Abstract
This narrative review explores the potential role of electrochemotherapy (ECT) in treating anorectal tumors, focusing on its effectiveness, feasibility, and associated toxicities. ECT, which combines chemotherapy with the application of an electric field to enhance drug uptake by tumor cells, has shown promise as a local treatment, particularly in cases where conventional therapies such as radiotherapy have been exhausted or are unsuitable. The review, conducted according to SANRA guidelines, included 18 studies, on ECT in anorectal tumors, ranging from preclinical trials in dogs to case reports and clinical studies in humans. The findings indicate that ECT can achieve high tumor overall response rates (70-100%) with minimal side effects, offering benefits such as tumor reduction and preserved organ function. These results highlight the potential of ECT to provide not only tumor reduction but also the preservation of vital organ function with a relatively low toxicity profile. However, further comparative research is necessary to substantiate its role as a standard therapeutic option. Moreover, the evidence is limited by significant heterogeneity across studies, small sample sizes, and a lack of comparative research with other local treatments like radiotherapy and cryosurgery. Consequently, while ECT appears to be a promising option, particularly for palliative care or in a neoadjuvant setting, it cannot yet be recommended as a standard treatment. Future research should focus on larger, more robust studies with standardized outcomes and explore the potential synergy between ECT and other therapies to establish its place in the treatment of anorectal tumors.
Keywords
Introduction
Tumors of the anorectal region most commonly present as adenocarcinoma or squamous cell carcinoma, although rare histological types such as melanoma 1 and porocarcinoma 2 can also occur. Locally advanced rectal tumors are typically managed with a neoadjuvant treatment regimen consisting of chemotherapy and radiotherapy, followed by total mesorectal excision.3‐5 In contrast, anal tumors are generally treated with definitive RT combined with CT, with surgery reserved for selected cases, such as abdominoperineal resections or wide local excision. 3
The multidisciplinary approach has become increasingly essential in personalizing treatment strategies for each patient. However, certain patients may not be suitable candidates for surgery or a multimodal approach due to comorbidities, procedural risks, or refusal of specific therapies. Additionally, in cases of heavily pretreated patients or tumor recurrences, all standard treatments may have already been exhausted. Furthermore, some patients require symptomatic treatment with a palliative intent to enhance their quality of life.
In these scenarios, local and palliative treatments play a crucial role in achieving effective management. Among these, electrochemotherapy (ECT) has emerged as a promising local treatment that may improve tumor control and symptom management with a favorable toxicity profile. ECT combines a chemotherapeutic agent, such as bleomycin or cisplatin, administered either intravenously or intratumorally, followed by the local application of an electric field via a specialized electrode. This technique facilitates the enhanced uptake of the chemotherapeutic drug into tumor cells, thereby enhancing its cytotoxic effects. Additionally, ECT is known to induce a “vascular lock,” which is particularly beneficial in managing bleeding lesions.6,7 Moreover, besides the wellknown ‘vascular lock’, a transient arteriolar vasoconstriction occurring within minutes of pulse delivery, ECT provokes a genuine vascular disrupting effect. Endothelial swelling, apoptosis and secondary thrombosis lead to an irreversible shutdown of tumor microcirculation over the ensuing hours, thereby prolonging intratumoral drug retention and amplifying cell death. 8
Currently, ECT is primarily applied in palliative settings or when other treatment options are contraindicated or unfeasible. Additionally, ECT has shown potential in managing bleeding9,10 or obstructing tumors, offering symptomatic relief and localized control, which can be particularly beneficial for anorectal cancer patients.
However, in the context of anorectal tumors, the published evidence is limited and heterogeneous, with no comprehensive reviews or meta-analyses available.
The aim of this narrative review is to explore the role and potential applications of ECT in the treatment of anorectal tumors, with a focus on oncological outcomes, toxicity, and patient quality of life, to enhance its utilization and availability in this patient population.
Materials and Methods
Team and Review Guidelines
This narrative literature review was conducted by a multidisciplinary team comprising radiation oncologists, surgeons, radiotherapy technicians, and physicists, all actively involved in the treatment of anorectal tumors. The review was planned, conducted, and reported in accordance with the Scale for the Assessment of Narrative Review Articles (SANRA). The completed checklist is available in Supplemental Materials - Table 1. 11 To maintain comprehensive coverage of the topic, a structured narrative review checklist was utilized (details provided in Supplemental Materials - Table 2).
Registration and Literature Search Strategy
Prior to the literature screening, the review protocol was registered on the PROSPERO international database in April 2021 (registration number: CRD42021240198). 12 The literature search was conducted across multiple databases, including PubMed, Scopus, and the Cochrane Library, with no restrictions on publication date. Search terms included combinations of the following keywords: “Electrochemotherapy,” “Electroporation,” “Electromagnetic Fields” in conjunction with “Anorectal Neoplasms,” “Rectal Neoplasms,” “Anal Neoplasms,” and “Colorectal Neoplasms.” To ensure a comprehensive retrieval of relevant studies, the search was further expanded using the snowball technique.
Review Endpoints
The primary endpoint of this review was to evaluate the tumor response after ECT in the treatment of anorectal tumors. Secondary endpoints included feasibility, toxicity, symptom relief, and quality of life.
Inclusion and Exclusion Criteria
Inclusion criteria encompassed all English-language studies with retrospective or prospective designs, case reports, case series, and clinical trials that explored the application of ECT for anorectal tumors of any histology. The main inclusion criterion was to select studies that reported tumor control after the application of ECT for anorectal tumors. Secondary considerations included studies addressing feasibility, toxicity, and quality of life.
Animal studies involving ECT for anorectal tumors were also included. Conversely, reviews, commentaries, letters to the editor, in vitro studies, and non-English language papers were excluded from this review.
Study Selection and Data Extraction
Study selection was independently conducted by two authors (MF, AA), with a third author (AGM) resolving any disagreements. From the selected studies, data were systematically extracted concerning the author, study aim, methods, the setting in which ECT was applied, tumor histology, tumor response, toxicity, main results, and conclusions.
Qualitative Synthesis
Given the heterogeneity and limited scope of the available data, statistical analysis was not deemed appropriate. Instead, the findings from the selected articles were qualitatively synthesized and presented in a narrative format, providing a comprehensive overview of the current knowledge on the application of ECT in the treatment of anorectal tumors.
Results
The literature search results are presented in the PRISMA flow chart (Figure 1). A total of 79 studies were excluded, primarily due to irrelevance to anorectal tumors, non-English language, lack of primary data (such as reviews and letters to the editor), and studies focusing solely on in vitro research.
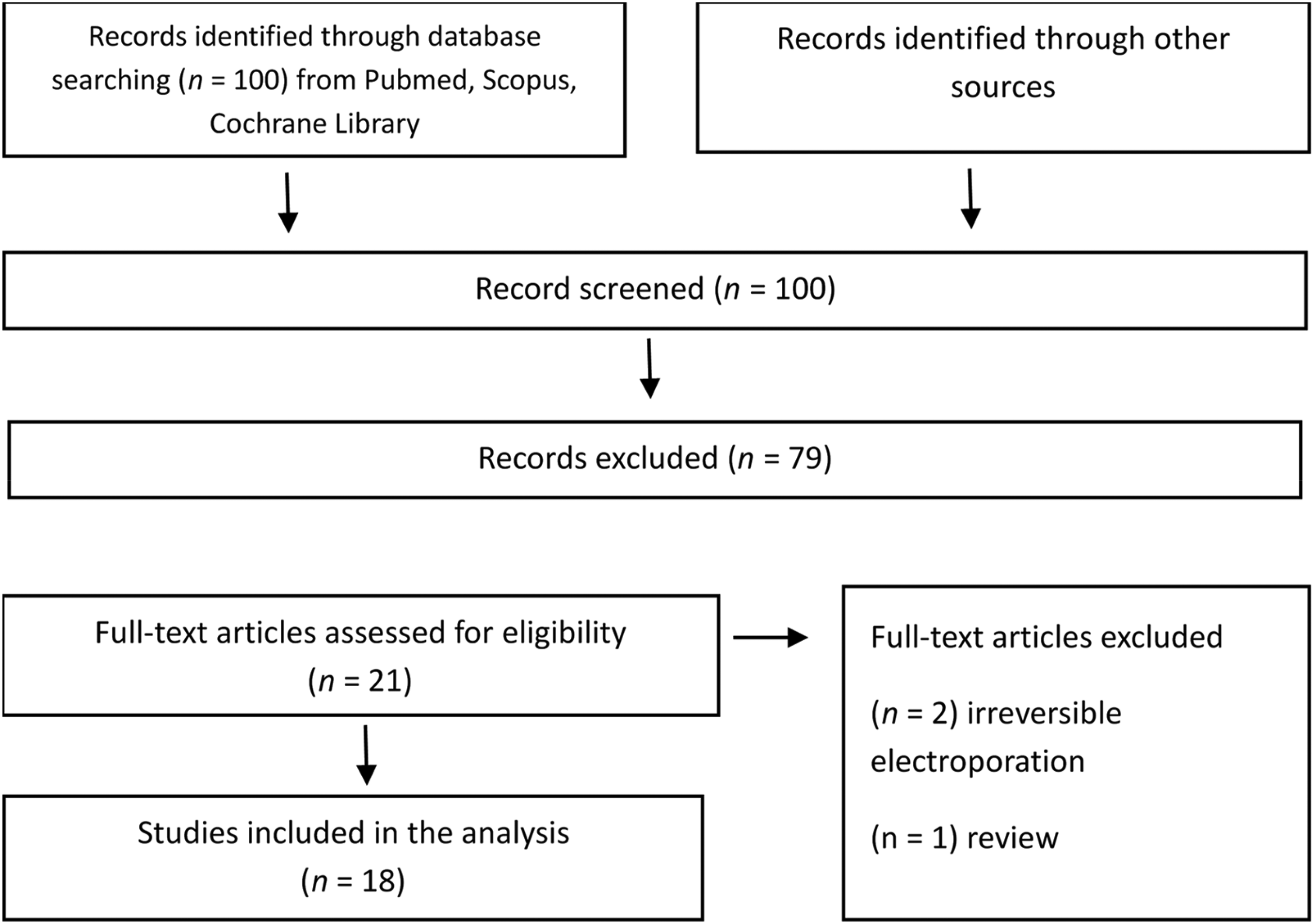
PRISMA Flowchart.
After removing duplicates, 18 studies were selected for the final analysis.13‐30 Among these, six studies focused on ECT in anorectal tumors in dogs13‐18 (Table 1), five were case reports19‐23 (Table 2), three were clinical studies24‐26 (Table 3), one was a study protocol 27 (Table 4), and three were registered ongoing clinical trials28‐30 (Table 5).
Studies on Dogs.

Legend: AGASAC: apocrine gland anal sac adenocarcinoma; CR: complete response; ECT: electrochemotherapy; PR: partial response, SD: stable disease.
Case Reports.

Legend: CR: complete response; ECT: electrochemotherapy; PR: partial response.
Clinical Studies.

Legend: AGASAC: apocrine gland anal sac adenocarcinoma; CR: complete response; ECT: electrochemotherapy; PR: partial response, SD: stable disease.
Study Protocol.

Legend: CR: complete response; ECT: electrochemotherapy.
Ongoing Clinical Trials.

Legend: ECT: electrochemotherapy.
Two commercial electroporation platforms have been adopted in the retrieved clinical studies:
Cliniporator™ (IGEA S.p.A., Modena, Italy) drives needle based electrodes either fixed linear arrays or the expandable STINGER® that are introduced transanally, laparoscopically, or percutaneously under imaging or endoscopic guidance. This set up is particularly suited to thick or extra-luminal lesions; pulses follow the ESOPE protocol (8 × 100 µs, 1 kV cm-1) delivered once tissue impedance stabilises. EndoVe® / ePORE® (Mirai Medical, Galway, Ireland) couples a flexible suctionchamber electrode to a standard colonoscope, providing circumferential contact with purely endoluminal access. After bleomycin administration the same pulse train (8 × 100 µs, 1 kV cm-1) is fired at 5 kHz; the chamber's negative pressure ensures stable electrode–tumor apposition and eliminates the need for transparietal needles”.
Of the selected studies, three papers19,22,27 and two ongoing trials28,29 reported the use of ECT in a neoadjuvant setting, one paper described ECT in an adjuvant setting, 17 four studies reported exclusive use of ECT for tumor recurrence,16,21,25,26 and nine studies focused on ECT for primary tumors, particularly in cases where other treatment options were unsuitable.13‐15,18,20,23‐26 Figure 2 shows a schematic representation of an electrochemotherapy treatment of an anorectal tumor.

Schematic Representation of an Electrochemotherapy Treatment of an Anorectal Tumor (in Red).
Studies on Dogs
The primary aim across the studies was to evaluate the effectiveness and safety of ECT in treating various types of anorectal tumors in dogs. Specific objectives included assessing the response rates of perianal tumors to ECT with different drugs and electrical pulses,13,14,18 palliating canine anal melanoma, 15 treating chemotherapyresistant Sticker's sarcoma, 16 and investigating the adjuvant use of ECT in incompletely excised apocrine gland anal sac adenocarcinoma (AGASAC).17,18
The methods involved treating dogs with ECT using either intratumoral or intravenous administration of chemotherapeutic agents (cisplatin or bleomycin), followed by the application of electric pulses. The studies included a range of tumor types and involved single or multiple ECT sessions. Most treatments were performed under sedation or general anesthesia, with biphasic electric pulses being a common technique.13,14,16,18 The studies varied in design, including case reports,15,17 retrospective evaluations, 18 and smallscale clinical trials.13,14,16
The results indicated high efficacy of ECT in treating anorectal tumors in dogs, with objective response rates ranging from 70% to 100% across different studies. Specifically, complete responses were observed in cases of perianal tumors and chemotherapyresistant Sticker's sarcoma.13,15,16 Partial responses and stable disease were also reported, particularly in studies involving AGASAC,17,18 with some dogs achieving long-term remission. 18 No significant systemic or local toxicities were reported in most cases.
The studies concluded that ECT is a safe, effective, and minimally invasive treatment option for various anorectal tumors in dogs. The findings suggest that ECT can be used both as a primary treatment and as an adjuvant therapy in cases where other treatments are unsuitable or incomplete. The results highlight the potential for ECT to be integrated into standard therapeutic protocols for treating canine anorectal tumors, with further research recommended to optimize treatment protocols and expand its use (Table 1).
Case Reports
The primary aim across the case reports was to evaluate the use of ECT as a treatment option for various anorectal tumors, with specific goals of reducing tumor size, avoiding invasive surgery, treating recurrences, and preserving organ function. Each case focused on a different tumor type or clinical scenario, including anorectal malignant melanoma, 19 perianal eccrine porocarcinoma, 20 recurrent tubulovillous adenocarcinoma, 21 recurrent rectal cancer near critical structures, 22 and obstructing rectal cancer. 23
The methods involved the application of ECT using either intratumoral or intravenous chemotherapeutic agents (such as cisplatin or bleomycin), followed by the administration of electric pulses to the tumor area. Treatment approaches varied, including preoperative ECT followed by surgery and brachytherapy, 19 standalone ECT for sensitive or recurrent tumors,20,21 and a combination of ECT with surgical excision 22 or systemic chemotherapy. 23 The procedures were performed under local or general anesthesia, depending on the case.
The results demonstrated that ECT effectively achieved partial or complete tumor responses in all reported cases.Tumor size was successfully reduced in cases requiring preoperative treatment, allowing for less invasive surgery and better preservation of sphincter function.19,21 Complete response (CR) was observed in cases where ECT was used as a primary or adjunctive treatment, with no relapse or significant side effects during follow-up.20,22 In the case of obstructing rectal cancer, a partial response was achieved, improving the patient's quality of life. 23
The case reports concluded that ECT is a valuable and effective treatment modality for various anorectal tumors, particularly in scenarios where conventional surgery is either too invasive or not feasible. ECT can be integrated into multimodal treatment plans to reduce tumor size preoperatively, enabling sphinctersparing procedures. 19 It also serves as a viable alternative to demolitive surgery in elderly or high-risk patients, offering high efficacy with minimal invasiveness. 20 Additionally, ECT shows promise as a nonsurgical alternative for small lesions and can be effectively combined with surgery or systemic chemotherapy to enhance local control and reduce recurrence risks.21‐23 (Table 2).
Clinical Studies
The primary aim of the clinical studies was to evaluate the safety and efficacy of ECT in treating colorectal and anorectal tumors. Specific objectives included assessing the use of ECT in patients ineligible for standard treatments, 24 evaluating its effectiveness in anorectal malignant melanoma, 25 and investigating its impact on primary and recurrent colorectal cancer. 26
The multicenter, phase I study from Falk Hansel et al 24 aimed to evaluate the safety and efficacy of electrochemotherapy (ECT) using a novel endoscopic electrode device for colorectal tumors. Seven patients with inoperable colorectal cancer were recruited from three centers: Mercy University Hospital (Ireland), St. Vincent's Hospital (Ireland), and Copenhagen University Hospital (Denmark). All patients had declined or were deemed ineligible for standard treatment options.
Patients received bleomycin either intravenously (IV) or intratumorally (IT) based on their clinical situation. Bleomycin was administered intravenously in a fast drip (15,000 IU/m² body surface) diluted in 250 mL of isotonic sodium chloride. The European Standard Operating Procedure on Electrochemotherapy (ESOPE) protocol was followed for drug administration. For intratumoral bleomycin, the dose was based on tumor size (eg, 250 IU/cm³ for tumors larger than 1 cm³). The endoscopic electrode (EndoVe device) was attached to a standard endoscope, and electric pulses were delivered to the tumor tissue after being suctioned into the electrode chamber. The pulses (eight pulses of 0.1 ms duration, frequency of 5 kHz, and amplitude of 1 kV/cm) were delivered by a pulse generator (Cliniporator).
Patients were evaluated using magnetic resonance imaging (MRI), computed tomography (CT), or endoscopy at baseline, 3, and 6 months post treatment to assess tumor regression. Safety was assessed by recording adverse events, including tissue perforation, inflammatory response, and any device-related issues. Follow-up endoscopies were performed at intervals of 6, 12, 18, and 24 weeks for biopsies and further assessment.
The results indicated a positive tumor response in the treated areas. Imaging and endoscopic evaluations showed that:
Tumor Regression: Tumor responses were observed in all patients, with no damage to the surrounding tissues. Five patients showed partial response, while two patients had a complete response. Safety: The treatment was well tolerated, with no severe adverse events or perforations reported. Only a few grade 1 adverse events (mild) were noted. Bleeding Control: Two out of three patients with rectal bleeding before treatment experienced cessation of bleeding, while one reported reduced bleeding.
The retrospective study of Farrincha et al 25 focused on the safety and efficacy of ECT in patients with non-metastasized primary or recurrent anorectal malignant melanoma. The cohort consisted of eight patients treated at various institutions in Portugal, Italy, and France.
All patients underwent electrochemotherapy with bleomycin administered systemically. The standard procedure for ECT involved intravenous injection of bleomycin (15,000 IU/m²) followed by the delivery of electric pulses using an electrode array positioned at the tumor site with the Cliniporator™ device (IGEA Ltd, Modena, Italy). Electric pulses were delivered based on the ESOPE guidelines. The electrode arrays were designed to ensure that the entire tumor area was exposed to electrical pulses, increasing cell membrane permeability and enabling chemotherapeutic drug entry.
Clinical follow-up assessments, including ultrasound and physical examinations, were conducted at 1 and 2 months post treatment to evaluate local control and disease progression. Adverse events were monitored, and response to treatment was classified as complete response, partial response, or stable disease. No serious adverse events were reported.
The results showed:
Tumor Response: At one month post treatment, 75% (6/8) of patients achieved a complete response, one patient showed partial response (12.5%), and one had stable disease (12.5%). These results were confirmed at two months follow-up. Local Control: Bleeding stopped in all patients, and pain levels were reported as mild to moderate. Survival: At one year of follow-up, 87.5% of patients (7/8) were alive, with 50% (4/8) being disease free and 37.5% (3/8) alive with disease. At the longest available follow-up (mean of 4.9 ± 2.0 years), 62.5% (5/8) of the patients were still alive.
The study of Rega et al 26 investigated the role of ECT as an adjunct to neoadjuvant chemoradiotherapy and for the treatment of local recurrences in colorectal cancer. The study included two patients treated for locally advanced rectal cancer (LARC) following neoadjuvant therapy and one patient with local recurrence after surgery.
For neoadjuvant therapy, the standard protocol included long-course radiotherapy (45-50 Gy, 1.8-2 Gy per session, five times a week) with concurrent chemotherapy (capecitabine 825 mg/m² twice daily). Patients underwent ECT 12 weeks after completing chemoradiation. Bleomycin was administered intravenously (15,000 IU/m²), and after 8 min, electric pulses were delivered using the Cliniporator™ device (IGEA Ltd, Modena, Italy). The electric pulses were delivered through electrodes (either expandable STINGER or fixed geometry electrodes), depending on the tumor's location and size. The procedure was minimally invasive and performed with a transanal laparoscopic approach when appropriate.
Patients were assessed with CT and MRI before and after ECT. These imaging modalities were used to monitor tumor response, including changes in tumor size and tissue characteristics (eg, fibrosis). The safety of the procedure was determined based on the absence of significant complications during or after treatment. Pain reduction and bleeding control were also monitored as part of the follow-up.
The results showed:
Tumor Response: Two patients with LARC showed stable disease after ECT, while one patient with a local recurrence achieved a complete response. Symptom Relief: Significant reductions in pain were reported in all patients. The patient with a recurrence also experienced reduced bleeding after treatment.
Local Disease Control: Tumor stabilization was maintained in follow-up, and there were no significant morphostructural changes in the lesion in the stable disease cases. The studies concluded that ECT is a safe and effective treatment option for anorectal tumors, particularly in patients ineligible for standard treatments or with recurrent disease. In fact, Falk Hansen et al 24 confirmed the safety and local efficacy of endoscopic ECT in elderly patients with multiple comorbidities. Moreover, Farricha et al 25 demonstrated that ECT offers good local control and is well tolerated in patients with anal melanoma. Finally, Rega et al 26 suggested that ECT improves quality of life by maintaining local control and reducing symptoms in patients with significant clinical responses post neoadjuvant therapy (Table 3).
Overall, considering case reports and clinical studies, the overall response rate was 19/23 (82.6%).
Study Protocol
The primary objective of the study of Rega et al is to evaluate whether ECT can increase the complete response rate following neoadjuvant treatment in patients with LARC and thereby improve the possibility of sparing organ function. 27
The study involves a Phase II randomized controlled trial. Initially, 28 patients are enrolled: 14 patients receive ECT four weeks after completing neoadjuvant treatment, followed by local excision, while the other 14 patients receive only the standard neoadjuvant treatment followed by local excision. The trial is designed to assess the potential benefit of adding ECT to the treatment regimen in terms of achieving higher complete response rates.
The trial is structured as a two stage randomized controlled trial. Depending on the outcomes of the initial group, a second stage may be initiated, potentially expanding the study to include an additional 42 patients, providing a more robust evaluation of ECT efficacy in this context. 23 (Table 4).
Ongoing Clinical Trials
The ongoing clinical trials aim to evaluate the efficacy and safety of electrochemotherapy (ECT) in the treatment of colorectal and rectal cancers, particularly focusing on neoadjuvant settings, downstaging, immune response enhancement, and treatment of inoperable tumors. Each trial targets different aspects of ECT application, from improving surgical outcomes to offering minimally invasive options for inoperable cases.
These trials are important for assessing the potential role of ECT in the treatment of colorectal and rectal cancers, particularly in the context of neoadjuvant therapy and in comparison, with electroporation alone. They also aim to explore its use in inoperable cases. The emphasis on minimally invasive procedures and comprehensive assessment methods, such as imaging and immunologic responses, highlights the need for further evaluation to determine the efficacy and safety of ECT in these clinical settings (Table 5).
Discussion
This narrative review aimed to evaluate the role and potential applications of ECT in treating anorectal tumors, focusing on its effectiveness, feasibility, and associated toxicities. The review followed SANRA guidelines and included 18 studies identified through a comprehensive literature search across multiple databases. We opted for a narrative review due to the significant heterogeneity across the included studies in terms of tumor types, treatment techniques, and clinical outcomes.
The findings suggest that ECT may be a promising treatment option for anorectal tumors, particularly in cases where standard therapies are unsuitable. However, caution is warranted, as the available data are insufficient to fully assess the safety profile and the potential toxicities associated with the treatment. Both preclinical studies in dogs and clinical reports in humans showed high response rates (70-100%), minimal side effects, and benefits such as tumor reduction and preserved organ function, with ongoing trials continuing to explore its broader efficacy.
The limitations of our review are primarily related to the significant heterogeneity across the included studies, both in terms of the types of tumors treated and the techniques employed for ECT. It is important to note that the management of various types of anorectal tumors, such as melanomas, squamous cell carcinomas, and adenocarcinomas, differs significantly, which may influence the reported outcomes of ECT. Moreover, the small number of studies, particularly in human subjects, limits the generalizability of the findings. Notably, only two prospective studies were conducted in humans, while the majority of reports are case studies or small case series, often involving fewer than 10 patients. Similarly, the preclinical studies on dogs involved very small sample sizes, ranging from 1 to 12 subjects, which increases the risk of publication bias. Additionally, most studies had a relatively short follow-up period, which constrains the ability to assess long-term outcomes and the durability of ECT effects. It is also important to note that the study outcomes assessed in preclinical studies (eg, tumor volume reduction in animal models) differ from those assessed in clinical studies (eg, patient survival or quality of life), making direct comparisons challenging.
Another significant limitation is the lack of comparative studies evaluating the response and palliative effects of ECT against other local treatments, such as external beam radiotherapy, 31 brachytherapy, 32 laser therapy, 33 and cryosurgery. 34 These factors collectively highlight the need for more robust, large-scale, comparative, and long-term studies to better understand the efficacy, safety, and relative benefits of ECT in treating anorectal tumors.
Given the current state of the evidence, which is limited and not robust, ECT cannot yet be recommended as a standard treatment for anorectal tumors. The existing studies, while promising, are small in scale and lack the comparative rigor needed to definitively establish ECT efficacy and safety compared to more traditional treatments like radiotherapy. Therefore, the use of ECT should be considered within a multidisciplinary framework, evaluated on a case by case basis, particularly for patients who have already undergone conventional treatments such as radiotherapy and are not suitable for further standard therapies. This tailored approach will help ensure that the potential benefits of ECT are maximized while minimizing risks to the patient.
Future studies should focus on establishing standardized protocols for ECT, including optimal timing, drug combinations, and integration with other therapies such as radiotherapy. Larger, multicenter trials are needed to confirm ECT efficacy and safety. These efforts will facilitate the transition of ECT from an experimental treatment to a potential standard option in routine clinical practice for anorectal tumors.Standardized methods for evaluating clinical response and symptomatic relief should be established to allow for more consistent and comparable outcomes across studies. Additionally, future research should explore the potential benefits of combining ECT with other treatments, such as radiotherapy, and specifically aim to define the optimal timing and sequence of these therapies to maximize their effectiveness. Furthermore, ECT could be tested as an alternative to radiotherapy, particularly in recurrent disease, due to its safety, repeatability, and minimal toxicity. Its potential role in neoadjuvant settings, either as an adjunct to or an alternative to radiotherapy, should also be considered in future studies. This approach will help clarify the role of ECT within the broader treatment landscape for anorectal tumors and may improve patient outcomes.
Conclusions
In conclusion, while ECT shows promise as a safe and effective treatment for anorectal tumors, particularly in cases where standard therapies are unsuitable, the current evidence is not robust enough to recommend it as a standard treatment. ECT should be considered within a multidisciplinary context and evaluated individually for each patient, especially when conventional treatments like radiotherapy have been exhausted. Future research with larger sample sizes, standardized evaluations, and longer follow-up is needed to better define the role of ECT and its potential combination with other therapies in treating anorectal tumors.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251349596 - Supplemental material for Electrochemotherapy for Anorectal Tumors: A Narrative Literature Review
Supplemental material, sj-docx-1-tct-10.1177_15330338251349596 for Electrochemotherapy for Anorectal Tumors: A Narrative Literature Review by Martina Ferioli, Alessandra Arcelli, Savino Cilla, Arina A Zamfir, Giorgio Tolento, Dajana Cuicchi, Milly Buwenge, Erika Galietta, Bruno Fionda, Luca Tagliaferri, Matteo Rottoli, Gilberto Poggioli and Alessio G Morganti in Technology in Cancer Research & Treatment
Footnotes
Author Contributions
MF, AA and AGM had the idea for the article; AAZ, GT, DC, MB and EG performed the literature search and data collection and analysis; MF, EG, and AGM drafted the manuscript; all authors critically revised the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
