Abstract
Introduction
According to recent cancer burden data, the most common cancer in China is lung cancer; which is also the leading cause of cancer death in China and in the USA.1,2 Among nearly two million new cases of lung cancer diagnosed every year, more than 85% of these cases were non-small cell lung cancers (NSCLCs) (which were identified by histopathological grading), including adenocarcinoma, squamous cell carcinoma, and large cell carcinoma.3,4 Regardless of clinical characteristics, various genetic screenings (such as for EGFR, ALK, BRAF) are currently recommended by ASCO in all patients with advanced lung adenocarcinoma. Cumulative data further showed that more than 40% of patients with NSCLC in the Asian population have epidermal growth factor receptor (EGFR) mutations, most of which are deletions in exon 19 (Ex19del) or exon 21 L858R point mutations.5-7 Depending on the involvement of EGFR receptor tyrosine kinase inhibitors (EGFR-TKIs) on EGFR mutations in 2004, various generations of EGFR-TKIs have been developed for the sequential administration of targeted therapy and have replaced traditional platinum-based chemotherapies, such as gefitinib, afatinib and osimertinib. 8 Indeed, these small-molecule drugs have been proved with significant clinical responses and reduced treatment-related toxicities, and become the standard of care for the treatment of NSCLC with EGFR mutation. 3 Unfortunately, most of patients will acquired drug resistance to EGFR-TKIs treatment and show disease progression within approximately 10 months, including the third-generation EGFR-TKI osimertinib. 9 These mutations associated resistance to EGFR-TKIs generally arise due to the drug selective pressure on tumor cells in the treatment through EGFR-dependent or EGFR-indepent resistance mechanisms. 9 Thus, it is a significant challenge to treat advanced NSCLC with mutation to avoiding or obviously delay the emergence of acquired resistance, and maximize the benefits from EGFR-TKIs in clinic. So, the combination of EGFR-TKIs with other target anticancer strategies, such as chemotherapy, radiotherapy, antiangiogenesis therapy and immunotherapy have been under consideration.
The cumulative data showed that EGFR-TKIs treatment has the regulation on the tumor immunological microenvironment and the EGFR activation up-regulates the expression of PD-L1 on tumor cells10-13 . So, immunotherapy using immune checkpoint inhibitors (ICIs) for tumors has attracted more attention due to its benefits in the clinic and has been integrated into the treatment of NSCLC. 14 Unfortunately, published data also have revealed that patients with EGFR-mutant NSCLC have a poor response to ICI monotherapy, such as anti-PD-1/PD-L1 therapy. 15 The potential mechanisms for the poor efficacy of anti-PD-1/PD-L1 therapy in EGFR mutant NSCLC include lower expression of PD-L1 in cancer cells and low tumour mutational burden (TMB).16,17 Moreover, multiple clinical trials of EGFR-TKI-immunotherapy combination therapy have been terminated because of increased toxicity or high incidence of adverse events (AEs), and indicated the limitation of anti-PD-L1 in the current treatment of NSCLC.18-20 Therefore, it is necessary to seek alternative strategy for immunotherapy in the treatment of these diseases with EGFR-TKIs.
Immune cells which with cytotoxicity to tumor cells attract attention in cancer therapy for past decades, such as CD8 T cells and NK cells. Generally, EGFR-mutant NSCLC has a noninflamed tumor microenvironment (TME) and infiltrated with low frequency of CD8+ T cells. But, tumor EGFR-TKIs treatment enhanced the immune recognition and lysis to tumor cells which mediated by antigen-specific T cells and NK cells.21-23 Moreover, published data showed that EGFR-TKIs treatment reversed immunosuppression by increasing cytotoxic CD8+ T cells and dendritic cells, which was accompanied with tumor shrinkage. Unfortunately, such EGFR-TKI treatment also consistently promoted the production of myeloid-derived suppressor cells (MDSCs), and increased the levels of IL-10 and CCL-2. 24 Thus, EGFR-TKIs treatment induced the change in tumor immunological microenvironment might bring about a window of opportunity for combination treatment with cellular adoptive immunotherapy in advance NSCLC. Different autologous or allogeneic immune cells, such as CD8+ tumour-infiltrating lymphocytes (TILs), NK cells, and genetically-modified immune cells (CAR-T and TCR-T cells) have been cultured in vitro for tumour immunotherapy since the 1980s.25–28 In a previous study, we also identified one CD1d-independent CD8+ NK-like T subset, also called NKT cell, that expresses both T-cell markers and NK-cell receptors, and has cototoxicity to tumor through killing MDSCs in vivo. 29 We also identified the equivalent CD8+ NKT (CD8 + CD56+) in the peripheral blood in humans and confirmed its anti-tumor capability (data not shown). These data further indicated its potential usage in the combination with EGFR-TKIs treatment in the advance NSCLC.
Although vaious EGFR-TKIs of different generations are currently used in clinic, gefitinib is a representative drug of first-generation EGFR-TKIs, and has been proven to have superior efficacy in EGFR advanced NSCLC treatment than platinum-based chemotherapy, with a response rate (RR) of 73% and PFS of 10.8 months.30,31 Thus, in this study, we designed a trial to explore the potential efficiency and safety of NKT cells adoptive immunotherapy in the combination with EGFR-TKIs in the treatment of the EGFR mutated advanced NSCLC.
Materials and Methods
Patients
In this clinical trial, we followed the guidelines of the Declaration of Helsinki, and it was approved by the institutional review board. Eligible male or female patients were aged between 18 and 75 years old, had a histopathological diagnosis of advanced NSCLC (clinical stage III/IV) with an EGFR exon 19 deletion mutation or EGFR exon 21 L858R substitution mutation, and one or more measurable lesions. In the trial period, the patients did not receive any additional chemotherapy or radiotherapy other than that specified in this study. Complete inclusion/exclusion criteria are reported in the trial protocol. 32 The trial was registered in the Chinese Clinical Trial Registry (ChiCTR-IIR-17013471) (http://www.chictr.org.cn/index.aspx). All patients signed informed consent forms before screening. The reporting of this study conforms to the CONSORT statements. 33
Study Design and Treatment
This study is prospective. Subjects were randomly divided into the gefitinib arm and gefitinib/NKT arm. After enrollment (and prior to any trial treatment), subjects were randomly assigned to the experimental group (gefitinib /NKT group) or the control group (gefitinib group) on a 1:1 ratio. And the group randomization method is used to randomly group the subjects. The subjects were divided into different block groups and then randomly assigned within each group, and tables of random numbers had been generated by statistical experts and recorded by the researchers. As the aim of this study was to explore the effects of adaptively transferred NKT cells on acquired gefitinib resistance in NSCLC, all volunteers were first treated with gefitinib for 8 weeks and then were screened the degrees of gefitinib response. Thus, the week-8 was designated as the baseline for this trial (Figure 1), and only the volunteers with no disease progression were enrolled in the study for further random allocation into the gefitinib arm and the gefitinib/NKT arm. Next, in the gefitinib arm, the patients were orally treated with 250 mg gefitinib once daily until disease progression. In the gefitinib/NKT arm, the patients were treated with gefitinib (as described in the gefitinib arm) but in combination with an infusion of a total of 1 × 1010 NKT cells every four weeks from the eighth week after gefitinib treatment. The diagram of the trial, the frequencies of gefitinib treatment and NKT-cell infusion are described in (Figure 1) and the protocol. 32

The schedule for the gefitinib treatment and NKT cell adaptive transfer in the trial.
NKT-Cell Preparation
The immediate family members of the patient with consanguinity served as the blood donors. All donors signed informed consent forms and underwent medical examinations to ensure that they met the inclusion criteria for donors in the protocol. 32 peripheral blood mononuclear cells (PBMCs) from blood donors are collected using a Fresenius blood cell separator, model COM.TEC (COM). TEC cell separator for NKT-cell preparation in the trial (COM). The TEC cell separator (Fresenius Haemocare, Bad Homburg, Germany) had a total circulating blood volume of approximately 4000 ml. All PBMC samples were analyzed to determine the total cell number and the percentage of CD8+ T cells and then cryopreserved in liquid nitrogen for further experiments.
CD8+ NKT-cell preparation was accomplished by culturing allogeneic PBMCs from donors with CD8+ NKT culture medium developed by the Institute of Cell Therapy of Tsinghua University (Beijing, China). PBMCs were differentiated in T75 flasks and transferred to 2 L culture bags on Day 4 for CD8+ NKT-cell expansion until cells were harvested on Days 12 and 15. The percentage of CD8+ cells among the expanded cells was greater than 60%, and representative flow cytometery graphs are shown in (Figure 2) the supplementary data. The total number of cells used for patient infusion was up to 1 × 1010. The final cell product for infusion was prepared in saline with albumin.

Flow analysis for cultured NKT cells.
Clinical Evaluation
According to the improved version 1.1 of the evaluation standard for solid tumours, 34 imaging evaluations were performed at baseline and every 2 months thereafter until progression. Reaction, progression-free survival (PFS) and overall survival (OS) were assessed by investigators. Adverse events were graded using the National Cancer Institute CTCAE. After progression, survival information was collected every 6 months, including post-treatment information, until death.
Study end Points
The primary end point was the time from randomization to the first recording of disease progression (imaging-based) or patient death comparing PFS between the gefitinib/NKT arm and the gefitinib arm. Secondary endpoints included OS, TTP, safety and adverse reactions. OS was defined as the time from randomization to death from any cause. The safety index is based on the CTCAE standard of the degree of adverse reactions, so adverse events are recorded regardless of the cause to ensure objective reporting.
Statistical Analysis
The primary end point PFS was first compared to verify superiority between the two arms according to the protocol. 32 The test was considered effective only when it was demonstrated that the PFS of the gefitinib/NKT arm was greater than that of the gefitinib arm. The Kaplan–Meier method was used to estimate the survival curve of PFS, TTP and OS, and the log rank test was used to analyse the confidence interval. The research team considered that adverse reactions related to NKT-cell infusion, such as shivering, dizziness, fatigue, and fever, were not related to clinical outcomes. The median quartile interval was used to compare CEA and ALT, and Fisher's exact test was used to compare the incidence of adverse reactions between the two arms.
Results
Patient Allocation
From July 2017 to June 2021, a total of 19 subjects were randomized. In both arms, 18 (94.7%) completed all the study visits, and 1 (5.3%) withdrew early from the study. Among them, 11 patients (57.9%, 11/19) were treated with gefitinib/NKT, and 8 patients (42.1%, 8/19) were treated with gefitinib alone. Of the 11 subjects in the cellular immunotherapy group, 10 (90.9%) completed all the study visits, and 1 (9.1%) withdrew from the trial for several reasons. Of the 10 subjects who completed the trial, 3 (30%) died of disease progression during the follow-up period after disease progression. Of the 8 subjects in the gefitinib arm, 8 (100.0%) completed all the study visits. During the follow-up period after disease progression, 1 (12.5%) died due to disease progression. Information on the patient registration and distribution is shown in (Table 1). The database was finalized on June 30, 2021.
Patient Accumulation.

*1. All patients were randomly divided into GEF Arm and GEF/NKT Arm.
2. During the follow-up period, 1 patient in the GEF Arm died and three patients in the GEF/NKT Arm died.
Demographic and Baseline Characteristics
The baseline characteristics of the patients are listed in (Table 2). There were 19 subjects, including 8 males and 11 females. In the gefitinib/NKT arm, there were 4 males (36.4%) and 7 females (63.6%). In the gefitinib arm, there were 4 males (50.0%) and 4 females (50.0%). Among the smoking status, 11 people had no smoking history in GEF/NKT Arm (100%), 7 people had no smoking history in GEF Arm (62.5%), and 2 people had smoking history (37.5%). A uniform genetic test was taken before starting treatment. These patients had different histological subtypes, with exom19 mutation in GEF/NKT Arm in 7 patients (63.6%) and GEF Arm in 2 patients (25.0%). Exon 21 Leu858Arg mutation was found in 5 persons (36.4%) in GEF/NKT Arm and 6 persons (75.0%) in GEF Arm. Smoking status, KPS score, location, pathological type, stage and EGFR mutation type were compared, and Chi-square test was performed on the data, however, the difference was not statistically significant (all P > .05).
Baseline Demographics and Clinical Characteristics.

*Abbreviations: KPS, Karnofsky, Physical condition rating sheet; EGFR, epidermal growth factor receptor.
Treatment Process
Efficiency
Although different degrees of response to gefitinib treatment were observed, all randomly assigned volunteers in both arms showed response to the gefitinib without significant difference in both arms in the first 8 weeks (Figure 3A and 3B). Next, the week-8 was designated as the baseline to explore the efficiency of adaptive transferred NKT cells in delaying the gefitinib resistance in the patinets in this trial, and the efficiency was shown below.
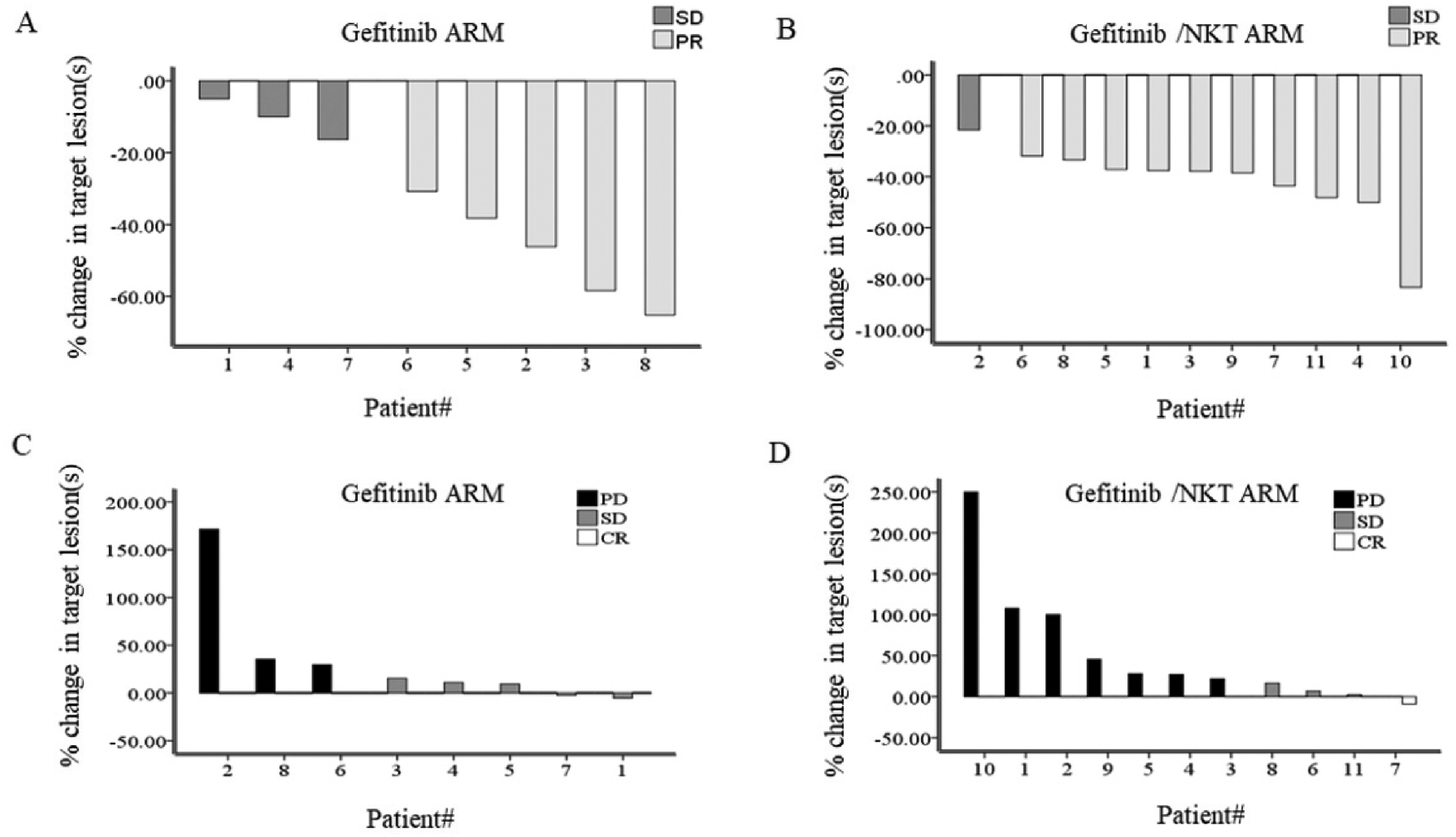
The response of the volunteers to gefitinib treatment at different stages.
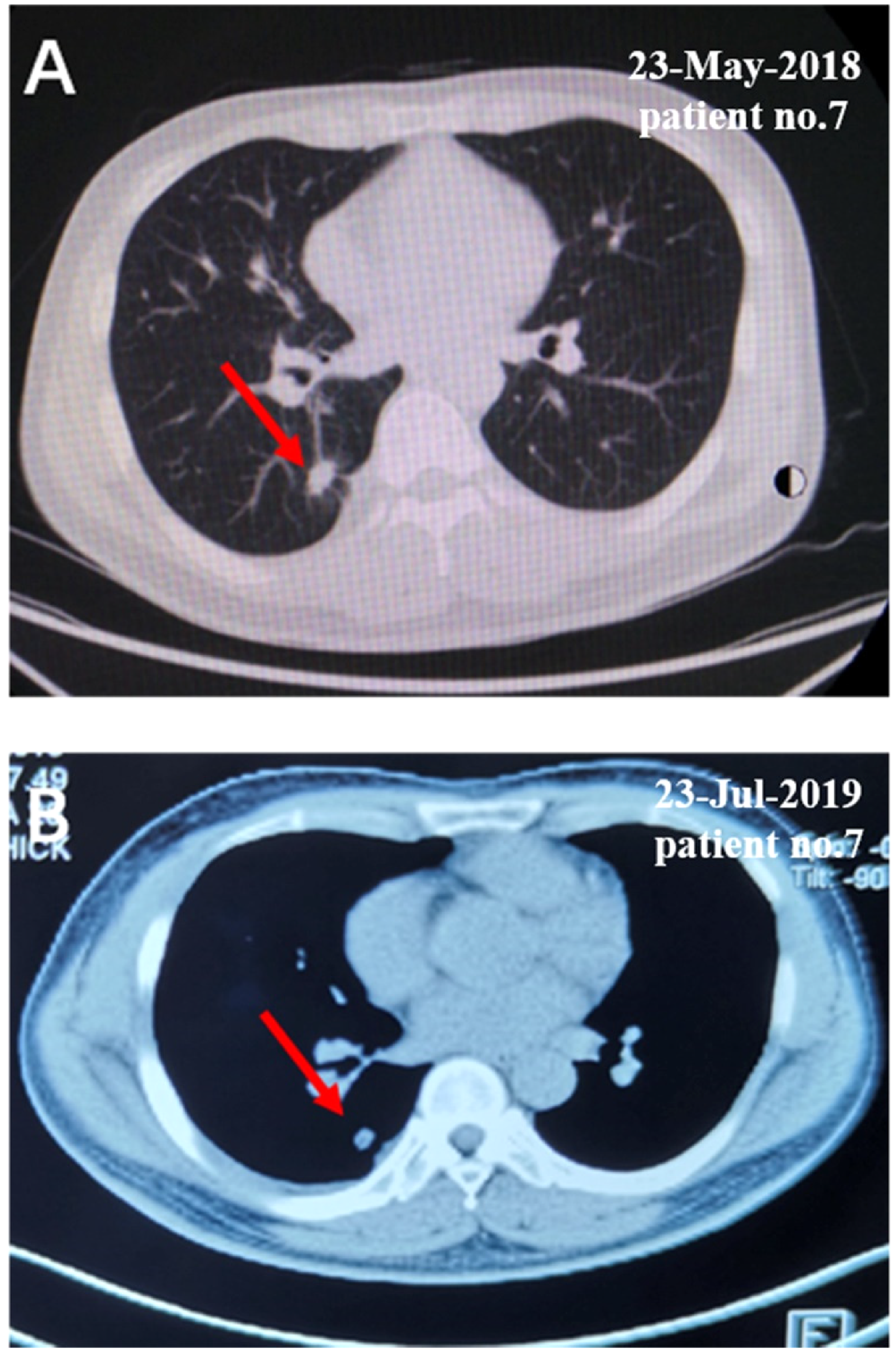
The CT scan data of the representative patient in the gefitinib/NKT arm after being treated with NKT-cell.
Efficacy Evaluation

PFS, TTP and OS of the volunteers.

The levels of CEA and ALT in the serum of the volunteers.
Common Adverse Events.
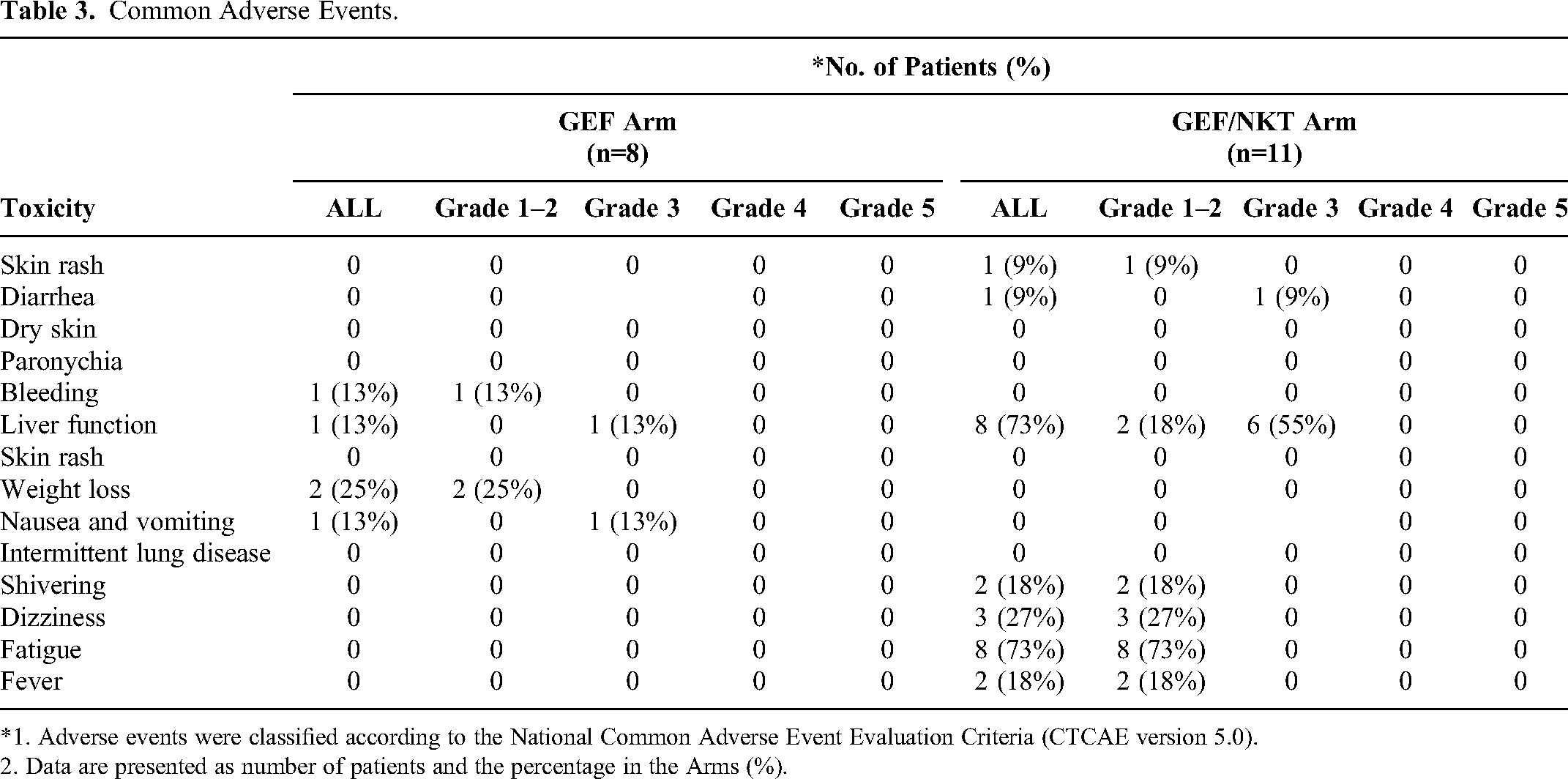
*1. Adverse events were classified according to the National Common Adverse Event Evaluation Criteria (CTCAE version 5.0).
2. Data are presented as number of patients and the percentage in the Arms (%).
The most common adverse event was abnormal liver function in the gefitinib/NKT arm, which mainly manifested as elevated transaminase levels (Figure 6B). Second, fatigue, dizziness and shivering appeared as immune activation reactions. The third most common adverse event included nausea, vomiting and diarrhoea in the digestive system. In the gefitinib arm, weight loss was the most common adverse event associated with the metabolic system. The next most common event was associated with the digestive system and involved liver dysfunction and nausea and vomiting. The third most common event was bleeding. With the exception of the cases described in the adverse events section, no other obvious abnormal results with clinical significance were found.
Discussion
In this study, we evaluate the safety and efficacy of allogenic cellular immunotherapy combined with EGFR-TKIs in the treatment of patients with advanced NSCLC and EGFR-sensitizing mutations compared with standard EGFR-TKIs monotherapy. By treating with the first generation EGFR-TKI gefitinib simultaneously, the adoptive transfer of allogenic CD8+ NKT cells dramatically increased the PFS time from 7 months in the gefitinib arm to 12 months in the gefitinib/NKT arm. Despite the lack of results regarding OS due to the limited numbers of cases, it should be noted that the patients in the gefitinib/NKT arm benefited from this immunotherapy without obvious or clinically relevant toxicity.
As a new kind of molecular targeted drug, EGFR-TKIs, including the first-generation gefitinib, represent a promising therapeutic option for NSCLC patients with EGFR mutations. 35 However, long-term treatment with EGFR-TKIs often results in drug resistance, which remains an unavoidable dilemma for NSCLC patients. Published data have shown that the median PFS times of patients treated with gefitinib vary from 8 to 9 months.36-38 In this study, 19 subjects (8 patients in the gefitinib arm and 11 in the gefitinib/NKT arm) were assessed for more than three years, and the median PFS time was 7 months, which was slightly different from that previously reported. These variations in PFS might be attributed to the different patient populations and the small size of the samples in the studies.
It is well known that the occurrence and development of tumours are the result of the immune escape of newly mutated tumour cells. Similar mechanisms were also found in the resistance to EGFR-TKIs treatment in the advanced NSCLC. Therefore, immunotherapy is regarded as a potential effective therapy strategy in these diseases. However, low PD-1 and PD-L1 expression levels and increased B7-H4 protein levels in tumour cells result in poor therapeutic efficacy in patients with advanced EGFR-mutant NSCLC.39-42 In this trial, we treated these patients by combining gefitinib with allogenic CD8+ NKT cells, which were amplified from PBMCs in vitro. The results showed that the median PFS time was extended from 7 to 12 months in the patients (Figure 5A). Moreover, the TPP analysis results of the secondary analysis index also showed that the TPP of the gefitinib/NKT arm was significantly longer than that of the gefitinib arm (Figure 5B). Therefore, these results showed that NKT cells combined with gefitinib partially improved the progression of EGFR-TKIs drug resistance caused by treatment with gefitinib alone in EGFR-mutant NSCLC patients.
CEA is an important tumour marker in lung cancer. 43 In this trial, the dynamic evaluation results of CEA showed that the CEA values in the gefitinib arm fluctuated greatly. After treatment with gefitinib alone for 7 months, the levels of CEA increased significantly and peaked at the 10th month, indicating disease progression in the patients. Conversely, the CEA values were well controlled in the gefitinib/NKT arm, and the curve was smooth until the end of the trial (Figure 6A). Thus, this CEA tumour index also reflects the good control effect of CD8+ NKT-cell immunotherapy on tumour load in patients.
To note, most patients in the gefitinib/NKT arm exhibited good medication compliance (80%) in the trial period. Unfortunately, during the COVID-19 epidemic in China, some patients could not go to the hospital to complete cell transfusions due to regional blockade policies in China from early March 2020. Therefore, these patients maintained gefitinib monotherapy at home, resulting in rapid progression of the disease and even death in the follow-up period. Therefore, these unpredictable force and majeure factors affected the efficacy evaluation in this trial to some degree. Moreover, the insufficient number of recruited volunteers in the trial directly impacted the OS evaluation.
This pilot study was also designed to assess the safety of CD8+ NKT-cell adoptive therapy. The clinical results indicated that this cellular immunotherapy offers potential clinical benefits with minimal adverse reactions. It has been reported that the most common side effects of gefitinib treatment include moderate degrees of rash, diarrhoea, nausea and vomiting, and these side effects typically occur in the first month of treatment. In this trial, no rash or diarrhoea occurred in the gefitinib arm, and only one patient developed nausea and vomiting. In the gefitinib/NKT arm, one patient developed skin rash and diarrhoea in the early gefitinib treatment period, and NKT-cell transfusion tended to aggravate diarrhoea and caused withdrawal from the trial. However, the direct effects and potential mechanisms of allogenic CD8+ NKT-cell treatment on diarrhoea need further exploration in the future. Moreover, in this study, chills (2 cases), dizziness (3 cases), fatigue (8 cases) and fever (2 cases) were observed in some patients in the gefitinib/NKT arm.
Published data have shown that gefitinib treatment alone typically has no obvious liver damage effects, 44 which was also confirmed by the data (Figure 6B) in this trial. Generally, our preliminary clinical data revealed no abnormal or intolerable changes in liver and kidney function caused by CD8+ NKT-cell infusion (data now shown). Herein, it should be noted that 8 patients in the gefitinib/NKT arm had elevated ALT levels (Figure 6B), indicating that this combined strategy caused mild liver injury. This might be due to the rich distribution of CD8+ NKT cells in the liver after donor infusion, which can cause cytotoxic activation.
CD8+ NKT cells are immune cells that express granzyme and perforin and exhibit high levels of IFN-γ secretion. Therefore, transferred CD8+ NKT cells exhibit direct cytotoxic effects to kill tumour cells and myeloid-derived suppressor cells (MDSCs) to inhibit tumour growth. 45 The impact of HLA allele mismatch on transplant outcomes is associated with an increased incidence of graft-versus-host disease in immunocompetent recipients and acute graft-versus-host disease (GVHD) in haploidentical stem cell transplantation. 46 These alloreactive reactions are mainly mediated by the activation of T cells from the donor and the host. Thus, allogenic CD8+ NKT-cell transfer can also induce alloreactivity in vivo to upregulate the efficiency of antitumor immunity and contribute to the benefits and adverse reactions of patients with NSCLC. Of note, the mismatch between the donor and the recipient potentially leads to hypersensitivity in the patients. Therefore, to prevent hypersensitivity, anti-allergic drugs should be simultaneously administered during cell transfusion. Fortunately, in this trial, no patients experienced serious allergic reactions, and only a few patients experienced low to moderate adverse reactions, such as fever, chills, and dizziness.
Since the end of our study is in 2021, most of the patients who participated in our study were infected with COVID-19. Although we did not actually study the relationship between COVID-19 and lung cancer progression, there is still some literature supporting the relationship between COVID-19 and lung cancer progression. So these results of the study were also potentially affected by COVID-19.
There have been other case reports where EGFR TKI's and ALK TKI's were continued in patients with active COVID 19 infections, but patients in these reports had mild infection and did not require intensive care, unlike patients in this study.47,48 The factors favoring the good response in patients could be the younger age and nonsmoker status in this study and results may be different for older patients who are smokers in many published works. However starting and continuing TKI therapy in driver mutated lung cancer warrants further studies. 49
On the other hand, due to the complexity of cell preparation, rigorous individualization, and early stage of clinical trials, only a small number of patients can be recruited for trials to evaluate the initial effectiveness and safety, resulting in a small sample size bias.
Currently, though the first generation (gefitinib and erlotinib), the second generation (afatinib and dacotinib) EGFR-TKIs and the third generation EGFR-TKIs, including osimertinib, have been developed, it is not easy to select the optimal therapy for the first-line treatment. Therefore, more and more attention has been paid to the orderly design of optimization strategies for various EGFR-TKI applications in clinical practice. 50 In oncology, a universally principle of “best therapy first” or “hit hard and early” have been accepted. At the earliest stage of EGFR-TKIs treatment in the advance NSCLC, these small molecular target drugs significantly induce tumor regression and gain disease control for a long period of time. Thus, such treatment provides one time window to receive efficacious combination therapy before patient status deteriorates. Considering the direct impact on the tumor immunological microenvironment after EGFR-TKIs treatment, a safe combination of immunotherapy and EGFR-TKIs is available in the first-line application. Unfortunately, a growing number of clinical trials for immunotherapy, such as using durvalumab and nivolumab, recruit the patients with well-tolerance to EGFR-TKIs, and such delaying treatment may limit overall benefit of the entire therapy.19,51 In this study, we combined CD8+ NKT cell adoptive transfer with EGFR-TKIs at the opening phase, and the data declared that this synergistic action lifted benefit of the entire therapy, including the safety and efficacy. Thus, it indicated that the treatment using CD8+ NKT cells adoptive transfer combine with EGFR-TKIs may be a potential strategy for the advanced NSCLC with EGFR mutation.
Conclusions
In summary, we combined allogenic CD8+ NKT-cell immunotherapy with EGFR-TKI (gefitinib) for EGFR-mutant NSCLC treatment and demonstrated that this synergistic effect benefit patients with prolonged PFS, and no serious adverse reactions. The data suggest the potential usage of this therapeutic regimen in EGFR-mutant NSCLC. However, more trials are still needed for confirmation, and the underlying mechanisms require further exploration.
Supplemental Material
sj-xlsx-1-tct-10.1177_15330338241273198 - Supplemental material for EGFR-TKIs Combined with Allogeneic CD8+ NKT Cell Immunotherapy to Treat Patients with Advanced EGFR-Mutated Lung Cancer
Supplemental material, sj-xlsx-1-tct-10.1177_15330338241273198 for EGFR-TKIs Combined with Allogeneic CD8+ NKT Cell Immunotherapy to Treat Patients with Advanced EGFR-Mutated Lung Cancer by Fei Ye, Xiao Yuan, Wanjun Yu, Yali Ma, Chaoming Mao, Xiaoqin Li, Jian Li, Chunhua Dai, Fenhong Qian, Junrong Li, Xiujuan Fan, Yuepeng Zhou, Dongfang Dai, Deqiang Wang, Deyu Chen, Sheng Xia and Minghui Zhang in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgements
We are indebted to the blood donors and the patients who participated in this study, as well as their families,thank all investigators who devote their time and passion in the implementation of this study.
Availability of Data and Materials
The raw data supporting the conclusions of this article will be made available by the authors with https://www.jianguoyun.com/p/DQ-_B3oQsJjtCRjOnqsE and ![]()
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jiangsu Provincial Key Research and Development Program, the National Natural Science Foundation, China, (grant number BE2017696, 81871234, 82172763, 82272824).
Ethics Approval and Consent to Participate
All subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki,Institutional review board approval was obtained for the trial from the Biomedical Research Ethics Committee of affiliated hospital of Jiangsu University (Approved No. of ethic committee: SWYXLL20170904). This Trial was registered on 21/11/2017 with ![]() , ChiCTR-IIR-17013471.Written informed consent is obtained from all participants.
, ChiCTR-IIR-17013471.Written informed consent is obtained from all participants.
Consent
All participants provide their written informed consent forms before their treatment of Gefitinib or Gefitinib/NKT cell. This study does not contain any individual person information of patients, and all patients have consented to participate in this study and publish this paper.
Trial Registration Number
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
