Abstract
Objective
To evaluate the killing effect of c-Met CAR-T on tumor cells with different degrees of c-Met expression. It was demonstrated that CAR-T autocrine PD-1 antibody could alleviate immune checkpoint inhibition and enhance the anti-tumor effect of T cells.
Methods
The specificity and clinical significance of c-Met and PD-L1 expression in various solid tumors were verified by bioinformatics analysis. c-Met specific CAR-T and c-Met specific CAR-T secreted by PD-L1 were synthesized, and c-Met CAR-T and c-Met/PD-1 CAR-T were prepared by constructing lentivirus. Flow cytometry was used to verify the positive rate and cell population of CAR-T, western blot was used to verify the secretion of PD-1 antibody, and cck-8 was used to detect the proliferation of CAR-T in tumor cells with different c-Met expression. LDH and ELISA further evaluated the antitumor effects of c-Met CAR-T and c-Met/PD-1 CAR-T in vitro.
Results
c-Met and PD-L1 were expressed in pancreatic cancer, ovarian cancer, esophageal cancer, bladder cancer, glioma and other tumors, and were associated with a variety of immune cell infiltration. Tumor cells with high expression of c-Met can strongly stimulate the proliferation of c-Met CAR-T, and c-Met CAR-T has strong cell lysis ability on tumor cells with high expression of c-Met. Autocrine PD-1 antibody can significantly improve the activity of c-Met CAR T cells, tumor lysis ability and cytokine secretion level.
Conclusion
The antitumor activity of c-Met CAR-T is positively correlated with the expression of c-Met. c-Met CAR-T secreted by PD-1 showed enhanced antitumor function in solid tumor treatment.
Introduction
About 10 million people die from malignant tumors every year in the world. The high morbidity and mortality of malignant tumors have become a major public health problem worldwide. 1 Traditional radiotherapy, chemotherapy, targeted therapy and other tumor treatment methods usually target tumor cells themselves. Although they have obvious effects in the early stage of tumor treatment, with the progression of disease and the prolongation of treatment time, problems such as tumor recurrence and drug resistance have gradually become prominent. 2 Therefore, the development of more effective anti-tumor drugs and new treatment methods is a problem that researchers pay close attention to. In recent years, chimeric antigen receptor T cell (CAR-T) therapy has attracted much attention in the field of tumor research and clinical treatment as an emerging immunotherapy method. 3 CAR-T cell immunotherapy is a cell adoptive therapy method that separates the peripheral blood T cells of the patient in vitro and transforms them by genetic engineering, and then reinfusion them into the patient ‘s body. 4 It gives T cells the ability to target and recognize tumor surface antigens, and is not restricted by MHC (major histocompatibility complex). 5 CD19-specific CAR-T for acute lymphoblastic leukemia has been approved for clinical use,6,7 but in the field of solid tumors, CAR-T research still needs to explore suitable tumor-associated antigens, 5 and overcome the tumor microenvironment and physical barriers.8–10 C-Met belongs to the tyrosine kinase receptor family and is mainly present in epithelial cells. 11 Hepatocyte growth factor (HGF) is a high affinity ligand of c-Met.c-Met dimerizes after binding to bioactive HGF, which promotes the recruitment of downstream signal effector molecules such as PI3K, PLC-γ1, STAT30 and GRB2.12,13 Under normal physiological conditions, HGF / c-Met plays a role in embryogenesis, tissue regeneration, wound healing, and nerve and muscle formation. Anti-c-Met monoclonal antibodies, such as Onartuzumab and Emibetuzumab, have been applied to tumors such as pancreatic cancer and esophageal cancer, and have shown good tolerance and clinical response rate.14,15 However, the interaction of c-Met with other carcinogenic molecules (such as EGFR and RON) can activate downstream pathways, thereby mediating tumor progression and drug resistance. 16 The efficacy of CAR-T-based therapy depends only on tumor-related targeting, not on the mechanism of tumor-associated antigens. In the previous study of our research group, it has been found that c-Met is a safe and effective target for CAR-T in the treatment of some solid tumors.17,18 At present, a number of studies have shown that c-Met-specific CAR-T can produce effective killing effects on malignant tumors such as liver cancer, 19 renal cancer 20 and nasopharyngeal carcinoma. 21 However, in the actual experimental operation, it is found that the killing process of the second-generation CAR-T on target cells is often accompanied by different degrees of effector cell depletion.
T cell exhaustion is a state of functional exhaustion in which T cells are continuously exposed to inflammation or antigen signals. It is characterized by the gradual loss of T cell effector function and self-renewal ability, high expression of multiple inhibitory receptors, metabolic changes, and changes in epigenetics and transcriptional profiles.22–25 Exhausted T cells have a unique molecular phenotype. Inhibitory immune molecules such as PD- (L) 1, CTLA-4 and TIM-3 can inhibit T cell immune response by up-regulating TIM.26–28 The PD-1 molecule on the surface of T cells can promote the apoptosis of T cells by binding to PD-L1 on the surface of tumor cells. At present, inhibitors that block the PD-L1 pathway have been widely used in clinical practice.29,30 However, it is not clear whether the c-Met-specific CAR-T secreting PD-1 antibody has a good anti-tumor effect on solid tumors, and whether the killing efficiency and proliferation activity are affected by the expression of c-Met and PD-1. Therefore, it is assumed that the blocking of PD-1/PD-L1 checkpoint can improve the therapeutic effect of CAR-T, and the tumor killing efficiency increases with the increase of c-Met expression.The activity of CAR-T cells increased with the increase of PD-1 expression. In this study, the fourth generation of PD-1 antibody secreted c-Met CAR-T was designed based on the previous second generation of c-Met CAR-T, and the expression of c-Met and PD-L1 in esophageal cancer, pancreatic cancer, bladder cancer and glioma cells was verified. In a variety of solid tumor cell models, we explored whether PD-1 antibody can be stably secreted by CAR-T and improve its anti-tumor activity. To determine whether the killing efficiency and cell proliferation activity of c-Met / PD-1 CAR-T on tumor cells are related to the expression of c-Met and PD-L1. This study will provide an experimental basis for the combined use of immune checkpoint blockade and CAR-T therapy for solid tumors.
Materials and Methods
Materials, Cell Lines and Culture
Aspc-1, EC9706, T24 and U87 cells were purchased from the cell bank of the Chinese Academy of Sciences, SKOV3 cells were retained by our research group, human lymphocyte separation solution was purchased from Soleibao Company, interleukin 2 was purchased from Beijing Tongli Haiyuan Biology, mouse anti-human CD3-APC, CD4-PE, CD8-Per / Pc5.5, c-Met-FITC and PD-L1-PE antibodies were purchased from Semmerfeld Company, RPMI1640 medium and fetal bovine serum were purchased from Gibco Company. Anti-CD3 / CD28 activation antibody and Retronectin concentrate were purchased from GK gene, and restriction endonuclease was purchased from Biyuntian company. CCK-8 reagent was purchased from BIOSHARP company, LDH release kit was purchased from Biyuntian company, and ELISA kit was purchased from Shanghai enzyme-linked biology. The cell line information are as follows: Aspc-1((ATCC CRL-1682, RRID: CVCL_0152); T24((ATCC HTB-4, RRID: CVCL_0554); SKOV3((ATCC HTB-77, RRID: CVCL_0532); U87((ATCC HTB-14, RRID: CVCL_0022); cell line EC9706 was obtained from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China).
Esophageal cancer, Ovarian cancer, Pancreatic cancer, Bladder cancer and Glioma samples were collected from the pathology department of the local cancer hospital. Aspc-1, EC9706, T24, beas-2b and U87 cells were purchased from the cell bank of the Chinese Academy of Sciences. SKOV3、A2780 cells were retained by our research group. SKOV-3 tumor cells were cultured in McCoy ‘s 5A culture medium (Sigma-Aldrich) with 10% fetal bovine serum (FBS) (GE Healthcare) ; Aspc-1, EC9706,A2780 and T24 tumor cells were cultured in 1640 (Gibco) supplemented with 10% fetal bovine serum. U87,beas-2b cells were cultured in DMEM (Gibco) supplemented with 10% fetal bovine serum. The retroviral synthesis services of c-Met CAR and PD-1 / c-Met CAR were provided by Jikai Gene.
Bioinformatics Analysis
Firstly, the c-Met mRNA expression of esophageal cancer, ovarian cancer, pancreatic cancer, bladder cancer and glioma and adjacent non-tumor tissues in TCGA database was compared. The expression of c-Met protein between tumor tissues and normal tissues was compared in GEPIA (Gene Expression Profiling Interactive Analysis) database (cancer-pku.cn). The online resource database Timer2.0 (HTTP : / / timer.cistrome.org /) was used to analyze the correlation between c-Met and PD-L1 expression. The correlation between c-Met expression and infiltration abundance of immune cells (B cells, CD8+T lymphocytes, CD4+T lymphocytes, macrophages, neutrophils and dendritic cells) was analyzed by genes in the immune association module. The effect of c-Met expression on the prognostic value of lung cancer was evaluated. Kaplan-meier survival curve was used to analyze the overall survival (OS) of c-Met in patients with solid tumors.
Immunohistochemical Staining
The specimens of esophageal cancer, ovarian cancer, pancreatic cancer, bladder cancer and glioma diagnosed by the pathology department of the local tumor hospital were used for c-Met staining in 40,30,40,50 and 40 cases, respectively, and for PD-L1 staining in 118,30,40 and 40 cases, respectively.Twenty adjacent tissues were taken as c-Met and PD-L1 staining control groups. All tissue samples were sliced, dried, dewaxed, hydrated and washed with deionized water. Paraffin sections were dehydrocarbonized in xylene and rehydrated with gradient ethanol concentration. Endogenous peroxidase activity was blocked by 3% H2O2 for 15 mins. Wake-up antigen retrieval was incubated with 2% Triton X-100, blocked and incubated with antibodies against c-Met or PD-L1 (servicebio, China, mouse antibody) for 30 mins, and then incubated at room temperature for 30 mins. The prepared goat anti-mouse Fab antibody (servicebio, China) was incubated with tissue sections. DAB chromogenic agent, hematoxylin staining, gradient ethanol dehydration after xylene transparent 2 times, sealing.Immunohistochemical staining
The stained samples were evaluated by two pathologists. In order to determine the expression status of c-Met and PD-L1, the samples were analyzed according to two groups of scoring methods : staining intensity (including complete basal and lateral membrane staining) and the percentage of tumor cells at different staining intensities, and the percentage of all tumor cells in the corresponding tissue samples. : 0 points, no staining ; 1 point, light yellow ; 2 points, brownish yellow ; 3 points, brown. The percentage of positive cells was 0% ∼ 10% for 0, 10% ∼ 25% for 1, 26% ∼ 50% for 2, 51% ∼ 75% for 3% and 76% ∼ 100% for 4. The final result was calculated by multiplying the number of positive cells by the staining intensity. 0 ∼ 1 is ‘ − ’, 2 ∼ 3 is ‘ ± ’ 4 ∼ 6 is ‘+’, 7 ∼ 9 is ‘ ++ ’, 10 ∼ 12 is ‘ +++ ’.
Peripheral Blood PBMC Extraction and CAR-T Preparation
10 ml of peripheral blood from healthy volunteers was diluted with the same amount of normal saline, and then slowly dripped into the upper layer of 1.2-fold volume lymphocyte separation solution, centrifuged at 500 g at room temperature for 30 min, and slowly increased and decreased. The white membrane layer of PBMC in the middle position obtained by centrifugation was collected, washed three times with PBS, and cultured in 1640 complete medium containing 10% fetal bovine serum containing IL-2. The cells were activated in CD3 / CD28 prediction plate for 48 h to obtain activated T cells.
The concentration of activated T cells was adjusted to 1 × 106/ml, and 150 μl was placed in a 48-well plate pre-coated with retronectin. Lentivirus was added according to the MOI value of 5, and polybrene, a lentivirus infection enhancer with a final volume of 0.04 times, was added. After 8 h of lentivirus infection, the equal volume of complete medium was supplemented. After 48 h, the infection was observed by fluorescence microscope. CAR-T cells were strictly counted and passaged, and the concentration was controlled at 0.5-1.5 cells / ml.
Lentivirus Construction
The antibody ScFv sequence targeting c-Met and the antibody ScFv sequence targeting PD-1 were obtained by patent search. The second generation c-Met CAR structure is composed of c-Met ScFv, CD8α hinge region and transmembrane region, 4-1BB costimulatory signal, CD3ζ chain intracellular signal domain. The fourth-generation c-Met / PD-1 CAR molecule was inserted into the secretory antibody ScFv sequence targeting PD-1 in the c-Met CAR structure. The second-generation c-Met CAR and the fourth-generation c-Met / PD-1 CAR molecules were inserted into the GV401 vector (Jikai gene) to complete the construction of the lentiviral vector.
The second-generation c-Met CAR and fourth-generation c-Met / PD-1 CAR lentiviral recombinant vectors were transformed and amplified in E.coli. The target plasmid was stably extracted from E.coli according to the instructions of plasmid extraction kit. The target plasmid was transfected into 293T cells with VSVG and PSPAX2 plasmids at a ratio of 4 : 3 : 2 by three-plasmid method, and Lipofectamin 2000 transfection reagent of 2.5 times plasmid volume was added. Three days after transfection, the lentivirus was concentrated by ultracentrifugation. 25 000 rpm, 2 h. The lentivirus packaged with non-target CD19 CAR molecule was used as a negative control. (This step is completed by Shanghai Jikai gene)
Detection of c-Met / PD-1 CAR-T Cells
Flow cytometry was used to detect the positive rate of CAR-T.Since the GV401 vector carried the eGFP marker gene, the flow cytometry FICT channel could be used to measure the CAR-T infection efficiency. The second-generation c-Met CAR-T and fourth-generation c-Met / PD-1 CAR-T were collected 2 × 105 each, and the negative control CD19 CAR-T and blank control activated T cells were collected 2 × 105 each. After PBS washing three times, flow cytometry was used to detect.
Western blot was used to detect the secretion of PD-1 antibody in the fourth generation of c-Met / PD-1 CAR-T.Since the secreted PD-1 antibody carries the His tag and forms a fusion protein with it, the secretion of PD-1 antibody can be verified by detecting the His tag in the cell supernatant.T cell supernatant cultured with commercial recombinant His-tag PD-1 ScFv antibody was added as positive control. The cell supernatant after 24 h of culture was centrifuged to remove the precipitate, and the protein was separated by PAGE gel electrophoresis. The target molecular weight protein was transferred to the PVDF membrane, blocked with 5% skimmed milk powder for 1 h, and incubated with mouse anti-human His primary antibody overnight at 4 °C. After washing, rabbit anti-mouse HRP coupled secondary antibody was incubated for 2 h, and the color solution was added dropwise and visualized by ECL exposure.
The distribution of CAR-T cell subsets was detected by flow cytometry. The second-generation c-Met CAR-T and the fourth-generation c-Met / PD-1 CAR-T were collected, 2 × 105 each. The negative control CD19 CAR-T and the blank control activated T cells were collected, 2 × 105 each. After washing with PBS, 5 μL of APC-CD3, PE-CD4 and Pre-PC5.5-CD8 antibodies were added to each group. At the same time, the activated T cells were used to set up three kinds of antibody single positive tubes to adjust the voltage, and incubated at 4 °C in dark for 30 min. Flow cytometry was used for detection. Analyze all data using Flowjo V12
Detection of CAR-T Cell Proliferation in Vitro
In the cytotoxicity experiment, five target cells were identified by flow cytometry results. Esophageal cancer cells EC9706 and pancreatic cancer cells Aspc-1 with high expression of c-Met and PDL1, The bladder cancer cells show moderate expression of c-Met and high-level expression of PD-L1., and glioma cells U87 with low expression of c-Met and PD-L1. Screen out tumor cells A2780 that do not express c-Met for cell function experiments to verify the specificity of CAR-T. Meanwhile, use bronchial epithelial cells beas-2b for cell function experiments to verify whether CAR-T causes damage to normal tissue cells. 1 × 104 tumor cells Aspc-1, EC9706, T24 and U251 with high expression of c-Met and PD-L1 were co-cultured with 1 × 104 effector cells for 24 h, 48 h and 72 h, and the final volume was 200 μl. After the culture, the suspended T cells and non-adherent cancer cell debris were washed away, and the medium was supplemented to a final volume of 200 μl. At the same time, the same number of co-culture unwashed holes were set up. All holes were added with 0.1 times the volume of CCK-8 reagent, and incubated at 37 °C for 2 h in the dark. The OD value of each measuring hole was detected by microplate reader, and the initial (0 h) OD value of each effector cell was detected. The number of cells at each time point in each group = 1 × 104 × (unwashed hole OD value-washed hole OD value) / 0 h effector cell OD value.
Detection of Anti-tumor Effect of CAR-T Cells in Vitro
The cytotoxicity of CAR-T cells was detected by lactate dehydrogenase release kit. The effector cells and tumor cells in each group were incubated in 2% horse serum 1640 medium for 6 h, and the final volume was 200 μl. The number of tumors was constant at 1 × 104, and effector cells were added to each group according to the gradient effect-target ratio (1 : 1, 5 : 1, 10 : 1 and 20 : 1) (experimental group 1 : c-Met CAR-T ; experimental group 2 : PD-1 / c-Met CAR-T ; control group 1 : CD19 CAR-T ; control group 2 : activated T cell), set up target cells, effector cells natural release holes, target cells maximum release holes and blank holes respectively, and 20 μl LDH release reagent was added to the maximum release hole of target cells 1 h before the end of co-culture. The detection solution was configured according to the instructions. The 120 μl cell supernatant and 60 μl detection solution were mixed in a new 96-well plate, incubated at room temperature for 30 min, and the OD value of each well was detected by a microplate reader.The formula is as follows : killing efficiency % = (OD value of co-culture hole-OD value of natural release hole of effector cells-OD value of natural release hole of target cells + OD value of blank hole) / (OD value of maximum release hole of target cells-OD value of natural release hole of effector cells).
The amount of cytokines IL-2, TNF-α and IFN-γ released by effector cells in the presence of target cells was detected by enzyme-linked immunosorbent assay. Each group was added with 1 × 106 effector cells and 1 × 106 tumor cells in a 96-well plate, respectively, and co-cultured for 24 h. The culture system was 5% fetal bovine serum + 95% 1640 medium, and the final volume was 200 μL. The supernatant of the cells was centrifuged and diluted 5 times, and the concentrations of IL-2, TNF-α and IFN-γ were detected according to the ELISA kit instructions.
Statistical Analysis of Data
The measurement data were expressed as mean ± standard deviation (x ± s), and the count data were expressed as percentage (%). SPSS 23.0 software was used for analysis. One-way analysis of variance was used for statistical analysis.
Results
Bioinformatics Analysis
The expression levels of c-Met protein in esophageal cancer, pancreatic cancer, ovarian cancer, bladder cancer and glioma tissues were analyzed by GEPIA database. The expression of c-Met protein in esophageal cancer, pancreatic cancer and ovarian cancer tissues was significantly higher than that in adjacent normal tissues (Figure 1A, P < .01).

Expression and Clinical Significance of c-Met Gene in a Variety of Solid Tumors Based on Bioinformatics Analysis. A: The Expression Level of c-Met Protein in Tumer Tissues and Adjacent Normal Tissues was Analyzed by GEPIA Database. * P < .01 Versus Normal Tissues. B:Kaplan-Meier Survival Curve of Human Cancers with High and Low c-Met and PD-L1 Expression Analyzed by the Timer2.0 Database; C: The Relationship Between c-Met Expression and PD-L1 Expression in Solid Tumor; D: Analysis of the Correlation Between c-Met Expression and Infiltration of B Cells, CD4+T Cells, CD8+ T Cells, Dendritic Cells, NK Cells and Macrophages in Solid Tumor Tissue; E: Analysis of the Correlation Between PD-L1 Expression and Infiltration of B Cells, CD4+T Cells, CD8+T Cells, Dendritic Cells, NK cells and Macrophages in Solid Tumor Tissue.
TIMER database was used to evaluate the relationship between c-Met protein expression level and PD-L1 expression. The expression of PD-L1 in bladder cancer (R = 0.46, P < .01), glioma (R = 0.296, P < .01) and pancreatic cancer (R = 0.397, P < .01) was positively correlated with the expression of c-Met (Figure 1B). The statistical results showed that the high expression level of c-Met in ovarian cancer, pancreatic cancer and glioma was significantly correlated with the survival time of patients (Figure 1C, P < .05). The high expression level of PD-L1 in bladder cancer, pancreatic cancer, glioma, ovarian cancer and esophageal cancer was not significantly correlated with the survival time of patients (Figure 1C, P > .01).
The correlation between c-Met and the degree of immune infiltration was analyzed based on the TIMER database. The results showed that the expression of c-MET in bladder cancer, pancreatic cancer and glioma was related to the infiltration of some immune cells. In pancreatic cancer, the correlation between c-Met mRNA and immune cell infiltration was CD4+T cells (R = 0.259, P < .01), CD8+T cells (r = 0.35, P < .01), macrophages (r = 0.142, P < .01), dendritic cells (r = 0.261, P < .01) and neutrophils (r = 0.21, P < .01). The correlation between c-Met mRNA and immune cell infiltration in bladder cancer was CD4+T cells (R = 0.142, P < .01), CD8+T cells (r = 0.322, P < .01), macrophages (r = 0.171, P < .01), dendritic cells (r = 0.383, P < .01) and neutrophils (r = 0.359, P < .01). The correlation between c-Met mRNA and immune cell infiltration in glioma was as follows : CD8+T cells (r = 0.163, P < .01), B cells (r = −0.219, P < .01), and neutrophils (r = 0.358, P < .01) (Figure 1C). The correlation between PD-L1 and the degree of immune infiltration was analyzed based on the TIMER database. The results showed that the expression of PD-L1 in bladder cancer, pancreatic cancer, glioma, esophageal cancer and ovarian cancer was related to the infiltration of some immune cells. The correlation between c-Met mRNA and immune cell infiltration in bladder cancer was CD4+T cells (R = 0.211, P < .01), CD8+T cells (r = 0.422, P < .01), macrophages (r = 0.171, P < .01), dendritic cells (r = 0.622, P < .01) and neutrophils (r = 0.643, P < .01). The correlation between c-Met mRNA and immune cell infiltration in esophageal cancer was dendritic cells (r = 0.259, P < .01) and neutrophils (r = 0.369, P < .01). The correlation between c-Met mRNA and immune cell infiltration in glioma was macrophages (r = 0.256, P < .01) and neutrophils (r = 0.242, P < .01).The correlation between c-Met mRNA and immune cell infiltration in ovarian cancer was CD4+T cells (R = 0.129, P < .01), CD8+T cells (r = 0.307, P < .01), dendritic cells (r = 0.374, P < .01) and neutrophils (r = 0.365, P < .01). The correlation between c-Met mRNA and immune cell infiltration in pancreatic cancer was B cells (R = 0.282, P < .01), CD8+T cells (r = 0.551, P < .01), macrophages (r = 0.171, P < .01), dendritic cells (r = 0.383, P < .01) and neutrophils (r = 0.359, P < .01). The statistical results showed that the high expression level of c-Met in ovarian cancer, pancreatic cancer and glioma was significantly correlated with the survival time of patients (Figure 1D, P < .05). The high expression level of PD-L1 in bladder cancer, pancreatic cancer, glioma, ovarian cancer and esophageal cancer was not significantly correlated with the survival time of patients (Figure 1E, P > .01).
Immunohistochemical detection was performed with anti-c-Met antibody. The c-Met protein was found in the cytoplasm and membrane of all tumor cells, while the expression of adjacent lung tumor tissues was less (Figure 2). Immunohistochemical assay in 40 cases of esophageal cancer specimens in pathological c-Met expression in 0 cases of negative expression ; in 40 cases of ovarian cancer specimens, the pathological c-Met expression score was positive in 22 cases and negative in 4 cases. According to the pathological PD-L1 score in 30 specimens, 25 cases were positive and 0 cases were negative. In 40 cases of pancreatic cancer specimens, the pathological c-Met protein was positively expressed in 25 cases and negatively expressed in 2 cases, and the pathological PD-L1 protein was positively expressed in 40 cases and negatively expressed in 0 cases. The expression of c-Met protein in 50 cases of bladder cancer was positive in 50 cases and negative in 0 cases. PD-L1 protein was positively expressed in 40 cases and negatively expressed in 0 cases. In 40 cases of glioma specimens, c-Met protein was positively expressed in 35 cases and negatively expressed in 2 cases. PD-L1 protein was positively expressed in 30 cases and negatively expressed in 0 cases (Table 1).

The Expression of c-Met and PD-L1 Protein in Pancreatic Cancer Tissue Samples was Detected by Immunohistochemistry. A: c-Met Immunohistochemical Staining of Clinical Specimens of Solid Tumors and their Clinical Specimens(×400); B: PD-L1 Immunohistochemical Staining of Clinical Specimens of Solid Tumors and their Clinical Specimens(×400).
c-Met and PD-L1 Expressionand in a Variety of Solid Tumors and Their Adjacent Tissues.

In order to clarify the expression of c-Met and PD-L1 on the cell surface of esophageal cancer, ovarian cancer, pancreatic cancer, bladder cancer and glioma cell lines. EC9706, SKOV3, ASPC-1, T24, U87, A2780, beas-2b cells were detected by flow cytometry. Each group of experiments was set up with 3 technique replicates, and their average values were taken as the final data of a single experiment. The data in the statistical chart represented three independent experiments (Figure 3). All tested Esophageal cancer cell line EC9706, Ovarian cancer cell line SKOV3, and Pancreatic cancer cell line Aspc-1 expressed high levels of c-Met.The expression level of c-Met in bladder cancer cell line T24 was medium, and the expression level of c-Met in glioma cell line U87 was low. Except for glioblastoma cells which have relatively low PD-L1 expression, the expression of PD-L1 in tumor cells of the other treatment groups is relatively high. The selected Non-target ovarian cancer cells A2780 do not express c-Met, and PD-L1 is expressed at a medium level in these non-target cells. The expression level of c-Met in the normal tissue cells, namely bronchial epithelial cells beas-2b, is relatively low, while PD-L1 is not expressed. Consistent with bioinformatics analysis and immunohistochemical results.

Flow Cytometry was used to Detect the Expression of c-Met and PD-L1 Protein on Aspc-1 Surface.
Preparation of Second-generation c-Met CAR-T and Fourth-generation c-Met / PD-1 CAR-T
In this study, a second-generation anti-c-Met CAR containing anti-human c-Met antibody, hinge and transmembrane domains, intracellular 4-1BB costimulatory domain and CD3ζ activation domain was constructed. In this study, we used the FP2A element as a linker between the c-Met CAR and the secretory sequence, and then used the His tag to prepare the c-Met CAR for secreting anti-pd1 scFv. All the target sequences were combined with the EGFP fluorescent reporter gene to evaluate the transduction efficiency (Figure 4A), and the plasmid was successfully constructed by enzyme digestion (Figure 4B). The lentivirus was coated and concentrated by three-plasmid method and high-speed centrifugation. The second-generation c-Met CAR-T, fourth-generation c-Met / PD-1 CAR-T and negative control CD19 CAR-T were successfully prepared after lentivirus infection of activated T cells. The EGFP-positive rate of each CAR-T cell was detected by flow cytometry. The results showed that the proportion of EGFP-positive expressed c-Met / PD-1 CAR-T was 30%, the EGFP-positive rate of the second generation c-Met CAR-T was 54.8%, and the EGFP-positive rate of CD19 CAR-T was 50.6% (Figure 4C).
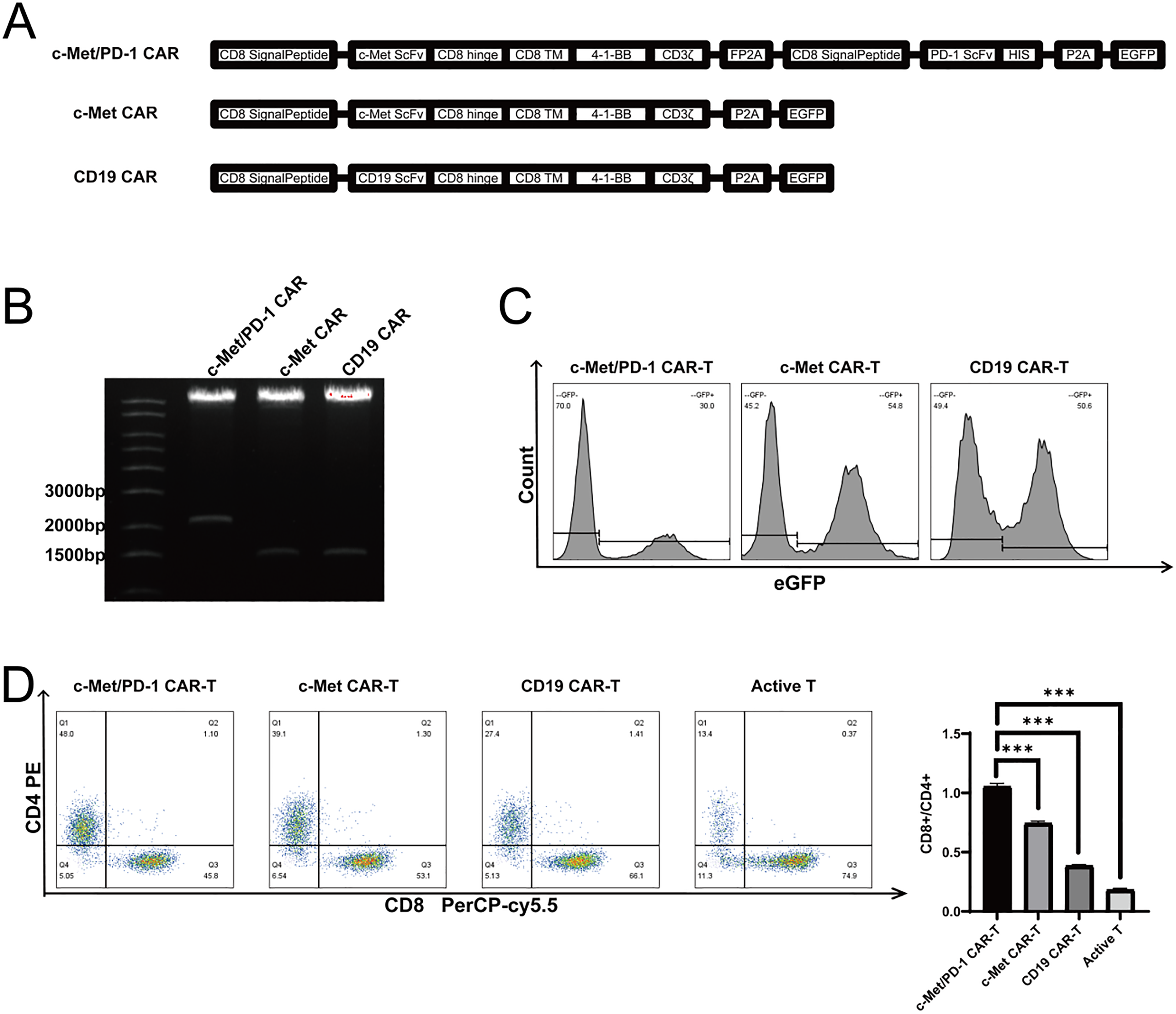
CAR-T Design, Preparation and Detection. A : CAR Core Structure Diagram ; B : BamHI Electrophoresis Exposure Map of CAR Plasmid ; C : CAR-T Transfection Positive Rate Histogram ; D : CAR-T Cell Subsets Distribution Map * * * P < 0.001 Versus PD-1/c-Met CAR-T.
Western blot was used to detect the PD-1 antibody secreted by PD-1 / c-Met CAR-T into the supernatant. The PD-1 antibody molecule carried the His tag. By detecting the His tagged protein in the supernatant, PD-1 / c-Met CAR-T could successfully secrete PD-1 antibody, but there was no PD-1 antibody in the supernatant of activated T cells, CD19 CAR-T and c-Met CAR-T cells. Flow cytometry was used to evaluate the changes of T cell subsets before and after lentivirus infection. Before lentivirus infection, T cell CD4+ subsets accounted for 13.4% and CD8+ subsets accounted for 74.9%. After lentivirus infection, CD19 c-Met CAR-T CD4+ subsets accounted for 27.4% and CD8+ subsets accounted for 66.1%. The proportion of c-Met CAR-T CD4+ subsets was 39.1%, and the proportion of CD8+ subsets was 53.1%. PD-1 / c-Met CAR-T CD4+ subsets accounted for 48.0%, and CD8+ subsets accounted for 45.8%. After lentivirus infection, CD8+ T cell subsets decreased and CD4+ T cell subsets increased (Figure 4D).
c-Met and PD-L1 Expression in Solid Tumor Cell Lines
Western blot results showed that His-labeled PD-1 antibody was present in the supernatant of PD-1/c-Met CAR-T and supernatant of T cells cultured with His labeled PD-1 ScFv positive control antibody, the molecular weight was in line with expectations (Figure 5), and c-Met CAR-T, CD19 CAR-T and activated T cell supernatant were not detected.

His-Labeled PD-1 Scfv Immunoblotting in the Cell Supernatant.
PD-1 Secreted Antibody can Effectively Improve the Proliferation Efficiency of CAR-T
The results of CCK-8 proliferation assay showed that the number of c-Met CAR-T cells was much higher than that of activated T cells and CD19 CAR-T at 24 h, 48 h and 72 h under the stimulation of esophageal cancer and pancreatic cancer. The number of c-Met CAR-T cells was much higher than that of activated T cells and CD19 CAR-T at 48 h and 72 h under the stimulation of ovarian cancer and bladder cancer. When stimulating glioma cells and Non target cell A780, there was no significant difference in the number of c-Met CAR-T cells compared with activated T cells and CD19 CAR-T cells within 72 h. However, after stimulating various cancer cells, the number of PD-1/c-Met CAR-T cells was higher than that of c-Met CAR-T cells within 72 h. After co-culturing bronchial epithelial cells with various effector cells for 72 h, there was no significant difference in the proliferation number of each effector cell.bHowever, this result was basically consistent with the expression of c-Met on the surface of target cells .Each group of experiments was set up with 3 technique replicates, and their average values were taken as the final data of a single experiment. The data in the statistical chart represented three independent experiments (Figure 6).

Proliferative Activity of Bi-Specific c-Met CAR-T and c-Met/PD-1 CAR-T Against Solid Tumor Cells by CCK-8.
In CAR-T therapy, the efficiency of CAR-T cells to lyse tumor cells and the ability to secrete cytokines are important basis for evaluating the efficacy of CAR-T therapy. The lytic efficiency of c-Met CAR-T and PD-1 / c-Met CAR-T on EC9706, SKOV3, Aspc-1, T24 and U87 cells was detected at different effector-target ratios. Our results showed that c-Met CAR-T effectively lyses tumor cells compared with activated T cells and CD19 CAR-T, and is related to the expression of c-Met.c-Met CAR-T cells showed weak cytotoxicity to glioma cells with low expression of c-Met. When the ratio of effector cells to ovarian cancer, pancreatic cancer and glioma was 10 : 1 and 20 : 1, the lysis efficiency of PD-1 / c-Met CAR-T tumor cells was higher than that of c-Met CAR-T when the ratio of effector cells to esophageal cancer and bladder cancer was 20 : 1. Under different effector-target ratios, there were no significant differences in the lysing efficiency of each effector cell on N0N target cells A2780 and bronchial epithelial cells beas-2b. Each group of experiments was set up with 3 technique replicates, and their average values were taken as the final data of a single experiment. The data in the statistical chart represented three independent experiments (Figure 7).
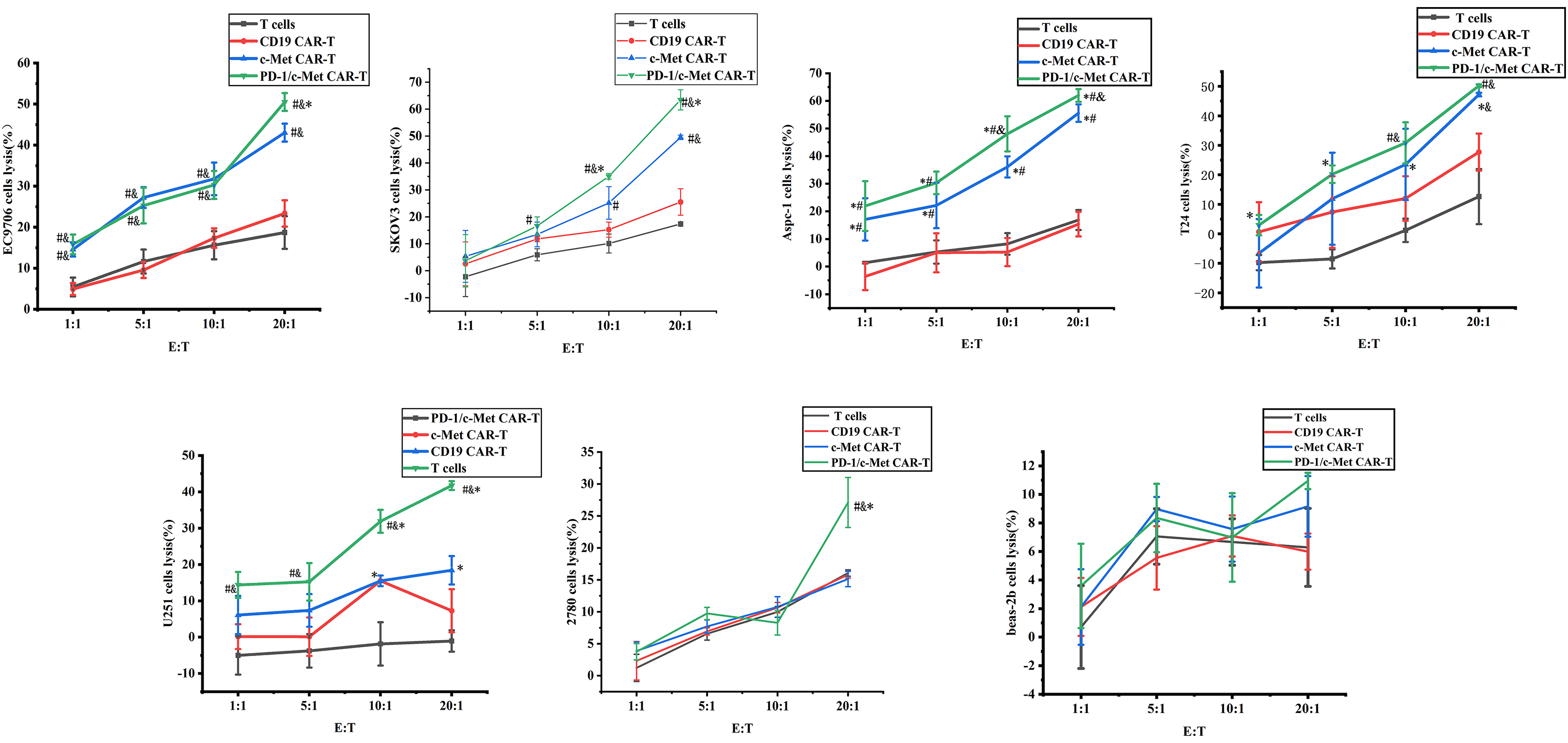
Cytotoxicity of c-Met CAR-T, c-Met/PD-1CAR-T and Control Groups on Solid Tumor Cells.
The level of cytokines secreted by CAR-T cells targeting cancer cells further indicates the activation and specific cytotoxicity of T cells. We will detect the content of IL-2, TNF-α and IFN-γ in the supernatant of tumor cell CAR-T co-culture for 24 h. Compared with activated T cells and CD19 CAR-T, the amount of cytokines secreted by c-Met CAR-T and PD-1 / c-Met CAR-T increased after co-culture of tumor cells. The vast majority of PD-1 / c-Met CAR-T secreted higher levels of cytokines than c-Met CAR-T.After co-culturing with Non target cell A2780 for 24 h, there was no significant difference in the amount of cytokines secreted by activated T cells, CD19 CAR-T and c-Met CAR-T. The amount of TNF-α secreted by PD-1/c-Met CAR-T was higher than that of the other effector cells. There was no significant difference in the amount of cytokines produced by each effector cell when co-cultured with bronchial epithelial cells. Each group of experiments was set up with 3 technique replicates, and their average values were taken as the final data of a single experiment. The data in the statistical chart represented three independent experiments (Figure 8).

Specific Cytokine Released by Bi-Specific c-Met CAR-T and c-Met/PD-1CAR-T Against Solid Tumor Cells.
Discussion
Immunotherapy is booming in the field of cancer treatment, in which CAR-T therapy and immune checkpoint inhibitors are two hot spots. 31 However, how to make CAR-T cells avoid the immune escape of solid tumors and better play the role of monoclonal antibodies in tumor therapy is an urgent problem to be solved. Therefore, we combined CAR-T therapy with immune checkpoint inhibitor therapy to construct PD-1 / c-Met CAR-T, which showed good anti-tumor activity in vitro and in vivo experiments. This study provides a new idea for immunotherapy of solid tumors and has a good clinical application prospect.
In this study, we aimed to enhance the therapeutic effect of CAR-T therapy on solid tumors by reshaping the tumor microenvironment. In the previous study, our group has proved that CAR-T targeting c-Met can be used for the treatment of solid tumors such as lung cancer and colon cancer, and has achieved good results. However, the relationship between the anti-tumor effect of c-Met-specific CAR-T and the expression of c-Met in tumor cells has not been studied. At the same time, it was found that CAR-T showed obvious apoptosis after a short period of 24-48 h high-speed proliferation under the stimulation of target cells. Through literature search and flow cytometry detection, it is speculated that immune checkpoint inhibition may lead to T cell apoptosis.
c-Met is a receptor tyrosine kinase that, upon binding to ligand hepatocyte growth factor (HGF), activates downstream signaling pathways (such as PI3K/AKT, MAPK), promoting tumor cell proliferation, migration, invasion, and angiogenesis. Studies have shown that abnormal activation of c-Met not only directly drives tumor progression but also promotes immune escape by upregulating immunosuppressive factors in the tumor microenvironment (such as PD-L1) [1]. For instance, activation of the c-Met signaling pathway may induce the expression of PD-L1 on the surface of tumor cells, thereby inhibiting T-cell activity and forming an “immune cold tumor” environment. Clinical data indicate that gliomas with high c-Met expression often accompany overexpression of PD-L1. Pearson correlation test shows a strong correlation between MET, PD-L1, and STAT pathways. This suggests that there may be a synergistic effect between c-Met and PD-1 in promoting cancer progression and immune escape. Studies have shown that c-Met is highly upregulated in pancreatic cancer [2] and esophageal cancer tissues [3], and is positively correlated with PD-L1 levels. Meanwhile, research has demonstrated that in lung adenocarcinoma, c-MET inhibits p-GSK3β, leading to the stabilization of PD-L1. In summary, a large number of research results indicate that there is a potential crosstalk between the abnormal expression of c-MET and the immune checkpoint inhibition of the PD-1/PD-L1 pathway, supporting the broad applicability of c-Met and PD-1 combined targeted therapy for solid tumors, and that combined targeted therapy has improved the immunotherapy efficacy of solid tumors. Currently, a small number of studies in the CAR-T treatment field have combined c-Met CAR-T with blocking the PD-1/PD-L1 pathway to verify its anti-tumor effectiveness in some solid tumors. However, the reported CAR structures are mostly c-MetScFv and PD-1 or PD-L1 ScFv linked as a dual-targeted CAR or PD1/CD28 chimeric switch receptor, which enhances the anti-tumor ability of c-Met CAR-T in solid tumors, but is still limited by the spatial conformation of CAR and is unfavorable for the distribution of PD-1 antibodies due to the diffusion of CAR-T. At present, PD-1 / PD-L1 checkpoint blockade combined CAR-T forms include co-expression of chimeric switch receptors 32 or knockout of PD-1 molecules in CAR-T cell genome. 33 Compared with T cells expressing CAR alone, the above methods have been shown to improve anti-tumor efficiency, cytokine secretion levels, and enhance CAR-T proliferation. However, this protective effect is limited to CAR-T cells themselves.However, the remaining lymphocytes in the tumor microenvironment still have PD-1/PD-L1-mediated inhibition ; in addition, the extracellular domain of PD-1 Scfv or PD-L1 Scfv in tandem with the target antigen Scfv, 33 but the spatial conformation is difficult to meet the simultaneous binding of immune cells (PD-1) and tumor cells (tumor-associated antigens), or simultaneous tandem of two tumor cells (PD-L1 and tumor-associated antigens, respectively). Given that PD-1 blocking scFvs secreted by CAR-T cells can bind to other immune cells except CAR-T, the local secretion of PD-1 Scfv may protect endogenous anti-tumor immune cells and inhibit tumor growth expressing target antigens. At present, PD-1 secreted antibodies are only used to enhance EGFR, 34 GPC3 35 and TREM2 36 specific CAR-T, and there is no report on the anti-tumor effect of c-Met CAR-T combined with PD-1 secreted antibodies.We inserted PD-1 secreted ScFv into c-Met CAR molecule and secreted it with the expression of c-Met CAR molecule. PD-1 scFv antibody entered TME and could bind to PD-1 antigen on T cell surface and block PD-1 / PD-L1 pathway. Different from the traditional PD-1 scFv directly expressed on the surface of CAR-T and PD-1 knockout, CAR-T cells expressed on the surface of PD-1 antibody must combine CAR-T with T cells and CAR-T with cancer cells to play a role. The efficiency of secondary lentivirus infection and the survival state of T cells after repeated infection should be considered when directly knocking out or down-regulating the PD-1 gene of CAR-T cells. The advantage of using secreted PD-1 scFv is that CAR-T cells play a role after recognizing c-Met, and the role is almost limited to the CAR-T cells themselves recruited near the tumor, which can avoid the occurrence of systemic inflammatory response to a large extent. This is also a new method to introduce PD-1 blockade into CAR-T therapy.
In this study, we obtained c-Met CAR-T cells capable of secreting PD-1 antibodies through lentivirus packaging and T cell transduction. PD-1 and c-Met were verified to express c-Met at different levels in the above five cancers by bioinformatics and clinical sample pathological detection, and both expressed PD-L1 at a high level, which was suitable as a cell model for studying the relationship between c-Met CAR-T and tumor c-Met expression and the effect of PD-1 on the anti-tumor effect of c-Met CAR-T. In this study, we used flow cytometry to detect the medium and high expression of c-cells on the surface of different types of tumors, and found that c-Met CAR-T cells could efficiently activate and kill tumor cells. In vitro functional verification experiments, the killing activity of PD-1/c-Met CAR-T was significantly higher than that of c-Met CAR-T at different effector-target ratios, and the IL-2 and TNF-α secreted by PD-1/c-Met CAR-T were significantly higher than those of c-Met CAR-T cells.At the same time, in the presence of target cells, PD-1/c-Met CAR-T cells have a higher proliferation rate than c-Met CAR-T cells. By setting up Non-target cell A2780 with medium expression of PD-1 and no expression of c-Met as the Non-target group, it was proved that c-Met/PD-1 CAR-T might not have a significant immediate killing effect on tumors that do not express c-Met. However, since PD-L1 is widely expressed in almost all solid tissues, or it can reduce the immune checkpoint inhibition of T cells by blocking the PD-1/PD-L1 pathway, it can reduce the exhaustion of T cells to a certain extent and thereby assist in the treatment of tumors. This study verified the safety of c-Met/PD-1 CAR-T in normal tissue cells using bronchial epithelial cells with low expression of c-Met.
The limitations of this study lie in that it mainly relies on in vitro cell models to evaluate the efficacy of CAR-T cells, without conducting functional validation in animal models. Although these preclinical methods provide valuable insights into tumor-specific targeting and cytotoxicity, the lack of in vivo data limits our ability to comprehensively assess key parameters, such as the migration, persistence, and dynamic interactions of CAR-T cells in the immunosuppressive tumor microenvironment (TME). Animal models, especially syngeneic models with intact immune function or humanized mouse systems, are crucial for evaluating the time- and context-dependent regulation, targeting/desensitizing toxicity, and systemic immune response of CAR-T cells. We stress that these limitations do not negate the mechanistic findings in this study. Instead, they highlight the necessity of conducting multi-level validations in the subsequent preclinical stage before proceeding to clinical translation. Currently, we are designing a comprehensive animal study that integrates bioluminescence imaging, single-cell transcriptomics, and histopathological analysis to address these deficiencies.
The positive rate of PD-1/c-Met CAR-T prepared by lentivirus method is less than that of c-Met CAR-T. In order to ensure the effect of positive rate on anti-tumor cell experiments, c-Met CAR-T positive rate was diluted to PD-1/c-Met CAR-T level by activated somatic cells in this experiment. Due to the low overall load of the lentiviral vector, the insertion fragment of more than 5 kb may cause a decrease in expression and viral titer after infection. In order to solve the technical problems of lentiviral load, CRISPR/CAS9 or transposon technology can be used to improve the later stage. The advantages of these two methods are not only to carry longer and more complex target genes, but also to insert CAR molecules at fixed points.For example, CRISPR/CAS9 can insert CAR at the TRAC site to construct CAR-T while removing the main recognition receptor of graft-versus-host disease, so as to achieve the purpose of constructing universal CAR-T, and further solve the problem that CAR-T therapy can only use autologous T cells. However, both CRISPR / CAS9 and transposon gene editing techniques need to use electroporation to construct CAR-T. Electroporation is more destructive to cells than virus method. How to improve the survival efficiency of T cells after electroporation may become another research hotspot.
Conclusion
This study demonstrated that PD-1 secretory CAR-T cells significantly enhanced the in vitro cytotoxic activity of c-Met CAR-T against various solid tumors by blocking the immunosuppressive signals in the tumor microenvironment through autocrine PD-1 antibodies. Experimental data indicated that the anti-tumor effect was positively correlated with the expression level of c-MET on tumor cells, suggesting that c-MET could serve as a potential biomarker for predicting the efficacy of this therapy. Additionally, no significant non-specific activation or toxicity was observed in normal tissues with PD-1/c-Met CAR-T, verifying its targeted safety. This finding provides a new theoretical basis for optimizing the combined CAR-T and immune checkpoint inhibition strategy for solid tumors. Future studies should further explore the in-depth experiments of PD-1 and c-Met as co-targets of CAR-T in animal or organoid models and promote the validation of this therapy in clinical translation.
Footnotes
Abbreviations
Acknowledgements
We thank Professor Ligao Wu for assisting in the immunohistochemical experiments.
ORCID iDs
Ethical Considerations
Studies involving human subjects are reviewed and approved by the Bengbu Medical University Ethics Committee [2022] No. 115.
Informed Consent
Written informed consent is not required for this study in accordance with national legislation and institutional requirements.
Consent for Publication
All authors have agreed to publish this manuscript.
Author Contributions/CRediT
Yang HR contributed to the conception and design, collection and/or assembly of data, data analysis and interpretation and manuscript writing. Zhang YJ,li YQ, Peng S, and Du NN contributed collection and/or assembly of data and cell culture . Min JT, Li ZH and Chu F contributed to the conception and design and provided fnancial support. An R and Cao JW contributed to the conception and design. All authors read and approved the fnal manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (No.81472656); Natural Science Research Project of Anhui Universities (No. 2023AH051991, 2022AH040224) ; National college students’ innovative entrepreneurial training program (202310367011).
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All data generated or analyzed during this study are included in this manuscript.
