Abstract
Purpose
To assess whether gray-scale ultrasound (US) based radiomic features can help distinguish HER2 expressions (ie, HER2-overexpressing, HER2-low-expressing, and HER2-zero-expressing) in breast cancer.
Materials and Methods
This retrospective study encompassed female breast cancer patients who underwent US examinations at two distinct centers from February 2021 to July 2023. Tumor segmentation and radiomic feature extraction were performed on grayscale US images. Decision Tree analysis was employed to simultaneously evaluate feature importance, and the Least Absolute Shrinkage and Selection Operator technique was utilized for feature selection to construct the radiomic signature. The Area Under the Curve (AUC) of the Receiver Operating Characteristic curve was employed to assess the performance of the radiomic features. Multivariate logistic regression was used to identify independent predictors for distinguishing HER2 expression in the dataset.
Results
The training set comprised 292 patients from Center 1 (median, 51 years; interquartile range [IQR]: 45-61), while the external validation set included 131 patients from Center 2 (median, 51 years; IQR: 45-62). In the external validation dataset, the radiomic features achieved AUC of 0.76 for distinguishing between HER2-low and positive tumors versus HER2-zero tumors. The AUC for differentiating HER2-low (1+) from HER2-zero tumors was 0.74, and for distinguishing HER2-low (2+) from HER2-zero tumors, the AUC was 0.77. In the multivariate analysis assessing HER2-low and HER2-positive versus HER2-zero tumors, internal echoes (P = .029) and margins (P < .001) emerged as independent predictive factors.
Conclusion
The radiomic signature and tumor descriptors from gray-scale US may predict distinct HER2 expressions of breast cancers with therapeutic implications.
Key Points
Breast gray-scale ultrasound scans, retrospectively obtained from 292 women diagnosed with breast cancer, facilitated the construction of a radiomic signature. This signature demonstrated accurate predictive capability for distinguishing between low or positive human epidermal growth factor receptor 2 (HER2) expressions versus zero expression, as evidenced in a prospective external test set comprising 131 patients, achieving an area under the receiver operating characteristic curve of 0.76.
Multiple clinical and pathological factors were found to be strongly associated with HER2 expression in the combined dataset (n = 423).
Summary
This study validates the efficacy of gray-scale ultrasound-based radiomic signatures in differentiating HER2 expressions in breast cancer, potentially guiding more personalized therapeutic approaches.
Introduction
Breast cancer (BC) is a heterogenous malignancy, and its management currently relies on TNM staging alongside traditional histopathological characteristics. 1 Within the realm of clinical practice, three molecular subtypes have been identified that carry significant prognostic implications: HER2-positive, luminal (hormone receptor-positive and HER2-negative), and triple-negative BC.2–4 The notable prognostic differences among these subtypes have prompted widespread investigation into the molecular diagnostics of BC. 5 It is estimated that between 10% and 15% of all BCs manifest overexpression of Human Epidermal Growth Factor Receptor 2 (HER2), which is associated with marked invasiveness, a tendency for early recurrence, and an unfavorable prognosis.6,7
According to the 2018 guidelines 8 by the American Society of Clinical Oncology and the College of American Pathologists (ASCO/CAP), HER2 status is determined by an immunohistochemistry (IHC) score of +3, or by a + 2 IHC score in conjunction with fluorescence in situ hybridization (FISH) demonstrating ERBB2 amplification. Tumors with an IHC result of 0, + 1, or +2 without FISH amplification are classified as HER2-negative. For tumors with IHC results of +1 or +2/FISH negative, defined as “HER2-low”. Some studies show that for patients with metastatic disease harboring HER2-low expression, the application of trastuzumab deruxtecan, a novel antibody-drug conjugate targeting HER2 has demonstrated encouraging clinical benefits in terms of progression-free and overall survival.9,10 In the landscape of BC research, it has been reported that approximately 55% to 60% of cases, including luminal or triple-negative BCs, are reported as HER2-low tumors, while 25% are reported as HER2-zero (ie, IHC score of 0).11,12 Only recently has there been an initiative to delve into the molecular and pathological distinctions among HER2-zero, HER2-low, and HER2-positive BCs.13,14
The aberrant activation of downstream signaling pathways induced by HER2 expression triggers a cascade of signal transduction events, leading to unregulated and uncontrollable cell proliferation. 15 It could be hypothesized that elevated levels of cell proliferation might manifest in the form of increased heterogeneity within the spatial relationships and voxel data as captured by magnetic resonance imaging (MRI) or ultrasound (US) imaging modalities. Notably, imaging has been identified as a means to capture tumor biology at both genetic and cellular levels. 16 Several studies have elucidated the correlation between imaging characteristics and the HER2-positive subtype of BC.17,18 Radiomics allows for the extraction of quantitative features from medical images, revealing hidden mathematical patterns that are not discernible to the human eye. 19 These features are repeatable, non-invasive, and can provide valuable insights into the underlying biology of tumors, particularly when pathological results are inconclusive or when biopsy procedures are limited.20,21
Significant strides have been made recently in exploring the association between MRI radiomic features and the expression of HER2 in BC. For example, studies have shown an association between multiparametric breast MRI radiomics of lesions and HER2 expression.22,23 However, investigations into the correlation between US radiomic features and HER2 expression in BC remain scant.
In light of the aforementioned findings, we aimed to assess whether US based radiomic features could help differentiate HER2-zero, HER2-low, and HER2-positive tumors in this study.
Materials and Methods
This retrospective dual-center study received approval from the institutional review committees of participating institution (Shenzhen People' s Hospital, approval Number: LL-KY-2022479-02; (Rizhao People' s Hospital, IRB-2022-024). Informed consent was not required because this study was retrospective. The methodology and execution of this study were meticulously aligned with the ethical principles delineated in the Declaration of Helsinki, alongside adhering to the Standards for Reporting of Diagnostic Accuracy Studies (STARD) guidelines. 24
Study Sample
Between February 2021 and July 2022, patients newly diagnosed with cancer at a primary tertiary care center (Center 1), exhibiting clinical stages T1–T4, NX, and M0 and undergoing gray-scale ultrasound (GSUS), were retrospectively enrolled into the training dataset. Exclusion criteria were meticulously defined to ensure the integrity of the dataset: (a) absence of invasive cancer in postoperative pathology, (b) incomplete clinical data or substandard ultrasonography quality, (c) lack of postoperative IHC or FISH verification of HER2 status, presence of heterogeneous FISH results, or an IHC score of 2 + without subsequent FISH confirmation, and (d) patients who received neoadjuvant chemotherapy or radiotherapy prior to surgery. Adhering to identical inclusion and exclusion criteria, consecutive patients treated at a secondary tertiary care center (Center 2) from August 2022 to July 2023 were incorporated into an external validation dataset (Figure 1).

Flowchart of patient inclusion and exclusion criteria. US = ultrasound, HER2: human epidermal growth factor receptor 2.
HER2 Score Testing
In accordance with the 2018 guidelines 8 issued by the ASCO/CAP, HER2 expression levels were meticulously assessed post-surgery by cancer-specialized pathologists. The determination of HER2 status was finalized using FISH, where a HER2/CEN-17 ratio of less than 2 was indicative of non-amplification, and a ratio of 2 or greater signified amplification (Appendix S1). Subsequently, HER2 expression outcomes were stratified into distinct categories: HER2-zero (IHC score of 0), HER2-low, or HER2-positive. The HER2-low category comprised tumors with an IHC score of 1 + or 2 + coupled with a negative FISH result. Conversely, the HER2-positive classification included tumors with either an IHC score of 3 + or an IHC score of 2 + in conjunction with a positive FISH outcome. Notably, for cases where there is no consensus between pathologists on the HER2 status, we employ a protocol that involves further evaluation by a senior pathologist with over 15 years of experience in HER2 assessment. For cases with artifacts, basolateral staining, or weak/incomplete staining, we will conduct a re-examination and, if necessary, collect new tissue samples for further evaluation.
US Images Acquisition and Segmentation
In this study, ultrasonography assessments were conducted using various instruments, including the GE LOGIQ E9 and Mindray Resona 7. High-frequency probe models were employed for conventional US examinations, ensuring optimal resolution. Image acquisition parameters were standardized across patients, with gain settings adjusted to approximately 50%, and image depth ranged from 3 to 6 cm to align focus parallel to the lesions. This protocol guaranteed the clear visualization of the largest portion of each lesion, with all US images archived in the Digital Imaging and Communications in Medicine (DICOM) format for uniformity and future reference. The US examinations were randomly assigned to two radiologists (H.T. and H.W.), both blinded to the clinical and pathological data, with five years of experience in breast US. Preoperative US-images were segmented using the ITK-SNAP software (version 3.8.0; [http://www.itksnap.org]), independently by the radiologists. Any discrepancies in segmentation masks were reviewed and confirmed by a senior radiologist (J.X.) with twenty years of expertise in breast MRI, ensuring accuracy through consensus-based discussions. Regions of interest (ROIs) were delineated manually on the transverse views of grayscale US-images in DICOM format, as shown in Figure 2, highlighting each tumor's contours. Furthermore, an additional over board-certified radiologist (Q.L.), with more than five years of experience in breast US, independently segmented 60 randomly selected lesions from the training set. This step was critical for evaluating inter-observer reproducibility using the two-way mixed absolute agreement intra-class correlation coefficient (ICC). The strength of agreement for the ICC was classified as follows: poor (<0.50), moderate (0.50-0.75), good (0.75-0.90), and excellent (0.90-1.00), facilitating a rigorous assessment of the consistency in tumor segmentation.
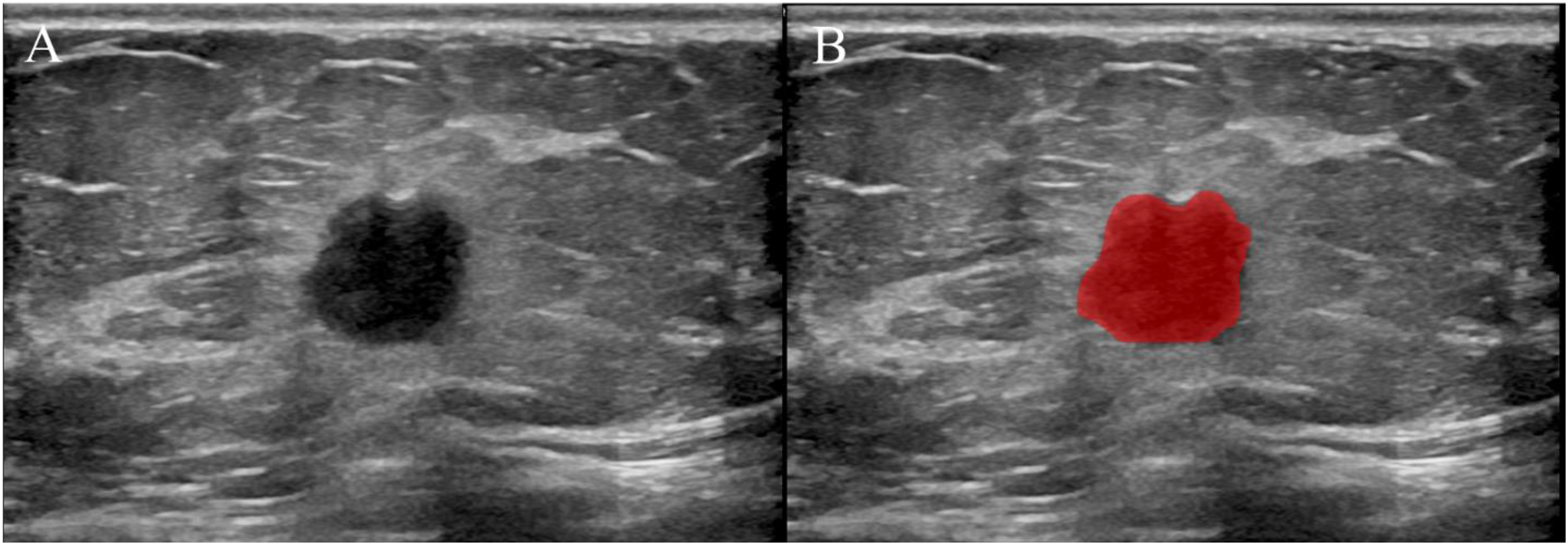
(A) 2D grayscale ultrasound image of breast lesion. (B) Manually outline the region of interest using software ITK-SNAP.
Radiomic Feature Extraction
In this investigation, quantitative radiomic features were meticulously extracted from the segmented regions of interest (ROIs) within the raw US images for each participant. This extraction process utilized the Pyradiomics package (version 3.0.1; Python Software Foundation), which adheres to the standards set forth by the Image Biomarker Standardization Initiative (IBSI). 19 Prior to feature extraction, a normalization step was implemented using Pyradiomics’ integrated functions to standardize the data, ensuring consistency across images. To accommodate the specific resolution requirements of ultrasonography imaging, voxel sizes were resampled to achieve an isotropic in-plane resolution of 1 mm × 1 mm. This resampling was critical for maintaining uniformity in the analysis of texture and shape features across different images. Moreover, to address the variability introduced by diverse imaging protocols and scanner equipment, the ComBat algorithm was employed for harmonizing features, effectively mitigating batch effects that could potentially confound the analysis. 25
From the two-dimensional US-images, a comprehensive suite of 1561 radiomic features spanning seven distinct categories was initially extracted. These categories encompass a broad spectrum of radiomic insights, including: (1) First-order statistics, capturing the basic intensity distribution within the ROI; (2) Gray Level Co-occurrence Matrix (GLCM) features, quantifying texture by assessing the distribution of co-occurring voxel intensities; (3) Gray Level Difference Matrix (GLDM) features, detailing texture through the differences between neighboring voxel intensities; (4) Gray Level Run Length Matrix (GLRLM) features, evaluating texture based on the length of consecutive voxels with the same intensity; (5) Gray Level Size Zone Matrix (GLSZM) features, analyzing texture by the size of homogeneous zones; (6) Neighbouring Gray Tone Difference Matrix (NGTDM) features, describing texture through the variation in intensity among adjacent voxels; and (7) Shape-based (2D) features, providing insights into the geometric properties of the ROI (Figure S1). First-order statistics: These features capture fundamental intensity characteristics of the tumor, which may reflect underlying tissue heterogeneity, a known marker for tumor aggressiveness. Texture features (GLCM, GLRLM, GLSZM): These features describe the spatial arrangement of pixel intensities, providing insights into the tumor microenvironment. Shape features: The geometric properties of the tumor, may indicate tumor invasiveness and are correlated with HER2-positive status.
Feature Selection and Classification Model Building
In the preparatory phase of our analysis, ultrasonography radiomic features underwent a rigorous screening process. Features demonstrating an interobserver intraclass correlation coefficient value below 0.75 among observers were excluded to ensure reliability and consistency. Feature normalization for each patient was achieved through Z-score and mean normalization techniques, effectively standardizing the data for subsequent analyses. To mitigate the issue of multicollinearity, features exhibiting a Pearson correlation coefficient greater than 0.80 were clustered and reduced to a single representative feature. This approach streamlined the feature set, enhancing the interpretability of our models. The optimization of our analysis pipeline utilized a 10-fold cross-validation technique, incorporating the Least Absolute Shrinkage and Selection Operator (LASSO) method for variable selection. 26 The LASSO technique applies a penalty to the size of the coefficients, thereby encouraging the selection of a sparse set of predictive features. A radiomic signature was obtained using logistic regression of the top seven LASSO selected features to most predictive between HER2-low and -positive and HER2-zero cancers and was assessed in the external testing set (Table S1). Feature importance was concurrently assessed using Decision Tree (DT) analysis, providing an additional layer of interpretability. Following the application of the LASSO methodology, DT analysis served to further refine the selection of features (Figure S2).
Statistical Analysis
Continuous variables were subjected to analysis using appropriate statistical tests, including the Wilcoxon rank-sum test, Mann-Whitney test, and Student's t-test, based on the distributional normality of the data. Categorical variables, on the other hand, underwent analysis utilizing the χ2 test or Fisher's exact test, as deemed suitable. Receiver Operating Characteristic (ROC) curves were constructed to assess the discriminatory ability of radiomic features in identifying HER2 expression. The AUC was computed for each feature, and the optimal threshold for HER2 expression discrimination was determined using the Youden index. Bootstrapped CIs were calculated. In univariate analysis, all variables demonstrating an association with HER2 expression at a significance level of P < .05 were included in subsequent multivariate logistic regression analysis. All statistical tests were two-tailed, and a P < .05 was considered indicative of statistically significant differences.
Results
Characteristics of the Study Sample
In this study, a cohort comprising 325 cancer patients and their corresponding breast ultrasonography images, sourced from Center 1, was included for analysis. Following stringent exclusion criteria, which encompassed patients who underwent preoperative neoadjuvant chemotherapy or radiation therapy (n = 9), those with incomplete clinical data or substandard ultrasonography quality (n = 15), individuals without invasive lesions (n = 3), and cases lacking confirmation of HER2 status through postoperative IHC or FISH assays (n = 6), a total of 292 patients (median, 51 years; interquartile range [IQR]: 45-61) were enrolled into the training dataset (Figure 1). Initially, 146 patients from Center 2 were considered for inclusion. However, after excluding 4 patients due to preoperative neoadjuvant chemotherapy or radiation therapy, 9 patients with incomplete clinical data or inadequate ultrasonography quality, and 2 patients without invasive lesions, a final cohort of 131 patients (median, 51 years; IQR: 45-62) from Center 2 was incorporated into the external testing dataset (Figure 1).
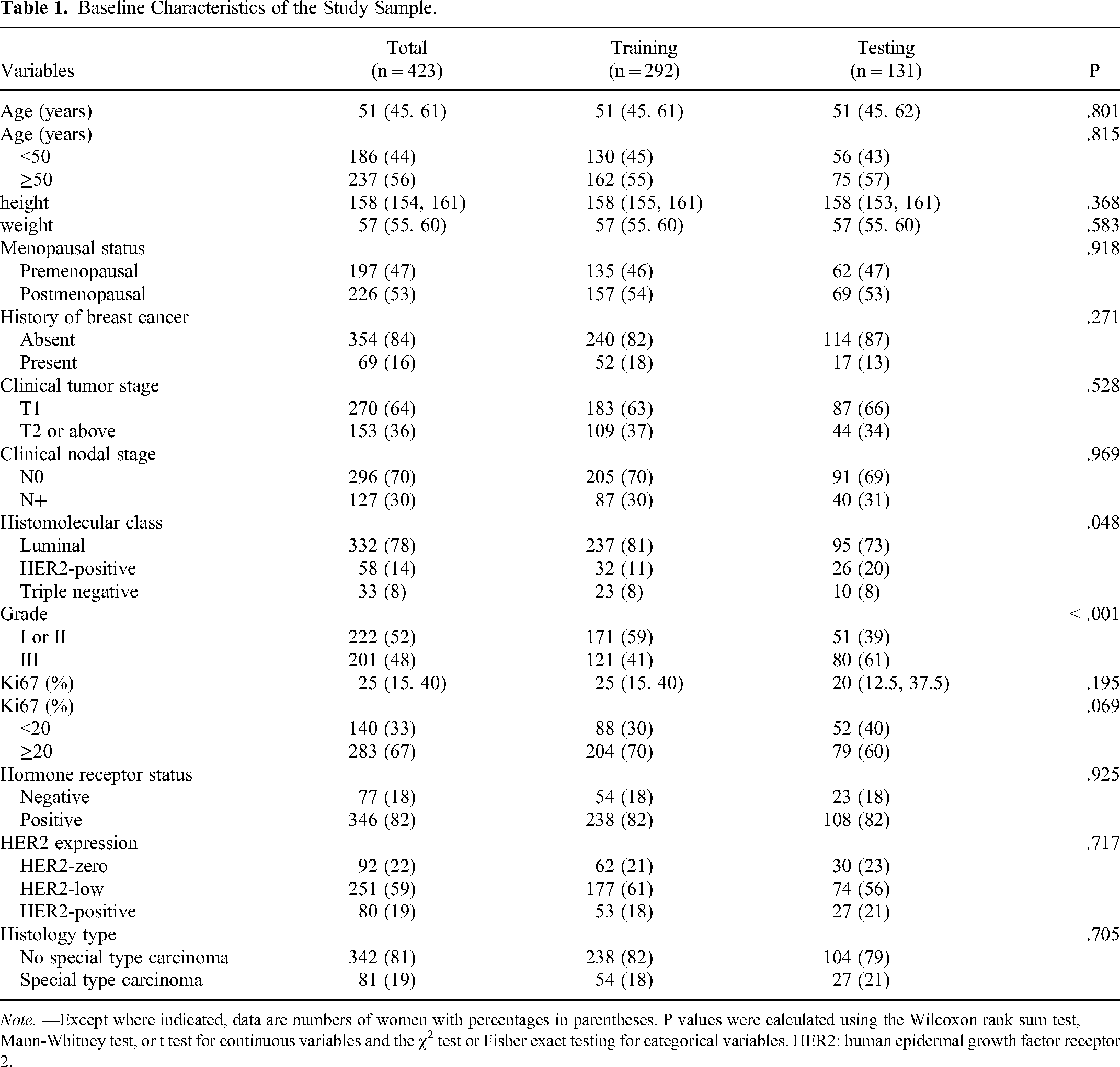
In comparison to an external testing set, patients derived from the training set exhibited a significantly higher incidence of Grade I or II BC (59% [171/292] vs 39% [51/131]; P < .001), as well as a predominance of the ductal carcinoma subtype (81% [237/292] vs 73% [95/131]; P = .048) (Table 1). Notable differences between the training and external testing sets were observed in ultrasonographic assessments, including the BI-RADS classification (P = .012), internal echoes (P = .032), and posterior acoustic features (P = .035) (Table 2). Within the training set, of the 292 patients, 53 (18%) were diagnosed with HER2-positive tumors, whereas 177 (61%) and 62 (21%) were classified as having HER2-low and HER2-zero BCs, respectively.
Baseline Characteristics of the Study Sample.
US Findings of the Study Sample.

Performance of Radiomic Signature for Distinguishing HER2-Low and -Positive versus HER2-Zero Cancers
In the training set, the radiomic features predictive of HER2-low/positive expression achieved an AUC of 0.86 (95% CI: 0.80-0.92). Within the subgroups of HER2-low and HER2-zero tumors, distinguished by IHC scoring, the radiomic signatures facilitated differentiation with notable efficacy. Specifically, the AUC for distinguishing between HER2-low (IHC score of 1+) and HER2-zero tumors was 0.83 (95% CI: 0.76-0.90), identical to the AUC for differentiating HER2-low (IHC score of 2+) from HER2-zero tumors. Additionally, when comparing tumors with HER2-low expression classified as IHC 1 + versus those classified as IHC 2+, the AUC was 0.76 (95% CI: 0.65-0.86) (Figure 3A).

Performance of the radiomic signature for differentiating human epidermal growth factor receptor 2 (HER2) expression levels in the training and external testing set. (A) Receiver operating characteristic curves of the radiomic signature in the training set show an area under the receiver operating characteristic curve (AUC) of 0.86 (95% CI: 0.80, 0.92) for distinguishing HER2-low and -positive versus HER2-zero tumors. (B) Receiver operating characteristic curves of the radiomic signature in the external test set show an AUC of 0.76 (95% CI: 0.65, 0.88) for distinguishing HER2-low and -positive versus HER2-zero tumors. Additionally, it is noteworthy that in the training set, the AUC for the HER2-Low (1+) versus HER2-zero model closely overlaps with the AUC for the HER2-Low (2+) versus HER2-zero model, resulting in overlapping ROC curves.
In the training set, a radiomic feature threshold of 0.77 demonstrated high diagnostic performance in differentiating between HER2-low and HER2-positive versus HER2-zero tumors, achieving a sensitivity (recall rate) of 87%, specificity of 82%, positive predictive value (precision) of 95%, accuracy of 86%, and an F1 score of 91%. Similarly, when employing a radiomic feature threshold of 0.55, the model exhibited a sensitivity of 89%, a precision of 82%, accuracy of 78%, and an F1 score of 83% for distinguishing between HER2-low (IHC 1+) and HER2-zero tumors. Furthermore, a threshold of 0.45 was associated with a sensitivity of 77%, specificity of 76%, accuracy of 77%, and an F1 score of 78% in discriminating HER2-low (IHC 2+) and HER2-zero tumors (Table 3).
Distribution of Diagnostic Efficacy of US Radiomic Models

In the external testing set, radiomic features demonstrated a discriminative ability to differentiate between HER2-low/positive and HER2-zero tumors, as evidenced by an AUC of 0.76 (95% CI: 0.65, 0.88). Specifically, when comparing HER2-low (IHC score of 1+) to HER2-zero tumors, the AUC was 0.74 (95% CI: 0.62, 0.86); for HER2-low (IHC score of 2+) versus HER2-zero tumors, the AUC improved to 0.77 (95% CI: 0.66, 0.89). The ability to distinguish between HER2-low tumors of differing IHC scores (1 + vs 2+) was represented by an AUC of 0.58 (95% CI: 0.44, 0.72) (Figure 3B). Furthermore, within this external testing set, the application of radiomic features yielded a sensitivity of 89%, specificity of 60%, precision of 88%, accuracy of 82%, and an F1 score of 89% for distinguishing between HER2-low and positive versus HER2-zero tumors. For differentiating HER2-low (IHC 1+) from HER2-zero tumors, the metrics were a sensitivity of 81%, precision of 73%, accuracy of 71%, and an F1 score of 77%. In the comparison of HER2-low (IHC 2+) against HER2-zero tumors, a sensitivity of 94%, accuracy of 71%, and an F1 score of 76% were achieved. Notably, the distinction between HER2-low (IHC 2+) and HER2-zero tumors was characterized by a high specificity of 95%, precision of 78%, and an F1 score of 76% (Table 3).
Assessment of Variables Associated with Increased Odds of HER2-Low and -Positive Cancers
Univariate analysis of data from two centers indicated a significant association between the presence of Color Doppler Flow Imaging (CDFI) versus absent and increased odds of HER2-low and positive tumors (P < .001). Tumors characterized by low internal echoes compared to other echo patterns were significantly associated with decreased odds of HER2-low and positive tumors (P = .02), as were tumors with clear versus indistinct margins (P < .001) and tumors classified as Grade I or II versus Grade III (P = .02). These variables were subsequently included in a multivariate analysis to further elucidate their relationship with HER2 expression levels. The multivariate analysis revealed that only internal echoes (Odds Ratio [OR] = 0.45; 95% CI: 0.22, 0.92; P = .029) and tumor margins (OR = 0.21; 95% CI: 0.09, 0.46; P < .001) retained a significant association with lower odds of HER2-low and positive tumor expression. These findings suggest that specific ultrasonographic features, namely internal echoes and tumor margins, may serve as non-invasive predictors of HER2 expression status in BC (Table S2).
Assessment of Variables Associated with Increased Odds of HER2-Positive versus HER2-Low Cancers
In a dataset encompassing two centers, preliminary analysis revealed significant associations between several variables and the likelihood of HER2-low and positive tumors. Specifically, hormonal receptor (HR) status (positive vs negative) and lesion orientation (vertical vs level) were inversely associated with the odds of HER2-low tumors (P < .001 and P = .01, respectively). Furthermore, lesions with the largest dimension ≤20 mm versus >20 mm (P = .03), the presence versus absence of calcifications (P = .01), the presence versus absence of CDFI data (P < .001), and Ki-67 labeling index ≤20% versus >20% (P = .02) were positively associated with higher odds of HER2-positive tumors. These findings prompted their inclusion in a multivariate analysis to ascertain their predictive value. The multivariate analysis identified the calcifications (presence vs absent; OR = 2.1; 95% CI: 1.2, 3.6; P = .015), the CDFI (presence vs absent; OR = 2.9; 95% CI: 1.4, 5.8; P = .003), HR status (positive vs negative; OR = 0.2; 95% CI: 0.1, 0.4; P < .001), and lesion orientation (vertical vs level; OR = 0.4; 95% CI: 0.2, 0.9; P = .024) as independent predictors of HER2-low and positive tumors (Table S3). These variables contribute significantly to the stratification of breast tumors by HER2 status, underscoring the potential of combining clinical and imaging markers to refine diagnostic accuracy.
Assessment of Variables Associated with Increased Odds of Luminal and Non-Luminal Breast Cancers
In a combined dataset from two centers, univariate analysis aimed at differentiating luminal from non-luminal BC revealed statistically significant associations for several variables. Specifically, the presence of low versus other internal echoes (P = .01), indistinct versus clear margins (P = .01), a Ki-67 labeling index of ≤20% versus >20% (P < .001), and cancer graded as I or II versus III (P < .001) were found to be of statistical significance, warranting their inclusion in a multivariate analysis. Subsequent multivariate analysis identified low versus other internal echoes (OR = 0.5; 95% CI: 0.3, 0.9; P = .031), a Ki-67 labeling index of ≤20% versus >20% (OR = 3.8; 95% CI: 1.1, 13.8; P = .042), and BC graded as I or II versus III (OR = 4.4; 95% CI: 2.5, 7.6; P < .001) as independent predictors (Table S4). These findings underscore the critical role of specific US features, alongside Ki-67 labeling index and histological grade, in the stratification of BC into luminal and non-luminal subtypes.
Discussion
Our study findings demonstrate the efficacy of grayscale US-based radiomic prediction models in identifying HER2 overexpression, HER2-low expression, and HER2-zero in BC, with an AUC ranging from 0.58 to 0.77 in the external testing set. Grayscale US-based radiomic models hold promise for non-invasively classifying HER2 status and may contribute to clinical decision-making in cancer management. In our analysis of data from two centers, we identified several US features and clinicopathological factors independently associated with alterations in HER2 expression patterns. Specifically, internal echoes (OR = 0.45; 95%CI: 0.22, 0.92; P = .029) and margins (OR = 0.21; 95% CI: 0.09, 0.46; P < .001) were independently associated with decreased odds of HER2-zero expression. Conversely, the calcifications (OR = 2.1; 95% CI: 1.2, 3.6; P = .015) and CDFI (OR = 2.9; 95% CI: 1.4, 5.8; P = .003) were independently associated with increased odds of HER2-positive expression. Moreover, the Ki-67 levels (OR = 3.8; 95% CI: 1.1, 13.8; P = .042) and the histological grades (OR = 4.4; 95% CI: 2.5, 7.6; P < .001) were independently associated with increased odds of non-luminal BC. These findings underscore the potential of US-based radiomic models and clinicopathological factors in refining the characterization of HER2 expression patterns and BC subtypes, providing valuable insights for personalized treatment strategies.
The efficacy of HER2-targeted therapies, such as trastuzumab, is well-established in patients with HER2-overexpressing BC, yet these therapies have shown limited effectiveness in those with HER2-negative BC. However, as highlighted by newly proposed classification systems, 27 traditional categorizations of HER2-negative BC encompass both HER2-low and HER2-zero expression subgroups. Notably, the advent of new HER2-targeted therapeutic approaches has significantly altered the clinical management paradigm for traditional HER2-negative BCs by demonstrating considerable clinical benefits in HER2-low expressing cancers. 28 A recent Phase III clinical trial has shown that trastuzumab deruxtecan, an antibody-drug conjugate, significantly extends survival in patients with advanced HER2-low expressing cancers compared to chemotherapy. 10 This underscores the clinical relevance of HER2-low protein expression and necessitates a reevaluation of this subgroup within the broader spectrum of traditional HER2-negative BCs.
Previously, a variety of radiomics models employing either single-parameter or multiparameter MRI techniques have been formulated to accurately distinguish between HER2-overexpressing and HER2-negative BC phenotypes.29–31 Recent investigations have highlighted the potential of multiparameter MRI radiomics models to effectively differentiate among HER2-overexpressing, HER2-low, and HER2-zero BC subtypes, demonstrating AUC metrics ranging from 0.73 to 0.82.23,32 Despite these advancements, radiomics research focusing on the correlation between imaging phenotypes and HER2 expression levels remains scarce, particularly in the application of GSUS to construct radiomics models for identifying the HER2-low category. In a recent advancement, Yan et al developed a radiomics model that leverages intratumoral US features and clinical characteristics to predict the HER2 status in patients with equivocal (2+) IHC results. 33 This model demonstrated robust performance in predicting equivocal HER2 status (IHC score of 2+), achieving an AUC of 0.86 in the testing set. Nevertheless, it did not extend to effectively identifying the more challenging HER2-low and HER2-zero tumors. In our study, we extracted high-throughput radiomic features from grayscale US images to construct a US-based radiomics model for distinguishing the newly proposed patterns of HER2 expression in BC. A total of 1561 radiomic features were extracted from each patient. We found that a radiomic signature built from tumoral features (based on US-images) capable of differentiating between each HER2 expression status, namely HER2-overexpressing, HER2-low, and HER2-zero expression states. Our findings reveal that the predictive model, based on US radiomic features, achieved an AUC of 0.76 in distinguishing HER2-zero from HER2-overexpressing and HER2-low expressing BCs. Moreover, the radiomics model demonstrated AUCs of 0.74 and 0.77 in the testing set for distinguishing HER2-zero from HER2-low expressing (IHC score of 1+, 2+) BCs, respectively. The performance of our US radiomics model is comparable to previously reported multiparametric MRI radiomics models23,32 and superior to earlier described single-parameter MRI radiomics approaches.30,31 These results suggest that features derived from US radiomics have the potential to serve as a non-invasive tool for identifying HER2-overexpressing, HER2-low, and HER2-zero expressing BCs. This could facilitate the identification of potential candidates for traditional anti-HER2 therapies, novel anti-HER2 antibody-drug conjugates, or those who may be excluded from anti-HER2 treatments.
IHC and FISH methodologies are recognized as the sole standardized and established techniques for evaluating HER2 expression, despite reported inconsistencies between central and local assessments of HER2 expression status. 34 These evaluations, typically based on sampling a small fraction of potentially heterogeneous tumors, involve labor-intensive and error-prone manual determinations of HER2 status, with diagnostic discrepancies among pathologists leading to misdiagnoses in a significant number of cases.35,36 By integrating radiomic features as a supportive diagnostic tool into their workflow, pathologists could become more alert to potential variations in tumor HER2 expression. Consequently, radiomic features may serve as a non-invasive biomarker for assessing tumor heterogeneity and assist in selecting biopsy targets most likely to exhibit HER2 expression across multifocal lesions. Moreover, this approach enables the repeated assessment of HER2 expression status in patients undergoing neoadjuvant chemotherapy, to monitor spatiotemporal biological changes in the tumor with each progression of the disease, and facilitates timely adjustments in treatment regimens for patients experiencing changes in HER2 expression status during neoadjuvant chemotherapy. Given the limited treatment options for the resistant luminal and triple-negative subtypes, employing imaging-guided re-testing for HER2-low expression could represent a critical strategy in ongoing clinical trials of anti-HER2 therapies.
There were some limitations in our study. Firstly, we observed certain disparities in characteristics among women in both the training and external testing phases. These differences, while potentially reflective of real-world clinical practices, caution against overly optimistic conclusions. Secondly, to compute the ROC curve for the tri-categorical event, we simplified the analysis by converting it into a binary classification to differentiate among various HER2 expression statuses. The adoption of more sophisticated statistical methods could potentially address this limitation. Furthermore, the segmentation of tumor Volume of Interest (VOI) was conducted manually, making the traditional radiomic feature extraction both time-consuming and complex. These challenges might be mitigated through the implementation of more automated segmentation approaches, such as those provided by deep learning frameworks. Lastly, the performance of our model, though promising, still falls short of the threshold for immediate clinical application. Future iterations of our research will aim to refine these aspects, enhancing the model's accuracy and utility in a clinical setting.
Conclusion
In summary, the results of this bicentric study indicate that radiomic features based on gray-scale ultrasound have the potential to accurately predict the expression status of Human Epidermal Growth Factor Receptor 2 (HER2) in breast cancer patients and may represent a non-invasive approach to identifying patients eligible for HER2-targeted therapy.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241292668 - Supplemental material for Ultrasound Radiomics for the Prediction of Breast Cancers with HER2-Zero, -Low, and -Positive Status: A Dual-Center Study
Supplemental material, sj-docx-1-tct-10.1177_15330338241292668 for Ultrasound Radiomics for the Prediction of Breast Cancers with HER2-Zero, -Low, and -Positive Status: A Dual-Center Study by Yunqing Yin, Sijie Mo, Guoqiu Li, Huaiyu Wu, Jintao Hu, Jing Zheng, Qinghua Liu, Mengyun Wang, Jinfeng Xu, Zhibin Huang, Hongtian Tian and Fajin Dong in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Availability of Data and Materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
This work was supported by the Shenzhen Science and Technology Program (GJHZ20240218114504009).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study involving human subjects, was thoroughly reviewed and approved by the Institutional Review Board of Shenzhen People's Hospital (Approval No. LL-KY-2022479-02) and the Institutional Review Board of Rizhao People's Hospital (Approval No. IRB-2022-024). Informed consent was waived due to the retrospective nature of the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
