Abstract
Immunotherapy has emerged as a pivotal advancement in oncological therapeutics, representing a paradigm shift from conventional treatment modalities including surgery, radiotherapy, and chemotherapy. This innovative approach demonstrates considerable clinical potential through its capacity to enhance systemic anti-tumor responses via active or passive immunomodulation. Compared to traditional therapies, immunotherapy offers distinct advantages such as broad applicability, rapid therapeutic onset, and reduced adverse effects. However, critical challenges persist in clinical implementation, particularly concerning treatment safety and efficacy optimization. Current limitations, including drug off-target effects and biological delivery barriers, frequently result in suboptimal therapeutic outcomes and severe complications such as autoimmune disorders and nonspecific inflammation. Recently advancements in drug delivery systems (DDS) present transformative solutions to these challenges. Sophisticated DDS platforms enable precise spatiotemporal delivery of tumor antigens, immunotherapeutic agents, and immunostimulatory molecules, thereby achieving targeted modulation of diverse immune cell populations. This technological innovation not only enhances therapeutic efficacy but also significantly mitigates adverse reactions, while facilitating synergistic combinations with conventional cancer treatments. In this review, we outline the application of new drug delivery platforms in major malignancies (including but not limited to melanoma, non-small cell lung cancer, hormone receptor-positive breast cancer, and hepatocellular carcinoma). We further propose evidence-based optimization strategies for next-generation delivery platforms, aiming to bridge the gap between preclinical development and clinical implementation in cancer immunotherapy.
Introduction
According to the report from the International Agency for Research on Cancer (IARC), there are approximately 20 million new cancer cases and 9.7 million cancer-related deaths globally each year. 1 By 2050, global cancer cases are projected to increase by 76.6%, with deaths rising by 89.7%. Lung cancer remains the most prevalent type in terms of both incidence (12.4%) and mortality (18.7%), while early-onset growth rates of breast cancer, esophageal cancer, and prostate cancer are particularly notable. 2 The rising cancer incidence has become a primary threat to global health. The conventional methods for cancer therapy include surgery, chemotherapy, and radiotherapy. For early-stage malignant tumors, complete resection can achieve a cure. Chemotherapy drugs directly kill tumor sites or disseminated cancer cells, but they also cause severe side effects or drug resistance issues. Radiotherapy destroys localized tumor cells through ionizing radiation; however, prolonged or high-dose radiotherapy may damage normal tissues and has limited efficacy against distant metastases. Cancer immunotherapy represents a transformative paradigm in oncology, employing strategic immune potentiation to eradicate malignant cells through enhanced host defense mechanisms. 3 Distinct from conventional cytotoxic approaches (eg, chemotherapy, radiotherapy) that directly eliminate tumor cells, this modality demonstrates reduced off-target effects through its intrinsic biological specificity. 4 Immunotherapy achieves more precise and durable anti-tumor efficacy, particularly offering new hope for patients with advanced or refractory tumors. Compared to the systemic toxicity of chemotherapy or the cumulative radiation damage from radiotherapy, immunotherapy exhibits a relatively lower incidence of side effects. Furthermore, combining immunotherapy with chemotherapy, radiotherapy, or targeted therapy can significantly enhance therapeutic outcomes. Clinical validation across diverse malignancies—particularly hematologic cancers and immunogenic solid tumors—has established immunotherapy as a cornerstone of precision oncology. Clinically, cancer immunotherapy mainly includes immune checkpoint inhibitors (ICI), lymphocyte-activating cytokines, anti-costimulatory receptor agonist antibodies, cancer vaccines, oncolytic viruses, and CAR-T cells and other cell therapies. 5 Among them, ICIs are the most clinically approved and widely used immunotherapy, which enhance the anti-tumor activity of immune cells by blocking specific immune checkpoints, such as primarily programmed cell death protein 1 / programmed death-ligand 1 (PD-1/PD-L1) and cytotoxic T lymphocyte-associated protein 4 (CTLA-4). 6 In recent years, cell therapy has made significant progress in the treatment of cancer diseases, especially in hematological cancers. The use of fluorescent and radiolabeled chimeric antigen receptor CAR-T cells for cell therapy is a successful strategy for the diagnosis or treatment of malignant tumors. 7 Although cancer immunotherapy has shown significant benefits, its clinical application still faces some challenges related to efficacy and safety. Critical limitations persist in therapeutic safety profiles. Systemic administration often leads to non-specific biodistribution, triggering immune-related adverse events (IRAEs) ranging from autoimmune sequelae to potentially fatal cytokine release syndromes. These safety concerns necessitate innovative strategies to optimize therapeutic indices through either combinatorial regimens or advanced delivery technologies.
In recent years, the development of DDS has received increasing attention, and improved delivery technologies can significantly increase the accumulation of immune drugs in tumor tissues, or target immune cells, and reduce off-target adverse reactions. These distinct advantages have garnered significant attention from researchers, driving further exploration into advanced nanocarrier designs and their clinical translation to amplify antitumor immune responses. The evolution of DDS since Paul Ehrlich's pioneering “magic bullet” concept has revolutionized the precision of oncology treatment. 8 Contemporary platforms—including engineered liposomes, exosome-based vectors, biomimetic nanoparticles, stimuli-responsive hydrogels, and programmable polymeric matrices are applied to immunotherapy.9-13 Studies have shown that DDS can ensure more targeted delivery of therapeutic drugs to target cells than chemotherapy drugs alone.14-16 Utilizing the special properties of the delivery system, such as pH sensitivity, light or ultrasonic stimulation, the therapeutic drug can be controlled in time and space, and the drug release is not active during the delivery process until it is rapidly released in the target cell.17,18 In addition, the development of local delivery platforms such as implants allows the drug to be released locally and slowly, minimizing the toxicity associated with systemic administration.19,20 Continuously improved delivery technology makes immunotherapy drugs safer, drugs can effectively target tumors or immune cells, and also provides a platform for the combined use of multiple drugs to improve the therapeutic effect of combined therapy. 21
In this review, we provide a brief overview on the application of DDS in several common cancers suitable for immunotherapy. Based on the epidemiological relevance, therapeutic priority, tumor biological characteristics and compatibility with DDS, we identified six common tumors, including lung cancer, hepatocellular carcinoma, melanoma, breast cancer, gastric cancer and brain cancer. We discussed the use of DDS for safe and effective cancer immunotherapy in specific tumors. Additionally, the opportunities and challenges faced by targeted DDS in cancer immunotherapy are also presented, and relevant recommendations and strategies are proposed.
Lung Cancer
Lung cancer remains the second most prevalent malignancy worldwide, accounting for approximately 11.4% of all new cancer diagnoses. 22 While conventional therapies (surgery, chemotherapy, radiotherapy) continue to serve as frontline interventions, delayed clinical presentation and diagnostic limitations frequently result in advanced-stage diagnoses with compromised therapeutic outcomes. 23 In the past few years, immunotherapy has gradually become a promising cancer treatment strategy, which has greatly improved cancer treatment. At present, various types of immunotherapy have been developed for the treatment of lung cancer. However, due to the relative advantages and disadvantages of each treatment method, and this clinical reality underscores the urgent need for innovative treatment paradigms with enhanced precision. 24 Nanoparticle-mediated drug delivery has demonstrated remarkable potential in augmenting the clinical implementation and therapeutic efficacy of lung cancer immunotherapy. Engineered nanoparticles exhibit dual-functional capabilities: (1) specific targeting of malignant cells through surface modification, and (2) synergistic enhancement of antitumor immunity via co-delivery of multiple immunotherapeutic agents, enabling simultaneous modulation of complementary oncogenic pathways. 25 The immunosuppressive tumor microenvironment (TME) constitutes a major barrier to effective immunotherapy. To address this challenge, Wan et al developed a biomimetic drug delivery platform utilizing cell membrane-derived vehicles for co-encapsulation of doxorubicin and sorafenib. This system demonstrated enhanced immunostimulatory effects through TME modulation, achieving 68% tumor growth inhibition in Lewis lung carcinoma models. 26 Building upon this approach, Guo et al engineered chlorin e6-loaded nanovehicles conjugated with sorafenib, where the photodynamic properties of Ce6 induced immunogenic cell death while sorafenib-mediated TME remodeling potentiated dendritic cell activation. 27 Gold nanoparticles (GNPs), characterized by their high biocompatibility and recognized as one of the most stable metal NPs, have been extensively utilized in detection, imaging, and tumor treatment. 28 In addition to enabling targeted anticancer drug delivery, GNPs serve as ideal nanoparticles for cancer immunotherapy across multiple applications. 29 Their adaptability for diverse adjuvants and antigen delivery facilitates immune-mediated cancer cell destruction by promoting or triggering cytotoxic T-lymphocyte responses. Consequently, GNPs are employed to construct artificial antigen-presenting cells (APCs), which may be functionalized with co-stimulatory chemicals and MHC complex proteins encapsulating antigenic peptides. Ultimately, GNPs can act as adjuvant nanoparticles by binding to dendritic cells and activating cytokine production. 30 Liu et al developed a novel nanoplatform, GNPs@PSS/PDADMAC, and successfully loaded human PD-L1 siRNA to form GNPs-hPD-L1 siRNA complexes. Both in vitro and in vivo studies confirmed that these complexes not only serve as siRNA delivery carriers to downregulate hPD-L1 expression but also function as photoacoustic imaging agents and photothermal agents for the photothermal therapy of human lung cancer cells. 31 Notably, Su et al synthesized glycosylated polyaniline-gold nanoparticles (Au@PG NPs) featuring M1 macrophage polarization capacity. These nanostructures enhanced cytotoxic T lymphocyte infiltration and secretion of immunogenic cytokines. 32 In addition, in order to improve the efficacy of PD-L1 inhibitors, Reda et al designed a dual-delivery nanosystem co-loading PLK1 inhibitor volasertib and anti-PD-L1 antibodies, which can reduce the effective dose of volasertib and PD-L1 antibodies. In addition, a lung tumor model insensitive to the combination of CTLA-4 and PD-1 inhibitors also showed efficacy. 33 Remarkably, exosomes possess inherent stability, low immunogenicity and compatibility, making them effective drug delivery nanocarriers, and are also widely used in real-time imaging and drug tracing. 34 In the mouse lung cancer model, the paclitaxel-encapsulated exosome drug delivery system can reduce the IC50 value of chemotherapeutic drugs and significantly enhance the inhibition rate of lung cancer transplanted tumors. 35 Compared with other systemic cancers, inhalation administration has different significance for respiratory cancers. Inhaled drug delivery is a non-invasive drug delivery pathway that helps to deliver anticancer drugs directly to tumor tissues. 36 Shukla et al developed cationic niosomes for inhaled nintedanib delivery, demonstrating that nintedanib-loaded niosomes showed enhanced internalization and obvious cytotoxicity to non-small cell lung cancer cells, and had appropriate aerosol properties, which could effectively transport the lung. 37 Similarly, Liu et al engineered inhalable afatinib-loaded immunoliposomes (AT-MLP-DPI), and proved that it had a significant effect on non-small cell lung cancer. The study of anti-tumor effect in tumor-bearing nude mice showed that inhalation of AT-MLP-DPI could effectively inhibit the growth of lung tumors. 38 Finally, as shown in Figure 1, the application of common DDS in the treatment of lung cancer is listed, along with its advantages.

A Broad Range of Applications of DDS in the field of Lung Cancer Treatment.
Liver Cancer
Hepatocellular carcinoma (HCC) ranks as the sixth most prevalent malignancy globally and constitutes the third leading cause of cancer-related mortality, with over 830 000 annual diagnoses worldwide. 22 The advent of tumor immunotherapy has positioned immunotherapy drugs as primary first- and second-line therapeutic options for hepatocellular carcinoma (HCC). However, their clinical efficacy remains suboptimal in most patients, particularly those with underlying cirrhosis-induced hepatic dysfunction. This compromised liver architecture significantly restricts drug biodistribution, leading to inadequate intratumoral drug accumulation. To address these limitations, advanced DDS capable of targeted tumor localization, enhanced cellular retention, and controlled drug release are urgently needed. Nanoparticle drug delivery systems demonstrate exceptional potential in this regard, offering superior drug-loading capacity, enhanced cellular internalization efficiency, and minimized off-target toxicity. 39 Through surface functionalization strategies, these nanovehicles can achieve tumor-specific targeting via receptor-ligand interactions, thereby concentrating therapeutic payloads within malignant lesions. He et al compared the in vitro and in vivo drug release levels of sorafenib loaded liposomes and free sorafenib. The results showed that the biocompatibility and anti-tumor effect of sorafenib-loaded liposomes were significantly improved compared with free sorafenib. 40 In addition, nano drug delivery platforms can achieve functional effects. Qiu et al developed a multifunctional nano-ultrasound contrast agent (ATO/PFH NPs@Au-cRGD) that synergizes with anti-PD-L1 immunotherapy. This combinatorial approach not only augments antitumor immune responses but also modulates macrophage polarization within the TME, achieving dual therapeutic effects. 41 Furthermore, sequential delivery platforms like the peptide/aptamer-modified lipid system developed by Lv et al effectively overcome TME-induced drug permeability barriers, demonstrating potent tumor-targeting capabilities and significant therapeutic efficacy in preclinical models. 42 Polymer micelles (PMs) is a potential nano-delivery carrier due to its small particle size, stable structure, solubilization and low toxicity. Zhang et al engineered glycyrrhetinic acid-modified carboxymethyl chitosan-thioketal-rhein conjugates that self-assemble into tumor-responsive micelles. These smart carriers maintain drug stability during systemic circulation while rapidly releasing payloads in the acidic, ROS-enriched TME, providing a rational strategy for delivering hydrophobic anticancer agents. 43 As a new type of communication and drug delivery medium, exosomes can transport a variety of bioactive molecules (proteins and nucleic acids) between cells and regulate the cellular microenvironment and immune system. Unlike other nanoparticle carriers, exosomes contain transmembrane proteins, which can enhance endocytosis and promote the delivery of their internal contents. Du et al developed CD47-functionalized exosomes (Er/RB@ExosCD47) loaded with ferroptosis inducers and photosensitizers. The surface CD47 decoration enables immune evasion from mononuclear phagocytes, while laser activation triggers tumor-specific ferroptosis. Compared to synthetic nanoparticles, these natural vesicles possess intrinsic transmembrane proteins that enhance cellular uptake and biocompatibility. 44 In addition, due to the limited efficiency of the drug through the cell membrane, it is often unable to target key cancer pathways, resulting in low drug efficacy. Creatively, Raman et al designed an intracellular delivery system based on Salmonella, using the natural invasion and survival mechanism of Salmonella, so that protein drugs can be self-targeted and directly deposited into cancer cells. 45 In addition, various nanotechnology platforms such as therapeutic and diagnostic integrated nanocarriers have considerable prospects as the next generation DDS, which can detect diseases early, monitor and treat at the same time, and target therapy with minimal toxicity to diagnose and treat liver cancer. As shown in Figure 2, the application of DDS immunotherapy for liver cancer and a series of classic pathways of action are shown.

Schematic Diagram of the Application of DDS in Liver Cancer Immunotherapy.
Melanoma
Melanoma, characterized by its aggressive metastatic potential and high mortality rate, remains one of the most lethal cutaneous malignancies. Traditional treatments, such as surgical resection of tumors, chemotherapy or radiotherapy, can cause serious side effects associated with immune responses. However, with the emergence of ICI, such as PD-1/PD-L1 blockade, the therapeutic prospect of melanoma has undergone tremendous changes from traditional treatment to immunotherapy. Nevertheless, clinical translation of immunotherapy faces dual challenges: inadequate T-cell infiltration within the immunosuppressive tumor microenvironment and systemic immune-related toxicities caused by off-target effects. To address these limitations, synergistic strategies integrating immunotherapy with drug delivery platform have emerged as promising approaches to enhance therapeutic efficacy while minimizing adverse events. GNPs can address the core challenge of targeted drugs struggling to reach tumor sites. Furthermore, utilizing GNPs to deliver PD-1/PD-L1 antibodies or siRNA represents a highly effective strategy to block the PD-1 immune checkpoint in tumors. Meir et al conjugated PD-L1 antibodies onto the surface of GNPs, and through non-invasive measurements of changes in intratumoral NP accumulation levels, the results demonstrated that these NPs effectively halted tumor growth with only one-fifth of the standard therapeutic dose. 46 Shi et al developed a dendritic nanoplatform fabricated using gold nanoparticles coated with partially zwitterionic dendritic molecules. By blocking the binding of dendritic cells (DCs) to programmed cell death ligand 1 (PD-L1) antibodies, this platform inhibits melanoma growth through the generation of an antitumor immune response. 47 As a new type of local drug delivery system, hydrogel has good biocompatibility, high drug loading and sustained release performance, which can protect drugs from harsh environments and can directly release embedded therapeutic agents into tumor tissues. In addition, their three-dimensional porous architecture not only protects therapeutic agents from enzymatic degradation but also enables spatiotemporal control of drug release within tumor tissues. 48 Meng et al prepared imiquimod nanocrystals embedded in chitosan thermosensitive hydrogels, which synergistically combined hyperthermia-induced immunogenic cell death with immune activation. This system significantly enhanced cytotoxic T-cell infiltration and suppressed melanoma progression and recurrence in preclinical models. 49 Similarly, Kim et al synthesized a novel thermosensitive hydrogel for co-delivery of BRAF inhibitors and immunotherapy checkpoint blocking antibodies for the treatment of BRAF-mutated melanoma. The results showed that the drug delivery system could significantly increase the local drug concentration in the tumor. 50 Yang et al engineered a stimuli-responsive DNA hydrogel system integrating multivalent functional modules, including photodynamic units, immune activation units, and exosome killing units. The results showed that in the mouse melanoma in situ model, the smart DNA hydrogel was used as a local therapeutic agent to achieve a combination of efficient immune synergistic therapy and photodynamic therapy, providing a potential drug-loading approach for future tumor immunotherapy. 51 Microneedle (MN) technology represents another breakthrough in transdermal drug delivery. By facilitating deep tumor penetration and minimizing systemic drug leakage, MNs not only act as physical adjuvants to stimulate antigen-presenting cells but also enable combinatorial therapies. 52 Soluble MNs (DMNs) are made of biocompatible and water-soluble materials that can usually be completely dissolved, and then dissolve and release the loaded compounds when implanted into the skin, which can further enhance the effect of chemotherapy immunotherapy. For instance, Tian et al designed dissolvable MNs loaded with pH-sensitive liposomes co-encapsulating doxorubicin and siPD-L1, which achieved tumor microenvironment-responsive release and demonstrated superior antitumor activity in murine melanoma models. 53 Furthermore, Li et al integrated self-assembling PD-L1-targeting micelles (FER) with MN systems, enabling sustained release of anti-PD-1 antibodies and resiquimod to potently inhibit tumor growth. 54 Sun et al reported a nano-drug delivery system with homologous tumor targeting, using zeolite imidazole framework-8 (ZIF-8) coated by cancer cell membrane as a carrier to deliver pyroptosis inducer oxaliplatin and immunomodulator imiquimod. After oxaliplatin-induced pyroptosis of tumor cells, the immune adjuvant imiquimod can enhance local anti-tumor immunity and achieve efficient combination of chemotherapy and immunotherapy. 55 Collectively, advanced drug delivery systems overcome the limitations of free therapeutics by enhancing bioavailability, reducing off-target toxicity, and enabling tumor-specific targeting. As shown in Figure 3, the application of DDS immunotherapy for melanoma and a series of classic pathways of action are shown.
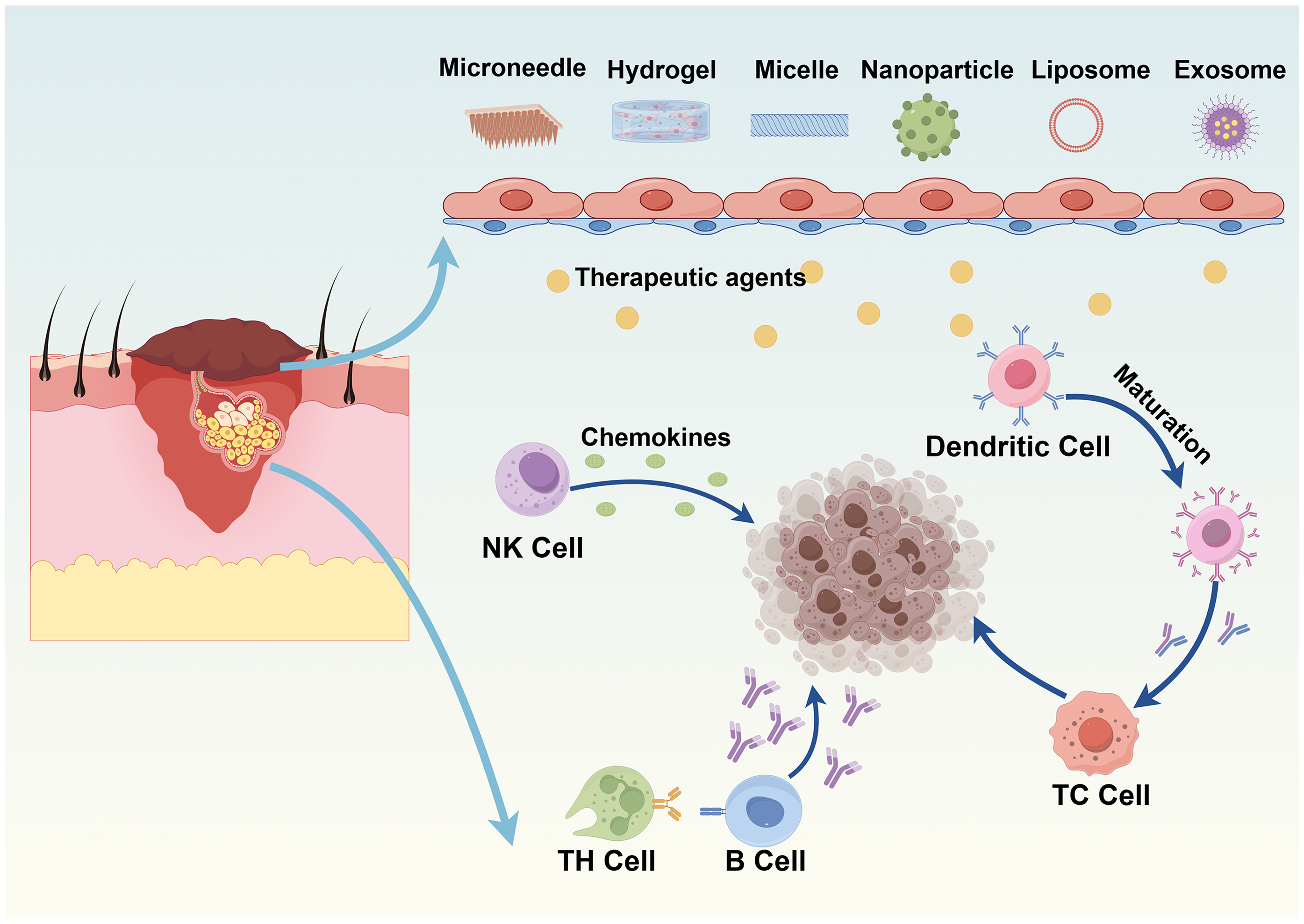
Schematic Diagram of the Application of DDS in Melanoma Immunotherapy.
Breast Cancer
Breast cancer is the most common cancer in women. Triple-negative breast cancer (TNBC), characterized by the absence of estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 expression, represents the most aggressive breast cancer subtype with 5-year survival rates below 30% for metastatic cases. Recently, atenlizumab combined with chemotherapy drug albumin-bound paclitaxel in the treatment of PD-L1 positive unresectable, locally advanced or metastatic TNBC has opened a new era of immunotherapy. However, immunotherapy as a single therapy has limited efficacy for TNBC. This limitation underscores the necessity for rational combination strategies integrating immunotherapy with chemotherapy, sonodynamic therapy (SDT), and photothermal therapy (PTT) through advanced nano-drug delivery systems, which can synergistically enhance therapeutic efficacy while mitigating systemic toxicity. Among them, PTT has achieved encouraging therapeutic effects. Photothermal can affect cell signal transduction of cell cycle and induce tumor cell death by activating autophagy and regulating mitochondrial function.56,57 Zhao et al constructed RIFe@TRM nanoplatform based on Fe3O4 nanoparticles loaded on breast cancer cell membrane, which showed excellent synergistic effect combined with photothermal therapy and significantly inhibited the growth of breast tumors. 58 Liposomal platforms exhibit superior biocompatibility and drug stabilization capabilities, making them ideal carriers for combination therapies. Chen et al developed cleavable immunoliposomes co-delivering paclitaxel and MMP-2-responsive anti-CD47 antibodies. This system can effectively polarize M2 macrophages into M1 phenotype, enhance macrophage phagocytosis of tumor cells and activate systemic T cell immune response. Synergistic inhibition of tumor cell proliferation and metastasis with paclitaxel. 59 Yang et al leveraged macrophage-mediated active tumor targeting by engineering biotin-avidin molecular docking systems to anchor doxorubicin-loaded liposomes on macrophage surfaces. The macrophage-liposome (MA-Lip) hybrid can significantly increase the accumulation of doxorubicin in tumor sites and enhance the anti-tumor immune response. 60 Hu et al constructed a tumor cascade targeted responsive liposome (NLG919 @ Lip-pep1) by combining peptide inhibitor (AUNP-12) with liposome and encapsulating indoleamine-2,3-dioxygenase inhibitor NLG919. In the mediation of AUNP-12, NLG919 @ Lip-pep1 targeted tumor cells with high expression of PD-L1. In this way, the precise blocking of the PD-1 signaling pathway can be achieved, and the activity of T cells can be restored, which further alleviates the immunosuppressive microenvironment. 61 Polymeric micelles, composed of amphiphilic block copolymers with hydrophilic shells encapsulating hydrophobic cores, offer programmable drug release profiles. Lan et al engineered pH-responsive polymeric micelles co-encapsulating doxorubicin (DOX) and 1-methyltryptophan. This system could increase the accumulation of drugs in tumors and prolong the circulation time. The co-released dox and 1-MT triggered immunogenic cell death, promoted the secretion of cytokines such as tumor necrosis factor-α and interferon-γ, and further promoted T cell-mediated immune response. 62 The hypoxic-sensitive block copolymer micelles designed by Liu et al can effectively deliver drugs to triple-negative 4T1 breast tumors, release chemotherapy drugs under hypoxic conditions, and jointly induce ICD of tumor cells. 63 In addition, emerging extracellular vesicle-based systems demonstrate exceptional tumor targeting through homologous recognition mechanisms. Bi et al extracted extracellular vesicles from mouse breast cancer cells to construct a dual-loaded melanin and paclitaxel albumin vesicle delivery system. In vivo and in vitro studies, the vesicle delivery system has stronger cytotoxicity than paclitaxel albumin. 64 Wu et al designed a metal-organic framework-based nano-delivery platform (MnCa-MOF), encapsulating the chemotherapeutic drug pyrotinib and the immunosuppressant PD-1/PD-L1 inhibitor 2 into MnCa-MOF, and coupling the target molecule triphenylphosphine, which can activate the tumor infiltration of T lymphocytes, enhance the accumulation and penetration of PD-1/PD-L1 immune agents, and ultimately enhance the immunosuppressive effect. 65 Despite these advances, critical challenges persist in NDDS development for TNBC, including precise control of spatiotemporal drug distribution, scalable manufacturing of complex delivery systems, and personalized therapeutic regimen optimization.
Gastric Cancer
Gastric cancer, ranking among the most prevalent gastrointestinal malignancies, exhibits particularly poor prognosis with over 50% of patients presenting metastatic dissemination at initial diagnosis. While emerging immunotherapies have demonstrated clinical promise for unresectable or metastatic cases, therapeutic efficacy remains constrained by molecular heterogeneity and immunosuppressive TME dynamics. Current strategies emphasize multimodal integration of immune adjuvants with complementary modalities to overcome these barriers. For instance, Zhang et al developed a novel nano-drug delivery system that synergistically combines hyperthermia, gene therapy, and immunotherapy. This system enables selective photothermal release of HER-2-targeted genes and immune adjuvant CpG sequences into gastric tumor cells. 66 Similarly, Meng et al designed a thermosensitive liposome-based platform (PTX-R837-IR820@TSL) co-delivering IR820, paclitaxel, and imiquimod, integrating photothermal/photodynamic therapy with adjuvant immunotherapy to establish diagnostic-therapeutic strategies for gastric cancer. 67 Furthermore, Li et al engineered iPSC-derived exosomes fused with dendritic cell exosomes, modified with anti-PD-1 antibodies, and loaded with chemotherapeutic agents for synergistic chemoimmunotherapy. Their results revealed that these exosomes vesicles significantly enhanced the in vivo efficacy of chemotherapeutic drugs while releasing PD-1-inhibited T lymphocytes to amplify antitumor immune responses. 68 However, the effectiveness of cancer immunotherapy is inherently limited by insufficient T cell infiltration into the TME. Compounding this challenge, tumor-associated macrophages actively participate in shaping the immunosuppressive landscape of the TME. Addressing this, Zhang et al innovatively utilized human serum albumin nanoparticles (HAS-NPs) to dual-target cellular components within the TME, combining chemotherapy and immunotherapy. Their approach successfully polarized M2 macrophages toward the pro-inflammatory M1 phenotype, offering a translatable strategy for clinical immunotherapy. 69 Hypoxia, a hallmark of the TME, is intricately linked to tumor progression and immune evasion. To target this feature, Wang et al developed TH-302-encapsulated mPEG-PLGA nanoparticles (TH-302 NPs) that selectively accumulate in hypoxic tumor regions. These NPs not only inhibited proliferation and induced apoptosis of gastric cancer cells under hypoxic conditions but also alleviated tumor hypoxia. Remarkably, combining TH-302 NPs with α-PD-1 antibodies synergistically enhanced PD-1 blockade efficacy and improved immunotherapy outcomes in vivo. 70 As an effective strategy for cancer immunotherapy, tumor vaccine can help patients actively produce immune responses against tumor-specific antigens and tumor-associated antigens. Kohnepoushi et al engineered poly (lactic-co-glycolic acid) nanoparticles (PLGA NPs) to encapsulate gastric tumor antigens, protecting them from proteolytic degradation while promoting dendritic cell maturation and T cell activation. Additionally, extracellular vesicle have emerged as promising natural nanocarriers. 71 Additionally, extracellular vesicles (EVs) have emerged as promising natural nanocarriers. You et al developed CD47-high-expressing engineered EVs that suppress YTHDF1 expression by inhibiting frizzled7 translation and inactivating the Wnt/β-catenin pathway. This innovative system facilitates immunogenic tumor cell self-presentation, thereby activating cytotoxic T lymphocyte responses and suppressing gastric cancer progression and metastasis. 72 Collectively, these advancements highlight the potential of multifunctional nano-delivery systems capable of co-delivering therapeutic agents and undergoing targeted modifications to address complex TME challenges.
Brain Cancer
Malignant brain tumors are classified as rare malignancies in adults, constituting merely 1.6% of all cancer types. 1 Nevertheless, they persist as one of the most lethal cancers. Among these, glioblastoma (GBM) stands out as the most prevalent and aggressive primary brain tumor in adults, characterized by high invasiveness and a marked propensity for recurrence. Despite the current standard therapeutic triad comprising surgical resection, radiotherapy, and chemotherapy, the median survival duration remains limited to 14-16 months, with a 5-year survival rate of merely 7.2%. 73 This dismal prognosis stems from GBM's intrinsic heterogeneity, immunosuppressive tumor microenvironment, and the protective blood-brain barrier (BBB). While immunotherapy has emerged as a transformative approach in oncology, its efficacy in GBM remains constrained by the aforementioned biological barriers. To address these limitations, recent investigations have focused on developing novel delivery systems leveraging endogenous receptor-mediated transport mechanisms, particularly targeting transferrin receptors, insulin receptors, and low-density lipoprotein receptor-associated protein 1(LRP1). A notable advancement was reported by Li et al, who engineered dual-responsive brain-targeting peptide-drug conjugates (PDCs). By conjugating camptothecin(CPT) and resiquimod (R848) to tandem peptides, they constructed supramolecular nanostructures (MAPDCs) capable of specifically recognizing vascular endothelial LRP1 for BBB penetration. In vivo evaluations demonstrated MAPDCs’ capacity to reprogram glioma-associated macrophages, amplify immune responses, and reactivate chemotherapy-mediated tumoricidal effects, thereby significantly enhancing antitumor efficacy. 74 Parallel innovations include pH-responsive nanoparticles developed by Wang et al, synthesized through free radical polymerization and conjugated with anti-PD-L1 antibodies. These nanoparticles exhibit superior BBB permeability and controlled drug release within acidic TME, potentiating immunotherapy outcomes. 75 Furthermore, emerging strategies aim to convert immunologically “cold” gliomas into “hot” tumors by modulating the immunosuppressive TME. Yu et al pioneered a sequential-release hydrogel system that epigenetically reprograms STING signaling through timed delivery of decitabine and STING agonists, effectively restoring antitumor immunity. 76 Complementary approaches, such as Mahmoud et al's CXCL12/CXCR4-axis-targeting AMD3100-loaded synthetic protein nanoparticles, enhance glioblastoma radiosensitivity while triggering T cell-mediated immune surveillance. 77 Multimodal therapeutic integration represents another promising frontier. Liu et al engineered multifunctional EVs capable of traversing the BBB to deliver PD-L1 inhibitors while rejuvenating exhausted CD8+ T cells within TME. 78 Similarly, Zhang et al devised redox-responsive micelles co-encapsulating paclitaxel and anti-PD-L1 antibodies, achieving synergistic reactivation of localized immune responses. 79 In addition,application of AuNPs as imaging probes, drug delivery systems, radiosensitizers, photothermal transducers, and multimodal therapeutic agents in malignant gliomas. 80 Chen et al developed a novel approach utilizing gold-core silica-shell nanoparticles (Au@SiO₂ NPs) combined with low-dose radiation to enhance the therapeutic efficacy of the immune checkpoint inhibitor atezolizumab against brain tumors. The combination of Au@SiO₂ NPs with low-dose radiation was shown to improve the clinical effectiveness of immune checkpoint inhibitors. 81 Moreover, drug-loaded AuNPs can overcome efflux transporters present on the BBB, by masking the drugs, leading to enhanced penetration of the chemotherapeutics into the brain. 82 Gao et al constructing acid-triggered self-assembled gold nanoparticles (D-A-DA/TPP) to achieve the selective delivery of doxorubicin (DOX) to glioma, combined with PD-1 checkpoint blocking to further activate T cells to stimulate anti-tumor immunity, more glioma cell apoptosis can be induced. 48 Ruan et al developed a gold nanoparticle-based drug delivery system (An-PEG-DOX-AuNPs). In vivo studies revealed that An-PEG-DOX-AuNPs exhibited significantly enhanced accumulation in gliomas compared to free DOX. This system enabled specific delivery and controlled release of DOX within glioma tissues, markedly prolonging the median survival time of glioma-bearing mice. 83 Although these advances underscore immunotherapy's potential in neuro-oncology, critical challenges persist. Foremost among these are the development of next-generation delivery systems to optimize BBB penetration and the rational design of immunotherapy-chemoradiotherapy combinations to amplify therapeutic synergies. Future directions should prioritize genomic profiling-driven patient stratification and biomarker-guided personalized immunotherapy regimens to advance precision neuro-oncology. In Figure 4, the application and advantages of DDS in breast cancer, gastric cancer and brain cancer immunotherapy are shown.

Schematic Diagram of the Application and Advantages of DDS in Breast Cancer, Gastric Cancer and Brain Cancer Immunotherapy.
Limitations and Challenges of DDS
Despite remarkable advancements in nanomedicine for targeted drug delivery, several intrinsic limitations and practical challenges must be acknowledged to guide future optimization and clinical translation. First, the biosafety of nanomedicine delivery systems requires in-depth evaluation. NPs can induce unintended biological effects, including oxidative stress, inflammatory responses, and organ-specific toxicity. For example, cationic polymers and metal-based nanomaterials inevitably release large amounts of metal ions, damaging vascular endothelial cells and leading to apoptosis and compromised vascular integrity. 84 Additionally, due to passive uptake by the mononuclear phagocyte syste, NPs frequently accumulate in non-target organs (liver and spleen), causing off-target effects and increasing hepatotoxicity risks. 85 Liposomal doxorubicin, for instance, significantly reduces cardiotoxicity but causes dose-dependent skin toxicity due to unintended extravasation. 86 Lipid-based NPs, such as PEGylated liposomes, have also been reported to trigger hypersensitivity or autoimmune reactions. 87 Notably, the tumor microenvironment can undergo dynamic remodeling during treatment. This adaptive alteration may gradually render the initial targeting strategies of nanocarriers ineffective, resulting in secondary therapeutic resistance. 88 Paradoxical effects, such as enhanced tumor metastasis via nanoparticle-induced endothelial leakage, have been reported in preclinical models. In addition, Complex synthesis processes hinder reproducibility, affecting drug loading efficiency and pharmacokinetics.
Nanomedicine development requires harmonized guidelines for physicochemical characterization, safety evaluation, and manufacturing practices. Regulatory agencies must collaborate to address the unique challenges of DDS, such as targeting inefficiency, aggregation in biological fluids and long-term toxicity. While nanodrug delivery systems hold transformative potential, their clinical success hinges on resolving these multifaceted limitations through collaborative research and rigorous safety assessments.
Conclusions and Future Directions
Despite significant advances in cancer immunotherapy, substantial challenges persist in clinical translation, particularly due to the complexity and heterogeneity inherent in the TME. Pathophysiological features of TME-including inadequate vascularization and stromal barriers-compromise drug penetration, leading to suboptimal delivery efficiency. Meanwhile, spatial and temporal heterogeneity of TME components frequently results in diminished targeting specificity and off-target effects. Emerging DDS demonstrate potential to address these limitations. Although the whole field of cancer immunotherapy is developing rapidly, the design of delivery technology in this field is still in its infancy. This review systematically evaluates novel DDS strategies for immunotherapy in six prevalent malignancies (non-small cell lung cancer, hepatocellular carcinoma, melanoma, breast cancer, gastric cancer and brain cancer), with Table 1 cataloging representative nanoplatform-based immune therapeutics and combination regimens. Advanced functionalization approaches, including encompassing active targeting ligands, bio-nanotechnology interfaces, exogenous physical triggers, and combinatorial therapeutic payloads, demonstrate enhanced tumor accumulation and improved modulation of immunosuppressive TME. These technological innovations potentiate the efficacy of diverse immunotherapeutic agents, including immune checkpoint inhibitors, cytotoxic drugs, cytokines, tumor-associated antigens, and immune adjuvants. Notably, current DDS development faces multifaceted challenges, including interspecies variability between preclinical models and human pathophysiology, intratumoral heterogeneity across disease subtypes, and patient-specific variations during disease progression. Despite these translational barriers, ongoing advancements in biomaterial science and pharmacokinetic optimization are poised to enable clinical implementation of next-generation delivery platforms. We anticipate that personalized DDS architectures will revolutionize immunotherapy paradigms, achieving enhanced therapeutic indices while mitigating systemic toxicity.
Summary of Drug Delivery Systems Applications in Different Systems.

Footnotes
Acknowledgements
This work was supported by Natural Science Foundation of Jiangxi Province (20224BAB216048) and grants from the National Natural Science Foundation of China (No.: 82260223, 82301322).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Jiangxi Province, National Natural Science Foundation of China (grant number 20224BAB216048, 82260223, 82301322).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
