Abstract
Objective
To develop and validate a CT-based radiomics model for predicting sequential immunotherapy response after concurrent radiochemotherapy (CCRT) in patients with unresectable stage III non-small cell lung cancer (NSCLC).
Methods
The study retrospectively included 71 patients who received sequential immunotherapy after concurrent chemoradiotherapy (CCRT) between January 2019 and December 2022, while prospectively including 17 additional patients between January 2023 and July 2023. The study documented each patient's tumor response and prognosis within two months of completing treatment. Patients were then categorized based on their treatment response, resulting in the identification of two distinct groups: treatment-responsive group and treatment-insensitive group. First, ITK-SNAP software was used to delineate the primary tumor lesions in the lung window and define a region of interest (ROI). Second, features were extracted using Python (version 3.6) and filtered using Least absolute shrinkage and selection operator regression. Third, radiological models were built using six machine learning algorithms: logistic regression (LR), discriminant analysis (DA), neural network (NN), random forest (RF), support vector machine (SVM) and K-Nearest Neighbour (KNN). Finally, Kaplan-Meier survival analysis was performed for high- and low-risk patients predicted by radiomic modeling.
Results
Based on the performance of radiomics models constructed by various machine learning algorithms in the prospective validation set, the LR with the highest AUC value (AUC: 90.00%) was finally selected, which also performed well in the independent test set (AUC: 84.96%). Risk stratification of patients based on the radiomic model constructed by LR was excellent for PFS (P = 0.001) and OS (P = 0.019) in the training set, PFS (P = 0.010) and OS (P = 0.028) in the prospective validation set, and PFS (P = 0.014) and OS (P = 0.041) in the test set.
Conclusion
Pretreatment CT-based radiomics model accurately and efficiently predicts treatment response and risk stratification in patients with unresectable stage III NSCLC treated with concurrent chemoradiotherapy and sequential programmed death-1 inhibitor therapy. Prior to prospective data collection, the study was registered with the China Clinical Trial Registry under the trial registration name: Prediction of concurrent chemoradiotherapy efficacy and its related molecular signaling pathway by medical artificial intelligence model based on CT of lung cancer, with the registration number: ChiCTR2100053175 (https://www.chictr.org.cn/showproj.html?proj = 136872).
Keywords
Introduction
The incidence and mortality of lung cancer remain high. 1 From a pathological perspective, lung cancer is categorized into two main subtypes: non-small cell lung cancer (NSCLC) and small cell lung cancer. NSCLC constitutes approximately 85% of all cases. About 30% of patients with NSCLC present with stage III disease at initial diagnosis, 2 with most ineligible for surgery. Unlike stage IV NSCLC, stage III NSCLC represents a highly heterogeneous group of patients with wide variations in tumor size and lymph node involvement, leading to significant differences in treatment modalities and prognosis. 3 However, there is hope for a cure in this group of patients without distant metastases, and selecting an effective treatment option is a significant challenge for all oncologists. For patients with inoperable stage III NSCLC, the traditional standard of care involves curative radiotherapy and chemotherapy. 4 Several studies have demonstrated that concurrent radiotherapy (CCRT) is superior to sequential radiotherapy in such patients,5,6 with a 5-year progression-free survival (PFS) rate of only 18.3% and a 5-year overall survival (OS) rate of 32.1%. 7 This indicates that CCRT alone is insufficient to address the treatment requirements of patients with inoperable stage III NSCLC. The advent of immunotherapy has led to large phase III trials, such as PACIFIC and GEMSTONE-301, which successfully established the standard of care for immune-consolidating therapy following radical radiotherapy and chemotherapy in inoperable stage III lung cancer.8–12 However, all patients are unsuitable for this treatment regimen, and in clinical practice, some patients may not respond to sequential programmed death-1 (PD-1) inhibitor therapy following CCRT. Besides, the sensitivity of patients to the treatment can significantly influence their prognosis. Previous research has shown that patients’ response to therapy is critical to their PFS and OS outcomes.13,14 Therefore, new biomarkers are urgently needed to predict the effectiveness of sequential PD-1 inhibitor therapy after CCRT and to identify suitable candidates.
In conventional medicine, imaging is an important part of the monitoring of the efficacy of treatments in cancer patients. As technology advances, artificial intelligence is increasingly applied to cancer medical imaging.15,16 However, much of the current research focuses on the therapeutic sensitivity of single radiotherapy, chemotherapy, or immunotherapy.17–22 Currently, no model exists to predict the treatment response in unresectable stage III NSCLC patients who receive CCRT followed by sequential PD-1 inhibitor therapy. In model selection for small-to-medium sample sizes, traditional machine learning algorithms (eg, logistic regression) can effectively prevent overfitting through regularization. In contrast, deep learning algorithms are prone to overfitting due to their high parameter complexity. Furthermore, deep learning has inherent black-box limitations: implicit layer features lack biologically interpretable mappings, and training requires Graphics Processing Unit clusters, which is computationally intensive. Conversely, traditional machine learning algorithms provide explicit feature weights to support clinical decisions while maintaining computational efficiency.
The aim of this study is to develop and validate radiomics-based machine learning models that predict the response of inoperable stage III NSCLC patients to sequential PD-1 inhibitors following CCRT, using preprocessed CT scans. These models aim to assist clinicians in identifying suitable candidates for this treatment strategy.
Materials and Methods
Patients
The training set consisted of 38 cases collected retrospectively from Hospital A (January 2019 to December 2022); the prospective validation set included 17 cases collected prospectively from Hospitals A, B, C, and D (January to July 2023); and the test set comprised 33 cases retrospectively collected from Hospitals B, C, and D (January 2019 to December 2022). The primary endpoint of this study was the tumor progression within two months after completing CCRT, followed by sequential PD-1 inhibitor therapy, while the secondary endpoints were the PFS and OS in patients. Due to the retrospective nature of data collection and the anonymization protocol approved by the ethics committee, verbal consent was deemed appropriate. Verbal consent was formally documented by research staff through audio recordings and written notes, confirming participants’ agreement to: use of their de-identified medical data for this study and future research; analysis of treatment outcomes without personal identifiers. For prospective data, written consent was obtained, specifying identical terms.
Study Design and Treatment
Figure 1 presents the experimental flow chart. Inclusion criteria: (1) NSCLC confirmed by pathological diagnosis; (2) no indication for surgery; (3) stage III NSCLC according to the eighth edition of the Tumor-Node-Metastasis classification 23 ; (4) no previous specialized cancer treatments (such as neoadjuvant chemotherapy, radiotherapy, targeted therapy, interventional therapy, or immunotherapy) before this study; (5) patients who received standard chest intensity-modulated radiotherapy (radiotherapy dose: 60-66 Gy/ 30-33 fractions) combined with a synchronous chemotherapy regimen (for squamous cell carcinoma, the chemotherapy regimen consisted of weekly paclitaxel combined with cisplatin; chemotherapy regimen for non-squamous cell carcinoma involved pemetrexed combined with cisplatin administered once every three weeks)24,25; (6) after completing CCRT, patients received at least 2 cycles of a PD-1 inhibitor (Tislelizumab or Sintilimab 200 mg/3 weeks)26,27; (7) patients underwent CT-enhanced imaging of the chest and imaging histology of other sites obtained one month before CCRT treatment and two months after immunotherapy. The exclusion criteria included the presence of other malignancies, incomplete or poor-quality CT images, and inadequate follow-up data. In this study, PFS was measured from the commencement of CCRT to either the first recurrence/ metastasis of lung cancer or the last follow-up on December 31, 2023. OS was characterized as the interval from the commencement of CCRT to death or the final follow-up date (December 31, 2023).

Experimental Flowchart.
Patient efficacy was assessed blindly within two months after treatment completion, according to Immune-related Response Criteria based solely on CT images and follow-up records, excluding model-predicted outcomes. 28 The treatment results were divided into a treatment-sensitive group (immune complete response/ immune partial response) and a treatment-resistant group (immune stable disease/ immune unconfirmed progressive disease/ immune confirmed progressive disease). To standardize the collected contrast-enhanced chest CT images, three-dimensional reconstruction was performed using MATLAB 2014b (https://ww2.mathworks.cn/), with the slice thickness adjusted to 1 mm. A radiotherapist with ten years of experience in cancer treatment delineated the primary tumor lesions on the CT images layer by layer (Window width −1600 Hounsfield units [HU] and window height −600 HU) using ITK-SNAP software (version 3.8.0; http://www.itksnap.org) following the guidelines of the International Commission on Radiation Units 83 report. 29 After completing the annotation for each layer, the corresponding region of interest (ROI) was designated.
Statistical Analysis
The radiomics features were extracted from the ROI of patients. Initially, the data were standardized for radiomics features using the Z-score method. Thereafter, Lasso regression with 5-fold cross-validation folds was used to select relevant features. Logistic regression (LR), Discriminant Analysis (DA), Neural Network (NN), Random Forest (RF), Support Vector Machine (SVM), and K-Nearest Neighbor (KNN) were applied to construct radiomics models using the selected features. The selection of tools for the construction of radiomics models was based on their performance in terms of area under the curve (AUC) values in the training set. Accuracy, precision, recall, specificity, F1-score, receiver operating characteristic (ROC), AUC, Clinical Decision Curve Analysis (DCA), and Calibration curve were used to evaluate the model's predictive performance. Patients were categorized into high-risk and low-risk groups using predicted probabilities and cutoff values from the radiomics model, and their PFS and OS were analyzed using the Kaplan-Meier method. The predictive model was developed using Python software (version 3.6; https://github.com/radiomics/pyradiomics). R software (version 4.3.0; http://www.R-project.org) and SPSS software (version 26.0; IBM Corporation, Armonk, New York, USA) were used for all statistical analyses. Additionally, PyRadiomics 3.0 was employed as the feature extraction framework, strictly adhering to its default parameters. The reporting of this study adheres to the Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD) guidelines. 30
Results
88 patients were eligible for inclusion in this study. The training set retrospectively collected data containing 38 Hospital A patients treated between January 2019 and December 2022. The prospective validation set collected 4 Hospital B patients, 4 Hospital C patients, 1 Hospital D patient, and 8 Hospital A patients treated between January and July 2023. The test set collected 15 Hospital B patients, 11 Hospital C patients, 7 Hospital D patients treated between January 2019 and December 2022. The results presented in Table 1 indicate that no statistically significant differences were observed between the two patient groups regarding treatment response, immunotherapy drugs, gender, ethnicity, age, smoking history, pathological type, T stage and N stage (P > 0.05). From each patient's ROI, 1834 radiomic features were extracted, encompassing shape, texture, first-order, and other radiomic features. Following Lasso regression screening, six radiomic features were identified as associated with treatment efficacy (Table 2 and Figure 2). Supplementary Table 1 presents the weights and clinical significance of the six screened radiomics features.

Lasso Regression Screening for Radiomics Features. (a) Lasso Coefficient Profile of Radiomics Features. (b) Cross Curve Validation.
Subgroup Difference Table.
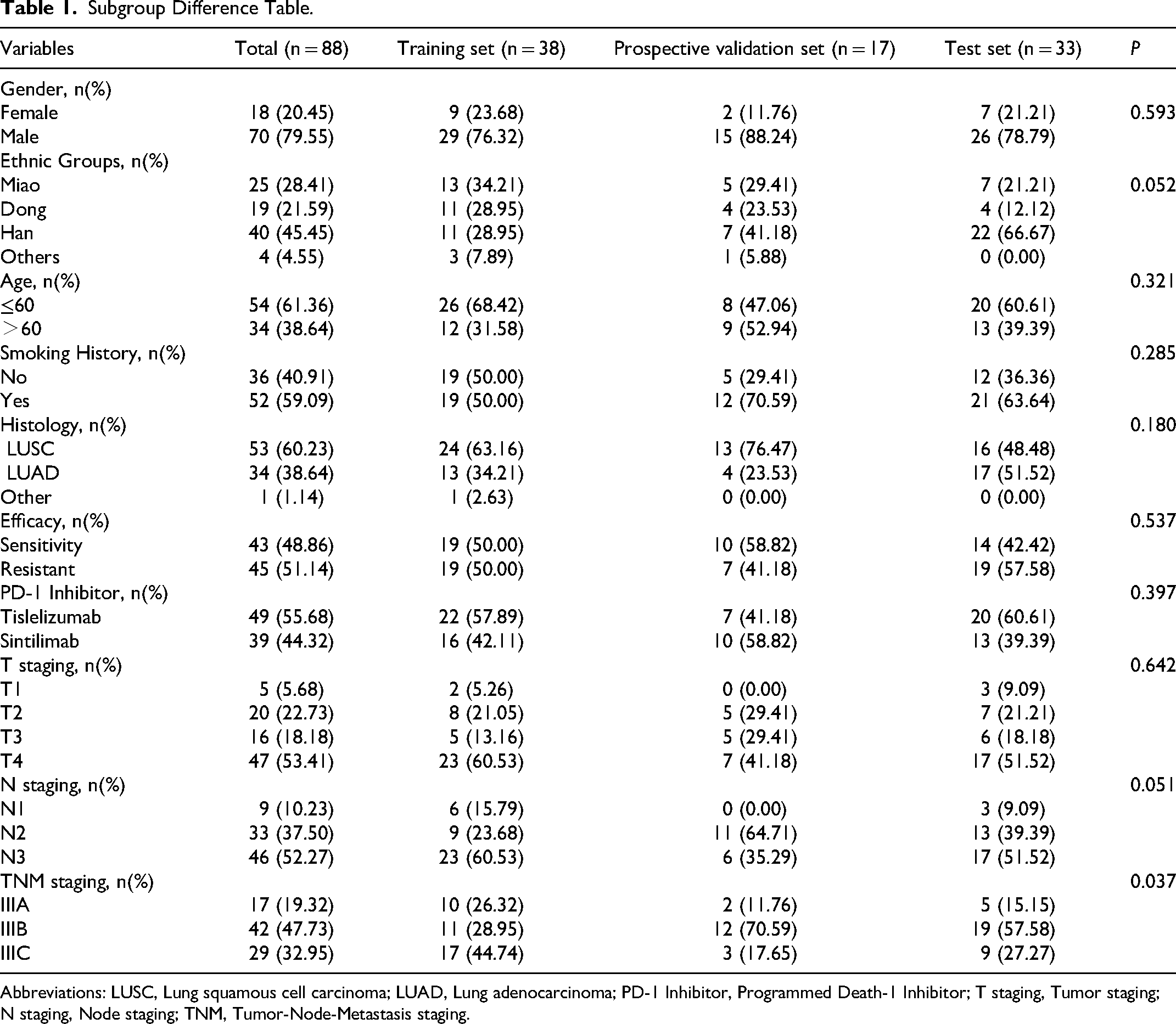
Abbreviations: LUSC, Lung squamous cell carcinoma; LUAD, Lung adenocarcinoma; PD-1 Inhibitor, Programmed Death-1 Inhibitor; T staging, Tumor staging; N staging, Node staging; TNM, Tumor-Node-Metastasis staging.
Statistical Analysis of Selected Radiomics Features.

Abbreviations: SD, standard deviation; Min, minimum; Max, maximum.
The DeLong test (Table 3) comparing ROC performance across algorithms in the prospective validation set showed no significant differences between KNN, LR, DA, RF, and SVM (P> 0.05), with the neural network (NN) exhibiting slightly lower performance. Given the limitations of the small sample size, the logistic regression model was ultimately selected for its optimal balance between interpretability and stability, and it demonstrated the highest AUC value in the prospective validation set. Figure 3 shows the ROC curves for the validation and test sets. As detailed in Table 4, the radiomics model developed via LR demonstrated strong predictive consistency across all datasets: training set (AUC = 0.895, 95% CI 0.79–1), prospective validation set (AUC = 0.900, 95% CI 0.75–1), and test set (AUC = 0.850, 95% CI 0.72–0.98). The model accuracy, precision, recall, specificity, and F1-score were all satisfactory. Given the limited sample size, we performed calibration-curve analysis to assess the reliability of the model's predicted probabilities (Figure 4) and clinical-decision-curve analysis to evaluate real-world utility (Figure 5).

ROC Curve. (a) ROC Curve for Validation Set. (b) ROC Curve for Test Set. KNN, K-Nearest Neighbor; LR, Logistic Regression; DA, Discriminant Analysis; NN, Neural Network; RF, Random Forest; SVM, Support Vector Machine.

Calibration Plot. (a) Training Set Calibration Plot. (b) Prospective Validation Set Calibration Plot. (c) Test Set Calibration Plot.

Analysis of Clinical Decision Curves Comparing Radiomics Model with TNM Stage in the Test Set.
DeLong Test p-Value Table.

Abbreviations: KNN, K-Nearest Neighbor; LR, Logistic Regression; DA, Discriminant Analysis; NN, Neural Network; RF, Random Forest; SVM, Support Vector Machine.
Predictive Performance of Models Constructed Using Logistic Regression.

Abbreviations: AUC, area under the curve.
In the entire cohort, 43 patients exhibited sensitivity to CCRT followed by sequential immunotherapy, whereas 45 patients demonstrated treatment resistance to this treatment regimen. Based on the most recent follow-up, a total of 60 patients were diagnosed with tumor progression, with a median PFS of 8.5 months (95% confidence interval [CI]: 7.0-11.0). Among these, 45 patients died, with a median OS of 12.0 months (95% CI: 10.0-17.0). Using a cutoff value of 0.602 derived from the training set and the predictive probabilities of the radiomics model to categorize patients into high- and low-risk groups, we observed significant differences in PFS and OS between risk groups: Figure 6a-b (training set), 6c-d (prospective validation set), and 6e-f (test set).

Survival Analysis Curve. (a)The Training Set Revealed a Significant Difference in PFS Between Patients in Low- and High-Risk Groups. (b) The Training Set Exhibited a Significant Difference in OS Between Patients in Low- and High-Risk Groups. (c) The Prospective Validation Set Revealed a Significant Difference in PFS Between Patients in Low- and High-Risk Groups. (d) The Prospective Validation Set Indicated a Significant Difference in OS Between Patients in Low- and High-Risk Groups. (e)The Test Set Revealed a Significant Difference in PFS Between Patients in Low- and High-Risk Groups. (f) The Test Set Exhibited a Significant Difference in OS Between Patients in Low- and High-Risk Groups.
Discussion
Our study demonstrated that the machine learning radiomics model based on pretreatment CT performed well in the training, validation and test sets to predict treatment response and survival risk stratification in patients with unresectable stage III NSCLC receiving CCRT followed by sequential PD-1 inhibitors (P < 0.05). This suggests that pretreatment CT examination can non-invasively and effectively predict patient response to sequential PD-1 inhibitor therapy following CCRT without the need for hematology tests and biopsy. This is the first study to evaluate a machine-learning radiomics model based on pretreatment CT for predicting treatment efficacy in patients with unresectable stage III NSCLC treated with sequential PD-1 inhibitors following CCRT.
Studies have indicated that approximately 30% of NSCLC patients are diagnosed with stage III at the time of their initial visit, 31 with the majority being ineligible for surgical intervention. For patients with unresectable stage III NSCLC, CCRT is the primary treatment option, 4 with a 5-year PFS rate of just 18.3% and an OS rate of 32.1%. 32 The PACIFIC trial series’ successive publications helped clinicians recognize immunotherapy's safety and effectiveness as a consolidation treatment after CCRT.8–11 Chemotherapeutic agents not only kill cancer cells but also act synergistically with immunotherapy by inducing immunogenic cell death, increasing cancer cell antigen expression, and regulating gene expression.33–35 Radiotherapy and immunotherapy also work synergistically. Radiation therapy enhances tumor immunotherapy by facilitating the release of tumor-associated antigens, optimizing the tumor immune microenvironment, and inducing hypoxia within tumor cells; immune checkpoint inhibitors (ICI) can eliminate radiation resistance induced by radiotherapy.36,37 Some studies have demonstrated that ICI can not only attack cancer cells by activating T cells but also regulate the tumor microenvironment by normalizing tumor blood vessels, thereby increasing radiation sensitivity.38,39 Consequently, combining radiotherapy, chemotherapy, and immunotherapy can produce stronger anti-tumor effects. Based on these research findings, durvalumab has been approved worldwide as the standard treatment for patients with stage III NSCLC who are ineligible for surgical resection. 25
However, due to individual variability, only certain patients may benefit from sequential PD-1 inhibitor therapy following CCRT, and if patients irresponsive to this regimen receive this therapy, it may result in both physical and financial losses, which can have a negative impact on the patient. Therefore, identifying sensitive patients who may benefit from sequential PD-1 inhibitors following CCRT will have important implications for individualized treatment. It has been proven through studies that radiomics is a non-invasive and accessible approach for predicting how a patient will respond to treatment.40,41 However, given that immunotherapy is a novel treatment option, a significant number of studies have largely focused on using radiomics to predict the effectiveness of single radiotherapy, chemotherapy, immunotherapy, or dual therapy.42–45
Due to the support of medical insurance policies in China, most patients requiring PD-1 inhibitors opt for tislelizumab and sindilimab, which have demonstrated comparable efficacy and safety to other PD-1 inhibitors.46–51 Therefore, all patients included in this study used these two immunotherapy drugs, and statistical analysis revealed no significant difference in efficacy between the two (P > 0.05). We screened six radiomic features and detailed their clinical implications in Supplementary Table 1. These features collectively suggest that increased intratumoral textural complexity is associated with enhanced treatment sensitivity. 52 The three most critical radiomic features are as follows: 1. “waveletLHH_firstorder_Skewness” : This feature correlates with density asymmetry in tumor gray-scale distribution. Predominance of low-density areas (eg, necrosis, edema) may indicate immunotherapeutic responsiveness, as necrotic antigen release activates T cells. 53 2. “lbp3Dk_glszm_SmallAreaLowGrayLevelEmphasis”: It quantifies 3D small low-density regions. Micro-necrotic zones release damage-associated molecular patterns (DAMPs, eg, HMGB1, ATP), activating antigen-presenting cells to promote T-cell recruitment. Studies suggest micro-necrosis also induces chemokines (CXCL9/10), directly attracting CD8+ T cells.54–56 3. “exponential_ngtdm_Complexity” : High values reflect local texture disorder, suggesting strong spatial heterogeneity. Elevated complexity is positively correlated with tumor mutational burden and potential sensitivity to ICI. 57
Radiomics modeling successfully predicted true response to sequential PD-1 inhibitors after CCRT in 82.35% (14/17) of patients in the prospective validation set and accurately identified true response in 75.76% (25/33) of patients in the test set. Clinical Decision Curve Analysis revealed that the radiomic model provided a significantly higher net benefit compared to TNM staging across the clinically relevant risk threshold range (0.1-0.4). This superiority indicates that the radiomic approach reduces overtreatment (ie, false positives) while maintaining effective interventions for high-risk patients (ie, true positives). Prognostic analysis following patient stratification into high- and low-risk groups based on the cutoffs calculated by the LR radiomics model demonstrated that patients were effectively stratified for PFS and OS (P < 0.05). This suggests that a better prognosis can be achieved for patients with unresectable stage III NSCLC who are sensitive to treatment with sequential PD-1 inhibitors following CCRT. It also indicates that the radiomics model can efficiently and accurately predict the efficacy of sequential PD-1 inhibitors after CCRT, as well as stratify risk for PFS and OS, which may help achieve individualized treatment.
This study has several limitations. First, to minimize bias, all ROIs were manually outlined by a radiation oncologist; however, this approach limits the ability to assess the reproducibility of radiological feature extraction. Future studies will involve multiple annotators to evaluate inter-observer variability. Second, existing studies suggest that machine learning algorithms require a minimum sample size of 80 to achieve a root mean square error < 0.01 and at least 30 samples to ensure statistical validity. Empirical data further indicate that the recommended number of features for binary classification models should be approximately one-tenth of the sample size. 40 Training Set (N = 38): Enhanced via 5-fold cross-validation (effective sample size = 190 iterations), meeting the ‘10 events per variable’ threshold (38/6 ≈ 6.3 events per feature). Validation (N = 17) & Test Sets (N = 33): Combined sample (N = 50) exceeds the minimum requirement (N = 30) for preliminary validation per FDA AI/ML guidance. 58 Although the feature count and sample size in this multicenter study met these general requirements, the focus on patients with unresectable stage III NSCLC inherently limited the cohort size. As an increasing number of stage IV NSCLC patients are now being treated with sequential PD-1 inhibitors following CCRT, additional data should be included in future studies to validate the generalizability and accuracy of the model. We plan to expand collaboration with multiple centers to establish a CT protocol-sharing platform and standardize scanning parameters. Third, this study is currently limited to radiomics data, and multimodal data such as tissue sections and genomics will be included in the future to further explore the potential mechanism of this therapy and to reveal the associations between the radiomics features and the biological properties of tumors. Fourth, existing hospital PACS systems lack native support for radiomics analysis, leading to two key bottlenecks: Data Format Compatibility: DICOM images must be converted to NIfTI or PNG formats prior to model input, prolonging preprocessing time. Real-Time Inference Delay: Manual export of images to external platforms for analysis fails to meet clinical real-time requirements. While fully resolving these challenges remains difficult, we are collaborating with PACS vendors to develop lightweight plugins for automated image retrieval and preprocessing.
Conclusions
In this study, a machine learning-based radiomics model was successfully developed and validated to non-invasively and accurately predict the efficacy of sequential PD-1 inhibitors in patients with unresectable stage III NSCLC undergoing CCRT with risk stratification to guide individualized treatment.
Hospital Pronouns
The Second Affiliated Hospital of Guizhou Medical University stands for Hospital A; Guiyang Pulmonary Hospital stands for Hospital B; Qiandongnan Prefecture People's Hospital stands for Hospital C; and Qiannan Prefecture Hospital of Traditional Chinese Medicine stands for Hospital D.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251351109 - Supplemental material for Pretreatment CT-Based Machine Learning Radiomics Model Predicts Response in Inoperable Stage III NSCLC Treated with Concurrent Radiochemotherapy Plus PD-1 Inhibitors
Supplemental material, sj-docx-1-tct-10.1177_15330338251351109 for Pretreatment CT-Based Machine Learning Radiomics Model Predicts Response in Inoperable Stage III NSCLC Treated with Concurrent Radiochemotherapy Plus PD-1 Inhibitors by Ya Li, Bachelor, Min Zhang, Bachelor, Yong Hu, MM, Bo Du, Bachelor, Youlong Mo, Bachelor, Tianchu He, MM, Mingdan Zhao, Bachelor, Benlan Li, Bachelor, Ji Xia, Bachelor, Zhongjun Huang, Bachelor, Fangyang Lu, MD, Zhen Huang, Bachelor, Bing Lu, MD, and Jie Peng in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgments
We thank all members for their invaluable contributions.
Ethics Statement
The present study was conducted in strict accordance with the principles of the Declaration of Helsinki. The study was approved by the Ethics Committee of the Second Affiliated Hospital of Guizhou Medical University (Approval No. 2020-LS-03), the Ethics Committee of Guiyang Pulmonary Hospital (Approval No. 2022-KY-24), the Ethics Committee of Qiandongnan Prefecture People's Hospital (Approval No. 2023-LS-21), and the Ethics Committee of Qiannan Prefecture Hospital of Traditional Chinese Medicine (Approval No. 2023-LS-12).
Author Contributions
Conception and design: YL, BL, and JP. Administrative support: BL, JP, and FY L. Provision of study materials and enrollment of patients: YL, MZ, YH, BD, YL M, TC H, and MD Z. Collection and assembly of data: YL. Data analysis and interpretation: YL, MZ, and JP. Manuscript writing: all authors. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by the Qian Dong Nan Science and Technology Program (No. qdnkhJz [2023] 14), Scientific Research Project of Guizhou Provincial Health and Wellness Commission (No. gzwkj 2024-099), and Spark Program (No. XHJH-0048).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Raw data supporting the conclusions of this paper are available from the authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
