary site of hematopoiesis and lymphocytes are highly radiosensitive, they concluded that minimizing radiation volumes and unintentional radiation of pelvic bone marrow might reduce lymphopenia and improve clinical outcome. Increased efforts to spare the bone marrow by implementing more stringent dose-volume parameters, including the V40, might thus reduce the hematological impact of WPRT.
Along with increased toxicity, comes a fear of inducing more secondary malignancies when applying WPRT. A retrospective analysis showed that the incidence of secondary malignancies after WPRT is 9.16% and comparable to the rate of secondary malignancy observed after PORT (8.88%) resulting in the adjusted odds ratio of 1.046 (95% CI: 0.968–1.130). 30
Our trial is not without shortcomings. First, we acknowledge the small sample size and short follow-up, with more missing blood samples for the later time points.
Second, within the trial, a hematological evaluation including RBC, WBC, and platelet count was requested but we did not ask for further WBC differentiation. Therefore, specific information on lymphocyte count lacks, which is the most important parameter so far to evaluate radiation effect. Since a decline in WBCs was observed and lymphocytes are the most sensitive type of WBCs, one can presume that the lymphocyte count was affected as well. However, whether this resulted in severe lymphopenia (< 500 cells/μL) was not evaluated in this trial.
Third, the delineation protocol of the LN areas is based on the RTOG consensus guidelines. At the time of designing the trial, the recent guidelines by Hall et al., which are more robust, were not published yet. 31 This will however not alter the results of this trial but makes dosimetric comparisons with other papers more difficult.
Also, evaluating more dose parameters for the different organs at risk could be of value but for clarity of the paper it was decided to limit the dose parameters to the ones that were implemented in our planning objectives. Current insights suggest that evaluation of the volume of bone marrow receiving >40 Gy is probably more relevant, although external validation is still lacking.
Conclusion
This study emphasizes the importance of selecting the PCa patients that truly benefit from WPRT since the incidental dose to the lymphatic areas becomes neglectable when PORT is delivered with modern radiotherapy techniques.
Hematological toxicity is very low when WPRT is delivered with VMAT and when bone marrow is considered an organ at risk in the treatment planning. On the other hand, even with VMAT, a large proportion of the bone marrow is irradiated with low to intermediate doses in WPRT, resulting in a decrease of RBCs and WBCs over time.
Supplemental Material
sj-xlsx-1-tct-10.1177_15330338231154088 - Supplemental material for Dosimetric and Hematologic Implications of Prostate-Only Versus Whole Pelvic Radiotherapy: Results of the Multicentric Phase 3 PROPER Study
Supplemental material, sj-xlsx-1-tct-10.1177_15330338231154088 for Dosimetric and Hematologic Implications of Prostate-Only Versus Whole Pelvic Radiotherapy: Results of the Multicentric Phase 3 PROPER Study by Valérie Fonteyne, Willeke Danckaert, Piet Ost, Charlien Berghen, Katrien Vandecasteele, Ben Vanneste, Kato Rans, Nick Liefhooghe, Steven Wallaert and Leen Paelinck in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338231154088 - Supplemental material for Dosimetric and Hematologic Implications of Prostate-Only Versus Whole Pelvic Radiotherapy: Results of the Multicentric Phase 3 PROPER Study
Supplemental material, sj-docx-2-tct-10.1177_15330338231154088 for Dosimetric and Hematologic Implications of Prostate-Only Versus Whole Pelvic Radiotherapy: Results of the Multicentric Phase 3 PROPER Study by Valérie Fonteyne, Willeke Danckaert, Piet Ost, Charlien Berghen, Katrien Vandecasteele, Ben Vanneste, Kato Rans, Nick Liefhooghe, Steven Wallaert and Leen Paelinck in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by an unrestricted research grant from Ipsen. Ipsen had no input into the study design, conduct or interpretation of results, decision to publish, or manuscript preparation. Ipsen conducted a courtesy review prior to submission but had no input into the content. Valérie Fonteyne is a postdoctorate clinical specialist funded by the Belgian Foundation against Cancer.
Ethical Approval
The study was approved by the ethics committee of Ghent University Hospital (EC/2016/0246) on March 24, 2016.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
