Abstract
The novel grading system developed by the International Association for the Study of Lung Cancer (IASLC) for clinical stage IA lung adenocarcinomas has demonstrated remarkable prognostic capabilities. Notably, tumors classified as grade 3 have been associated with poor prognostic outcomes, thereby playing a crucial role in the formulation of personalized surgical strategies. The objective of this study is to develop a radiomics nomogram that utilizes the optimal volume of interest (VOI) derived from high-resolution CT (HRCT) scans to accurately predict the presence of grade 3 tumors in patients with clinical IA lung adenocarcinomas.
In this multi-center, large-population study, clinical, pathological, and HRCT imaging data from 1418 patients who were pathologically diagnosed with lung adenocarcinomas were retrospectively collected. The data was obtained from four hospital databases between January 2018 and May 2022. From this patient cohort, 1206 individuals were screened from three databases and randomly divided into training and internal validation datasets in a 7:3 ratio. An additional dataset consisting of 212 individuals was used for external validation dataset. Radiomics features were extracted from HRCT images at various scales, including VOI−2mm, VOI entire, VOI +2mm, and VOI +4mm. To reduce dimensionality, select relevant features, and build radiomics signatures, the maximal redundancy minimal relevance (mRMR) and least absolute shrinkage and selection operator (LASSO) algorithm were utilized. Univariate and multivariate logistic regression analyses were conducted to identify independent clinic-radiological (Clin-Rad) predictors. Receiver operating characteristic (ROC) curves and corresponding area under the curve (AUC) were used to evaluate the diagnostic efficiency. A nomogram predicting the risk of grade 3 in clinical stage IA lung adenocarcinoma was constructed based on multivariate logistic regression, combining independent predictors and the optimal radiomics signatures.
Multivariate logistic regression revealed that males exhibited a higher prevalence of grade 3 tumors, and solid nodules were frequently observed through radiological assessments. The utilization of radiomics features extracted from the VOI entire resulted in significant improvements in predictive performance, as evidenced by AUC values of 0.900 (0.880-0.942), 0.885 (0.824-0.946), and 0.888 (0.782-0.993) for the training, internal validation, and external validation datasets, respectively. Furthermore, the nomogram that combined VOI entire -based radiomics signatures and Clin-Rad characteristics, exhibited remarkable predictive performance. This was indicated by AUC values of 0.910(0.873-0.942), 0.891 (0.845-0.937), and 0.905 (0.846-0.964) for the training, internal validation, and external validation datasets, respectively.
The extraction of radiomics features from both the indented and peri-tumoral regions does not offer any additional benefits in predicting grade 3 tumors according to the IASLC system. However, when combining the VOI entire-based radiomics model with Clin-Rad characteristics, the resulting integrated nomogram exhibited remarkable predictive performance.
Keywords
Introduction
Lung adenocarcinomas, even at clinical stage I, exhibit heterogeneous histologic features, with approximately 80% to 90% displaying a combination of various patterns. 1 The histological classification of adenocarcinomas established by World Health Organization (WHO) in 2015 characterized these tumors according to their distinct growth patterns, which include lepidic, papillary, acinar, micropapillary, and solid-predominant types. Research has demonstrated that these patterns are associated with patient prognosis.2–5 In 2020, the Pathology Committee of the International Association for the Study of Lung Cancer (IASLC) proposed a refined three-tier grading system that considers both the predominant histologic subtype and specific high-grade components, such as micropapillary or solid -predominant. 6 According to the updated grading system criteria, grade 1 now includes a lepidic-predominant component with less than 20% high-grade patterns, grade 2 comprises a papillary or acinar-predominant component with less than 20% high-grade patterns, and grade 3 encompasses any component with 20% or more high-grade patterns. 6 Subsequently, the fifth edition of the WHO thoracic tumor classification, released in 2021, incorporated this revised grading system. 7 Further validation studies have confirmed that this comprehensive approach improves prognostic efficacy compared to the WHO 2015 classification, which was based solely on the predominant subtype.8–11
The IASLC system provides important guidance for determining the most effective treatment regimens for lung adenocarcinomas. In case of grade 3 tumors, which are characterized by their aggressive behavior and poor prognosis, the utilization of segmentectomy is restricted. Instead, lobectomy combined with systemic mediastinal lymph node dissection is recommended.12,13 Additionally, these patients may benefit from adjuvant chemotherapy. 14 Therefore, preoperative diagnosis of grade 3 tumors could facilitate surgeons in selecting the most suitable surgical interventions.
Currently, puncture biopsy and intraoperative rapid frozen pathology are important approaches in diagnosing lung adenocarcinoma. However, these techniques may lead to inadequate preoperative diagnosis and false negative outcomes, primarily due to limited sample sizes and variations in operator expertise. Additionally, common complications such as pneumothorax, hemorrhage, and pleural effusions should not be overlooked. 15 Studies have indicated that clinic-radiological (Clin-Rad) characteristics, including air bronchogram, consolidation/tumor ratio, and margin assessment, can help predict the IASLC grading system preoperatively.16,17 Meanwhile, Fujikawa et al 18 identified a higher maximum standardized uptake value (SUVmax) on PET as a significant preoperative predictive factor for grade 3 tumors, achieving a prediction rate exceeding 90%. However, it is important to note that Clin-Rad and PET SUVmax may have a certain level of subjectivity, lack representativeness, and incur substantial costs can be costly.
The radiomics approach, which quantifies the size, shape, margin, and internal density of tumors as imaging biomarkers, has shown potential in various aspects of lung cancer research, including tissue classification, oncogenetic correlations, treatment response, and disease prognosis.19,20 Yang et al 21 found that a nomogram integrating CT-based intratumoral radiomics and Clin-Rad resulted in improved predictive performance for the preoperative assessment of the IASLC grading system in lung adenocarcinoma.
Invasion and metastasis represent critical biological characteristics of malignant tumors. Consequently, peritumoral radiomics features have gained increasing attention due to their potential to capture proliferation, migration, invasion, and microenvironmental changes, which are common characteristics of malignancies. Several studies have successfully extracted radiomics features from both intratumoral and peritumoral regions, leading to improved diagnostic efficiency in predicting various aspects of lung adenocarcinoma, such as invasiveness in ground-glass nodules (GGNs), 22 preoperative lymphovascular invasion, 23 lymph node metastasis, 24 and EGFR mutation assessment. 25 However, there is currently a lack of literatures systematically evaluating the radiomics model of multi-scale peritumoral ranges of lung nodules for the preoperative prediction of IASLC grading in clinical stage IA lung adenocarcinomas.
To achieve successful radiomics outcomes, it is crucial to develop predictive models that are reliable, highly accurate, and efficient. In pursuit of this objective, we conducted a multi-center study involving a large population to ensure the robustness and predictive capacity of our models. Our focus was on deriving radiomics features from both intratumoral and peritumoral regions using various volumes of interest (VOIs). To determine the optimal VOI, we performed a comprehensive comparison and selected the most effective combination for constructing a nomogram, thereby enhancing the accuracy of predicting IASLC grading in clinical stage IA lung adenocarcinomas.
Method
Enrollment of Patients
In this multi-center, large-population study, a retrospective analysis was conducted on the clinical, pathological, and HRCT images of patients who underwent surgical resection for lung adenocarcinomas between January 2018 and May 2022 across four hospitals. The inclusion criteria were as follows: (1) Clinical stage IA (cT1N0M0) with a maximum nodule diameter of ≤3 cm; (2) patients who had undergone chest CT within 7 days prior to surgery; (3) availability of HRCT with specified slice thickness; (4) no prior history of chemotherapy or radiotherapy. The exclusion criteria included: (1) the presence of multiple lesions; (2) tissue samples obtained via biopsy rather than surgical resection; and (3) the concurrent presence of other malignant tumors.
Subsequently, after a thorough screening process, a total of 1206 individuals were recruited from three distinct databases: Database 1 (n = 576), Database 2 (n = 439), and Database 3 (n = 191). These individuals were then randomly allocated to form both the training (n = 844) and internal validation (n = 362) datasets, adhering toa ratio of 7:3. Additionally, an independent validation dataset consisting of 212 individuals was established from Database 4 (Figure 1). This study received approval from the local research ethics committee, with the protocol identification numbers: KY2020147, 2021-07-009, 22/244-3446, 2023YJSLL-121, and KY-2024-231. Given the retrospective nature of our study, informed consent was waived.

Schematic representation of the screening process for enrolled patients.
Histopathology Re-Evaluation
The hematoxylin- and eosin-stained slides from all enrolled patients were re-evaluated and graded according to the IASLC 2020 classification. The assessment was conducted by two experienced pathologists at each participating institution, each with over a decade of expertise in pulmonary pathology, using a multi-head microscope. Consensus on the evaluation was reached through collaborative discussion. In addition to calculating the percentages of the five primary histological subtypes in 5% increments according to the 2015 WHO guidelines, we also examined non-traditional complex glandular patterns. 2 These patterns included cribriform formations, characterized by nests of neoplastic cells exhibiting sieve-like perforations, and fused gland structures, which are defined as poorly developed fused glands that lack intervening stroma or appear in a ribbon-like arrangement with irregular boundaries, accompanied by single cells infiltrating desmoplastic stroma. Individuals were graded according to the IASLC 2020 classification system 6 and categorized into two groups based on histopathology evaluation results (Group1: grade1-2; Group2: grade3). Furthermore, other aggressive pathological features such as visceral pleural invasion (VPI), spread through air spaces (STAS), mediastinal lymph node metastasis (LN), and vascular invasion, were also collected.
HRCT Radiological Features Evaluation
A flowchart illustrating the workflow of the study is presented in Figure 2. For details regarding the acquisition of HRCT images, please refer to Supplemental Digital Content 1. Non-enhanced chest HRCT scans in DICOM format were imported into the RadiAnt DICOM Viewer (https://www.radiantviewer.com) to visualize the radiological features of the tumor through multi-planar reconstruction (MPR) and maximum intensity projection (MIP). Two thoracic radiologists, with 10 and 15 years of experience, respectively, assessed and measured the tumors both qualitatively and quantitatively. In case of disagreements regarding qualitative indicators, a consensus was reached through consultation. Quantitative parameters were analyzed based on the average measurements provided by the two radiologists. If any discrepancies arise during the discussion, a consensus will subsequently be achieved. HRCT images were utilized to characterize the lesions in terms of sizes density, shapes boundary, spiculation, lobulation, vascular convergence sign, and vacuole sign. For a comprehensive understanding of the HRCT radiological features, please refer to Figure 3, which summarizes their respective definitions.

Workflow diagram illustrating the methodological approach employed in the study.

Compendious overview of High-Resolution Computed Tomography (HRCT) radiological features along with their respective definitions.
CT Images Preprocessing and Tumor Segmentation
The ITK-SNAP software (version 4.0, www.itksnap.org) was utilized for the slice-by-slice delineation of the gross tumor volume along the tumor boundary, with the 3D region defined as the VOIentire. The initial delineation of the VOI was conducted on CT images under a lung-specific CT window (width: 1500 HU; level: −500 HU). The tumor boundary was identified by a certified cardiothoracic radiologist with 10 years of experience, and the VOI was manually segmented across all two-dimensional sections of the nodule. Subsequently, another radiologist with 15 years of experience in the same field reviewed and adjusted the lesion delineation as necessary.
Previous research has established that an extension of 2 to 4 mm from the apparent border of a tumor is considered as the zone of microscopic disease extension and is recognized as an aggressive region. It has been observed that beyond this distance, there is no significant enhancement in diagnostic performance.26,27 Therefore, in this study, the multi-scale peritumoral ranges were defined by shrinking or expanding distances of −2 mm, 2 mm, and 4 mm from the tumor boundary, designated as VOI−2mm, VOI + 2mm, and VOI + 4mm, respectively. These regions were generated using pixel filtering on a Python platform (version 3.11, https://www.python.org). During the process of obtaining these VOIs, vessels, pleura, the chest wall, mediastinum, and bronchus were intentionally excluded based on appropriate pixel thresholding. Artificial exclusion of these structures was conducted for the defined VOIs when necessary.22–27
Image Preprocessing and Radiomics Feature Extraction
After incorporating CT images from multiple hospitals and utilizing various CT protocols, we implemented a comprehensive image preprocessing pipeline aimed at mitigating the variability of associated with different CT scanners and acquisition parameters. This pipeline included several key steps. First, we performed image resampling using the PyRadiomics package within the Python programming environment to standardize voxel spacing to 1 × 1 × 1 mm³ (x, y, z). The resampling algorithm employed was based on the B-spline curve, with the interpolator set to sitkBSpline (= 3), ensuring smooth and accurate interpolation of the image data.
Next, we adjusted the bin width to partition the pixel intensity range of 0–255 into five intervals. This binning approach effectively discretized the intensity values, thereby reducing the dimensionality of the feature space and facilitating subsequent analysis.
Subsequently, for each VOI, radiomics features were extracted from the original images and those processed using Laplacian of Gaussian (LoG) filtering with sigma values of 2, 3, 4, 5, as well as wavelet filtering applied in various directions. The extracted radiomics features included first-order statistics, shape-based metrics, the gray level dependence matrix, the gray level co-occurrence matrix, the gray level run length matrix, the gray level size zone matrix, and the neighboring gray tone difference matrix. In total, 1239 radiomics features were extracted. The images processed with LoG and wavelet filtering are considered derived images, and the corresponding radiomics features are regarded as derived features. Detailed description of the feature types is presented in Figure 4, as illustrated in the preceding reference.19,20 Following this, all radiomics features were normalized using the z-score transformation.
28

Detailed elucidation of radiomics features, including terminology and interpretative framework.
Where χ represents each radiomics feature, μ represents the mean of all features, σ represents standard deviation of all features.
Radiomics Feature Selection and Model Construction
To mitigate the risk of overfitting, we employed the maximal redundancy minimal relevance (mRMR) algorithm and the least absolute shrinkage and selection operator (LASSO) method to reduce the dimensionality of the radiomics features in the training dataset. Specifically, the mRMR algorithm was utilized with a threshold set at 100, aiming to select features that exhibit minimal redundancy and maximal relevance to the target variable. This process was conducted using the ‘mRMRe’ package in R software, which facilitated the efficient implementation of the algorithm.
Subsequently, we applied the LASSO method to further refine feature selection by penalizing the absolute values of the regression coefficients, thereby promoting a sparse model. This step was performed using the ‘glmnet’ package in R software. To determine the optimal hyperparameter λ that minimizes prediction error, we employed a 10-fold cross-validation process within the LASSO method. This cross-validation approach ensures the robustness and generalizability of our radiomics model by evaluating its performance across multiple subsets of the data.
Features exhibiting non-zero coefficients at the optimal value of λ were selected as the final candidate radiomics signature (Figure 5). Following feature selection, the rad score was established by combining the selected features with their corresponding weights in a linear manner, according to the formula:

Depiction of the feature selection process utilizing Lasso regression and a 10-fold cross-validation technique.
Four distinct types of VOIs were generated to calculate the various rad scores, leading to the development of four radiomics models: VOI entire, VOI−2mm, VOI + 2mm, and VOI + 4mm.The diagnostic efficacy of these models was evaluated using receiver operating characteristic (ROC) curves, and the optimal radiomics model was determined based on the highest area under the curve (AUC).
Statistical Analysis
The Shapiro-Wilk test was used to evaluate the normality of the numeric variables. Numeric variables that exhibited a normal distribution were presented as mean ± standard deviation, and comparisons between the two groups were conducted using two independent samples t-test. For data that did not conform a normal distribution, the median along with 25th and 75th quartiles were utilized, and the Mann-Whitney U test was applied for analysis. For categorical variables, the chi-squared test was applied. Univariate logistic regression analysis was employed to identify predictive factors associated with outcome risk, with a significant threshold set at P < .05. Following this, multivariate logistic regression analysis was conducted to determine independent factors that demonstrated significance in the univariate analysis, also at P < .05. The results were presented as odds ratios (OR) with their 95% confidence intervals (CI) and corresponding P-value. A nomogram was subsequently generated based on the coefficients obtained from the multivariate logistic regression results. To evaluate the predictive performance of the model for the IASLC grading system, ROC curves and their associated AUC values were utilized, in addition to decision curve analysis (DCA) and calibration plots. These assessments were conducted across the training, internal validation, and external validation datasets. All statistical analyses were conducted using the R version 4.3.1, with a two-sided P-value of less than .05 considered statistically significant.
Results
Comparison of Baseline Characteristics among Enrolled Patients
A total of 1418 patients with clinical stage IA lung adenocarcinomas were enrolled in this study. Among them, 505 patients were pathologically diagnosed as grade 1, 804 as grade 2, and 109 was grade 3. The clinical-pathological characteristics corresponding to grades 1 through 3 of the IASLC grading system for clinical stage IA lung adenocarcinomas are presented in Table 1, showing that grade 3 patients are more likely to exhibit aggressive pathological features such as VPI, STAS, and LN compared to grades 1 and 2.
Clinical-Pathological Characteristics Across Grade 1-3 IASLC Grading System in Clinical IA Lung Adenocarcinomas.

Abbreviation: VPI, Visceral pleural invasion; STAS, Spread through air spaces; LN, Mediastinal lymph node metastasis; IASLC, international association for the study of lung cancer.
Detailed clinical and radiological characters were recorded and compared among the training, internal validation, and external validation datasets (Table 2), and there were no statistically significant differences (P = .074-.935) were observed among these datasets.
Comparative Analysis of Radiological Features Across Training, Internal Validation, and External Validation Databases.

Abbreviation: IASLC, International Association for the Study of Lung Cancer; GGN, ground-glass nodule; SN, solid nodule; SSN, subsolid nodule; LUL left upper lobe, LLL left lower lobe, RUL right upper lobe, RML right middle lobe, RLL right lower lobe.
Clin-Rad Model Building
Table 3 presents a comparative analysis of Clin-Rad characteristics between grade 1-2 and grade 3 lung adenocarcinomas within the training datasets. Patients classified as grade 3, compared to grades 1-2, demonstrate t a significantly higher likelihood of exhibiting aggressive radiological features, such as lobulation, spiculation, pleural indentation, and a predominance of solid nodules (SN) density, when compared to those in grades 1-2 (all P < .05). Additionally, a male predominance, advanced age, and larger tumor size were also observed in grade 3 patients (all P < .05).
Comparative Analysis of Radiological Characteristics Between Grade 1-2 and Grade 3 Within the Training Database.

Abbreviation: GGN, ground-glass nodule; SN, solid nodule; SSN, subsolid nodule; LUL left upper lobe, LLL left lower lobe, RUL right upper lobe, RML right middle lobe, RLL right lower lobe.
Multivariate logistic regression analysis identified sex and CT density as independent predictors of grade 3 lung adenocarcinomas, as defined by the IASLC grading system, in patients with clinical stage IA disease (Table 4). Notably, female gender was found to be associated with a decreased risk, yielding an OR of 0.946 (95% CI: 0.913-0.98). In contrast, a higher CT density was significantly correlated with an increased risk of grade 3 lung adenocarcinomas, with an OR of 1.215 (95% CI: 1.16-1.273), using GGN as the reference point. The Clin-Rad model developed by these two independent predictors showed AUCs of 0.855 (95% CI: 0.814-0.896), 0.797 (95% CI: 0.728-0.865), and 0.853 (0.788-0.918) in the training, internal validation, and external validation datasets, respectively.
Univariate and Multivariate Logistic Analysis of Clin-Rad Characteristics for the Prediction of Grade 3 According to the IASLC System in Patients with Clinical Stage IA Disease.

Performance of Multi-Scale VOI Radiomics Model
The Rad score for each VOI subgroup was constructed using radiomics features selected through LASSO regression. which further facilitated the construction of various VOI−2mm-based, VOI entire-based, VOI + 2mm-based, and VOI + 4mm-based radiomics models. Notably, grade 3 achieved higher Rad scores across all the VOI subgroups when compared to grades 1 and 2, as evidenced in the training, internal validation, and external validation datasets (Figure 6). Overall, the VOI entire-based radiomics model led to substantial predictive performance, as indicated by AUCs of 0.900 (95%CI: 0.880-0.942), 0.885 (95%CI: 0.824-0.946), and 0.888 (95%CI: 0.782-0.993) in the training, internal validation, and external validation datasets, respectively (Figure 7, Table 5).

Comparative analysis of Rad scores across various VOI subgroups within the training, internal validation, and external validation datasets.

Comparative assessment of Receiver Operating Characteristic (ROC) curves for each VOI subgroup across the training, internal validation, and external validation datasets.
Performance Evaluation of Radiomics Models Utilizing Multi-Scale VOIs for Predicting Grade 3.

Abbreviation: AUC, Area under curve; ACC, Accuracy; SEN, Sensitivity; SPE, Specificity; VOI, Volume of internet.
Hybrid Model Construction and Clinical Application of the Nomogram
Given that the VOI entire-based radiomics model exhibited superior predictive performance for grade 3 according to the IASLC system in patients with clinical stage IA disease, a hybrid model was subsequently developed, integrating the VOI entire-based radiomics with the Clin-Rad model, which incorporates variables such as sex and CT density. The hybrid model demonstrated outstanding predictive performance, as evidenced by AUC values of 0.910 (95%CI: 0.873-0.942), 0.891 (95%CI: 0.845-0.937), and 0.905 (95%CI: 0.846-0.964) for the training, internal validation, and external validation datasets, respectively (Figure 8). Details of the AUCs for the hybrid, radiomics, and Clin-Rad models are presented in Table 6. The DCA indicated that the hybrid model yielded the highest net benefits within a threshold probability range of >8% to <55% in the training dataset, >9% to <38% in the internal validation dataset, and <75% in the external validation dataset. These ranges collectively encompass the broadest spectrum of probabilities (Figure 9).

Comparative evaluation of ROC curves for the Clin-Rad model, radiomics model, and hybrid model across the training, internal validation, and external validation datasets.

Comparative evaluation of DCA curves for the Clin-Rad model, radiomics model, and hybrid model across the training, internal validation, and external validation datasets.
Performance Assessment of Radiomics, Radiological, and Hybrid Models in Predicting Grade 3.
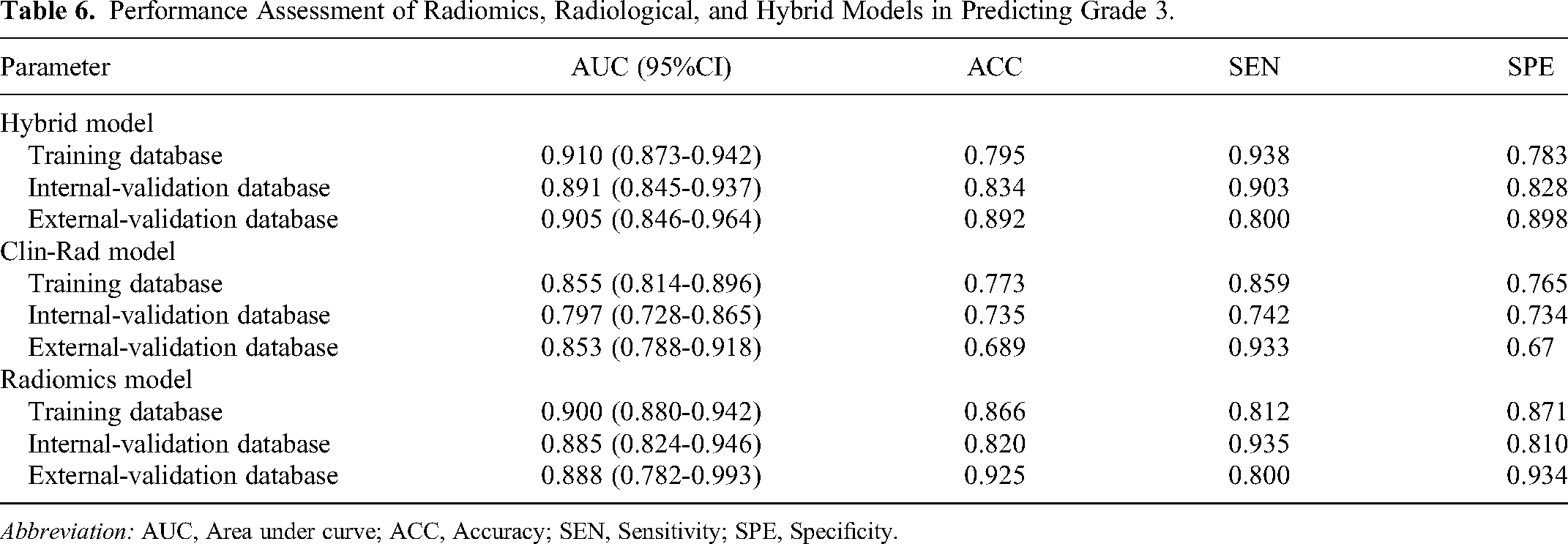
Abbreviation: AUC, Area under curve; ACC, Accuracy; SEN, Sensitivity; SPE, Specificity.
The hybrid model was transformed into a nomogram that incorporates Rad score, sex, and CT density to quantify the risk of grade 3 in clinical stage IA lung adenocarcinomas (Figure 10). Additionally, the nomogram demonstrated strong calibration capabilities, as shown by the standard curve fitting well with the predicted curve across the training, internal validation, and external validation datasets, respectively (Figure 11).

Radiomics nomogram for preoperative prediction of IASLC grading in clinical stage IA lung adenocarcinomas.

Calibration plots for the Clin-Rad model, radiomics model, and hybrid model across the training, internal validation, and external validation datasets.
Discussion
In this multicenter, large-scale study, we conducted a multiscale region-based radiomics analysis to predict the IASLC grading system in clinical stage IA lung adenocarcinomas. Our findings suggest that extraction of radiomic features from both the indented and peritumoral regions does not provide additional benefits in predicting grade 3 according to the IASLC system. The Clin-Rad model, which integrates includes sex and CT density, was identified as an independent predictor of the IASLC grading system in clinical stage IA lung adenocarcinomas. Importantly, the VOI entire-based radiomics model, when combined with the Clin-Rad model, resulted in an integrated nomogram that demonstrated remarkable predictive performance, with strong discriminative and calibration capabilities.
In comparison to grades 1 and 2, grade 3 patients are more likely to exhibit aggressive pathological features such as VPI, STAS, LN, and vascular invasion, according to the IASLC grading system for clinical stage IA lung adenocarcinomas. This indicates that grade 3 tumors exhibit more aggressive biological behavior, which aligns with findings from previous studies.8–11 In the present study, the Clin-Rad model incorporates factors such as male predominance and elevated CT density, indicating that male patients with grade 3 tumors exhibit a slight yet statistically significant increase in risk compared to their female counterparts. Additionally, these tumors primarily characterized as SN in CT image findings, a result that corroborates the outcomes of previous investigations.8,21
To further enhance diagnostic capabilities, our study explored the impact of multi-scale ranges on the preoperative prediction of IASLC grading by radially adjusting the distances at −2, 2, and 4 mm from the tumor boundary. Our findings suggest that the VOI entire-based radiomics model demonstrates robust predictive capability in distinguishing between grade 3 and grades 1-2. Furthermore, the VOI entire-based radiomics model outperforms the Clin-Rad model, indicating that the radiomics model provides additional insights that visual assessments alone may not provide. In contrast to previous research,22–25 the integration of peritumoral radiomics features did not enhance the performance beyond exclusive tumor features. This finding suggests that the diagnostic benefits provided by peritumoral radiomics are adaptive and closely associated with various factors such as nodule type, peritumoral range, and tumor malignancy. Wu et al 26 concluded that extending the analysis to a peritumoral region of 5 mm does not provide any additional advantage in distinguishing invasive adenocarcinoma from adenocarcinoma in situ or minimally invasive adenocarcinoma when compared to the 2 mm region. Additionally, another study. 29 found that the radiomics features of the surrounding parenchyma of the nodule did not improve the effectiveness of the diagnostic model in distinguishing between benign and malignant solid pulmonary nodules smaller than 2 cm.
The hybrid model, which incorporates the VOI entire-based radiomics and Clin-Rad characteristics, exhibits remarkable predictive performance. This approach enables a more comprehensive extraction of information from multiple dimensions. By integrating quantitative imaging features derived from radiomics with Clin-Rad characteristics, the model captures a broader spectrum of disease characteristics, thereby enhancing both accuracy and robustness in diagnostic processes. Additionally, this multimodal fusion helps overcome the limitations of relying solely on either diagnostic model, potentially uncovering concealed patterns or relationships that may not be apparent when utilizing only one type of data. Ultimately, this integration facilitates a more holistic and precise diagnostic assessment.
This study has shown promising results but also has several limitations. Firstly, as a retrospective study, it is susceptible to selection bias, which may have led to a lower-than- anticipated representation of grade 3 patients, potentially underestimating the actual positive rate. To mitigate this concern, future research could improve sampling methods or increase sample sizes. Secondly, although preoperative imaging was utilized to predict grade 3 of the IASLC system, long-term follow-up is essential to elucidate the relationship between Clin-Rad features, radiomics features, outcomes, and survival, thereby improving the understanding of the prognostic significance of radiomics features. Thirdly, there is currently no standardized method for delineating tumoral and peritumoral VOIs for the extraction of radiomics features and the prediction of the IASLC grading system in clinical stage IA lung adenocarcinomas. To address this challenge, further comparative studies with pathological assessments are warranted.
Conclusion
The extraction of radiomic features from both the intratumoral and peritumoral regions does not offer additional benefits in predicting grade 3 according to the IASLC system. Furthermore, when the VOI entire-based radiomics model is combined with Clin-Rad characteristics, the resulting nomogram exhibits remarkable predictive performance. The multicenter, large-scale nature of this study further enhances its robustness and accuracy, thereby minimizing redundancy and improving reliability.
Supplemental Material
sj-tif-1-tct-10.1177_15330338241300734 - Supplemental material for Radiomics Nomogram Based on Optimal Volume of Interest Derived from High-Resolution CT for Preoperative Prediction of IASLC Grading in Clinical IA Lung Adenocarcinomas: A Multi-Center, Large-Population Study
Supplemental material, sj-tif-1-tct-10.1177_15330338241300734 for Radiomics Nomogram Based on Optimal Volume of Interest Derived from High-Resolution CT for Preoperative Prediction of IASLC Grading in Clinical IA Lung Adenocarcinomas: A Multi-Center, Large-Population Study by Zhichao Zuo, Guochao Zhang, Shanyue Lin, Qi Xue, Wanyin Qi, Wei Zhang and Xiaohong Fan in Technology in Cancer Research & Treatment
Supplemental Material
sj-doc-2-tct-10.1177_15330338241300734 - Supplemental material for Radiomics Nomogram Based on Optimal Volume of Interest Derived from High-Resolution CT for Preoperative Prediction of IASLC Grading in Clinical IA Lung Adenocarcinomas: A Multi-Center, Large-Population Study
Supplemental material, sj-doc-2-tct-10.1177_15330338241300734 for Radiomics Nomogram Based on Optimal Volume of Interest Derived from High-Resolution CT for Preoperative Prediction of IASLC Grading in Clinical IA Lung Adenocarcinomas: A Multi-Center, Large-Population Study by Zhichao Zuo, Guochao Zhang, Shanyue Lin, Qi Xue, Wanyin Qi, Wei Zhang and Xiaohong Fan in Technology in Cancer Research & Treatment
Footnotes
Author Contributions
Zhichao Zuo, Guochao Zhang, and Shanyue Lin contributed equally to this work. Zhichao Zuo primarily contributed to the writing. Guochao Zhang and Shanyue Lin were responsible for data collection and analysis. Qi Xue and Wanyin Qi participated in the conceptualization and design of the study. Wei Zhang and Xiaohong Fan provided critical revisions and approved the final manuscript. All authors have reviewed and approved the final version of the manuscript.
Compliance with Ethical Requirements
The Ethics Committees of the participating hospitals approved the study (reference numbers: KY2020147, 2021-07-009, 22/244-3446, 2023YJSLL-121, and KY-2024-231) in accordance with the Declaration of Helsinki.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by The National Key R&D Program of China (2022YFC2407404, Qi Xue); Beijing Natural Science Foundation (7232134, Guochao Zhang); National High-Level Hospital Clinical Research Funding (2022-PUMCH-C-043, Qi Xue); Beijing Municipal Science & Technology Commission (Z211100002921058, Qi Xue); Beijing Hope Run Special Fund of Cancer Foundation of China (LC2021L01, Qi Xue).
Informed Consent Statement
Given the retrospective nature of our study, informed consent was waived.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


