Abstract
With the progress of molecular diagnosis research on non-small cell lung cancer (NSCLC) cells, four identified categories of microRNAs have been found to be related to disease diagnosis, diagnosis of treatment resistance, prediction of prognosis, and drugs for treatment. To date, nine target mRNA/signal pathways have been confirmed for microRNA drug therapy both in vitro and in vivo. When microRNA drugs enter blood vessels, they target the tumor site and play a similar role to that of targeted drugs. However, whether they will produce serious off-target effects remains unknown, and further clinical research is needed. This review provides the first summary of microRNA therapy for NSCLC.
Introduction
Exosome and microRNA Therapies
Exosomes are subcellular vesicles composed of a lipid bilayer membrane and cytoplasm, which can protect internal RNAs. Almost all cell types can secrete exosomes, which can be used to transfer signaling molecules between cells to regulate physiological conditions. There are two ways by which exosomes can transfer signal molecules 1 : (1) via receptor/ligand recognition and the subsequent release of contents to target cells and (2) by selective pinocytosis and phagocytosis of target cells. In the tumor microenvironment, tumor-derived exosomes transfer signaling molecules between tumor cells and stromal cells to induce tumor metastasis and drug resistance, and aid in the cancer escape immune response.2,3 In contrast, immunocyte-derived exosomes inhibit tumor proliferation and metastasis. 4 Thus, exosomes function as a double-edged sword in tumor immunity.
The initial exosomal therapy for non-small cell lung cancer (NSCLC) uses a variety of natural exosomes from immune cells. These exosomes can play an anti-tumor role similar to that of immune cells, but will not induce significant graft rejection or graft versus host reaction. 5 In particular, the exosomes released by mature dendritic cells contain high levels of major histocompatibility complex and costimulatory molecules, which endow them with strong NK and T cell-dependent antitumor activity. 6 Moreover, CAR-T-derived exosomes have been shown comparable efficacy to that of CAR-T cells. 7 In exosomal therapy for NSCLC, exosomes can also be used as a delivery method for chemotherapy drugs or microRNA by modifying the cells used for extracting exosomes or directly modifying exosomes. The cellular source of exosomes is no longer limited to a variety of immune cells, but can also include tumor cells or stem cells, which are more optimal cell types for exosome production. Based on the research progress made in the application of exosomal microRNAs for the treatment of NSCLC, several microRNAs have been shown to have preventive, controlling, or therapeutic effects on NSCLC. This article reviews the recent progress in this field using various human NSCLC cell lines (ie, A549, H460, H1299, H1975, PC9, SPC-A1, and LLC) and NSCLC patients. In recent years, miRNA-based therapies have been reported in vivo animal studies with significant efficacy on the treatment of NSCLC, and corresponding clinical studies are at different stages of research. The adverse events of miRNAs are called “too many targets for miRNA effect,” which are inevitable because of the special complementary nucleotides between miRNAs and their targets. 8
Ideal Delivery Modes of microRNAs
microRNAs are usually protected by exosomes to avoid RNase degradation in the extracellular fluid. 9 Exosomes are extracellular vesicles of 30–150 nm, with the same lipid bilayer structure as their parental cells, which can cross various physiological barriers in the body and transmit information between cells. Therefore, the microRNAs carried by exosomes can penetrate neighboring cells and control the translation of their target mRNAs. 10 Mesenchymal stem cell (MSC)-derived exosomes are currently the most widely used type of exosomes, being suitable for local administration, with low immunogenicity. 11 Furthermore, MSC-Exos have similar functional elements to their parent MSCs, which can naturally target inflammatory sites and readily fuse with inflammatory cells, 12 apoptotic cells, the vascular endothelium, and tissue-derived stem cells, to modulate their behavior. 13 Notably, owing to their nanosize and biological origin, MSC-Exos possess outstanding capabilities to penetrate the stratum corneum barrier and reach the deep epidermis, dermis, and subcutaneous tissue. 14 When studying the function of exosomes, the exosomes inhibitor GW4869 is often used to inhibit the release of exosomes by MSCs and thereby reverse the therapeutic effect. 15
Although there are many types of active ingredients in the exosome, their content is usually low. The techniques to increase the abundance of microRNAs in exosomes include both viral and nonviral methods. 16 Most current applications of exosomal microRNA therapy for disease treatment use lentiviral vector-mediated gene delivery. MSCs are easily transfected by lentiviruses, which is more efficient than nonviral methods and ensures stable and long-term transcription of microRNAs. 17 Meanwhile, lentiviral vectors show better safety after insertion into the MSC genome than other virus types. 18 Lentivirus transfection is highly efficient for microRNA expression; however, the production costs are high and they are associated with adverse immune responses. 19 In contrast, nonviral transfection is easy to operate, with low immunogenicity, and the common types are electroporation and liposomes. 20 The electroporation method uses a high-voltage electric pulse to reversibly perforate the cell membrane and introduce the microRNAs into the cytoplasm or nucleus. 21 Liposomes are nanoscale vesicles with a structure similar to cell membranes, which encapsulate microRNAs and fuse with MSC 22 or MSC-Exos 23 to introduce microRNAs into the target cells.
Promising microRNA Markers for the Treatment of NSCLC
NSCLC exosomes contain several types of RNA (eg, mRNA, microRNA, lncRNA, and circRNA), of which microRNA is considered to be the most valuable for diagnosis and treatment. MicroRNAs are small non-coding RNAs that can specifically bind to the mRNA of a given target gene to prevent its translation. Exosomal microRNAs are recruited via a specific sorting mechanism according to the functional requirements of the NSCLC cells from which they originate. 24 The lipid membrane of NSCLC exosomes protects anti-apoptosis, metastasis-promoting, and drug-resistant microRNAs, transporting them to surrounding NSCLC cells. 25
Some plasma exosomal microRNAs have been found to be highly expressed in NSCLC and can be used as specific biomarkers for NSCLC diagnosis and prognosis. miR-451a 26 and miR-21 27 have both shown a significant positive correlation with the diagnosis of NSCLC. In squamous cell carcinoma, miR-21-5p/miR-93-5p/miR-106a-5p/miR-181a-5p 28 and miR-10b-5p/miR-320b/miR-486 29 are significantly upregulated. In adenocarcinoma, miR-30a-3p/miR-30e-3p/miR-361-5p 29 are significantly upregulated. miR-21/miR-155 30 and miR-21/miR-4257 31 both exhibit a significant negative correlation with the progression-free survival of patients. miR-10b-5p/miR-21-5p/miR-23b-3p, 32 miR-210, 33 and miR-494-3p 34 are all significantly negatively correlated with the overall survival of patients.
Some NSCLC exosomal microRNAs have been found to participate in the resistance to standard therapies. For example, miR-4443 has been associated with resistance to the chemotherapy drug, cisplatin. 35 In addition, miR-1246 augments radioresistance by directly targeting death receptor 5, 36 and miR-547-5p suppresses protein tyrosine phosphatase receptor type U (PTPRU), which weakens the effect of tyrosine kinase inhibitors. 37 Furthermore, miR-21 activates the PI3 K/Akt pathway to weaken the effect of epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI). 38
Potentially Effective microRNA Drugs for the Treatment of NSCLC
Other microRNAs have been found to exhibit low expression in therapy-resistant NSCLCs, and have the potential to reverse therapeutic resistance. Some examples include miR-30d-5p/miR-34a/miR-126/miR-144/miR-302a-5p/miR-433/miR-497 in NSCLC cells, miR-433 in chemotherapy-resistant NSCLC, miR-218-5p in radiotherapy-resistant NSCLC, miR-7/miR-7-5p in targeted drug-resistant NSCLC.
Inhibition of NSCLC Proliferation
Integrins are intercellular adhesion molecules that mediate cell-cell and cell-matrix communication and can promote NSCLC proliferation. Integrin α6 (ITGA6) is a member of the integrin family that performs a key role in NSCLC proliferation and invasion. 39 The study by Li et al 40 reported that the level of serum miR-126 expression in patients with advanced NSCLC was significantly lower than that in healthy controls. In addition, loading miR-126 in A549/H460 cells was shown to inhibit cellular proliferation and induce apoptosis. The overexpression of miR-126 also blocked the growth of A549 cells in nude mice, which was achieved by targeting and suppressing the translation of ITGA6 mRNA. 40 Chen et al 41 used similar research methods and found that miR-302a-5p can also inhibit A549/H1299 and A549 cells in nude mice by binding to ITGA6 mRNA.
The PTEN/PI3 K/AKT pathway is known to be a crucial and common pathway for improving NSCLC growth and metabolism. 42 Song et al 43 reported that miR-126 overexpression in A549 cells could block the PTEN/PI3 K/AKT pathway and suppress A549 cellular proliferation and invasion. Nie et al 44 further confirmed that miR-126 could inhibit NSCLC proliferation and metastasis by blocking the PTEN/PI3 K/AKT pathway in nude mice inoculated with A549 cells.
Cyclin E1 and E2 are both important factors that can regulate NSCLC cell entry into the S and G1 growth phases. The study by Liang et al 45 found that miR-144 was poorly expressed in resected NSCLC tissues from patients and A549/H1975/H1299/SPC-A1 cells, and the overexpression of miR-144 can inhibit A549 cell proliferation and colony formation and A549 in nude mice. miR-144 achieves this effect by targeting and downregulating both cyclin E1 and E2 expression. 45 Similarly, the study by Chen et al 46 found that miR-30d-5p overexpression could directly target cyclin E2 and suppress the proliferation, invasion, and migration of A549/H1299/SPC-A1 and A549 cells in nude mice. Moreover, Han et al 47 demonstrated that the overexpression of miR-34a and miR-497 can synergistically suppress the growth of A549/H460/H1299 and A547 cells in nude mice by targeting cyclin E1.
NSCLC cells actively release various angiogenic factors to promote the formation of blood vessels and provide oxygen and nutrients. Such angiogenesis serves to strongly improve NSCLC proliferation and metastasis. 48 Jeong et al 49 demonstrated that miR-497 has synergistic inhibitory effects on A549 growth and angiogenesis through the suppression of various genes, including yes-associated protein (YAP), cyclin E1, vascular endothelial growth factor-A (VEGF-A), and hepatoma-derived growth factor (HDGF). It was further demonstrated that miR-497 binds directly to the mRNA of all the above genes and thereby regulates the respective protein translation. 50
Reversal of Chemotherapy Resistance
Transmembrane p24 trafficking protein 5 (TMED5) functions as an oncogene, 51 and Wnt/β-catenin signaling represents an important pathway associated with NSCLC proliferation and invasion. 52 Liu et al 53 reported that low miR-433 expression was common in patients with large, advanced NSCLC and a poor prognosis. Moreover, miR-433 overexpression can suppress NSCLC proliferation by blocking the cell cycle progression of A549/H1299 cells, as well as reverse chemoresistance to cisplatin in A549/H1299 cells and LLC in B6 mice. 53 miR-433 mediates this effect by targeting TMED5 mRNA to indirectly suppress the Wnt/β-catenin signaling pathway. 53
Reversal of Radiotherapy Resistance
Long-term radiotherapy can induce genetic neo-mutations, and an increasing number of studies have revealed that NSCLC can acquire resistance to radiotherapy. 54 Chen et al 55 found that miR-218-5p was downregulated in NSCLC patients following radiotherapy, and miR-218-5p overexpression could accelerate the DNA damage, apoptosis, and radiosensitivity of radioresistant cell lines (A549R and H1299R). Mechanistically, miR-218-5p targets the protein kinase, DNA-activated, catalytic polypeptide (PRKDC) mRNA, which is closely related to the maintenance of chromosome stability. 56
Reversal of Targeted Drug Resistance
Gefitinib is a type of EGFR-TKI that can be used for EGFR-mutated NSCLC; however, over half of all cases eventually develop gefitinib resistance due to a T790 M mutation. 57 The study by Chen et al 58 demonstrated that the level of miR-7 expression was significantly higher in gefitinib-sensitive PC9 cells compared to that in Gefitinib-resistant H1975 cells (T790M-positive). In addition, miR-7 overexpression promoted the apoptosis of H1975 cells induced by Gefitinib treatment. The associated mechanism was revealed to be achieved through miR-7 targeting of YAP mRNA, which indirectly suppressed pro-apoptotic Bcl-2-modifying factor (BMF) and limited drug-induced apoptosis. 59
While Everolimus can inhibit NSCLC growth by inhibiting the PI3K-AKT-mTOR pathway, it promotes the excretion of miR-7-5p-loaded exosomes, thereby activating the MNK/eIF4E pathway to promote NSCLC growth. 60 Liu et al 61 found that the overexpression of miR-7-5p could inhibit the proliferation and metastasis of A549/SPC-A1 cells and exhibit a synergistic anticancer efficacy when used in combination with Everolimus. This effect is mediated via miR-7-5p targeting of MNK, which activates eIF4E to promote Everolimus resistance. 62
Conclusions
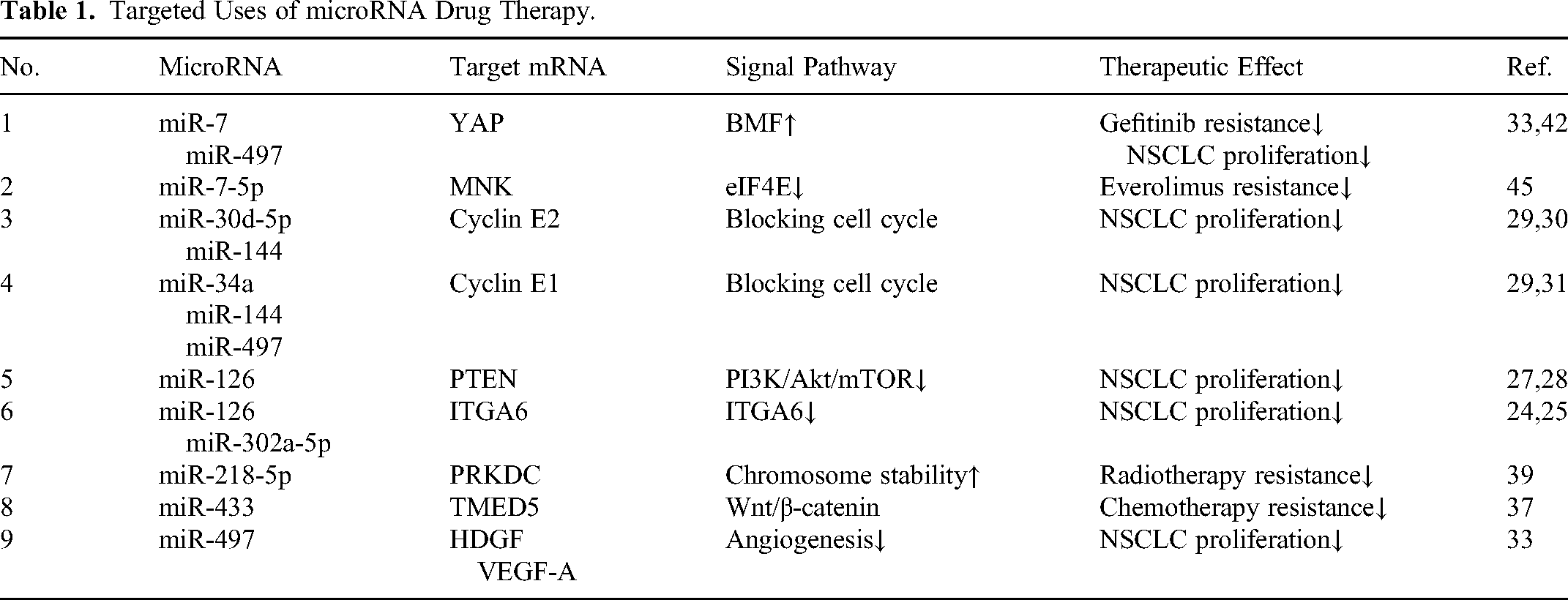
The microRNAs related to NSCLC found in research can be divided into four categories: (1) diagnosis of disease; (2) diagnosis of treatment resistance; (3) prediction of prognosis; and (4) effective drugs for treatment. Although there have been several reviews on the first three types of microRNAs, therapeutic microRNA drugs have not been summarized to date. The latest literature indicates that proliferation-related signaling pathways in NSCLC are highly complex. There are currently nine target mRNA/signal pathways that can be used for microRNA drug therapy (Table 1 and Figure 1).

Current target mRNA/signal pathways that can be used for microRNA drug therapy.
Targeted Uses of microRNA Drug Therapy.
The administration of microRNA drugs is primarily mediated through an intravenous injection of exosome carriers. These exosomes can be derived from tumor cell lines or mesenchymal stem cells, and microRNA can be loaded into exosomes and can be stable transfection or transient transfection. After microRNA drugs enter blood vessels, they can target the tumor site and play a similar role to that of targeted drugs. However, it remains unknown whether they will produce serious off-target side effects. Potential areas of future research may focus on the diagnosis or treatment of microRNAs in clinical trials (Table 2), but the number is still small and the outcome is still uncertain.
Clinical Trials of microRNAs for NSCLC.

The limitations of the present literature are that the treatment target is tumor cell lines rather than heterogeneous tumor masses, and the difficulty of treatment is far lower than the actual situation. In general, the delivery of specific microRNA to tumor cells provides novel insight and methods of supplementing existing standard NSCLC therapies. Moreover, exosomal microRNA therapy has the potential to become a convenient non-invasive therapy for the prevention, control, and treatment of NSCLC.
Footnotes
Acknowledgements
We would like to thank the native English-speaking scientists of Elixigen Company (Huntington Beach, California) for editing our manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Guangzhou Science and Technology Plan (grant number 202102010059), the Guangxi Chinese Medicine Administration Special Fund for Chinese Medicine Inheritance and Innovation Development (Construction of Translational Medicine Center of Ruikang Hospital 2023), and the Guangxi Science and Technology Base and Talent Project (grant number AD22035122).
