Abstract
There are no standard third-line or beyond treatments for patients with driver mutation-positive advanced lung adenocarcinoma (LUAD). Anlotinib was approved as a third-line multitarget drug in China in 2018. Limited data are available regarding the efficacy and safety of anlotinib compared with chemotherapy. To investigate the efficacy and safety of anlotinib compared with traditional chemotherapy in patients with epidermal growth factor receptor (EGFR)-positive advanced LUAD. We conducted a retrospective study of 83 EGFR mutation-positive patients with advanced LUAD between 2011 and 2022. Progression-free survival (PFS) and overall survival (OS) were the primary endpoints, whereas the objective response rate (ORR) and disease control rate (DCR) were the secondary endpoints. Anlotinib-related adverse events (AEs) were recorded to evaluate the safety of anlotinib. 39 patients with LUAD received anlotinib and 44 patients with LUAD received chemotherapy were enrolled in the study. Patients treated with anlotinib exhibited longer PFS (11.2 vs 4.5 months, P < .01) and OS (18.8 vs 15.8 months, P < .05) than patients treated with chemotherapy. There were no significant differences in ORR (7.9% vs 20.5%, P = .129) or DCR (100% vs 93.2%, P = .120) between the two groups. Anlotinib-related AEs grading 3-4 level were observed in 2 (5.1%) patients, no anlotinib-related death was recorded. Cox regression analyses of PFS and OS showed that brain metastases and age < 30 years at diagnosis had negative effects on clinical outcomes. Anlotinib is effective and safe in patients with EGFR-positive advanced LUAD. Patients without brain metastases had better clinical outcomes.
Introduction
In 2020, lung cancer was the second most commonly diagnosed cancer and the leading cause of cancer death in the world. 1 Lung adenocarcinoma (LUAD) was the most common histological type of non-small cell lung cancer (NSCLC). 2 Non-small cell lung cancer is commonly diagnosed with distant metastases, which leads to poor survival. 3 Epidermal growth factor receptor (EGFR) mutations are among the most common driver gene mutations of NSCLC 2 and are more frequent in females and never-smokers.4,5 Tyrosine kinase inhibitors (TKIs) are the mainstream treatment agents used in patients with NSCLC harboring EGFR mutations. 6 Unfortunately, almost all patients acquire resistance to TKIs, primary or acquired.7,8 Extensive efforts have been made to elucidate the mechanisms underlying TKI resistance; however, our understanding remains limited. The exon 20 T790M mutation in EGFR is a well-described associated mechanism. 7 Several third-generation TKIs, such as osimertinib, have been designed to target the T790M mutation. 8 However, resistance to third-generation TKIs may occur, and platinum-based dual-drug chemotherapy is generally selected as a subsequent treatment.9,10
Anlotinib is a novel TKI targeting the vascular endothelial growth factor receptor, fibroblast growth factor, platelet-derived growth factor receptor, c-Kit, and Ret, 11 and it has been approved as a third-line treatment for advanced NSCLC in China since 2018. Phase II (ALTER0302) and phase III (ALTER0303) clinical trials have shown the efficacy of anlotinib, in comparison with placebo, in patients with advanced NSCLC who had received targeted drugs and/or at least two lines of systemic chemotherapy.12,13 The efficacy and safety of anlotinib in patients with refractory NSCLC have been demonstrated in retrospective and meta-analysis studies.14–21 However, associated real-world studies are limited. Because of the placebo used in the control group in clinical trials and the descriptive nature of most retrospective studies, the advantages of the clinical outcomes of patients receiving anlotinib compared with those of patients receiving chemotherapy are unclear.
Our study was designed to investigate the efficacy and safety of anlotinib compared with traditional chemotherapy in patients with EGFR-positive advanced LUAD.
Materials and Methods
Patients
We conducted a retrospective study of 83 EGFR-positive patients with advanced LUAD who were treated with anlotinib or chemotherapy at the Hospital, between May 2011 and July 2022. This study was approved by the Clinical Research Ethics Committee of the First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou (Number: 2023-0467) on June 14, 2023. The reporting of this study conforms to STROBE guidelines. 22 All the patient details had been de-identified. The eligibility criteria were as follows: a) histologically or cytologically confirmed LUAD grading IIIC, IVA, or IVB; b) EGFR mutation-positive; c) Eastern Cooperative Oncology Group performance status of 0–2; and d) anlotinib or chemotherapy was administered with disease progression after treatment with EGFR mutation-targeted drugs (disease progression after osimertinib treatment if the T790M mutation was positive). The exclusion criteria were as follows: (a) anaplastic lymphoma kinase (ALK), ROS1, KRAS, v-raf murine sarcoma viral oncogene homolog, c-mesenchymal epithelial transition factor, or rearranged during transfection mutations were positive; and (b) chemotherapy was concurrently administered with anlotinib.
Treatments
The patients were divided into anlotinib and chemotherapy groups (n = 39 and n = 44, respectively). Anlotinib was administered orally at 12, 10, or 8 mg quaque die (QD) on days 1–14 of 21-day circles in the anlotinib group. The patients in the chemotherapy group were administrated with pemetrexed 500 mg/m2 or pemetrexed 500 mg/m2 plus either cisplatin 75 mg/m2 or carboplatin target area under the curve 5 every 3 weeks for four cycles and maintenance with pemetrexed until progression/discontinuation.
Assessment
Anlotinib-related or chemotherapy-related adverse events (AEs) were graded according to the Common Terminology Criteria for Adverse Events v5.0. The smoking index (SI) was used to measure smoking status. Objective tumor responses, including complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD), were defined according to RECIST v1.1. Objective response rate (ORR) was defined as the sum of CR and PR. The disease control rate (DCR) was defined as the sum of CR, PR, and SD. Progression-free survival (PFS) and overall survival (OS) were the first endpoints. PFS was defined as the interval from the beginning of treatment to tumor progression or patient death. OS was defined as the interval from the beginning of treatment to patient death. The secondary endpoints were the ORR and DCR.
Statistical Analysis
All the statistical analyses were performed using SPSS version 26 (IBM Corp., Armonk, NY, USA). The Mann–Whitney U test and Student's t-test were used to compare continuous variables. The Kruskal-Wallis test and chi-square tests were used to compare categorical variables. Kaplan-Meier curves were used to visualize and compare the PFS and OS. Cox regression analysis was used for univariate and multivariate analyses. Missing data were deleted and excluded from the statistical analysis. All tests were two-tailed, and the statistical significance level (P) was set at .05.
Results
Patient Profiles
We retrospectively enrolled 83 patients at the First Affiliated Hospital, College of Medicine, Zhejiang University between May 2011 and July 2022. There were 39 (47.0%) and 44 (53.0%) patients in the anlotinib and chemotherapy groups, respectively. The patient characteristics are summarized in Table 1. There were missing SI data for two patients with a smoking history in the chemotherapy group. There was a missing EGFR mutation type in one patient in the anlotinib group, but the EGFR mutation was confirmed positive. 1 (2.6%) patient had received two lines of systemic chemotherapy and 4 (10.3%) patients had received one line of systemic chemotherapy before anlotinib was administrated in anlotinib group. After disease progression after treatment with EGFR mutation-targeted drugs and/or clinical trials (disease progression after osimertinib treatment if the T790M mutation was positive), 34 (87.2%) patients in anlotinib group were treated with anlotinib with systemic chemotherapy skipped. Anlotinib was the second-line, third-line, and fourth-line and beyond in 20 (51.3%), 9 (23.1%), and 10 (25.6%) patients, respectively. Anlotinib was administered at 12, 10, and 8 mg in 1 (2.6%), 35 (89.7%), and 3 (7.7%) patients, respectively. Systemic chemotherapy was administrated as the second-line, third-line, and fourth-line and beyond in 24 (54.5%), 19 (43.2%), and 1 (2.3%) patient, respectively. The distribution of age, gender, smoking history, SI, TNM, brain metastases, and EGFR mutation except for the T790M mutation and T790M mutation in exon 20 showed no significant differences between the anlotinib and chemotherapy groups (all P > .05).
Characteristics of Patients with Adenocarcinoma who Were Treated with Anlotinib or Underwent Chemotherapy.
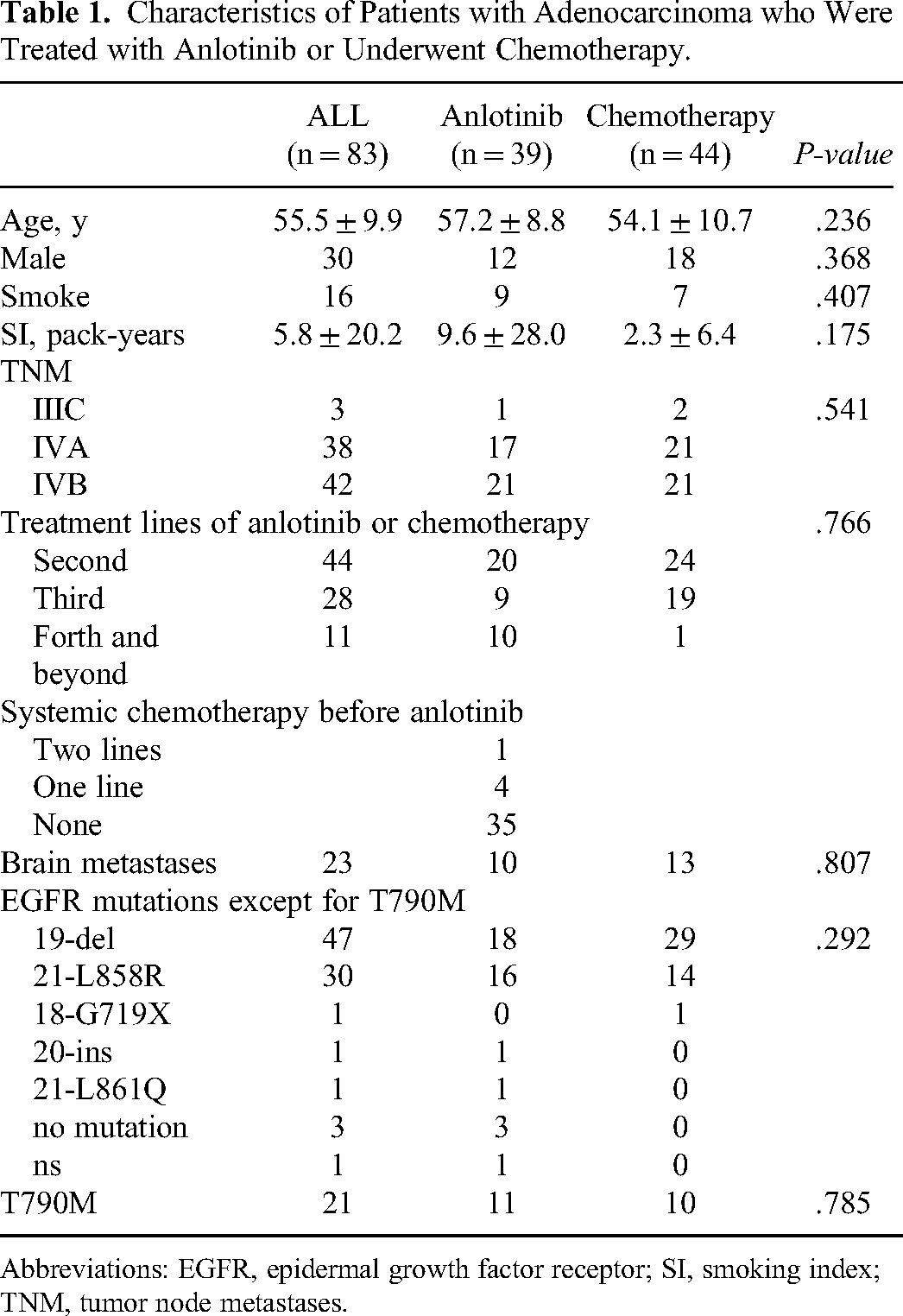
Abbreviations: EGFR, epidermal growth factor receptor; SI, smoking index; TNM, tumor node metastases.
Clinical Outcomes
There was one missing data point on treatment response in the anlotinib group, while 3 (7.9%) patients achieved PR and 35 (92.1%) had SD (Table 2). In the chemotherapy group, 9 patients achieved PR (20.5%), 31 (70.5%) patients had SD, and 4 (9.1%) patients had PD. The ORR were 7.9% and 20.5% (P = .129) in patients treated with anlotinib and chemotherapy, respectively. The DCR were 100% and 93.2% (P = .120) in the anlotinib and chemotherapy groups, respectively. The univariate and multivariate analyses of PFS and OS are summarized in Tables 3 and 4, respectively. Compared with the patients younger than 30 years old, the HR of OS in patients aged 50-70 and older than 70 years old were 0.06 (95% CI 0.01-0.75, P < 0.05) and 0.06 (95% CI 0.00-0.64, P < .05). The HR of PFS and OS in patients with brain metastases were 1.90 (95% CI 1.07-3.30, P < .05) and 2.90 (95% CI 1.41-5.96, P < .01). The median PFS in patients treated with anlotinib was 11.2 months (95% CI 10.6-13.9) and 4.5 months (95% CI 3.7-5.4) in patients treated with chemotherapy (HR = 0.2, 95% CI 0.11-0.36, P < 0.01, Figure 1 and Table 3). The median OS in patients treated with anlotinib was 18.8 months (95% CI 26.1-37.7) and 15.8 (95% CI 12.6-26.7) in patients treated with chemotherapy (HR = 0.43, 95% CI 0.21-0.89, P < .05, Figure 1 and Table 4). The PFS and OS of patients with exon 20 insertion, exon 18 G719X mutation, and exon 21 L861Q mutation of EGFR were 3.1 and 3.2 months, 4.9 and 13.5 months, and 11.0 and 19.4 months, respectively. There were no significant differences in the comparison of the effects of different treatment lines and doses of anlotinib on the clinical outcomes of the patients (Figure 2).

Overall survival (OS) and progression free survival (PFS) in patients who were treated with anlotinib or underwent chemotherapy.

Overall survival (OS) and progression free survival (PFS) in patients treated with anlotinib as the first-line, second-line, third-line, or fourth-line and beyond treatment.
Secondary Endpoints of Patients Receiving Anlotinib or Chemotherapy.

Abbreviations: CR, complete response; DCR, disease control rate; ORR, objective response rate; PR, partial response; PD, progressive disease; SD, stable disease.
Univariate and Multivariate Analyses of Progression Free Survival.
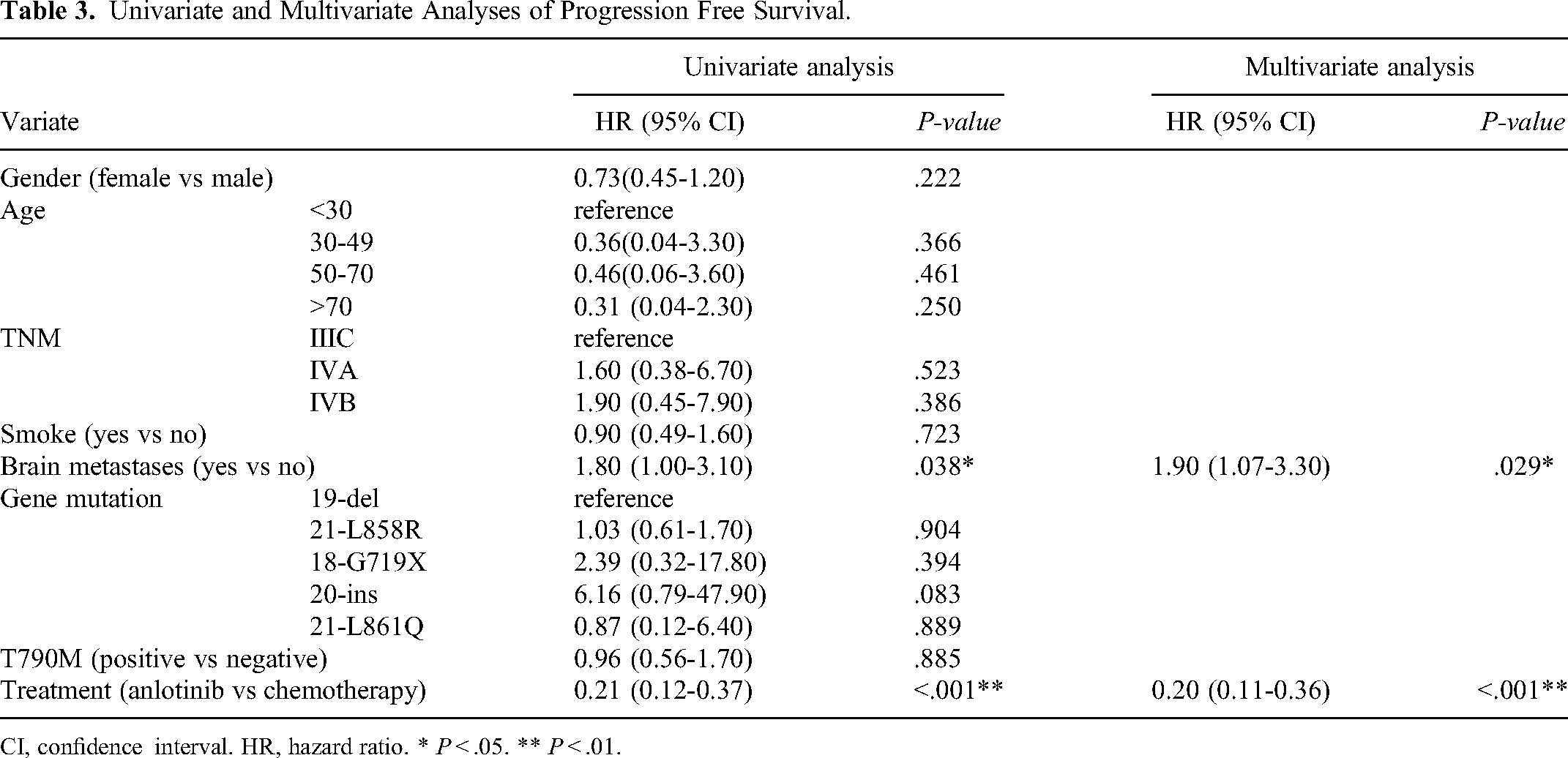
CI, confidence interval. HR, hazard ratio. * P < .05. ** P < .01.
Univariate and Multivariate Analyses of Overall Survival.

Abbreviations: CI, confidence interval; HR, hazard ratio. * P < .05. ** P < .01.
Safety
Adverse events were observed in 18 (46.2%) and 39 (88.6%) patients in anlotinib and chemotherapy groups respectively (P < 0.01), whereas 8 (20.5%) and 30 (68.2%) patients experienced more than one type of AEs respectively (P < .05). Adverse events grading 3-4 level were observed in 2 (5.1%) and 15 (34.1%) patients in anlotinib and chemotherapy groups respectively (P < .01). Diarrhea (25.6%) and decreased white blood cell count (45.4%) were the most common anlotinib-related AEs and chemotherapy-related AEs respectively. The AEs are summarized in Table 5. Due to AEs, anlotinib dose adjustment (one from 10 mg to 8 mg QD for swollen and painful gums grading 3, one from 8 mg QD to 8 mg quaque omni die for diarrhea grading 2) and terminal discontinuity (for rash grading 3) were observed in 3 patients (7.7%) and 1 patient (2.6%), respectively. No anlotinib-related death was recorded.
Adverse Events Associated with Anlotinib or Chemotherapy.

Abbreviations: AEs, adverse events; TSH, thyroid stimulating hormone.
Discussion
Since 2018, anlotinib has been approved in China as a third-line treatment for patients with advanced NSCLC after at least two lines of systemic chemotherapy or targeted drugs, and two lines of systemic chemotherapy if EGFR and/or ALK is positive. However, clinical data on anlotinib, especially its efficacy compared with that of traditional chemotherapy, are limited. The study was designed to help clarify the effectiveness and safety of anlotinib and add more clinical data to the literature. We found that the PFS and OS of patients treated with anlotinib were significantly longer than those of patients treated with chemotherapy. Cox regression analyses revealed that patients with brain metastases had shorter PFS and OS.
The efficacy of anlotinib has been confirmed in previous descriptive studies23–25 and placebo-controlled clinical studies.12,13,26 However, limited data have been reported regarding comparisons between the efficacy of anlotinib and chemotherapy in EGFR-positive advanced LUAD after disease progression with targeted drugs and/or chemotherapy. In our study, patients treated with anlotinib had longer PFS and OS than those patients treated with chemotherapy, indicating the advantages of anlotinib over traditional chemotherapy in terms of the clinical outcomes in EGFR-positive advanced LUAD. The median PFS and OS of the patients treated with anlotinib in our study were remarkably longer than those in former studies. The patients included in our study were all EGFR-positive while previous studies tended to have no restriction on the EGFR mutation status of the patients included. Anlotinib was administrated to patients who failed at least two kinds of systemic chemotherapy and/or intolerance of targeted drugs (third line or beyond) in former studies. However, the majority of patients who were resistant to targeted drugs in our study were treated with anlotinib directly with systemic chemotherapy skipped. That may explain the better efficacy of anlotinib in our study than the former ones. The ORR of anlotinib was similar to that reported in a previous study, whereas the DCR was higher in our study.12,18,27 However, no significant differences in ORR or DCR were observed between the anlotinib and chemotherapy groups. The treatment lines and doses of anlotinib seemed to have no impact on the clinical outcomes of the patients, from which limited information could be obtained, owing to the small sample size of the subgroups.
Cox regression analyses of PFS and OS showed that patients with brain metastases had worse clinical outcomes, which is similar to the findings of previous studies.20,25,28 The patients seemed to have worse OS if they were younger than 30 years at the time of advanced LUAD diagnosis. The T790M mutation in exon 20 of the EGFR gene appears to have no negative influence on the clinical outcomes of patients. Deletion in exon 19 (19-del) and L858R mutation in exon 21 (21-L858R) were the most common EGFR mutations in our study, and the number of other EGFR mutation types was too small to draw a significantly reasonable conclusion. However, a tendency to worsen clinical outcomes was observed in patients with exon 20 insertion of the EGFR mutation, which was consistent with the characteristics of patients harboring the exon 20 insertion reported before. 29
The incidence of anlotinib-related AEs was 30.8%, and some patients experienced more than one type of AEs which was similar to previous research.12,18,30,31 Based on the retrospective nature of our study, the incidence of AEs may have been underestimated. The majority of AEs were graded 1–2 and were well-tolerated in most cases. However, adjustment of the dose and termination discontinuity were recorded in a minority, indicating that attention was needed during the treatment period.
The following are the limitations of our study. (a) This was a retrospective study and the conclusion derived from this study needs to be confirmed in the future. (b) The sample size was small. (c) All patients came from one center. (d) the previous targeted therapy or chemotherapy before the anlotinib or pemetrexed plus either cisplatin or carboplatin may have unconfirmed effects on the tumor control which may confuse the result of our study. More prospective multicenter and well balanced studies should be performed to verify the efficacy of anlotinib in advanced EGFR-positive LUAD compared to traditional chemotherapy.
Conclusion
Anlotinib is effective and safe in patients with advanced EGFR-positive LUAD. Diarrhea was the most commonly observed anlotinib-related AE in our study. Patients without brain metastases had better clinical outcomes. Patients with an exon 20 insertion of the EGFR mutation and those diagnosed with advanced LUAD at an age younger than 30 years tended to have worse clinical outcomes.
Footnotes
Acknowledgements
We express our gratitude to Jianying Zhou for the application of ethics approval document.
Authors Contributions
Cuihong Cai contributed to the design of the study, acquisition of the data, analysis of the outcomes, and drafting of the manuscript. Qian Shen contributed to the design of the study, acquisition of the data, and analysis of the outcomes. Jianya Zhou, Jingjing Shao, Jingjing Qu, and Shuangshuang Zhou contributed to the study design, provision of study patients, data analysis and interpretation, and critical suggestions of the manuscript. All authors have agreed to the final version of the manuscript. All authors take responsibility for the reality and accuracy of the data and data analysis.
Availability of Data and Materials
The datasets used during the current study are available from the corresponding author upon reasonable request.
Consent to Participate
Informed consent exemption was obtained for this study from Clinical Research Ethics Committee of the First Affiliated Hospital, Zhejiang University School of Medicine.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
All experiments were performed in accordance with relevant guidelines and regulations (such as the Declaration of Helsinki). This study was reviewed and approved by the Clinical Research Ethics Committee of the First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou (Number: 2023-0467) on June 14, 2023.
Funding
This work was supported by the Zhejiang Key Research and Development Program (2019C03042) and the Natural Science Foundation of Zhejiang Province (LGF22H160005).
