Abstract
The battle against cancer remains a top priority for society, with an urgent need to develop therapies capable of targeting challenging tumours while preserving patient's quality of life. Hadron Therapy (HT), which employs accelerated beams of protons, carbon ions, and other charged particles, represents a significant frontier in cancer treatment. This modality offers superior precision and efficacy compared to conventional methods, delivering therapeutic the dose directly to tumours while sparing healthy tissue. Even though 350,000 patients have already been treated worldwide with protons and 50,000 with carbon ions, HT is still a relatively young field and more research as well as novel, cost-effective and compact accelerator technologies are needed to make this treatment more readily available globally. Interestingly the very first patient was irradiated with protons in September 1954, the same month and year CERN was founded. Both of these endeavours are embedded in cutting edge technologies and multidisciplinary collaboration. HT is finally gaining ground and, even after 70 years, the particle therapy field continues innovating and improving for the benefits of patients globally. Developing technologies that are both affordable and easy to use is key and would allow access to more patients. Advances in accelerator-driven Boron Neutron Capture Therapy (BNCT), image-guided hadron beams delivery, clinical trials and immunotherapy, together with the recent interest and advances in FLASH therapy, which is currently an experimental treatment modality that involves ultrahigh-dose rate delivery, are just a few examples of innovation that may eventually help to provide access to a larger number of patients.
Keywords
Premise for This Review
Right from the outset, it should be declared that the roots of all the authors involved this review are embedded at CERN in particle therapy, detector and accelerator technology and, above all, global collaboration. One of the authors who was previously at Lawrence Berkeley was particularly interested in writing this review in 2024, as it marks 7 decades from the first patient treated with protons in Berkeley as well as the foundation of CERN and motivated the other authors to write this paper. The difference between the development of CERN becoming a world leading laboratory and the challenges faced by hadron therapy becoming global also emphasises the time it takes to overcome the challenges of translating technologies into a clinical environment. The paper is not intended to be an exhaustive review of hadron therapy from the beginning but more to focus on the importance of multi-disciplinary collaboration in the field and of continuing technology developments, by illustrating with three new examples, in which the authors are playing a key role, the challenges and the time scales faced by new advanced technology to enter into clinical practice.
Introduction
Cancer imposes an enormous economic burden worldwide. The total cost of cancer to the global economy will reach 25.2 trillion US dollars between 2020 and 2050, according to an analysis of 29 cancers across 204 countries. 1 Previous estimates cost of cancer, the leading cause of death worldwide, had mainly looked at a selected number of cancers. This study also found that the cost of certain cancers differed around the world: breast and cervical cancer will have the largest economic impact in sub-Saharan Africa, whereas wealthy countries will have the highest costs from cancers that mainly affect ageing populations, such as lung cancer.
According to data of the World Health Organization (WHO), cancer remains one of the most significant causes of death globally – accounting for nearly one in every six deaths globally in 2020. 2 In 2022 alone, 20 million new cancer cases and 10 million cancer deaths were registered. 3 Throughout the years various regions around the world have seen an increase in the incidence rates, with current estimates predicting an increase of almost 3 times by year 2050–58.6 million cases globally. 4 With global cancer burden expected to grow, effective cancer management strategies have to be considered in healthcare systems and novel treatment methods to be explored and researched. Out of the 3 primary methods for cancer treatment – surgery, chemotherapy and radiotherapy (RT) – RT, as treatment modality in course of care, is recommended for more than 50% of patients. 5
Radiation Therapy (RT)
The goal of RT is to deliver the necessary radiation dose to destroy the tumour while minimizing the damage to healthy tissues.
Conventional x-ray RT
The roots of RT date back to the discoveries of x-rays and radium made by Roentgen and the Curies in the years 1895–1897. As far as x-ray radiotherapy is concerned, while kilovoltage source were already used since the beginning of the 1900s, around 1950 “megavoltage” tubes were built and cobalt-60 machines entered clinical practice. The surprise was that, due to the higher energy and longer electron ranges, these “high-energy” radiations spared the skin much better than “orthovoltage” x-rays.
After a short period in which x-rays of energy larger than 5 MeV were produced with medical “betatrons”, electron linear accelerators (linacs) became the instrument of choice. The first electron linac of energy above 1 MeV was built in the 1950s at Stanford by Bill Hansen and collaborators for research purposes and was powered by a klystron produced by Varian Associates. 6
RT is a critical tool for treatment for over half of all cancer patients with the number of patients increasing. In the face of this growing burden there is a global shortage in access to RT, especially in Low-Middle-income Countries (LMICs) where 70% of cancer patients live.7,8 This lack of access to RT in LMICs was articulated in 2015 by the Lancet Oncology Commission on Radiation Therapy
9
and again in the latest
Today, only around 15,000 linacs 13 are installed in facilities all over the world and there is a huge shortfall with more than10,000 RT machines needed. In this scenario, any significant improvements in RT could have a dramatic positive impact on patient survival, quality of life and global economic burden.
Research and innovation efforts have been carried out worldwide to improve the effectiveness of RT. The main goal of advanced RT treatment is to maximize the damage of ionizing radiation to the tumour cells while minimizing the exposure of the surrounding normal tissues and critical organs thereby enhancing the likelihood of tumour destruction while the side effects of the treatment are minimized. To achieve this goal, RT has progressed considerably with the development of new technologies and methodologies able to increase the conformity of the dose delivered especially to deep-seated tumours. While the most frequently used modern RT modalities still rely on high energy (MeV) x-rays, there is a rapidly growing interest in the curative effects of accelerated charged hadrons, ie, protons and heavier ions such as carbon.
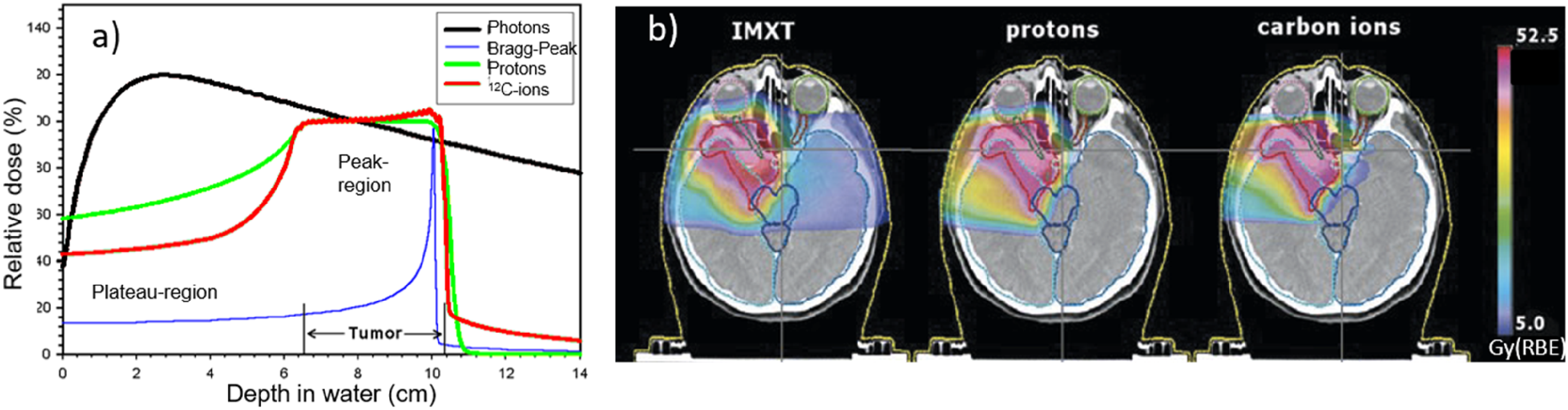
As shown by the black curve of Figure 1(a), the depth−dose distribution of a conventional x-rays beam (photons), after reaching a maximum at a few cm depths (the so-called build-up region), is characterized by an almost exponential attenuation and absorption of the dose, and consequently delivering the maximum dose near the beam entrance, but continuing to deposit significant amounts of energy at distances beyond the cancer target until it exits the body.
Hadron Therapy (HT)
HT can offer superior tumour-dose conformality (“conformality” or “conformity” stands for the dose shaped to the shape of the tumour size and volume) with a reduced number of treatment fields (ie irradiation delivered from different angles) compared to conventional x-ray radiation, mainly due to the favourable depth-dose deposition of ions in tissue, presented in Figure 1. However, despite the considerable recent progress of HT, numerous challenges and new opportunities are yet to be addressed to maximize clinical outcome and cost-effectiveness of this advanced RT modality for improved and uniformly accessible healthcare.
The idea of hadron therapy dates back to 1946 when Robert Wilson, physicist, and founder of Fermilab, was the first to propose using hadrons (and hadron accelerators) for cancer treatment. 14 As already pointed out, the year 2024 is doubly significant year for accelerator technology development and hadron therapy since the first patient was treated with protons in Berkeley by the Lawrence brothers and collaborators in September 1954, the same month and year that CERN was founded 15 (see Figure 2).

(Top-left) E. Lawrence, the inventor of the cyclotron with his first 4.5 inches cyclotron model built with his student Livingston. 16 (Top-right) R. Wilson, another student of Lawrence, who first proposed in 1946 the use of protons and light ions for tumour treatment. 14 (Bottom-left) In the spring 1954 the groundbreaking of CERN started in Geneva (Switzerland). (Bottom-right) In September 1954, the European Organization for Nuclear Research officially came into being with the signatures of the 12 founding states. 15
As originally highlighted by Wilson, particle therapy has an advantage over x-ray delivery methods due to the physical properties of charged particles, which deposit the main portion of their dose in the Bragg peak (Figure 1), minimizing the dose to tissue in the entrance channel and delivering minimal dose beyond the distal edge of the Bragg peak (ie the part further away from the peak, at a greater depth).
Particle therapy delivered either with protons, carbon or other ions has unique biological effects when compared to photon therapy. In order to optimize the clinical outcomes of particle therapy, it is important to understand the biological characteristics of particle beams, how these relate to physical factors and to introduce this knowledge into the treatment planning process. This is still an active field of study. Currently protons are believed to have biological effects relatively similar to photons, while carbon ion treatments incorporate more detailed biological information within the medical dose prescription. The enhanced biological effects produced by particles are quantified by their Relative Biological Effectiveness (RBE), which defines the dose modification necessary to achieve the same biological endpoint. RBE is defined as the dose of the reference radiation divided by the particle dose required to achieve the same biological effect.
The dose profile can be more precisely shaped using narrowly focused and scanned pencil beams of variable penetration depth, thus reaching better treatment effectiveness for the patient and reduced radiation exposure compared to x-rays. Due to the higher density of ionization events along the particle track, heavier ions, such as carbon, exhibit a higher RBE than x-rays or protons, especially in the Bragg peak region, making them prime candidates for the treatment of radio-resistant tumours.
A major part of the early scientific research and the rationale for the use of high-energy charged particles for cancer therapy was carried out at Berkeley. 17
There have been many centres showing the success of proton radiotherapy after the treatment of the first proton patient in Berkeley (1954): the Uppsala accelerator in Sweden (where the first patient in Europe was treated in 1957), 18 the Harvard Cyclotron in Boston, MA (USA), the Dubna, ITEP, and St. Petersburg facilities in Russia, the Chiba and Tsukuba accelerators in Japan, the Clatterbridge machine in UK and the Paul Scherrer Institute in Switzerland. The first dedicated hospital-based proton therapy centre was built in Loma Linda, California (USA) with help of Fermilab and the first patient was treated in 1990. 19
Soon after, in 1994 carbon ions treatment began in Chiba, Japan, for the first time with a dedicated heavy-ion accelerator for medical purposes as a part of a national 10-year strategy for cancer control. 20
Boron Neutron Capture Therapy (BNCT)
Another game-changing technique is BNCT, which is a dual therapy: this technique integrates the principles of chemotherapy with the cornerstone of RT (the capability of targeting tumour volume on a spatial basis). The principle of BNCT is to selectively kill tumour cells, sparing the surrounding normal tissues. The BNCT mechanism of action relies on a neutron capture reaction requiring two steps. First, the non-radioactive isotope 10B atoms (contained in the drug molecules – such as boronophenylalanine BPA) accumulates in the nuclei of malignant cells and then as a second step, comes the exposure to a neutron irradiation field: the treatment relies on the fact that the 10B nucleus has a very high affinity for neutron capture (with a large thermal neutron cross section of 3800 barn) and promptly decomposes, predominantly via α emission. The recoiling nucleus of 7Li and 4He (α particle) have a high Linear Energy Transfer (LET) and a short range almost equal to the diameter of cancer cells (Figure 3).

The non-radioactive isotope 10B atoms accumulate in the nuclei of malignant cells. Then, they absorb low-energy (<0.5 eV) thermal neutrons, breaking up into an α particle (4He) and a recoiled lithium nucleus (7Li). The reaction produces high Linear Energy Transfer (LET) alpha particles (≈150 keV/μm) and 7Li ion (≈175 keV/μm). Their peculiarity is to distribute a high-energy gradient along a very short pathway (<10 μm), limiting the energy deposition to the diameter of a single cell. 21
The treatment also relies on finding a boron-containing pharmaceutical with a marked preference for uptake by cancer cells rather than by normal cells. In other words, the selectivity of BNCT relies more on the boron compound's exact distribution than on the beam's precision, thus underlying its unique bridging position between chemo and RT. In this perspective, BNCT could be considered as a “cellular” RT contrary to conventional RT, it can selectively hit the tumour, saving the surrounding normal tissue (Figure 4).

The mechanism of selective killing of malignant cells with BNCT. 18
BNCT was first proposed as early as 1936. Research on BNCT has progressed slowly but steadily. Transformative technologies are seldom produced by individuals. Rather, success is built on the stepwise iterations of their predecessors. BNCT is no exception. The earliest conceptual work for neutron capture therapy was first theorized in the mid-1930s by the astrophysicist Gordon Locher. He hypothesized that neutrons emitted from a radium source captured by beryllium could be used to selectively kill tumour cells.
In 1951,22,23,24 neurosurgeon William Herbert Sweet and physicist Gordon Lee Brownell, of Massachusetts General Hospital (MGH) and Massachusetts Institute of Technology (MIT), respectively, conducted the first clinical trials involving BNCT, using the Brookhaven Graphite Research Reactor. After performing the primary debulking craniotomy on patients with high-grade glioma in Cambridge, Massachusetts, Sweet transported patients to Upton, New York, for the intravenous infusion of 10B-enriched borax (sodium tetraborate). Then, he exposed his patients to a single fraction of thermal neutrons from the experimental research reactor. None of the patients treated in these first clinical trials survived beyond 1 year and all experienced severe toxicities. It is important to note that thermal neutrons do not penetrate very deep.
The clinical BNCT neutron beam should ideally consist of epithermal neutrons (from 0.5–1.0 eV up to 10–30 keV) with minimum contamination by fast neutrons, thermal neutrons and photons. 25 However, the neutrons produced from 2.5 MeV protons on lithium have a maximum energy just under 800 keV. A static arrangement of carefully chosen materials called a Beam Shaping Assembly (BSA) is required to adjust the original neutrons into a clinically useful neutron source, via judicious moderation and reflection of neutrons, as well as absorption of thermal neutrons and photons exiting the surface. The total mass of the BSA exceeds 3 tons, primarily due to the large amount of lead used both as a reflector and as photon shield.
In 1968, Sweet's mentee, Hiroshi Hatanaka, 26 started a Japanese clinical program. Between 1968 and 1993, Hatanaka treated approximately 120 patients with high grade gliomas using a combination of surgery and BNCT. The results obtained by Hatanaka were favourable, suggesting the effectiveness of BNCT.
Nuclear reactors have been the only fission neutron sources for BNCT for decades. Most pre-existing reactors have been adapted to host the equipment for patients’ treatment in suitable rooms built within the reactor hall. The few exceptions are in-hospital-built reactors specifically constructed for medical applications. They include the Massachusetts Institute of Technology Research Reactor, the Brookhaven Medical Research Reactor, the in-hospital neutron irradiator realized in Beijing, and the project in Nakhon Ratchasima, Thailand. The last two examples, which are the more modern, are based on miniature reactor neutron sources (MRNS). Besides these, only a few reactors dedicated to BNCT are active (eg, those located in Taiwan, Japan, Argentina, and China). Most of the other reactors had been dismissed for the use of BNCT.
So far, BNCT has been used for more than 2000 cases worldwide, and the efficacy of BNCT in the treatment of head and neck cancer, malignant meningioma, glioblastoma, cutaneous melanoma and hepatocellular carcinoma has been reported, although large prospective BNCT data remain sparse. 27
Collaborations
All modalities of RT require multidisciplinary collaboration all the way from fundamental physics and chemistry to medicine. In the following section are some examples of collaborations in the RT field.
The Proton Ion Medical Machine Study (PIMMS) collaboration
With the increasing interest in hadron therapy at the beginning of the 1990s, Ugo Amaldi (CERN and founder of TERA Foundation, Italy) and Meinhard Regler (MedAustron, Austria) convinced CERN management to establish a study to identify the technologies available and to be developed to meet the needs of this emerging treatment modality. This resulted in PIMMS. The PIMMS project 28 was carried out under the technical leadership of CERN and coordinated from 1996–2000 by Phil Bryant in collaboration with MedAustron and TERA. The study group worked for a while in collaboration with GSI (Germany) and was later joined by Onkologie 2000 (Czech Republic).
The PIMMS mandate was to design a light-ion hadron therapy centre made of a combination of systems, optimized for the medical application, without considering any financial and/or space limitation.
The outcome of the four-year study was a design that combined many innovative features, capable of providing an extracted pencil beam of particles whose energy can be varied electronically, is very uniform in time and can be easily adjusted in shape, based on a synchrotron accelerator.
The PIMMS design includes several basic elements, and the general structure of the complex is shown in Figure 5. The beams are generated by two ion sources producing protons and carbon ions that are pre-accelerated to an energy of 7 MeV/u by a linac injector. Next, the beams are injected into the synchrotron which has a diameter of about 25 m and accelerates the beams to the pre-set clinical energy, ranging from 60 to 250 MeV for protons and 120 to 400 MeV/u for carbon ions (12C6+).

Schematics of the PIMMS design study 1996–2000 for a light-ion hadron therapy centre 28 ). The main accelerator (a synchrotron) accelerates protons and ions. It receives the pre-accelerated beam from an injection chain, made by a linac which is fed by a source. Ideally two dedicated injector chains, one optimized for protons and one for ions, can be built. The beam from the synchrotron is extracted toward the treatment rooms.
The PIMMS study envisaged three rooms for proton therapy and two rooms for the irradiation of deep tumours with carbon ions. One of them featured a rotating cabin referred to as the Riesenrad gantry. 28 The Italian and Austrian facilities have based their design on the results of the PIMMS studies, which was further optimised by TERA and then implemented at two treatment centres: CNAO (Centro Nazionale Adroterapia Oncologica) in Pavia (Italy), which opened in 2011 29 (see Figure 6), and MedAustron in Wiener Neustadt (Austria), which treated its first patient in 2016 30 (see Figure 7).

CNAO facility in Pavia based on PIMMS design started treating patients in 2011. 29 It is an example of one of the first-generation hadron therapy centres.

Medaustron facility in Wiener Neustadt also based on the PIMMS design treated its first patient in December 2016. 30 It highlights the scalability of the design for advanced treatment.
While the PIMMS study was being pursued, GSI's (GSI Helmholtzzentrum für Schwerionenforschung in Darmstadt, Germany) pilot project for irradiation unit for experimental patient treatment using the heavy ion accelerator SIS was well underway and in December 1997 the first two patients were treated using GSI's raster scanning technique.31,32 This pilot project laid the foundation for the Heidelberg Ion Therapy (HIT) centre (Figure 8) in Heidelberg (Germany), that started treating patients in 2009 and which is equipped with one of the few rotating gantries for carbon ions (there are only three in the world), which allows delivering the dose to the patient from various angles.

Layout of Heidelberg ion therapy (HIT) centre in Heidelberg (left and carbon-ion gantry at HIT, one of the few rotating gantries worldwide, enabling flexible dose delivery from multiple angles. (right). 33
ENLIGHT: Collaboration is key
The idea of creating a multi-disciplinary and transnational platform for researchers and experts involved in hadron therapy was born in 2001 when the PIMMS was presented and the whole idea of setting up specialised centres providing multiple radiation modalities was taking off in Europe. 34 The creation of ENLIGHT (European Network for Light Ion Hadron Therapy) was, indeed, the result of the work of a few visionary people who could see the power of collaboration and knowledge sharing.
ENLIGHT had its inaugural meeting at CERN in February 2002. 35 About 70 specialists from different disciplines, including radiation biology, oncology, physics, and engineering attended this first gathering. 36 At that time, ‘multidisciplinarity’ was not yet a buzzword and the network was a real pioneer in the field. 37
Since its foundation, ENLIGHT, in order to harness the full potential of particle therapy, has relied on the variety of partners and skills of its members to be able to identify and tackle the technical challenges, train young researchers, support innovation and lobby for funding involving industrial partners and clinical centres (see Figure 9).

ENLIGHT is a multidisciplinary collaboration network bringing together physicists, physicians, radiobiologists, engineers, and information technology experts, as well as collaborations among academic, research, and industrial partners in particle therapy (left) and the personalized treatment approach enabled by ENLIGHT's interdisciplinary framework (right).
One of the most enlightening initiatives that the network supported was the organization of conferences devoted to blending scientific backgrounds and expertise with the aim of creating a new culture of collaboration and sharing. Although, at first sight, large accelerators and giant detectors do not seem to have much in common with precision tools that medicine needs, physics is not new to producing applications for life sciences. Several detection techniques are used in diagnostic instruments, and both RT and HT were born in physics laboratories. When ENLIGHT started, there were very few hadron therapy initiatives in Europe. Now the field is blooming and there are around 30 hadron therapy centres in operation in Europe 38 and 125 worldwide (see Figures 10 and 11).

Number of hadron therapy centres and their geographical distribution in Europe in 2023. It can be seen from this map that both the South- Eastern Europe region and the Baltic region do not have a hadron therapy facility.

Map showing the global distribution of particle therapy facilities currently in clinical operation (2023). There are 125 facilities in total of which 110 are proton facilities (in red) and 15 are carbon therapy facilities (in blue). 35
At the end of 2023, the number of treated patients had grown to more than 400,000 (350,000 with protons, 57,000 with carbon). 38 This is due primarily to the greater availability of centres. Until recently, very few randomized studies had been initiated to compare the results of hadron therapy with conventional x-ray therapy.
Role of ENLIGHT in Building Collaboration, Education and Training
The ENLIGHT collaboration inspired a first of its kind conference at CERN called Physics for Health in Europe (PHE) in 2010. Following the great success of PHE, in 2012 the ICTR-PHE conference brought together the International Conference on Translational Research in Radio-Oncology (ICTR) and the Physics for Health in Europe conference which became a winning alliance aiming to unite physics, biology and medicine for better healthcare and to review the most recent advances in translational research. The aim being that developments in basic research are “translated” into means for improving health, namely how to translate from “lab to bed” as quickly as possible.
ENLIGHT's commitment to education features a comprehensive spectrum of initiatives designed to empower the next generation of experts. 39 Through the PARTNER 40 and ENTERVISION Marie-Curie training programs, ENLIGHT has cultivated a vibrant research exchange, encouraging mobility and collaborative learning. The cornerstone of the educational structure includes annual European meetings and specialized training days, which are complemented by competitive poster sessions that spotlight emerging scientific talent. These gatherings not only serve as knowledge hubs but also as arenas for recognizing and nurturing future leaders. Towards the culmination of these efforts, ENLIGHT Highlights and Heavy Ions Therapy Research Integration plus (HITRIplus) seminars 41 stand out as platforms for extensive global information sharing, reflecting ENLIGHT's dedication to a universally informed and interconnected scientific community.
Advances in HT and BNCT Accelerators
Proton therapy is now a fairly mature field while the use of carbon and other ions is still not as widespread as would be hoped, with currently only 15 functioning centres in the world, mostly in Japan (7), the first country to implement carbon ion therapy, China (2), Taiwan (China) (1), Europe (4), and S. Korea (1). Currently, proton therapy is delivered mostly by industrially manufactured 230 MeV cyclotrons and compact synchrotrons of less than 6 × 6 m2. For carbon ions, since the energy required is higher, ie 430 MeV/u, and the beam rigidity is higher by a factor 2.7, the magnets need to be proportionally more powerful, or the machines larger compared to those for protons.
Since the GSI and PIMMS designs, particle accelerator technology has progressed remarkably, especially in the development of high-field superconducting magnets and high gradient cavities. Several designs have been studied in the last 20 years 42 as alternatives to the relatively large synchrotrons with warm magnets, among which are Fixed Field Alternating Gradient (FFAG) accelerators, rapid-cycling synchrotrons, laser-based accelerators and high frequency linacs.
In recent years, the number of reactor-based BNCT facilities has decreased. However, accelerator-based neutron sources have improved and neutron intensities in the order of 1 × 1013 −1014 n/sec or more can be obtained. Because accelerator-based BNCT facilities can be constructed near hospitals, many have been planned and constructed around the world. Proton accelerators with energies ranging from approximately 1.5 MeV to 30 MeV have been used for BNCT.
ABNS for BNCT are structured as follows (Figure 12):
a hardware to generate the high-charged particle beam a neutron-producing target and target heat removal system (HRS) a moderator/reflector assembly to optimize the neutron energy spectrum for patient irradiation
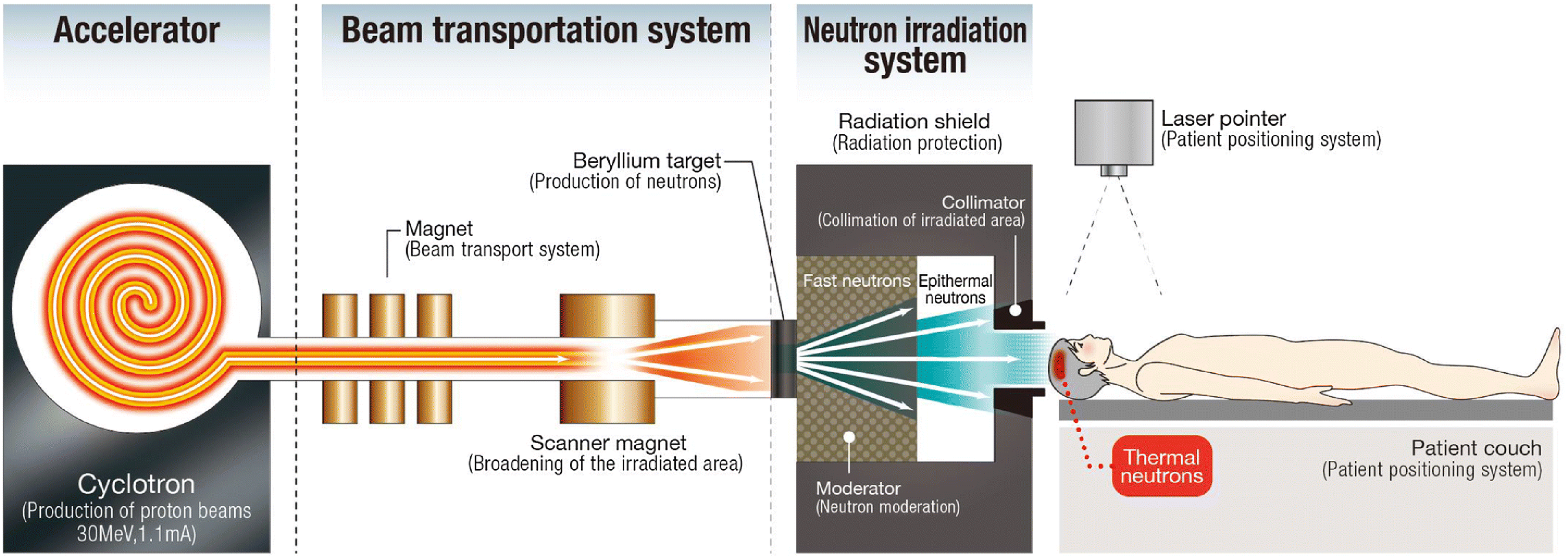
Schematic diagram of an ab-BNCT system showing the principal components of the accelerator, beam transport, and the target and moderator inside the shielded neutron irradiation system emitting a therapeutic neutron ‘beam’ for a carefully positioned patient. 43
The performance of a specific ABNS relies on the reaction among charged particles, which generates neutrons and the clinical requirements of field characteristics. ABNS with a beryllium target requires more effort than a lithium target to get the appropriate field target. On the contrary, lithium targets are more challenging to cool. A valuable option, which has recently been developed, is HRS based on submerged impingement and microchannels. Furthermore, the ABNS performance requirements are proportional to the intensity and quantity of the neutron field. Theoretical calculations have estimated that an ABNS can generate a neutron field of significantly better quality than standard reactors. Moreover, the treatment time is acceptable for the desired boron concentration (about 30 min for a beam current of 10 mA).
ABNS began seeing clinical use and validation at Kyoto University Research Reactor Institute by Sumitomo Heavy Industries Ltd, using a cyclotron and a beryllium target. Based on this experience, a hospital-based system was installed at the Southern Tohoku General Hospital in Koriyama (Fukushima Prefecture) and the same system is now treating patients at the Kansai BNCT Medical Center at Osaka Medical College. Another system using a linear accelerator and a lithium target is from Cancer Intelligence Care Systems, Inc. They are currently performing clinical trials for validation at the National Cancer Center Hospital in Tokyo. In Europe, one facility from Neutron Therapeutics Inc is under commission at Helsinki University Hospital and recently, the Italian government decided that the National Hadron Therapy Center in Pavia would house a BNCT facility. More details about the 25 facilities under development worldwide can be found in ref. 44
Examples of Present and Future Developments
In the following sections there is a look into the future describing both for existing facilities, those which are going to implement new devices and adopting novel treatment options, and for emerging and newly conceived facilities, profiting from the history already depicted by the current hadron therapy centres. The authors have chosen three examples of current innovation to illustrate projects with varying timescales of implementation, maturity level and purpose. CNAO, one of the original first generation large HT centres as well as one of the longstanding key players in the ENLIGHT network, it is now going through extensive upgrades and expansion, including accelerator-based BNCT. The proton therapy linac machines are an example of accelerators that after being developed through international collaboration in research centres, are being implemented in industrial settings. On a longer timescale, second generation large hadron therapy centres are being developed, helped by the collaboration of scientific and medical communities to become centres of excellence for research and therapy for the South-East Europe region 45 and the Baltic region. 46
Expansion Project at the National Centre for Oncological Hadrontherapy (CNAO)
The Italian HT facility, CNAO, is based on a strong collaboration network. In over 10 years of its clinical activity, CNAO has treated more than 5000 patients and has acquired an outstanding profile and visibility on the international community for hadron therapy and precision radiotherapy. From 2022 CNAO premises are expanding with a new building constructed next to and integrated with the existing building that is ready to house a modern system for protons using the latest gantry technology in an area reserved to oncological research. 47 For the first time in Italy, a particle accelerator for the production of neutron beams will be installed in a space dedicated to clinical and medical research. The neutron accelerator, part of TLS's Alphabeam™ Neutron System, will be used to deliver BNCT.
The Expansion Project also offers a unique opportunity to implement the actual area devoted to research.
In the framework for research purposes, a third ion source has been designed, 48 installed and will be exploited soon to provide oxygen, lithium, helium and iron beams, not only for radiobiological and clinical research, aiming to personalize tumour treatments, but also for the aerospace industry that is interested in iron beams, suitable to simulate very high dose radiation environments.
With the implementation of the new technologies included in the Expansion Project, CNAO will offer the most advanced and effective therapy against challenging cancers and will leverage interest in the scientific community for a complete spectrum of particle therapy options, promoting a multidisciplinary approach for personalized and optimized treatment of specific tumours, linked to their histology and main characteristics, teaming up with the key research institutes and promoting a multiparticle therapy approach as a successful paradigm in the near future.
BNCT: a new Treatment Option at CNAO
The system to be installed in CNAO (schematically represented in Figure 13) consists of a neutron source produced by a high current (nominally 10 mA) beam of protons at a nominal operational energy of 2.5 MeV and a lithium target. A compact tandem accelerator Vacuum Insulated Tandem Accelerator (VITA), created at the Budker Institute of Nuclear Physics in Novosibirsk, Russia) 49 will produce a DC current at the required energy and intensity.

The alphabeam™ TAE life science technology for BNCT to be installed at CNAO. The system integrates a compact tandem accelerator with advanced lithium targets for precise neutron production.
The accelerator is called “tandem” because it uses the same accelerating voltage twice. Negative hydrogen ions are injected into the entrance and accelerated by a positive potential gradient toward the centre of the cylindrical geometry. A charge exchanger located at the centre of the accelerator strips both electrons from the negative ions, converting them to positively charged protons. The same voltage gradient that accelerated the negative ions towards the centre now accelerates the protons away from the centre to the exit on the opposite side from the entrance. The accelerating voltage is reduced to half the nominal voltage, allowing a more compact accelerator by simplifying the electrostatic insulation requirements. The accelerator voltage gradient is created with a series of nested electrodes and a high voltage power supply, connected by a high voltage feedthrough column. The power supply and lower portion of the feedthrough are housed in an electrostatic vessel (ELV) that extends below the tandem accelerator. Electrostatic breakdown in the accelerator and ELV is prevented using a combination of vacuum and dielectric gas (SF6).
The facility configuration at CNAO requires a series of two 90-degree bending (dipole) magnets: a single beam splitter sending either the beam left or right from a single magnet is not possible due to neutrons back streaming from each target to the opposite room. The beam dump is designed to accept the full beam current as needed in the event of magnet power loss and is also used during system commissioning.
Particle Therapy and High Frequency Linacs
The Rationale of High Frequency Linacs for Protons and Light Ions
One example of technological developments in proton and hadron therapy is represented by the high-frequency Radio Frequency (RF) linac, where the same technology used for conventional radiotherapy linacs has been adapted and tailored for the purpose of proton and light ions acceleration.
While cyclotrons and synchrotrons have been used in the past seventy years to accelerate protons and light ions beams for tumour treatments - first in physics laboratories and since the nineties in dedicated treatment centres – only recently have linacs been designed for such a purpose. 50 This idea arose from two considerations: i) the linac technology is widely used in x-ray clinical machines and ii) beam currents of few nA are enough to treat deep seated tumours.
Considering the first point, the linac technology is widespread in hospitals worldwide - but in Low-and-Medium Income Countries (LMIC) the access to this technology is still inadequate.9–12 Originally developed in the late ‘40 s, the modern linacs used in conventional x-rays therapy are based on normal conducting RF accelerating structures made of copper and working at a frequency of about 3 GHz. These linacs are used to accelerate electrons up to 12–18 MeV that then hit a target made of high Z material (typically a tungsten alloy) to produce megavoltage x-rays beams (Figure 14).
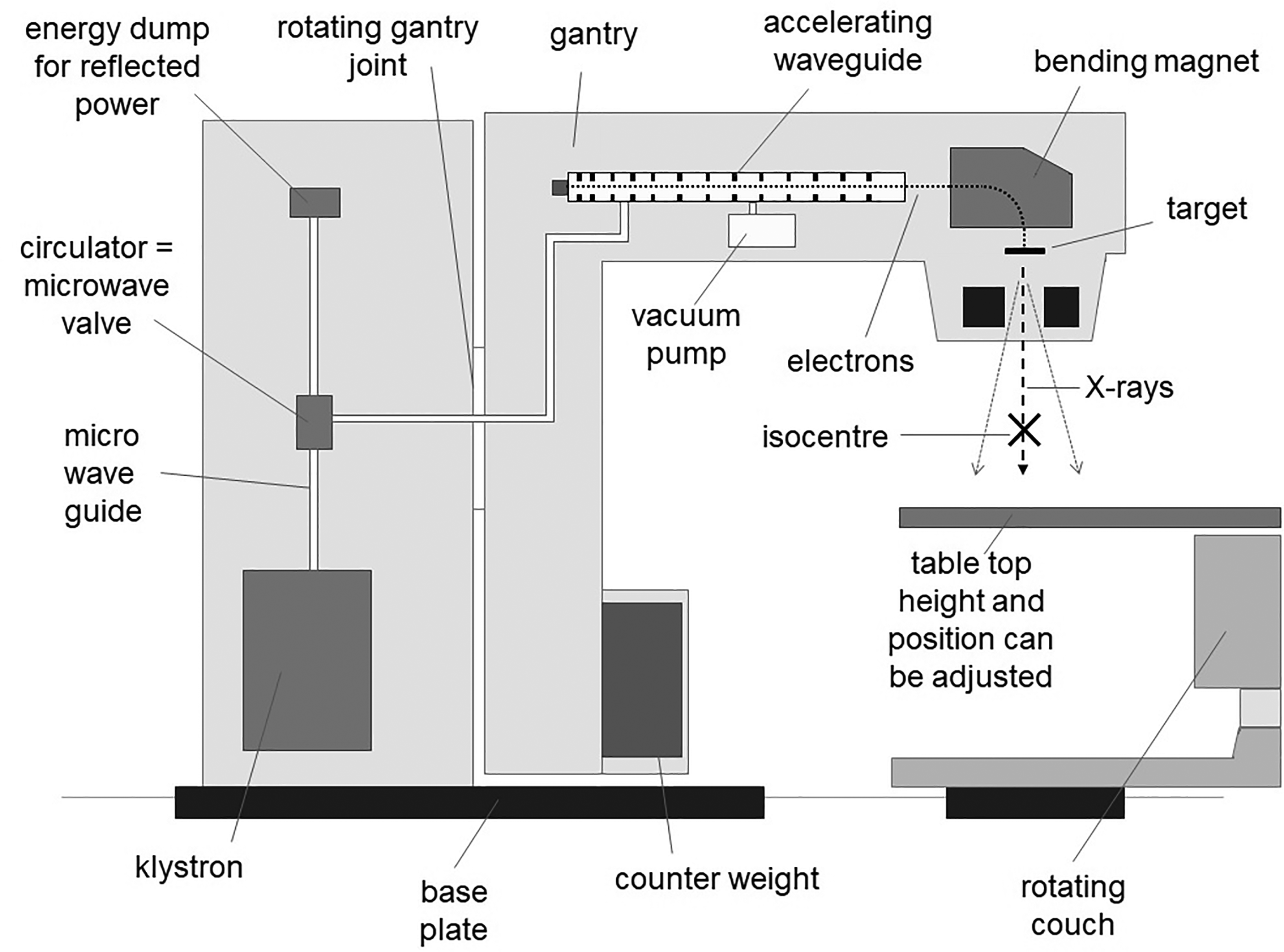
Schematic of modern radiotherapy linac design mounted on a gantry. This structure accelerates electrons to produce megavoltage x-ray beams for conventional radiotherapy (source IAEA).
Related to the second point, the linacs for protons and light ions typically run at frequencies of few hundred MHz. This is because they are designed for high current applications and typically require a large bore hole aperture. However, for tumour therapy average beam currents of the order of 1 nA for protons and 0.2 nA for carbon ions are needed. This allows the choice of small-bore hole diameters (5–8 mm) and the use of high frequency RF structures. Starting from this initial consideration, the same technology of conventional RT machine has been proposed in the early nineties for the use with proton and ion beams.51,52
A design based on normal conducting RF cavities with frequencies as high as the typical 3 GHz used in electron linacs (and in certain cases even higher), allows to keep the transverse size of the modules rather small and to achieve accelerating gradients of the order of 15–20 MV/m. With such a gradient inside the copper cavities, and accounting for a filling factor of accelerating structures along the linac of about 70% (ie the fraction of space occupied by the RF cavities with respect to the total linac length), the beam can be accelerated up to its full energy of 230 MeV for protons in a length of about 25 m.
Instead of one accelerating structure (as in the RT linacs), the proton therapy linacs are made of several accelerating modules that are designed to follow the increasing protons speed from the initial energy from the source up to about 60% of the speed of light, ie 180 m/µs, at maximum energy. The modularity of the linac represents one of the advantages of this approach, compared to other accelerator technologies. The linac can be installed module by module and deployed in stages, starting with the low energy section (for ocular and paediatric tumours) and then completed up to full energy.
Another aspect related to the linac modularity is the possibility of coupling it with a cyclotron, in what has been named cyclinac approach. In this configuration, the linac is used as energy booster of a low energy cyclotron. The cyclotron beam (at around 30 MeV) can be used for radioisotope production used in medical imaging, while the linac is used to reach the higher energies required to treat deep seated tumours.
Apart from the modularity and flexibility offered by the linac design, the most important feature of this approach is the fast and active beam energy modulation. The high frequency linac modules are powered independently by klystrons pulsed at few hundreds Hz. This allows to achieve variable energy beams that can be electronically modulated between 70 and 230 MeV on a pulse-to-pulse basis (within few ms) without the need for an absorber or degrader and enabling fast longitudinal tumour painting
More details about the rationale and the basic design concept of hadron therapy linacs are described in article. 53 For the future, there are still opportunities for improvement for proton therapy linacs, in particular to reduce the power consumption (by the use of cutting edge technological developments on high-efficiency klystrons) or to fully exploit the fast energy modulation capability (by combining the linac with high momentum acceptance transfer lines). 54
Synergies and Challenges in Translation from Research to Commercial and Clinical Implementation
The development of proton therapy linacs is being initiated in research laboratories as an R&D proposal and has advanced through small unit testing and prototyping. From the first prototypes sub-units and modules, it has then passed to industry.55,56
In the late 80 s and in the early 90 s, few proposals were made for the design of linac solutions for proton therapy. At the end of the 90's the TERA Foundation built the first prototype of a high-frequency proton linac in collaboration with CERN and INFN. 57 This proof of principle module was called LIBO (LInac BOoster). It consisted of a 3 GHz Cell Coupled Linac made of 4 “tanks”, in which the average accelerating field of 16 MV/m accelerated protons from 62 to 74 MeV in about 1 meter. The module was tested experimentally with beam in a cyclotron + linac setup formed by the INFN superconducting cyclotron in Catania and LIBO. 58
Following this first demonstration, several designs and construction projects were launched. Three full scale machines have been designed and partially built in the past decade: IMPLART (Intensity Modulated Proton Linear Accelerator for Therapy) at the ENEA laboratories in Frascati, where in 2020 protons have been accelerated to 35 MeV. 59 In southern Italy, near Bari, the company LinearBeam (spin-off from ITEL) has developed and implemented the Enhanced Radiotherapy with Hadrons (ERHA) system (Figure 15), an innovative proton therapy system for cancer treatment. 55 Finally the company Applications of Detector and Accelerators to Medicine (ADAM-Geneva) - a subsidiary of Advanced Oncotherapy (AVO-London) – has built LIGHT (Linac for Image Guided Hadron Therapy) as a commercial high-frequency proton linac (Figure 15) inspired by technologies developed by TERA and CERN. 56 A first working prototype 60 was commissioned in Geneva in 2018 up to an energy of 53 MeV and a full-scale machine has been installed in Daresbury (UK) on the campus of the Science and Technology Facility Council (STFC) laboratories and reached 230 MeV in September 2022. In 2023 the LIGHT project was stopped due to lack of fundings.

(Top) LIGHT linac from AVO-ADAM, an innovative high-frequency proton linac. (Bottom) ERHA system from Linear Beam, representing advancements in compact linac designs for cancer treatment. 55
All these projects, that span over more than a decade, show the difficulty in bridging the gap from a working prototype to an industrial medical device product, where regulatory and safety aspects impose important constraints and ask for a considerable financial effort to support both the hardware and software design and integration activities. The importance of collaborating and finding synergies between public and private sector is crucial for such endeavours.
Despite these challenges, the linac technology for hadrontherapy continues to improve and some recent developments include the adaptation of the 750 MHz Radio Frequency Quadrupole (RFQ) design from CERN to the acceleration of fully stripped carbon ions (C6+, Z/A = 6/12 = 1/2) as a joint collaboration between CERN and CIEMAT, 61 as well as novel designs developed within the NIMMS (New Ion Medical Machine Study) collaboration at CERN. 62
Furthermore some key characteristics of the high frequency linac beams, like the reduced beam emittance (thanks to the absence of absorbers and degrader), make the linac option very valuable for novel approaches such as mini-beam particle therapy or more generally spatially fracitonated particle therapy, that are still at the pre-clinical stage.63,64
Another important development area for high frequency hadron linacs is the use for Ultra-High Dose Rate (UHDR) or FLASH therapy, 65 with dose rates above 40 Gy/s and delivery times of few hundred ms. The peak current within each pulse allows achievement of an extremely high dose rate during the pulse, which would be enough for delivery of small volumes (∼cm3) at FLASH regime with the Bragg Peak (rather than in transmission mode). Further improvements are needed to increase the duty factor and to extend this modality to larger volumes. The research and development that is ongoing in the monitoring and control of pulsed charged beams for FLASH Very High Energy Electrons (VHEE) projects would also be important and could be transferred to proton and light ions linacs.
Next Generation Ion Synchrotrons for EU Partner Consortia
The flexibility of a synchrotron allows for the possibility to accelerate different species of ions, of interest for therapy and for research, among which protons, helium, carbon and oxygen ions. It can switch between different species within less than a minute. Moreover, as for linacs, they provide the option to change the beam energy without the need of beam degraders, thus avoiding transmission losses and radio-protection constraints.
The development is taking place within the NIMMS initiative 62 and its core team based at CERN which, in addition to the TERA Foundation and its Swiss spring Tera-Care, includes possible partners which are interested in implementing the design in their region.
Here we discuss three synchrotron solutions, developed within the EU Integrating Activity HITRIplus 66 and NIMMS, and most important, the consortia potentially interested in adopting these developments.
High Intensity Synchrotron for the SEEIIST International Consortium
The first of these partners is the South East Europe International Institute for Sustainable Technology (SEEIIST), a consortium of the countries in the SEE region to build a common facility and to federate among a common endeavour. 67
SEEIIST follows the same model of “Science for Peace” and “Science for Diplomacy” of CERN and SESAME, 68 which are large-scale scientific infrastructures to promote peace, collaboration and scientific excellence.
The three main goals of SEEIIST are 69 : to foster scientific and technological excellence in South-Eastern Europe and help reverse the brain drain, to build a culture of peace through scientific collaboration in a region torn apart by the Balkan war and to enhance the tools and knowledge needed to fight cancer at European level.
From Figure 10, one can clearly see that there are no hadron-therapy centres in South East Europe with a population of over 40 million even though the number of such facilities is growing worldwide. Patients needing such treatment have to be sent abroad.
Seven out of the ten countries in the SEEIIST initiative are middle-income countries and are facing common challenges in fighting cancer due to the lack of national and regional strategies, organized reliable screening programs for early cancer detection, adequate state-of-the-art diagnostic and treatment equipment and advanced treatment. SEEIIST will foster innovative RT research and technology in the region to address the current challenges. 69 It has to put in place schemes to attract junior engineers and physicists from the SEE region, so that they can receive training and eventually constitute the core of the technical team that will build and ensure the facility's successful operation.
The baseline chosen by SEEIIST is an advanced synchrotron with a similar layout as the PIMMS design but with a factor of 20 larger intensity42,45 such that it will store and accelerate in a single cycle up to 2 1010 carbon ions and equivalent intensities for all the other therapy ions, corresponding to a radiation dose of 2 Gy in a 1-liter target. This allows faster extraction modalities such as the Multi-Energy Extraction, pioneered in Japan 70 to maximize the duty cycle by avoiding the 2-s waiting time between synchrotron fillings with the possibility to do FLASH extraction.
Contrary to the other hadron therapy facilities, for which the daytime is fully dedicated to treatment, SEEIIST's distinctive characteristic is that it will be also a research institution, attracting a community of users in the field of radiobiology, material science and medical physics research while providing RT to cancer patients in the SEE region.
Figure 16 shows the preliminary layout of the SEEIIST facility, 71 whose footprint is about 6500 m2.

Preliminary bunker layout of the SEEIIST facility. 71 the main accelerator is an advanced synchrotron with warm magnets, fed by a linac and 3 sources: for protons, ions and high-intensity carbon ions. The beam is sent to 3 treatment rooms or to an experimental Hall that can be reconfigured according to the needs.
The main principle adopted is to separate the clinical and experimental areas. The facility features three treatment rooms, one equipped with a horizontal beamline, one with an horizontal and a vertical beamline, and one with a carbon-ion rotating gantry, as shown in Figure 16. For the experimental area the chosen solution is a reconfigurable hall with two beamlines with the possibility to extend them and/or split them with further magnets and shielding blocks that can be rearranged according to the requirements of the experiments.
The facility is based on an advanced synchrotron with resistive (warm) magnets, fed by a 5–7 MeV/u injector linac and three ion sources, providing ions for treatment (proton, carbon, helium, and oxygen) and for experiments (up to argon).
Superconducting Magnet Compact Ring for SEEIIST Advanced Option
Reducing the dimensions of an ion accelerator from a circumference of about 70 m (such as the PIMMS design), down to 35 m requires the introduction of technologies that are presently beyond state-of-the-art, such as strongly curved and fast ramping superconducting magnets. Superconductivity is well known for large accelerators, such as the CERN Large Hadron Collider, but needs substantial development in the design and manufacturing of more than 300 bent magnets and ramping within few tens of seconds, ie with ramp rates of at least 0.4 T/s.
In 2018, a design of a compact gantry and synchrotron based on superconducting technology was developed by the TERA Foundation, 72 based on 900 magnets at 3.5 to 4 T field. For this specific application, the chosen technology is Canted-Cosine-Theta magnets including nested Alternating-Gradient quadrupoles (AG-CCT) for combined deflection and focusing based on a development going on at LBNL for proton therapy gantries. 73
The development is ongoing within HITRIplus for what concerns the synchrotron 74 and within both HITRIplus and another EU program for the R&D on the superconducting magnets. 75
Compact Helium-ion Synchrotron for the Baltic Countries
The region of North-East Europe, ie the Baltic region, also does not have any particle therapy facility. The consortium of the three Baltic countries is already used to working together within scientific projects, eg in CERN. It is preparing to gather together to build a facility for therapy and research in the Baltic region following the example of SEEIIST, but focusing more on the technological and innovation development of the area. 46
The consortium will exploit the potential of He-ion therapy by making use of the concept of a compact facility based on a synchrotron optimised for helium beams74,76 recently developed by the NIMMS collaboration with the contribution from HITRIplus.
The accelerator concept is like the one developed for carbon-ions using superconducting technology. It has a triangular layout, but with warm magnets, which implies that the beam size in the vertical plane needs to be smaller than in the horizontal plane (for the SC-magnet version the aperture was round). The main challenge is compactness, ie to reduce the size so as to be able to fit all the essential hardware within the available length. Figure 17 shows a sketch of the layout with an overall “circumference” of 33 m.

Sketch of the he-ion synchrotron layout 74 design which is taken as baseline for the Baltic project. 46 in blue are the bending magnets, in green the quadrupoles and in light blue the sextupoles. The injection (ESI, MSI) and extraction septa (ESE, MSE) (electrostatic and magnetic) are in orange and the RF cavity in red.
To develop this innovative collaborative facility, the Baltic countries have started exploring the feasibility of such advanced particle centre from three perspectives: i) clinical and medical needs, ii) technological and technical integration, iii) socio-economical synergies. These will be the main topics for a feasibility study in the coming years. 46
Current Topics for RT and HT
After 70 years, HT is still an active and innovative field and many new advances are happening as this review is being written. The recent interest and advances in FLASH, Mini beams, Radioactive beams (theragnostics for simultaneous imaging and treatment), BNCT (see Section on BNCT), helium-ion facilities (see Section on Synchrotron and the future) and SC-magnet-guided proton therapy could be real break-throughs in the field and push it forward beyond current state-of-the-art to optimally and effectively deliver HT.
FLASH Therapy
The FLASH effect is a fairly recently discovered phenomenon observed in numerous in vivo radiobiological and animal studies which have demonstrated that delivering the prescribed dose of radiation at an UHDR causes less damage to healthy tissue than when it is delivered at conventional dose rates, while still maintaining the same tumour control efficacy.77,78 Determining the dose delivery parameters at which the FLASH effect is observable is still a huge area of research. Current preclinical data seem to suggest that delivering doses in excess of 10 Gy within a total delivery time of <500 ms, at mean dose rates of > 40 Gy/s, are reasonable values to ensure a significant FLASH effect. 79 FLASH effect offers a promising new paradigm in radiotherapy. New opportunities for improved treatment outcomes with FLASH have recently emerged in connection with new biological mechanisms for improving the radiation effectiveness while decreasing the normal tissue damage and expanding the portfolio of usable radiation beams beyond protons and carbon ions. 80
Research and preclinical studies have shown that FLASH radiotherapy is less toxic to normal tissues and is as effective as conventional radiotherapy at destroying tumours. It therefore has the potential to revolutionize radiotherapy – such that higher doses could be delivered safely to tumours or established doses delivered with fewer fractions resulting in reduced toxicity to OARs. Therefore, FLASH therapy could reduce radiation treatment times allowing higher patient through-put. At a more fundamental level, FLASH irradiation might also trigger different immune pathways and gene expression, creating novel opportunities for radiation-drug?? combinations. However, FLASH therapy is not ready to be deployed in practice but holds promise in the near future.
Image-Guided RT
Following the MRI-guided linac RT, we now have MRI-guided proton therapy developed by the Dresden group allowing radiotherapy that is based on real-time and on-line patient anatomy. Thisallows the clinical team to maximize the dose to the target volume while minimizing th dose to OARs, a big step towards personalised treatment.
Immunotherapy and RT
Another hot topic in cancer treatment is the combined-modality synergies of integrating immunotherapy and radiotherapy. Formenti's group 81 is one of the main-movers behind a paradigm shift in radiobiology with their efforts elucidating the role of ionizing radiation on the immune system while demonstrating the efficacy of combined radiotherapy–immunotherapy regimes in the treatment of solid tumours.
Clinical Data
A number of randomized phase III clinical trials with protons are accruing data or have recently closed for cancers including lung, oesophageal, liver, head-and-neck and the brain. Meanwhile, patient data are also being accrued to evaluate proton treatment in routine clinical practice for patients with breast and prostate cancers.
The Future for ENLIGHT
It is clear that the focus of R&D for hadron therapy has shifted since the birth of ENLIGHT, if only for the simple reason that the number of clinical centres (in particular for protons) has dramatically increased. Currently, in Europe there are around 30 centres and a similar trend is reflected globally where there are around 130 hadron therapy centres, only 15 of which are carbon centres including 4 located in Europe (see Figure 10).
Over its 20 years of life, ENLIGHT has shown a remarkable ability to reinvent itself while maintaining its cornerstones of multi-disciplinarity, integration, openness and attention to the future generations. ENLIGHT is ready to face the evolving challenges of a frontier discipline such as hadron therapy.
Conclusion
It is important to note that, while technological developments are still needed to ensure safer and more cost-effective treatment, proton therapy is now solidly in the hands of clinicians supported by industry. The advent of single-room PT facilities is bringing proton therapy to patients in smaller hospitals and clinical centres. From the clinical standpoint, the major challenge in the coming years will be to catalyse collaborative efforts in defining a roadmap for randomised trials to study in detail the issue of RBE. Concerning technology developments, efforts will continue on quality assurance through imaging and in the design of compact accelerators and gantries for ions heavier than protons.
Information technologies and Artificial Intelligence (AI) will take centre stage as data sharing, data analytics and decision-support systems become key topics for innovation and development. As this review highlights, the role of multidisciplinary collaboration is fundamental—not only in driving innovation but also in ensuring equitable access to these advanced hadron therapies worldwide. By leveraging collective expertise and fostering global partnerships, the field of hadron therapy will continue to build on its strong foundation, addressing current challenges and opening new horizons for cancer treatment. 82
Footnotes
Abbreviations
Author's contributions
All authors contributed to the conceptualization, drafting and preparing and finalising the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
