Abstract
Introduction
The utility and application of endocervical curettage (ECC) during colposcopy remain controversial. This study optimized ECC application for primary human papillomavirus (HPV) screening in patients with high-risk (HR)-HPV.
Methods
This retrospective study included patients with HR-HPV, who underwent subsequent cervical biopsy and ECC from January 1, 2014, to December 31, 2020. Logistic regression was used to calculate the odds ratios (ORs) and 95% confidence intervals (CIs). The prediction model was presented as a nomogram and evaluated for discrimination and calibration.
Results
The additional detection rate of cervical intraepithelial neoplasia 2 + lesions with ECC was 2.0% (77/3887) in patients with HR-HPV. In multivariate risk factor analysis, HPV 16 infection presented a high risk of positive ECC, followed by HPV 33, HPV 58, and HPV 31. Irrespective of the abnormal cytopathological results, positive ECC was significantly increased (all P < .001). Females with acetowhite changes on colposcopy, transformation zone (TZ) type II, TZ type III, colposcopic impression of high-grade squamous intraepithelial lesion, or cancer were at a high risk of positive ECC. The final prediction model included significant variables from risk factor analysis, and had excellent calibration and classification capabilities, with an area under the receiver operating curve of 0.902 (95% CI, 0.881-0.922). Additionally, calibration analysis suggested consistency.
Conclusion
As the additional detection value of ECC is limited. A satisfactory prediction model was designed to optimize ECC application in patients with HR-HPV infection.
Keywords
Introduction
Cervical cancer has the second highest morbidity and mortality among gynecological malignancies globally. 1 Colposcopically directed biopsy (CDB) is a vital tool for the early detection of cervical precancerous lesions and malignancies.2,3 However, in patients with a history of cervical treatment that destroys the transformation area or in older females with the transformation area transferred to the cervical canal, determining the state of endocervical lesions using a colposcopy is difficult.
Endocervical curettage (ECC) can be used to obtain tissue samples from the cervical canal for histopathological evaluation, which can improve the detection rate of cervical lesions. However, the utility and application of ECC remain controversial.4‐6 A real-world study reported that ECC increased the diagnosis rate of cervical intraepithelial neoplasia (CIN) 2 + lesions by only 1.01%, 6 indicating the limited significance of routinely performing ECC during colposcopy. According to the current American Society for Colposcopy and Cervical Pathology (ASCCP) guidelines, 2 no clear recommendations for ECC use during colposcopy exist, and the decision to perform ECC depends primarily on the experience of clinicians. But not all females benefit from ECC at colposcopy. Performing colposcopy and ECC based on clinician's experience, rather than evidence-based ECC, may lead to a higher incidence of adverse outcomes. 7
Since the release of the 2015 ASCCP interim guidelines, 8 primary human papillomavirus (HPV) testing has become the dominant method for cervical cancer screening in China. The increase in primary HPV testing has significantly raised the number of patients referred for colposcopy and ECC owing to its high sensitivity but low specificity. 9 The overuse of ECC wastes resources and increases economic burden. Furthermore, ECC is frequently the most uncomfortable step in colposcopy for most patients.10,11 Therefore, decisions regarding the necessity of performing ECC should not be made lightly, and there is a requirement for an alternative approach to enable early identification of females infected with HR-HPV who require this procedure.
During primary HPV screening, we aimed to conduct risk stratification for positive ECC (histopathologically CIN 2 or worse) and develop and validate prediction model for ECC use in females with HR-HPV infections.
Methods
Patients and Study Design
Females who underwent a primary HPV genotyping test between January 1, 2016, and December 31, 2020, were recruited for this study. Meanwhile, patients were excluded if they were pregnant, had a history of other malignancies or severe immunodeficiency diseases, or had received the HPV vaccine. Eligible women referred for routine colposcopy with CDB and ECC, following abnormal screening results (including HPV and ThinPrep cytological test [TCT] results) based on clinical guidelines were included. For subgroup analysis, the included individuals were divided into two groups according to their HR-HPV infection status: the HPV 16/18 infection and non-16/18 HR-HPV infection groups.
The ethics committee of the hospital approved all protocols and informed consent procedures. Owing to the retrospective observational design of this study data anonymity, the requirement for written informed consent was waived.
Colposcopy, Cervical Biopsy, and ECC
Pre-colposcopy, basic information, including gravidity, parity, medication history, cervix-related lesions, other malignancies, and clinical manifestations (contact bleeding, irregular vaginal bleeding, and increased leukorrhea) were recorded.
During colposcopy, the cervix and vagina were first examined by gross visualization under bright light, without the application of any solutions. Then, a solution of 3%–5% acetic acid, followed by Lugol's solution, was applied to the cervix. A green filter on the colposcope was used to identify accentuated abnormal blood vessels, and CDB was performed by targeting visible acetowhite areas. If colposcopy showed no lesions, four quadrants perforated biopsy was randomly performed. Then, ECC was performed after cervical biopsy to allow for clear directed biopsy, without blood from the ECC obscuring the biopsy site. Under colposcopy guidance, an in-and-out and rotating motions with a Kevorkian curette were used to ensure comprehensive sampling of the full canal circumference. Electronic colposcopy (SLC-2000; Goldway, China) was used to visually evaluate the cervix, including the transformation zone (TZ) type (I, II, and III), acetowhite changes, Lugol's staining, punctate blood vessels, and abnormal blood vessels. A standardized documentation form was used to consistently document colposcopic findings. All colposcopic examinations, CDB, and ECC procedures were performed by the same experienced gynecologist. The least favorable pathological result from CDB and ECC was considered as the final diagnosis. Colposcopic diagnoses were categorized as normal, low-grade squamous intraepithelial lesion (LSIL), high-grade squamous intraepithelial lesion (HSIL), or cancer.
HPV Genotyping Test, Cytopathology, and Histopathology
The genotyping test was conducted with PCR-reverse dot blot (Yaneng® Biosciences, Shenzhen, China). 12 Fourteen HR-HPV genotypes were identified using this technique, namely HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68. All reactions were performed according to the manufacturer's instructions.
The cytopathological diagnoses were based on the 2014 Bethesda System. 13 The cytopathological classifications were: negative for intraepithelial lesions or malignancy (NILM), atypical squamous cells of undetermined significance, LSIL, atypical squamous cells cannot exclude an HSIL (ASC-H), HSIL, squamous cell carcinoma (SCC), atypical glandular cells not otherwise specified, atypical glandular cells-favor neoplasia, adenocarcinoma in situ (AIS), and adenocarcinoma of the cervix (AC).
Histopathological diagnoses by two gynecological pathologists blinded to the cytopathological findings included suspicions of NILM, CIN 1–3, SCC, AIS, or AC. If a sample was primarily diagnosed as a CIN 2 + histopathologically, it was then reviewed by an independent pathologist. Differences were discussed and resolved through additional histopathological examination until consensus was reached.
Development, Validation, and Presentation of the Predictive Model
All variables included were analyzed using univariate analysis to determine the HR factors for positive ECC, while calculating the odds ratios (OR) and 95% confidence intervals (CI). Variables considered statistically significant in multivariate logistic regression analysis were then selected for the predictive model.
The Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis guidelines were used to report this study. 14
To ensure the precise estimation of key parameters in the prediction model, the sample size was calculated following the specific criteria proposed by Riley et al 15 Based on the formulae reported, positive ECC incidence was 11.9%. 16 The required calculated sample size was at ≥577 (14 candidate predictors).
To evaluate the performance of the predictive model, we drew a calibration curve using predicted versus actual results. Furthermore, a receiver operating characteristic (ROC) curve with sensitivity and specificity was used to determine the model's discrimination, which was quantified using the area under the ROC curve (AUC). An AUC exceeding 0.7 was classified as reasonable, and exceeding 0.8 as strong. A decision curve was constructed to demonstrate the clinical utility of the predictive model. Internal verification was performed to evaluate the stability of the predictive model employing 10-fold cross-validation and 100 bootstrap re-samplings. The 10-fold cross-validation was used to randomly select a subset of 90% of participants, and the remaining 10% were used to test validity. This test was repeated 10 times to assess the predictive accuracy of the model and determine whether the extent was overoptimistic. The model was replaced with a development sample that was modified to assess the optimism of model performance.
The final model, which include d statistically significant variables, was presented as a nomogram for clinical application, with each predicted value corresponding to a weight; therefore, the total score is equivalent to a linear predictor. For this prediction model, logistic regression outcomes were transformed into the linear predictor to produce estimates of the probability of positive ECC in females with HR-HPV infection.
Statistical Analysis
A positive ECC result was defined as a histopathology of CIN 2 or worse, and negative ECC was defined as normal histology or CIN 1 on ECC. Continuous and categorical variables of the two groups were compared using t-tests and chi-square tests, respectively. Univariate and multivariate logistic regression analyses were performed using the IBM SPSS statistical package (version 22.0; IBM Corporation, Armonk, NY, USA). R software (version 3.6.0; R Foundation for Statistical Computing, Vienna, Austria) was used for nomogram calculations and validation. All statistical tests were two-sided, and P < .05 was considered statistically significant.
Results
Patient Characteristics
Overall, 4677 females who underwent primary HPV testing for HR-HPV infection were referred for colposcopy. Of these, 2730 had non-16/18 HR-HPV infection (2730/4677; 58.4%), and 1947 had HPV 16/18 infection (1947/4677; 41.6%). All participants underwent ECC and CDB (Supplementary Figure 1). Figure 1 shows a comparison between the cervical biopsy and ECC histopathology results. The total detection rate of positive ECC was 8.4% (392/4677) in all patients with HR-HPV. The detection rate was 12.7% (248/1947) in the HPV 16/18 infection and 5.3% (144/2730) in the non-16/18 HR-HPV infection groups. The probability of the additional detection of CIN 2 + lesions was only 2.0% (77/3887). In the HPV 16/18 infection group, the rate was 2.6% (37/1437), whereas it was 1.6% (40/2450) in the non-16/18 HR-HPV infection group. The demographic and clinical characteristics of patient positive for HR-HPV in the negative and positive ECC groups are listed in Supplementary Table 1.

Comparing the consistency of cervical biopsy histopathology and ECC histopathology. A, All included individuals. B, HPV 16/18 infection group. C, Non-16/18 HR-HPV infection group. Abbreviations: HR-HPV, high-risk human papillomavirus; ECC, endocervical curettage; CIN, cervical intraepithelial neoplasia.
Risk Factors Analysis of Positive ECC Histopathology
Table 1 presents the risk factor analysis for positive ECC histopathology in females with HR-HPV. Using the multivariable logistic regression analysis, compared with females aged <25 years, those >35 years were an independent risk factor for positive ECC. Furthermore, females >65 years were at the highest risk of positive ECC (OR, 6.437; 95% CI, 1.324-31.289; P = .021). In addition, contact bleeding was associated with positive ECC (OR, 1.876; 95% CI, 1.218-2.889; P = .004).
Risk Factors Analysis of ECC Histopathological CIN 2 + in Patients Positive for HR-HPV (n = 4677).
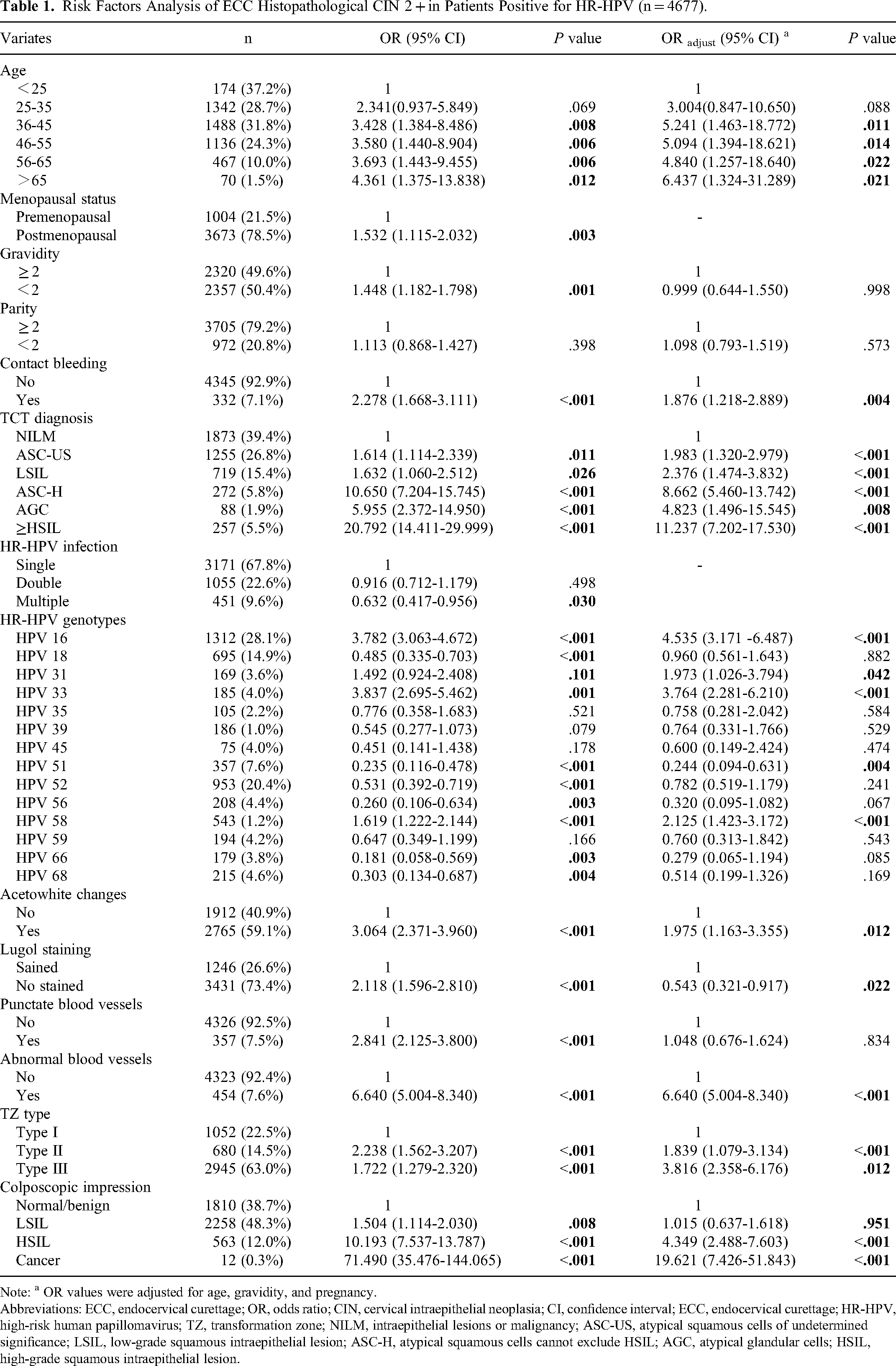
Note: a OR values were adjusted for age, gravidity, and pregnancy.
Abbreviations: ECC, endocervical curettage; OR, odds ratio; CIN, cervical intraepithelial neoplasia; CI, confidence interval; ECC, endocervical curettage; HR-HPV, high-risk human papillomavirus; TZ, transformation zone; NILM, intraepithelial lesions or malignancy; ASC-US, atypical squamous cells of undetermined significance; LSIL, low-grade squamous intraepithelial lesion; ASC-H, atypical squamous cells cannot exclude HSIL; AGC, atypical glandular cells; HSIL, high-grade squamous intraepithelial lesion.
Additionally, an abnormal TCT result was the primary reason for colposcopy referral. Abnormal cytopathological results increased the probability of positive ECC (P < .001). Compared to individuals with cytopathological NILM, those with cytopathological result greater than HSIL had an 11.2 times (95% CI, 7.202-17.530; P < .001) higher risk of positive ECC. In specific genotypes, HPV 16 (OR, 4.353; 95% CI, 3.171-6.487; P < .001), HPV 33 (OR, 3.764; 95% CI, 2.281-6.210; P < .001), HPV 58, (OR, 2.125; 95% CI, 1.423-3.172; P < .001), and HPV 31 infection (OR, 1.973; 95% CI, 1.026-3.794; P = .042) were risk factors for pathological ECC CIN 2 + . Notably, double or multiple HR-HPV infections were not associated with a higher likelihood of positive ECC.
Females with acetowhite changes on colonoscopy (OR, 1.975; 95% CI, 1.163-3.355; P = .012) had a higher risk of positive ECC. Lugol's staining, punctate blood vessels, and abnormal blood vessels was discovered to have no association with risk. TZ types II (OR, 1.839; 95% CI, 1.079-3.134; P < .001) and III (OR, 3.816; 95% CI, 2.358-6.176; P < .012) were significantly associated with ECC CIN 2 + and compared to those with TZ type I. Furthermore, compared with a normal/benign impression in colposcopy, patients with an impression of LSIL, HSIL, and cancer had 1.015 (95% CI, 0.637-1.618, P = .951), 4.349 (95% CI; 2.488-7.603; P < .001), and 19.621 (95% CI, 7.426-51.843, P < .001) times greater risks of positive ECC, respectively.
Subgroup Analysis of Individuals with HPV 16/18 Infection and Other Types of HR-HPV
Multivariable logistic regression analysis of patients infected with HPV 16/18, demonstrated statistically significant differences in variables between 36 and 65 years. Furthermore, symptoms of contact bleeding, TCT ≥ ASCUS other than AGC, HPV 33 infection, acetowhite changes, TZ type III, and colposcopic impression (Supplementary Table 2). However, in patients infected with other types of HR-HPV we observed that TCT ≥ ASC-H; HPV 31 and HPV 33 infections, acetowhite changes, TZ type III, and colposcopic impression were statistically significantly associated with positive ECC (Supplementary Table 3).
Model Design, Performance, and Validation
Variables determined to be significant in multivariate risk factor analysis of positive ECC histopathology, including age, symptoms of contact bleeding, HPV genotypes, TCT, TZ type, acetowhite changes, and colposcopic impression were included in the final model. Figure 2 provided the weighted nomogram and risk prediction scores that allowed estimation of individual probabilities of identifying positive ECC at colposcopy. When the grading value of the ten predictors is determined, the total score can be obtained by adding them together and the probability of ECC positivity in patients with HR-HPV infections can be calculated.

Nomogram predicting ECC positivity in patients with hr-HPV infections. Abbreviations: HR-HPV, high-risk human papillomavirus; ECC, endocervical curettage; CIN, cervical intraepithelial neoplasia; NILM, intraepithelial lesions or malignancy; ASC-US, atypical squamous cells of undetermined significance; LSIL, low-grade squamous intraepithelial lesion; ASC-H, atypical squamous cells cannot exclude HSIL; AGC, atypical glandular cells; HSIL, high-grade squamous intraepithelial lesion; TZ, transformation zone.
Figure 3A displayed the ROC curve, with the sensitivity and specificity of the nomogram were 83.7 and 81.3%, respectively, and the AUC was 0.902 (95% CI: 0.881-0.922). The calibration plots of the observed versus predicted probabilities by cross-validation and bootstrapping are shown in Figure 3B and C, and the predicted positive ECC rate was consistent with the actual value. Then, the overfitting was assessed, and found to be negligible. The original C-statistic of the final model was 0.866, and performing three-fold cross-validation yielded a modified C-statistic of 0.860. Meanwhile, during bootstrapping, the C-statistic was 0.865. Thus, model optimism was found to be negligible in 10-fold cross-validation and bootstrapping.

Performance of the predictive model. A. Receiver operating curve of the nomogram. B Calibration plots of the predicted and actual probabilities of the nomogram by cross-validation. C, Calibration plots. Abbreviations: AUC: area under the receiver operating characteristic curve.
Discussion
In clinical practice, the correct application of ECC can reduce the rate of missed diagnoses and improve the detection rate of cervical lesions. However, by aiming to completely eliminate missed diagnoses in clinical practice, the rate of overtreatment and financial implications of excessive testing was inevitably increased. 6 No previous studies identified the real beneficiaries of ECC from the large cohort of patients with HR-HPV referred for colposcopy. Therefore, in this study, risk factor analysis was performed, and an effective prediction model was constructed to individualize the risk of histopathological ECC CIN 2 + in individuals with HR-HPV infection.
In individuals with HR-HPV infection, the additional detection rate of CIN 2 + lesions with ECC was only 2.0%. Although it increased to 2.6% in individuals with HPV 16/18 infection, the diagnostic utility of ECC alone for cervical HSIL remain limited. These findings are similar to those reported previously.17,18 One study demonstrated the limited benefits of ECC in patients with normal or satisfactory colposcopy with visible abnormal lesions. 4 However, clinical gynecologists in China prefer to perform ECC during colposcopy to avoid omissions, despite their knowledge that only some patients will benefit from it. 19 Therefore, individualized risk stratification of positive ECC in the HR-HPV infection cohort referred for colposcopy is an urgent concern.
We identified 10 powerful predictors in our prediction model to optimize the ECC strategy in patients with HR-HPV infections, including age, symptoms of contact bleeding, HPV infection, acetowhite changes, TZ types, and colposcopic impression. All of which are easy to assess in clinical practice.
As the first steps in cervical cancer screening, HPV infection and cervical cytopathology are the primary reasons for colposcopy referral. However, correlations between specific HPV genotype infections and ECC positivity have rarely been reported. As various genotypes have been reported to have wide-ranged levels of carcinogenicity, data suggest that the most pathogenic HPV genotypes can distinguish those at a high risk for CIN 3.20,21 The current study revealed that patients with HPV 16, HPV 33, HPV 58, and HPV 31 were associated with the potential for histopathologically positive ECC. These findings underscore the significance of ECC in individuals infected with distinct HPV genotypes. Notably, HPV 18 has no association with ECC CIN 2 + due to CIN 2 + being the study endpoint, whereas HPV 18 infection is closely related to the incidence of cervical adenocarcinoma.22‐24 The clinical manifestation of cervical adenocarcinoma is atypical. Therefore, vigilance is required in clinical practice regarding HPV 18 infection because of its close association with cervical adenocarcinomas.
With the widespread application of primary HPV testing, cervical cytopathology has become essential for colposcopy referral for patients positive for HR-HPV. However, studies report varying conclusions regarding the role of abnormal cytopathological results in predicting ECC. Xue et al 25 claimed that only TCT ≥ HSIL strongly correlated with ECC, and Liu et al 4 reported a 13.0% CIN 2 or worse ECC yield in females with ASCUS or LSIL cytopathology. Using multivariate logistic regression analysis, we revealed a statistically significant association between abnormal cytopathology and an increased risk of positive ECC, even in cases with the mildest form of ASCUS, underscoring the importance of conducting additional reflex triage testing in those testing positive for primary HPV screening per recommendations. 2
Age was found to be a significant factor in determining the requirement for ECC; the older the individual, the higher the risk of positive ECC, with those aged ≥65 years having the greatest risk. These age-related trends correspond with the findings of Pretorius et al, 19 who reported that limiting ECC use in females aged ≥25 years resulted in 29.3% (5433/18,537) fewer ECCs. Herein, older females benefited more from endocervical sampling due to challenges in completely visualizing the cervical squamous-columnar junction. 26 However, the TZ type, which also affects adequate visualization of the cervix surface, that would likely benefit from ECC remain debatable.27,28 One study claimed that TZ type III is a crucial reference index for occult cervical lesions, 27 while others suggested that those with TZ types I and II should be encouraged to undergo ECC due to possible positive ECC findings. 28 Our study confirmed that TZ types II and III, which are the origins of cervical carcinogenesis, were both important references for ECC decision-making, and were included in model construction.
In addition, clinical manifestations and colposcopy results are important reference factors for ECC decision-making, although they have not been described in the guidelines. As contact bleeding is the most common clinical manifestation of cervical cancer, a previous study suggested that females with contact bleeding should be referred for colposcopy to identify potential cervical cancer. 26 Our study also found that contact bleeding was associated with positive ECC. Therefore, ECC should be performed in females with HR-HPV and contact bleeding to assess for underlying positive ECC lesions. The severity of colposcopic impression may also influence the clinician's decision to perform ECC. A study reported that a colposcopy diagnosis ≥ HSIL was an independent risk factor for pathological CIN 2 + in ECC. 25 Additionally, acetowhite changes were reported as a risk factor of positive ECC. 29 Our results verify these previous studies' findings, and these factors were included as important indicators in prediction model construction due to their strong correlation with positive ECC. Nevertheless, we found that Lugol's staining, punctate blood vessels, and abnormal blood vessels on colposcopy were not associated with an increased risk of positive ECC.
In the context of primary HPV screening, an increasing number of people are being referred for colposcopy and ECC due to positive HPV results. Identifying groups that will benefit from ECC has become an urgent concern. Previous studies that attempted to develop predictive models for ECC in individuals referred for colposcopy,25,29 did not incorporate HPV genotypes as predictors, despite these being the predominant etiology of cervical cancer. Our study attempted to fill this gap by developing and validating a predictive model to identify individuals at high risk of positive ECC in patients with HR-HPV and enhance the clinical management of ECC procedures. During clinical colposcopic examination of patients with HR-HPV, clinicians comprehensively chose a wait-and-watch approach for females at low risk, and referred those at high risk for ECC with the assistance of an accurate, established model, thereby reducing unnecessary ECC as well as minimizing physical and psychological harm. This predictive model has good recognition and calibration capabilities, and its indicators are easy to obtain in clinical practice, making the model convenient for clinical application.
This study has several strengths. First, to our knowledge, no prior study has examined the relationship between specific HR-HPV genotypes and ECC results. Second, in the context of primary HPV screening, we are the first to develop and internally validate a model that optimizes variables for ECC application in females with HR-HPV infection. Third, the predictors in the derived model are easy to assess, and the model can assist in clinical decision-making regarding the necessity of ECC in females with HR-HPV. However, this study had some limitations. First, as this was a retrospective study, it is less effective than a prospective study. Second, although this study has several advantages, only preliminary suggestions were provided because the focus was on females in China. Finally, due to limited data, we could not evaluate the additional diagnostic role of positive ECC in patients where diagnosis was missed by biopsy alone.
Conclusions
The additional diagnostic value of ECC in females infected with HR-HPV is limited. In the context of primary HPV screening, we developed an easy-to-use clinical prediction model with good discrimination and calibration capabilities for patients with HR-HPV. This model assisted in evaluating the individual risk of positive ECC and aided clinicians in determining the necessity of ECC during colposcopy. Thus, the overuse of ECC can be reduced, and unnecessary physical and psychological harm may be minimized. Prospective studies are required to further explore the efficacy of this model in clinical practice.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241312573 - Supplemental material for Risk Factors of Positive Endocervical Curettage and Predictive Model Construction Based on Primary Human Papillomavirus Screening
Supplemental material, sj-docx-1-tct-10.1177_15330338241312573 for Risk Factors of Positive Endocervical Curettage and Predictive Model Construction Based on Primary Human Papillomavirus Screening by Hangjing Gao, Guanxiang Huang, Binhua Dong, Ye Li, Hongning Cai, Xianqian Chen, Tingting Jiang, Kelvin Stefan Osafo, Dabin Liu, Jiancui Chen, Huihua Ge, Diling Pan, Huifeng Xue and Pengming Sun in Technology in Cancer Research & Treatment
Footnotes
Acknowledgment
The authors thank all the participants for their great effort.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The present study complied with the Declaration of Helsinki and was approved by the ethics committee of Fujian Maternity and Child Health Hospital (2019-033).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the following grants: the National Natural Science Foundation of China (grant no. 82271658), the Fujian Provincial Natural Science Foundation of China (grant no. 2022J011040), the Major Scientific Research Program for Young and Middle-aged Health Professionals of Fujian Province, China (grant no. 2021ZQNZD011), the Fujian Province Central Government-Guided Local Science and Technology Development Project (grant no. 2023L3019), the Fujian Clinical Medical Research Center for Gynecologic Oncology (grant no. 2021Y2013), the Fujian Provincial Science and Technology Innovation Joint Fund (grant no. 2021Y9171, 2021Y9166, and 2021Y9176), and Fujian Province's Third Batch of Flexible Introduction of High-Level Medical Talent Teams (grant no. TD202307).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
