Abstract
Introduction
Primary unresectable rectal cancer is found in 10% to 15% of newly diagnosed patients (pts) with rectal tumors and it is still a challenge for all clinicians.1,2 The large tumor volume and the infiltration of adjacent organs always pose the questions: is it still possible to cure the patient and what could be the complication of aggressive treatment for this frail, often elderly group of pts?
There is a paucity of data regarding unresectable rectal cancer. Because of relatively low incidence, literature data is scarce. Fixed rectal tumors grow locally, worsening quality of life, and palliative treatment does not lead to long-term relieving of symptoms. Yet, the appropriate radical neoadjuvant treatment of even large tumors can lead to successful surgery. The standard of care in primary unresectable rectal cancer is chemoradiotherapy (CRT); nowadays implemented as total neoadjuvant treatment (TNT): long course of chemoradiotherapy and subsequent 12 to 16 weeks of chemotherapy, but for many years, it was long course of chemoradiotherapy only. The aim of this treatment is to convert the tumor into the resectable one. Another recognized neoadjuvant treatment option with a similar aim is a short course of hypofractionated RT with 12 to 16 weeks of chemotherapy. 3 Nevertheless, some pts are not suitable for chemotherapy, or cannot finish this long, demanding treatment. It is worth noticing that the TNT method is not yet adopted in many institutions, and chemoradiotherapy still remains standard. The results of this neoadjuvant procedure are still not satisfactory and around 25% to 30% of pts remain unresectable.4,7
In our cancer center, different schemes of neoadjuvant treatment were implemented, from long course radiochemotherapy (CRT) through RT alone either conventionally fractionated or hyperfractionated. The choice of treatment depended on a current institution protocol at a specified time period, patient's performance status (PS), comorbidities, courtesy of attending radiation oncologist, and pts’ preferences. We published our results concerning the outcome of pts with resectable rectal cancer before, both on hyperfractionation and on establishing alpha/beta ratio for rectal cancer.7,9
Hence, the aims of the present study were to investigate whether RT alone in pts could be as effective as CRT, a widely approved standard in primary unresectable rectal cancer. Second, to evaluate the role of accelerated hyperfractionation, a novel scheme in rectal cancer pts, which may have value for a certain group of pts. Third, to imply prognostic and predictive factors to clinical practice and additionally to analyze the role of primary tumor volume on treatment results.
Material and Methods
Group Characteristics
Material consisted of 145 consecutive pts with primary unresectable, locally advanced rectal cancer (cT4N0/N + /M0) treated in our hospital from 2000 to 2016. Data was collected retrospectively, by analysis of pts’ medical records and RT treatment plans. Data of all pts was de-identified. Every patient gave informed written consent for RT with or without chemotherapy as a standard treatment per protocol of our institution.
Study Inclusion Criteria
The study inclusion criteria were: fixed or tethered rectal tumor described as primary unresectable by attending surgeon, exclusion of the presence of distant metastases, good or very good pts PS—WHO 0 to 2. The maximum distance from the anal verge was 15 cm and mesorectal fascia involvement was accepted. Moreover, involvement of surrounding organs was also accepted: uterus, prostate, seminal vesicles, bladder, urethra, muscles, and sacral bone . The exclusion criteria were the diagnosis of local recurrence, the presence of distant metastases, fistulas, or poor PS of WHO ≥3.
Rectal tumor was considered fixed when there was no mobility on digital rectal examination (DRE) or an unresectable infiltration of the adjacent organs was visualized on imaging studies (mostly computed tomography [CT] or magnetic resonance imaging [MRI]). In contrast, tethered rectal tumors revealed mobility in at least one direction on DRE and there was no adjacent organs infiltration on imaging studies. Forty (28%) pts underwent the attempt of primary radical surgery in other hospitals before referral to our center that revealed an intraoperatively unresectable tumor and those tumors were also considered fixed.
Diagnostic Work-up
The initial diagnostic work-up included: colonoscopy and/or rectoscopy, abdomen and pelvic CT and/or MRI, chest x-ray, blood count, biochemistry, and carcinoembryonic antigen (CEA) serum concentration measurement. In all the cases, the diagnosis was based on diagnostic imaging and pathologic examination of the tumor tissue samples obtained during a biopsy or surgery. WHO scale was used to classify pts’ PS.
Primary Neoadjuvant Treatment
In general, two schemes of neoadjuvant treatment were used: RT-only or CRT.
In the RT group, there were two types of fractionation: conventional (CFRT) or accelerated hyperfractionation (HART). 8 The total dose (TD) in the HART group was 66 Grey (Gy) delivered in 1.5 Gy fraction doses (fd) twice a day (break at last 6 h) and the conventional TD was 60 to 66 Gy in 2 Gy fd delivered once a day. Pts were treated 5 days a week. In the CRT group, TD of 54 Gy with fd of 1.8 Gy conventionally fractionated was given with concurrent 2 courses of chemotherapy with 5-Fluorouracil (5-Fu) 325 mg/m2 or 375/m2 + leucovorin (LV) 20 mg/m2 in weeks 1 and 5 of the neoadjuvant therapy. The interval between completion of the neoadjuvant treatment and surgery was 6 to 8 weeks.
Radiotherapy Treatment Planning
In all pts, CT in the treatment position (prone) was performed. The irradiated volume was similar in both groups: gross tumor volume (GTV) included visible tumor and suspected lymph nodes. The margin of 3 to 5 cm covering the whole mesorectum was added to create clinical target volume-tumor (CTV-T). The elective clinical target volume-nodes (CTV-N) covered mesorectal nodes (with upper limit at sacral promontory) and obturator, internal iliac, and lower common iliac nodes. In all the pts with involvement of pelvic organs, the external iliac nodes were also included in CTV-N. 14 If there was an involvement of the external anal sphincter, anal canal, or the one-third lower part of vagina, the inguinal nodes were also incorporated into CTV-N. Then, 0.5 to 1 cm of margin was added to CTV-T and CTV-N to form planning target volume (PTV). In the first phase of RT in all the treatment schemes, PTV was irradiated to the TD of 42 to 45 Gy. In the second phase, dose was delivered to GTV and suspected/enlarged lymph nodes with 0.5 to 1 cm margin and TD ranged from 54 (CRT group) to 60 to 66 Gy (RT group). Three-dimensional conformal radiotherapy (3D-CRT) or dynamic techniques: Intensity-modulated radiotherapy (IMRT) and volume-modulated arc therapy (VMAT) were used. Bowel and especially small bowel were outside high dose area, as the majority of tumors were low rectal cancers. Pts were irradiated with 6 and/or 20 MV photons generated by the linear accelerator. The dose was specified at the isocenter at 100% isodose. International Commission on Radiation Units and Measurements (ICRU) criteria were used in the treatment planning depending on the technique used. Verification of patient and target position was performed with the use of portal or kilovoltage (kv) imaging.
Toxicity Evaluation
The Common Toxicity Criteria (CTC) version 3.0 was used to evaluate toxicity. 15 The early toxicity was evaluated from the start of neoadjuvant treatment to the date of the surgery or up to 3 months if the surgery was not performed. Late toxicity was scored after surgery or over 3 months after the end of the neoadjuvant treatment.
Neoadjuvant Treatment Evaluation
Four to 6 weeks after the end of neoadjuvant RT or CRT, the pts had tumor evaluated in the physical examination and imaging studies (pelvic CT or MRI) were performed (to rule out local progression of disease or metastases). The regression of the tumor was not mandatory to qualify for the surgery. For tumors situated in the lower and mid-rectum (up to 10 cm from the anal sphincter), a total mesorectal excision was performed. Pts with tumors located higher (between 10 and 15 cm from the anal sphincter) had subtotal mesorectal excision (ie 4 cm below the tumor). The type of surgery, ie the abdominoperineal resections or the anterior resections was chosen depending on the tumor location and the surgeon's decision.
Study Endpoints
The primary aim of neoadjuvant treatment was pursuing to the radical surgery and achieving long-term local control (LC) and overall survival (OS). The outcomes were evaluated based on four endpoints. Two early endpoints were as follows: objective response (OR) being the sum of complete and partial regression measured according to the RECIST (Respone Evaluation Criteria in Solid Tumors) criteria version 1.1 and tumor resectability (RR). 16 Late endpoint was LC. Lack of LC, ie local failure was defined as the local recurrence within tumor bed or regional lymph nodes after radical surgery, or lack of radical surgery and complete tumor regression (CR) after neoadjuvant treatment. Second late endpoint was OS, which was measured from the day of the start of neoadjuvant RT to the date of the last information about patient or patient death. Missing dates of deaths were obtained from the Polish National Cancer Registry.
The treatment efficacy was compared between the RT and CRT and then in the three subgroups with different fractionation schemes (eg CRT, CFRT, and HART). The influence of different clinical factors and RT parameters on treatment efficacy was analyzed. Among analyzed clinical factors there were pts’ age, PS, sex, tumor volume, tumor mobility (fixed vs tethered), location of the tumor, the reason of irresectability (infiltration of surrounding organs vs bones), the exploratory laparotomy before neoadjuvant treatment, and the status of regional nodes. The analysis of the influence of treatment parameters are the RT technique (3D-CRT vs dynamic techniques), the method of verification of the patient positioning (portal images vs 2D-2D kV films), and the TD of RT and the length of chemotherapy cycles were evaluated.
The reporting of this study conforms to STROBE (The Strengthening the Reporting of Observational Studies in Epidemiology) guidelines.17
Statistical Analysis
In the statistical analysis, OS was estimated using the Kaplan–Meier method. Median follow-up was estimated by the Kaplan–Meier analysis with the reversed meaning of the status indicator. Comparisons were made with the use of the log-rank test. The Cox proportional-hazards model for the univariate and multivariate analyses of the prognostic factors was applied. Variables with a P value of <.1 in the univariate analysis were used in the multivariate Cox analysis. Statistical methods used for OR and RR were chi-square test and logistic regression. P value of <.05 was considered statistically significant.
Results
Group Characteristics
Out of 145 pts, there were 105 men (72%) and 40 women (28%). Most of the pts were in very good or good PS (52% and 46%, respectively) with only 3 (2%) in poor PS (WHO 2). Tethered tumor was described in 54 (37%) pts and fixed in 91 (63%). In 20 cases (14%), tumor was fixed to the nearby bones. In 102 pts (70%), tumor was situated in the lower rectum (0-5 cm from anal verge), while only in 7 cases (5%), it was located in the upper rectum (10.1-15 cm from anal verge). The regional lymph nodes were radiologically suspected in 73 pts (50%): measurable nodes with short axis ≥15 mm.16 Detailed characteristics of the study group with a subdivision into the CRT and RT subgroups are shown in Table 1. The tumor volumes (GTV) were evaluated with the use of RT planning system in 114 (79%) of cases and in 31 (21%) diagnostic CT was used because of the inaccessibility of the planning imaging (due to the change of the treatment planning system in 2004). The tumor volumes ranged between 31.4 cc and 858 cc with a mean and median volume of 215 cc and 180cc, respectively.
Patients (pts) and tumor characteristics in the radiotherapy (RT) and radiochemotherapy (CRT) group.

Neoadjuvant Treatment
Two main neoadjuvant treatment schemes were applied: 72 pta (50%) had CRT and 73 (50%) RT-only. Treatment choice depended on pts’ performance; we omitted chemotherapy in medically unfit pts. Second, in early 2000, our hospital treatment protocol allowed RT with dose escalation instead of CRF. The two groups were well balanced in most of the analyzed clinical factors. The only significant difference concerned the presence of suspected pelvic lymph nodes, that were more frequent in the CRT group (61% vs 40%, p = .010), and there was a statistical trend toward better PS in this group (p = .055). What is more, the choice of neoadjuvant treatment depended also on pts’ and physicians’ preference, and on institution treatment protocol that may vary at a specific time.
In the RT group, 37 pts had CFRT and 36 had HART. In the CRT group,16 pts (22%) had 5 days Fu-LV chemotherapy cycles, 50 (69%) had 3 days 5-Fu-LV cycles and 9% had other schemes. In the RT group, in 70 pts (96%) 3D-RT techniques were applied and positioning verification was based on portal imaging films. In the CRT group, 49 pts (68%) had dynamic techniques, and in 39 pts (54%) positioning verification was performed with kV-kV orthovoltage films.
Early Treatment Outcome
A total of 100 pts (69%) from the whole group (n = 145) underwent surgery. Out of 45 who had no surgery, the most frequent cause of not performing the resection was local progression of the disease in 14 pts (31%) followed by metastatic disease in 9 pts (20%). The other causes were worsening of a PS in 5 pts (11%), refusal to the surgery in 7 cases (15%), and 10 ptss (22%) were lost to follow-up.
Among 100 pts who underwent surgery in 74 radical resections was performed. Thus, the RR after neoadjuvant treatment was 50% in the whole group and 37% and 75% in RT and CRT groups, respectively. An abdominoperineal resection was performed in 37 pts (26%), an anterior resection in 31 (21%) and the Hartmann resection in 6 pts (4%).
The OR was observed in 67% of the pts in the whole group and in 60% and 75% in the RT and CRT group, respectively. The factors which had a statistically significant impact on OR were the tumor unresectability revealed in an earlier exploratory laparotomy and the tumor status—fixed versus tethered (Table 2).
The influence of various clinical factors on objective response (OR) and resection rate (RR) in all patients (pts).

Pts in better PS had a higher radical resection rate (RR) (64% vs 37% for pts in PS WHO 0 vs 1, respectively). Tumor mobility (tethered vs fixed) also influenced RR. Finally, the neoadjuvant treatment modality was also associated with an RR: pts after CRT presented a higher RR than after RT alone (Table 2).
Tolerance of Neoadjuvant Treatment
Almost all the pts completed neoadjuvant treatment. Only 6 (4%) had interrupted the treatment; 2 due to the early grade 3 hematological toxicity, 1 because of small bowel obstruction (grade 4), and 2 because of cystitis (grade 2), all in the CRT group. One patient refused treatment. The incidence of other early toxicities in CTC v 3.0 scale is shown in Table 3. 15
Early toxicity in radiotherapy (RT) and radiochemotherapy (CRT) groups according to Common Toxicity Criteria (CTC) version 3 classification. 10

Late toxicity was generally infrequent (Table 4). Because of overlapping of symptoms after neoadjuvant treatment and surgery, no specific scale for late toxicities was used. Two pts (2%) in the CRT group experienced small bowel obstruction. Out of the whole group, 7 (5%) had fistula in perineal scar, 6 (4%) had other fistulas, 2 (1%) presacral abscesses, 2 (1%) peritoneal inflammation, and 1 patient (0.7%) was diagnosed with ureter necrosis. Late toxicity was similar in RT and CRT groups.
Late toxicity in radiotherapy (RT) and radiochemotherapy (CRT) groups.

Local Control and Overall Survival
The median follow-up was 5.7 years (ranged from 0.12-13.48 years). Overall 5-year LC and OS in the whole group was 50% and 37%, respectively. The actuarial 5-year LC in the RT and CRT groups was 37% and 68%, respectively (P = .0002). The actuarial 5-year OS in the RT and CRT groups was 27% and 52%, respectively (P = .002). The clinical factors which had statistically significant influence on the LC and OS are shown in Table 5. Among them, WHO PS reached statistical significance and 5-year LC was 62% in pts with WHO PS of 0 versus 39% for PS WHO 1 (P= .003).
Long-term results: influence of various clinical factors on 5-year local control (LC) and 5-year overall survival (OS).
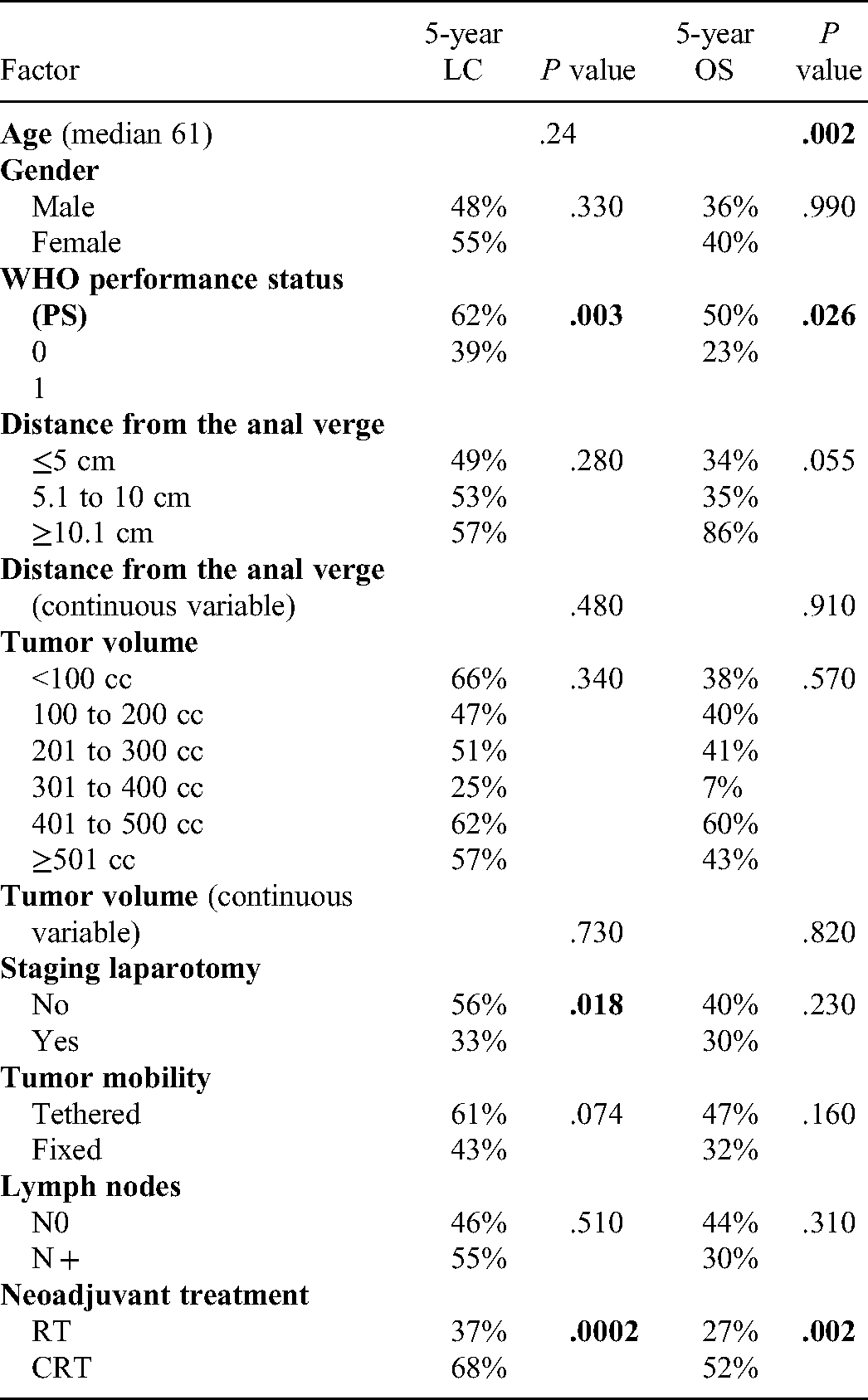
Five-year OS was 50% for pts with WHO PS of 0 versus 26% for pts with WHO PS 1 (P = .026). Moreover, pts who had the tumor unresectability revealed in an earlier exploratory laparotomy, had lower rate of LC (33% vs 56%, with P = .018). The duration of 5-Fu-LV cycles and the tumor volumes had no influence on any of the analyzed endpoints.
Multivariate Analysis of Prognostic Factors
The factors that confirmed their impact on OR were as follows: an earlier laparotomy (HR = 0.28, p = .01) and CEA serum concentration before the neoadjuvant treatment (Hazard Ratio = 0.68, P = .01). The factors that confirmed their impact on RR as independent prognostic factors were the tumor mobility (HR 0.38; P = .030), the type of neoadjuvant treatment: CRT versus RT (HR 2.74; P = .020) and the WHO PS 1 versus 0 (OR = 0.30, P = .003). Moreover, WHO PS 0 versus 1 (HR = 1.64; P = .049) and the type of neoadjuvant treatment: RT versus CRT (HR = 0.57; p = .034) were independent prognostic factors for LC. Contrary, for OS, older age was the only significant factor in multivariate analysis (HR = 1.03; P = .004).
The Detailed Evaluation of the Treatment Schemes
Taking into account treatment method and fractionation, three modalities were compared: HART, CFRT and CRT. The OR in the CRT group was better than in CF (75% vs 53%, P = .025). In the HART group, OR was described in 68% of the pts, without statistical differences between other irradiation schemes.
The evaluation of RR showed that CRT had much better results than RT (65% vs 27%, P = .025), whereas HART had intermediate effectiveness, not significantly worse than CFRT or CRT (RR = 47%, P= .070).
The evaluation of the long-term results showed that CRT proved to be more effective neoadjuvant treatment than the other two schemes. Five-year LC after CRT, HART, and CFRT was 68%, 42%, and 25%, respectively (P = .0002). Five-year OS after CRT was much better than after CFRT (52% vs 17%, P = .0002). Comparing CRT and HART only a trend toward better OS in the CRT group was observed (52% vs 36%, P = .057). Surprisingly, in the CRT group in those pts who underwent subsequent radical surgery, no local recurrences were noted further in a follow-up contrary to the other two schemes. These results are shown in Figures 1 and 2.

Local control (LC) in three groups.

Overall survival (OS) in three groups.
Discussion
Despite retrospective nature of the study, the groups in the present study were well balanced in both the No. and clinical characteristics. On the whole, it is a numerous group taking into account a small percentage of pts with primary unresectable rectal cancer. In other studies, the No. of pts varies between 20 and 100, and only one author reported the outcome of 207 pts.4,7,18,23 Moreover, in contrary to other studies, pts with unresectable local recurrences were excluded to make the groups more homogenous.19,21
The limitations of our study are its retrospective and observational character, which may be the source of potential selection bias. Because of the size of the group and retrospective character of the study, no initial assumptions nor power calculations were done. Therefore, we may expect that it could be underpowered to detect some differences. The choice of treatment type was made by attending oncologists, according to standard protocols in our center at a specific time and taking into account pts will.
All the pts were in very good or good PS; with only 2% with WHO PS of 2 (all in the RT group). This is in contrast with reports of other authors like the one conducted by Engineer et al. in which as many as 11 pts (25%) in the RT group and 4 (9%) in CRT had WHO PS of 2. 18 This observation suggests that pts with worse PS will rather be given RT than CRT, and it should be kept in mind when interpreting the results. According to the Sankt Gallen Expert group recommendations, MRI and TRUS are strongly recommended in the evaluation of local stage of rectal cancer. 23 Unfortunately, because of the low availability of MRI during the study period, the majority of pts had only contrast-enhanced CT of the pelvis and abdomen (89%). Nevertheless, diagnostic imaging studies showed that in the evaluation of locally advanced rectal tumors with the infiltration of adjacent organs, both CT and MRI have similar (76%) specificity rate. 24 In the evaluation of the involvement of regional and distant lymph nodes, several meta-analyses showed that both methods had similar specificity and sensitivity rates.24,27
Among other pretreatment factors, serum CEA concentration over 5 ng/mL before neoadjuvant therapy was found to be prognostic for pathological CR and OS.28,30 In this study, we found it also significant for OR and confirmed its role in the multivariate analysis. One of the negative prognostic factors in pts with rectal cancer is nodal infiltration. In pts with positive lymph nodes, the risk of local and distant recurrence is 70% and 33%, respectively. 31 In our study, 50% of the pts had clinically positive pelvic lymph nodes although no impact on LC or OS was found. One of the reasons for the lack of such correlation could be that the clinical assessment of the status of lymph nodes is based on radiological estimation which is unsure.31,34 Second, the most important aim in pts with unresectable rectal cancer is to lead to resectability of primary tumor irrespective of a lymph node status, so lymph node involvement could possibly have less importance.
The tumor location in rectum was found to be prognostic for OS.34,37 In study by Chan et al., pts with upper rectal tumors (≥10 cm from the anal sphincter) had better 5-year OS (71% vs 57%, p = .042) and cancer-specific survival (77% vs 60%, p = .028) than those with lower and medium location. 35 Low rectal tumor location is a negative prognostic factor that is confirmed also in other studies.34,37 We did not observe such a relationship, probably because of the low percentage of pts with lesions located in the upper part of the rectum (5%) and the majority of pts with low located tumors (70%). A tethered tumor was described in 54 (37%) pts and fixed in 91 (63%). Pts with tethered tumors had higher RR than those with the fixed ones. What is more, 5-year LC was also higher in tethered tumor group. Similar results were observed by Chan et al. who reported 5-year LC of 63% for pts with fixed tumors and 98% for tethered (p = .000001). 35
Primary tumor volume could have crucial significance on treatment results, especially in a definitive CRT. Surprisingly, it turned out that it had no impact on either of the studied endpoints. Possible explanation is rectal tumor heterogeneity and preoperative character of RT with the leading role of surgical treatment. We divided volumes by 100 cc to form relatively homogenous and larger subgroups of pts for comparisons. Although dividing by median volume seems to be more reasonable, two groups with different volumes would be too heterogeneous, taking into account the range of volumes (31.4 cc-858 cc). As far as we know, this is the first study analyzing rectal tumor volume in pts with unresectable lesions.
Despite the retrospective nature of this study, the timing between the end of neoadjuvant treatment and surgery was kept to 8 weeks’ maximum similarly as recommended in studies of other authors.36,39 Some studies actually show that prolonging the interval between treatment and surgery may be valuable in terms of tumor regression and sphincter preservation, so now we would be more eager to make this time longer, but at that point, we believed that 6 to 8 weeks is enough.
An exploratory laparotomy revealing unresectable tumor which preceded RT or CRT was a negative prognostic factor for OS and 5-year LC, probably because it is more unambiguous than indirect imaging studies and DRE. It is worth noticing that in the study by Braendengen et al. high RR was reached (84% and 68% after CRT and RT, respectively), but over 25% of all pts had an exenteration surgery, which was not performed in our hospital. To compare, we achieved RR of 64% and 37% for CRT and RT, respectively. In terms of LC, we similarly revealed better outcomes in the CRT group (68% vs 37% for CRT and CFRT). 6 What is important, OS was also better after CRT (5-year OS of 52% and 27% CRT and RT groups, respectively). In the literature, the RR was in a wide range from 45% to 90%.5,7,18,21 Such outcome might be the result of a retrospective character of the study, typical patient population outside of a clinical trial, and the fact that neoadjuvant RT or CRT was offered as a rescue treatment (alternative to palliative treatment).
The type of neoadjuvant treatment had the strongest correlation with the pts’ outcome. In the RT group, dose was escalated up to 66 Gy in some pts, supposing dose–effect relationship and assuming better local effect of higher doses. However, CRT confirmed higher effectiveness, despite lower RT dose, similarly to the studies of other authors. 6 In the study by Braendengen et al. 50 Gy was administered with or without CTH. 6 After CRT, higher rates of LC (82% vs 67%), time to treatment failure (63% vs 44%), and cancer-specific survival (72% vs 55%) were observed. The RR was also higher in the CRT group (84% vs 68%). 6
In the randomized study by Frykholm et al. who compared RT and CRT no difference in OS was found, but the RR of 74% in the CRT group and 64% in RT was described. 19 They reported very high rates of local recurrences of 44% pts after RT and radical surgery compared to only 17% after neoadjuvant CRT.
A question still remains: how to improve the results? One way is to introduce new chemotherapy schemes. For example, Bujko et al. achieved a high RR of 70% in pts receiving neoadjuvant 25 Gy in 5 Gy fd concurrently with FOLFOX 4 or 50.4 Gy in 1.8 Gy fd concurrently with 5-FuLV with oxaliplatin.40,41 Although the preferred scheme was short RT + FOLFOX 4, because of 8% difference in terms of OS, in favor of this scheme.
The updated results presented on ESTRO 38 in 2019 did not reveal any further difference. 41 Chemotherapy regimens with irinotecan, oxaliplatin, antiangiogenic drugs, or monoclonal antibodies were introduced in metastatic rectal cancer and gradually in studies with neoadjuvant treatment.42,50 Recently, newly published trials had proven effectiveness by adding FOLFIRINOX in neoadjuvant settings like PRODIGE 23, 51 or by adding CAPOX or FOLFOX in pts after a short course of RT (RAPIDO). 52 Results from the OPRA trial showed that neoadjuvant chemotherapy, as consolidation instead of an induction regimen, results in more pts being offered a watch-and-wait strategy (58% vs 43%, p = ·01). 53 On the other hand, CAO/ARO/AIO-12 study revealed that CRT followed by consolidation chemotherapy led to higher rates of pathological complete response compared with induction. 54 Aforementioned clinical trials are designed for pts in good PS. Such prolonged TNT may be for some pts unbearable. In addition, the results of TNT are very recent and need to be checked in everyday clinical practice. Thus, the scheme of chemotherapy used in our study (5-Fu-LV) could be still regarded as representative for actual standards.
Another option to improve the treatment results may be the intraoperative RT (IORT) boost as described by Valentini et al. 4 In this 14 year study, 10 to 15 Gy intraoperative boost was delivered after 50.4 Gy of CRT. In this group, LC was 100% compared to 81% in pts without IORT. The other way could be related to the role of time factor, which is well established in RT.8,10,12 However, its role is mostly proven in head and neck cancer pts, where the loss of RT effect due to the prolonging of the treatment time above 4 weeks was found as much as 0.6 Gy per each break day. 11 For other cancers, it can be lower, for example, 0.23 to 0.36 Gy/day for urothelial bladder cancer 13 or 0.37 Gy/day for rectal cancer.8,55,56
Clinical studies in which shortening the total treatment time in rectal cancer was performed were conducted with the most known hypofractionation, 57 but also with HART schemes.8,55,56 HART was introduced as an alternative to CRT, because of radiobiological studies from our institution on radiobiology and accelerated hyperfractionation in head and neck or bladder cancer, which showed its effectiveness.10,13 That is why those schemes were applied to other cancer sites. Moreover, studies showed that HART is a valuable method in resectable rectal cancer, and analysis of HART in comparison to other fractionation schemes led to discovery of alpha/beta for rectal cancer8,9 . The dose in RT-only group was escalated to compensate for 2 chemotherapy cycles by making the TD higher, similarily as it was presented in esophageal cancer study by Herskovitz. 58 Taking into account radiosensitivity of rectal cancer and unresectable tumor, the dose should be relatively high. In an innovative study by Coucke et al., HART was applied with fd of 1.6 Gy delivered twice a day to the TD of 41.6Gy. The excellent treatment results of 92% 5-year LC were described, but the material consisted of pts with primary resectable rectal cancer. 55
In a retrospective study by Suwinski et al. after HART, a lower recurrence rate of 4.3% was observed when compared to CF. 8 As described above, a possible explanation of higher effectiveness of HART can be lowering the risk of accelerated repopulation by shortening the treatment time. Moreover, during the routine 6-hour gap between fractions, no full regeneration of tumor clonogenic cells may occur, because rectal cancer cells have slow proliferation dynamics. Radiobiological formula used for comparing different fractionation schemes to the standard dose of 2 Gy is called linear quadratic isoeffect model (LQED2Gy). Taking into account time correction (T-time of treatment in days), this formula is: LQED2Gy −0.6 Gy (T-7). 56 LQED2Gy for HART TD of 66 Gy twice a day in fd of 1.5 Gy is 55.89 Gy, and for TD of 66 Gy in 2 Gy fd delivered once a day is 46.89 Gy 8 assuming α/β for rectal cancer = 5. This could be a possible explanation of the intermediate effectiveness of accelerated hyperfractionation compared to conventional fractionation and CRT in our study.
The novelty our study adds to literature is the analysis of the role of accelerated hyperfractionation, which is not a standard in rectal cancer.
Conclusions
Neoadjuvant CRT in the treatment of pts with primary unresectable rectal cancer was more effective than CFRT and HART in all analyzed endpoints: treatment response, RR, LC, and OS and is not characterized by higher toxicity.
Treatment results depend on clinical factors like PS and age and on factors characterizing the local disease extent: the unresectability stated on an exploratory laparotomy and the tumor mobility. CEA serum concentration before treatment was also found as a prognostic factor.
The lack of influence of tumor volume on all analyzed endpoints may indicate the need for neoadjuvant treatment regardless of rectal tumor dimensions and undeniable role of appropriate surgical treatment.
In the case of medical contraindications to CTH, accelerated hyperfractionation may be an optimal strategy, because of its higher effectiveness than conventional fractionation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of MSC National Research Institute of Oncology Gliwice Branch (number KB/430-01/22).
Study Type
Retrospective study.
