Abstract
Introduction
Pancreatic cancer is one of the most lethal solid malignancies, with an overall 5-year survival rate of less than 10%. 1 It is considered as the second leading cause of cancer-related death in developed countries. 2 The incidence and mortality rates of pancreatic cancer nearly mirror each other. The global age-standardized incidence of pancreatic cancer is 4.8 per 100,000 persons, and the mortality rate is 4.4. 3 Alarmingly, the median time from diagnosis to death from untreated advanced pancreatic cancer is about 3.5 months. 4 More effective prevention and treatment strategies are urgently needed.
As known, diabetes is a risk factor for pancreatic cancer. The incidence of pancreatic cancer is higher in patients with type 2 diabetes mellitus (T2DM), 5 as a result of high level of insulin and insulin-like growth factor (IGF-1), which activates PI3K/Akt/mTOR and AMP-activated protein kinase (AMPK) signaling pathways, enhances growth and metabolism of cancer cells. 6 In addition, pancreatic cancer–related death in T2DM patients goes up steeply to 30% to 40% compared with those without diabetes. 7
Interestingly, several studies reported that metformin exerted antitumorigenic effects via inhibiting the LKB1-AMPK-mTOR signaling pathway, suppressing the JNK pathway, and lowering insulin and IGF-1 levels.8–10 Metformin, a relatively cheap, safe, and well-tolerated medication in the biguanide class, is used as an oral antidiabetic drug for the treatment of T2DM. In the past decade, many epidemiologic studies reported that metformin rendered anticancer effects in several types of cancers, such as breast cancer and colorectal cancer.11–13
Nonetheless, its benefit on pancreatic cancer was controversial.14–17 Two prospective cohort studies supported that metformin users had a relatively lower risk for pancreatic cancer compared to metformin nonusers.15,16 Conversely, a large cohort study from the Netherlands did not detect a reduced risk of gastrointestinal cancers in current metformin users compared with those users of other nonsteroidal anti-inflammatory drugs, including pancreatic cancer. 14 Toriola et al also reported that there were no associations between metformin use and survival in patients with pancreatic ductal adenocarcinoma (PDAC) among African American patients. 17
Herein, we retrospectively comprised a cohort of pancreatic cancer patients who were exposed to metformin before cancer diagnosis to evaluate the effect of metformin use on the prognosis of pancreatic cancer. In this study, we investigated the overall survival (OS), progression-free survival (PFS), and disease-free survival (DFS) outcomes in diabetic patients with and without metformin treatment.
Materials and Methods
Participants
A retrospective cohort study was conducted using The Chinese Medicine Information Retrieval System (https://dc.wzhospital.cn/vpn/index.html) of The First Affiliated Hospital of Wenzhou Medical University. The analysis included patients diagnosed with pancreatic cancer from January 1, 2012, to December 31, 2020. All pancreatic cancer patients were diagnosed by pathological biopsy. The vital status of the participants was determined directly by their own medical records.
From the electronic medical record management system, we could get relevant information such as gender, age at diagnosis, date of diagnosis, tumor TNM classification, the primary tumor site (head, body, or tail of pancreas), the treatment modality (surgery, chemotherapy, targeted therapy, immunotherapy, or radiotherapy), and the history of drinking and smoking.
Metformin users were defined as participants who had been prescribed continuous oral metformin before pancreatic cancer diagnosis. Participants who were never prescribed metformin were defined as nonusers. The date of prescription was compared with the date of diagnosis to assess whether metformin was administered before or after diagnosis.
Patients excluded were as follows:
Follow-up less than 1 month after diagnosis. Incomplete medical records. Diagnosis as metastatic lesion from another primary cancer. Suffered from multiple primary cancers. Post-diagnostic metformin use. Died from other specific causes (not from pancreatic cancer).
The participants were selected randomly according to the following procedure (Figure 1). A total of 892 patients with pancreatic cancer were identified from The Chinese Medicine Information Retrieval System of The First Affiliated Hospital of Wenzhou Medical University. After application of the inclusion and exclusion criteria, the participants, in the final analysis, included 76 metformin users and 92 metformin nonusers.
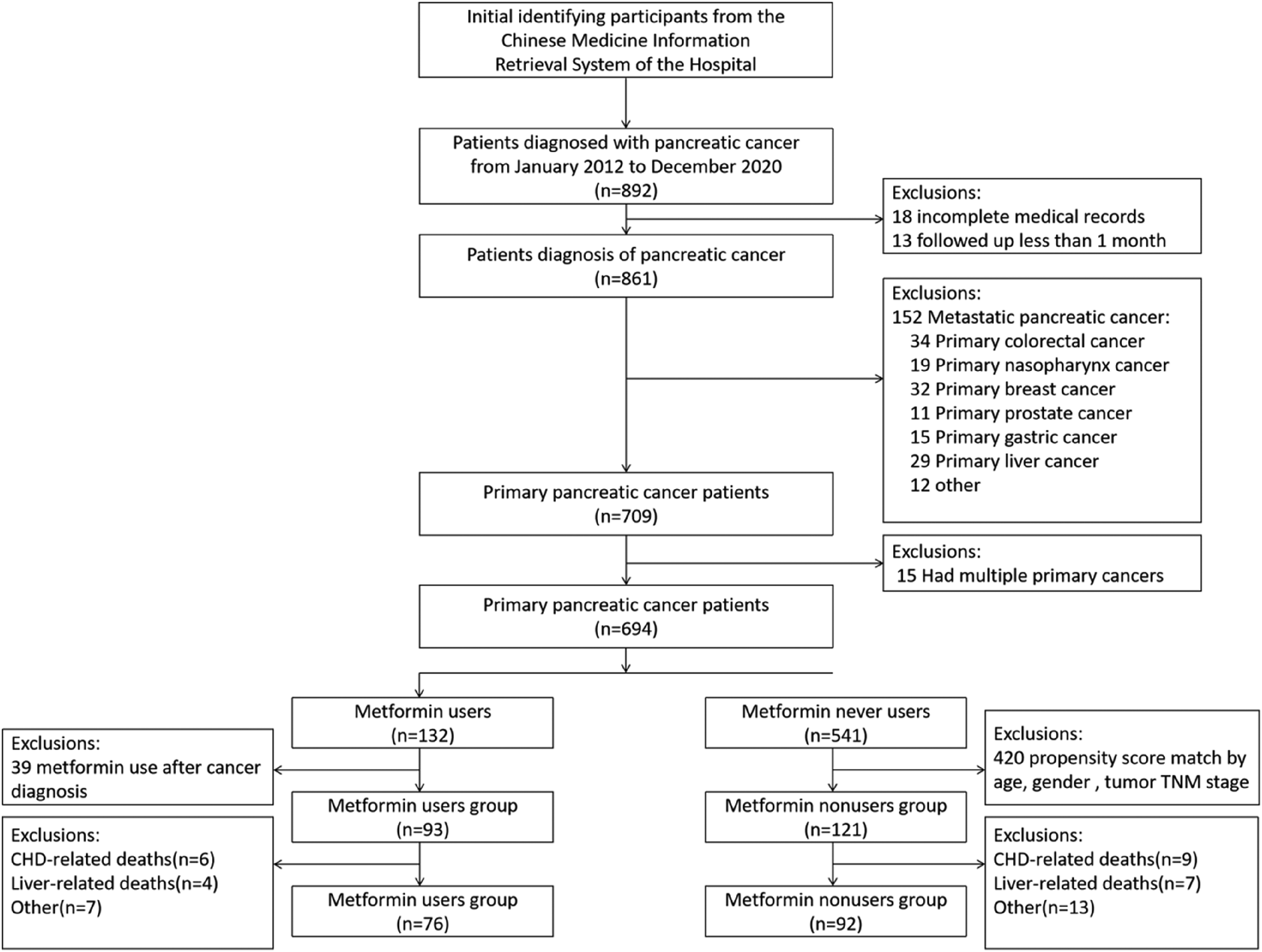
The selection process of the study population.
Exposure
To further analyze the relationship between metformin use and pancreatic cancer survival, and to reduce or avoid the confounding effects of other anti-hyperglycemic drugs, in this retrospective cohort study, patients who had been prescribed at least one dispensing record of oral metformin before pancreatic cancer diagnosis were defined as metformin users, while concurrent or previous use of other anti-hyperglycemic agents, including sulfonylureas, thiazolidinediones, alpha-glucosidase inhibitors, D-phenylalanine derivatives, dipeptidyl peptidase 4 inhibitors, incretin mimetic agents and insulins were excluded. According to the dispensing record, the metformin dosage form was extended-release tablets, with the dose ranging from 250 to 1000 mg orally once a day. Patients who never prescribed metformin before pancreatic cancer diagnosis were defined as nonusers.
Outcome Measures
Follow-up of the cohort started on the date of diagnosis of pancreatic cancer, and ended at the time of death, or the deadline of follow-up (October 22, 2021). The survival outcomes in pancreatic cancer patients were analyzed based on OS as the primary outcome, which measured from the date of diagnosis of pancreatic cancer to the date of death, or the last contact date (for the 18 patients who were lost to follow-up). Patients who did not die were censored at the close date of follow-up (October 22, 2021). Progression-free survival was analyzed as a secondary outcome and defined as the time from the first chemotherapy to disease progression or death from any cause. The last outcome was DFS, which was defined as the interval between the date of surgery and the date of the last follow-up or recurrence/progression. Progression or recurrence was assessed with radiologic images.
Statistical Analysis
PASS software (PASS 15. NCSS, LLC.) was used to calculate the minimum required sample size. Categorical variables, chiefly baseline characteristics, were analyzed and compared by Chi-square test, including Pearson Chi-square test, continuity correction Chi-square test, and Fisher exact test. Rank-sum test was applied to examine the differences in grade variables. Continuous variables were analyzed and compared by independent sample t test. We used Kaplan-Meier plot to do univariate survival analyses, with comparison by log-rank test. Median OS, DFS, and PFS were estimated at the same time. Cox proportional hazards regression model for univariate and multivariate analyses was used to measure the association between metformin use and tumor survival after adjustment, and the effect of metformin on tumor survival was valued by hazard ratio (HR) and 95% confidence interval (CI). Subgroup analyses were also carried out with this model. A 2-sided P value <.05 was defined as statistically significant. Statistical analyses were done by SPSS® version 25 (IBM) and R version 4.1.0 (R Foundation for Statistical Computing).
Ethical Statement
The reporting of this study conformed to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 18 We obtained the patients' verbal consent to use their data for this cohort study, and all patients were anonymous and arranged with ID for the purpose of protecting their privacy.
Results
Baseline Characteristics of the Cohort Study
One hundred sixty-eight patients with pancreatic cancer (median age = 63.3 ± 10.7) were recruited in total, consisting of 76 cases in the group of metformin use (45.2%) and 92 cases of metformin nonuse (54.8%). They were 57 female patients (33.9%) and 111 male ones (66.1%). The baseline characteristics of participants with pancreatic cancer who used or did not use metformin are presented in Table 1. Between the two groups, there were no significant differences in baseline age, gender, tumor stage, primary tumor site, treatment modality, comorbid disease, smoking, and drinking status. Seventy-five cases (44.7%) were diagnosed as stage I-II pancreatic cancer, 31 cases (18.5%) as stage III pancreatic cancer, and 56 cases (33.3%) as stage IV pancreatic cancer. In this cohort, metformin users were significantly more likely to be diagnosed with advanced pancreatic cancer than nonmetformin users. In addition, in both groups, only a few participants received radiotherapy (6.0%), immunotherapy (1.8%), and/or targeted therapy (3.0%).
Baseline Characteristics of the Study Population.a

Abbreviation: SD, standard deviation.
Data are number of participants enrolled/number eligible (%).
P value indicated the difference between baseline data of the two groups.
cNumber of “Unknown”: the number of cases whose stage or tumor location could not be determined.
Sensitivity Analysis
In univariate analysis, age, tumor stage, and radical surgery were more likely to be associated with survival outcomes in patients with pancreatic cancer (Table 2). Of note, metformin exposure was associated with a trend toward a lower risk of death (HR = 0.71, 95% CI = 0.48-1.04, P = .081). In the Cox proportional hazards model, 113 deaths (67.3%) were observed at the end of follow-up, while 37 cases (22.0%) were still alive. Metformin use was significantly associated with lower overall mortality when adjusted for tumor stage, age, and radical surgery (adjusted HR = 0.50, 95% CI = 0.33-0.76, P = .001).
The Factors Associated With OS.a

Abbreviations: CI, confidence interval; HRs, hazard ratios.
Values of HR and P values were obtained by univariate or multivariate Cox proportional hazards regression. If a variable had a relatively great significance in univariate analysis, or might be related to prognosis medically, it would be included in multivariate analysis. HR equaled to 1 indicated that this group was set as the reference. More specifically, for reference groups of these variables in the table, no metformin use, female, stage I, head and neck, no corresponding treatments, no corresponding comorbid diseases, and no smoking or drinking were set as the reference groups.
Survival Outcomes of the Cohort Study
A Kaplan-Meier curve can be seen in Figure 2. The earliest time of death was day 64, and the last observed time of failure was day 1941. The median OS in the group of metformin use and metformin nonuse was 481 days (95% CI = 295-667) and 345 days (95% CI = 279-410), respectively. As to the median DFS, it was prolonged by 167 days in the group of metformin use, compared with that of metformin nonuse (Figure 3). Moreover, the median PFS was significantly longer in patients who received metformin than those did not, that was 219 days and 136 days, respectively (Figure 4). In conclusion, our findings suggested that metformin use was associated with improved survival outcomes in patients with pancreatic cancer.

Kaplan-Meier curves for overall survival (OS) in the metformin-treated analysis. Log-rank test was carried out to estimate overall survival in Kaplan-Meier curves.
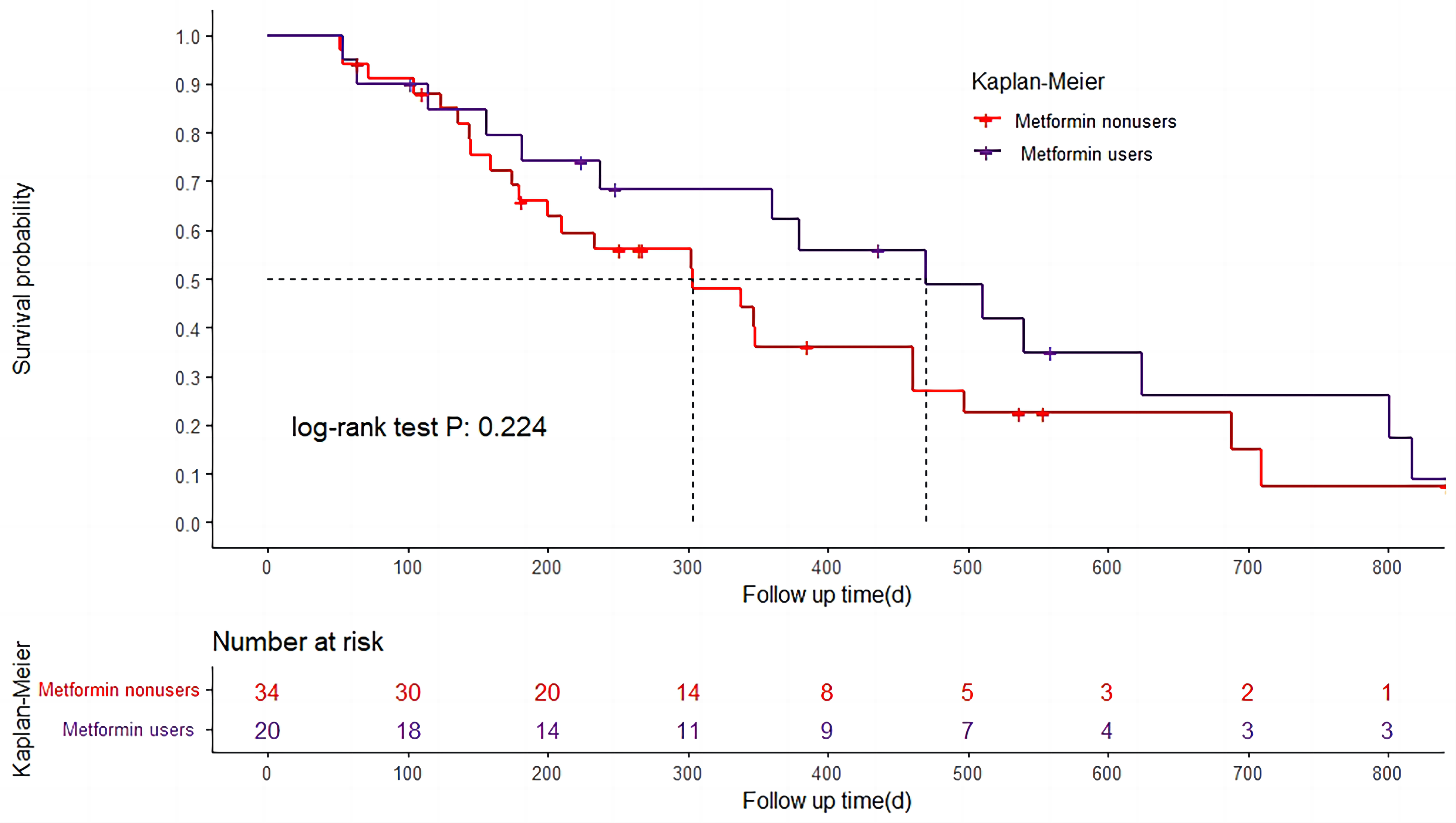
Kaplan-Meier curves for disease-free survival (DFS) in the metformin-treated analysis. Log-rank test was carried out to estimate disease-free survival in Kaplan-Meier curves.

Kaplan-Meier curves for progression-free survival (PFS) in the metformin-treated analysis. Log-rank test was carried out to estimate progression-free survival in Kaplan-Meier curves.
Subgroup Analysis
Furthermore, after stratification for various factors, it was found that metformin users still held a better OS (Table 3). There was a strong association between metformin exposure and survival among participants with lower disease stage, male and the elders. In subgroup analyses according to the clinical cancer stage, metformin exposure in stage I-II pancreatic cancer resulted in favorable survival outcomes (adjusted HR 0.47, 95% CI = 0.25-0.91) compared with those with advanced stage (adjusted HR for Stage IV = 0.62, 95% CI = 0.33-1.19). The efficacy of metformin was found to be more pronounced in patients over 60 years of age (HR = 0.37, 95% CI = 0.22-0.60). The association between metformin use and tumor survival persisted in individuals undergoing radical surgery (adjusted HR = 0.40, 95% CI = 0.20-0.79).
Subgroup Analysis of Overall Survival.a

Effect of patient subgroup on OS. Adjusted for age, clinical stage, and radical surgery.
Unknown cases of stage were excluded in the analysis of stage subgroup.
Discussion
Metformin, a recognized prescription drug for the prevention and treatment of T2DM, has attracted worldwide attention for its anticancer activity. Although some epidemiologic researches have reported a protective effect of metformin on pancreatic cancer risk,19,20 the relationship between metformin use and the survival of pancreatic cancer is still controversial. Some studies revealed positive effects,15,16,21 whereas others demonstrated different outcomes.17,22,23 Of note, a nationwide cohort study from New Zealand showed insulin use was associated with lower mortality risk in individuals with pancreatic cancer–related diabetes. 24 Lee et al also found that metformin, thiazolidinedione, and dipeptidyl peptidase-4 inhibitor exposure were associated with decreased risk for future pancreatic cancer. 25 So, in order to eliminate the confounding factors of other anti-diabetic drugs, we used data on dispensed drugs to determine concomitant use. In our study, metformin users were defined as participants who had been prescribed continuous oral metformin before pancreatic cancer diagnosis (those who concurrently or previously used other antihyperglycemic drugs were excluded), which would guarantee the exclusive use of metformin during the entire course of cancer treatment. Notably, we found that pancreatic cancer patients who received metformin had improved survival outcomes compared with patients who did not.
In subgroup analysis based on the clinical stage of pancreatic cancer, the survival benefits of metformin use were statistically significant in the I-II Stage subgroup (adjusted HR for I-II stage = 0.47, 95% CI = 0.25-0.91). These findings were in concordance with the study conducted by Shi et al 26 and Sadeghi et al. 16 In contrast to our study, a retrospective cohort study in South Korea suggested that metformin exposure was associated with survival benefits in patients with pancreatic cancer and preexisting T2DM, especially among those with an advanced cancer stage. 27 In the subgroup analysis of age, we found that the relative benefit of metformin may be higher in patients older than 60 years of age (adjusted HR 0.37, 95% CI = = 0.22-0.60). We hypothesized that the treatment modality and age could affect the antitumor effect of metformin. Patients at early tumor stage had more opportunity for radical surgery, so they usually survived and lived longer, and the cumulative effect of metformin was stronger than in patients with advanced-stage cancer. In principle, metformin may help to alter the tumor immune microenvironment and improve the efficiency of immunotherapy. 28 Of note, metformin may mitigate the effects of chemotherapy by decreasing reactive oxygen species, 29 which may relatively weaken its anti-cancer role in advanced cancer patients. Additionally, the cancer of advanced stage is relatively denser than that of the early stage in biology, which makes the entry or accumulation of metformin in cancer cells became more difficult, thereby affects its anticancer action.29,30 Moreover, we found that metformin seemed to have a better antipancreatic cancer effect in the aging male. Due that our current study was only observational, it was difficult to make an expanded explanation of why there was an association, and more prospective clinical trials would be needed to definitively address this issue.
Needless to say, there are some limitations to this study. First, given the small sample size in the subgroup analysis, the results had to be interpreted with caution, especially for stage III pancreatic cancer. Second, although PDAC accounts for more than 90% of pancreatic cancer cases in this cohort, some other histological types were also included. At last, data were limited to prescribed drugs, it was not possible to obtain information regarding metformin use at home.
In conclusion, our retrospective study of patients with pancreatic cancer elucidated that there was a strong association between longer survival and metformin exposure, especially for patients with early tumor stage. These suggested metformin use could be an adjuvant for pancreatic cancer treatment. However, more high-quality randomized controlled trials and prospective cohort studies are needed to confirm the conclusion.
Footnotes
Abbreviations
Authors’ Note
Ethical approval for this study was granted by the Ethics Committee in Clinical Research (ECCR) of the First Affiliated Hospital of Wenzhou Medical University (No. KY2022-R131).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the funding of the National Natural Science Foundation of China (grant number 81802328 to Gu DN & No. 82073203 to Tian L) and the Young Talents Program of the First Affiliated Hospital of Wenzhou Medical University (grant number qnyc094).
