Abstract
Accelerated partial breast irradiation is an attractive alternative to conventional whole breast radiotherapy for selected patients. Recently, CyberKnife has emerged as a possible alternative to conventional techniques for accelerated partial breast irradiation. In this retrospective study, we present a dosimetric comparison between 3-dimensional conformal radiotherapy plans and CyberKnife plans using circular (Iris) and multi-leaf collimators. Nine patients who had undergone breast-conserving surgery followed by whole breast radiation were included in this retrospective study. The CyberKnife planning target volume (PTV) was defined as the lumpectomy cavity + 10 mm + 2 mm with prescription dose of 30 Gy in 5 fractions. Two sets of 3-dimensional conformal radiotherapy plans were created, one used the same definitions as described for CyberKnife and the second used the RTOG-0413 definition of the PTV: lumpectomy cavity + 15 mm + 10 mm with prescription dose of 38.5 Gy in 10 fractions. Using both PTV definitions allowed us to compare the dose delivery capabilities of each technology and to evaluate the advantage of CyberKnife tracking. For the dosimetric comparison using the same PTV margins, CyberKnife and 3-dimensional plans resulted in similar tumor coverage and dose to critical structures, with the exception of the lung V5%, which was significantly smaller for 3-dimensional conformal radiotherapy, 6.2% when compared to 39.4% for CyberKnife-Iris and 17.9% for CyberKnife-multi-leaf collimator. When the inability of 3-dimensional conformal radiotherapy to track motion is considered, the result increased to 25.6%. Both CyberKnife-Iris and CyberKnife-multi-leaf collimator plans demonstrated significantly lower average ipsilateral breast V50% (25.5% and 24.2%, respectively) than 3-dimensional conformal radiotherapy (56.2%). The CyberKnife plans were more conformal but less homogeneous than the 3-dimensional conformal radiotherapy plans. Approximately 50% shorter treatment times and 50% lower number of delivered monitor units (MU) were achievable with CyberKnife-multi-leaf collimator than with CyberKnife-Iris. The CyberKnife-multi-leaf collimator treatment times were comparable to 3-dimensional conformal radiotherapy, however, the number of MU delivered was approximately 2.5 times larger. The suitability of 10 + 2 mm margins warrants further investigation.
Keywords
Introduction
Breast-conserving surgery, resection of the primary breast tumor and a rim of surrounding normal tissue, is the standard of care for early-stage patients. 1 Conventional adjuvant therapy for post-lumpectomy patients consists of whole breast conformal radiation therapy with doses of 45 to 50 Gy delivered over 5 to 6 weeks. The rationale for whole breast irradiation is the eradication of residual disease after lumpectomy. However, in addition to treating the entire breast, this form of radiotherapy exposes the skin, chest wall, lung, and, on occasion, heart to high doses of radiation.
Studies have shown that the majority of local recurrences occur within the same quadrant as the lumpectomy site. 2,3 These findings indicate that treating the lumpectomy cavity plus a margin, partial breast irradiation, may be as effective as treating the whole breast, with the advantage of less toxicity to normal tissue. Treating limited breast volumes with radiation could allow for higher doses per fraction and thus shorter treatment times, typically much shorter than the conventional treatment period of 5 to 6 weeks. This approach, accelerated partial breast irradiation (APBI), is an attractive alternative for patients who may find extended weeks of radiation cumbersome.
Several nonrandomized studies using various brachytherapy-based APBI techniques have shown that the rate of local recurrences remain comparable to those in a highly selected group of patients with low-risk disease who receive conventional treatment. 4 In addition, randomized studies using intraoperative approaches with similar eligibility and goals show comparable results. 5,6 However, it should be noted that these studies are all limited by a short mean follow-up time (<10 years). Large studies have also reported acceptable cosmetic outcomes, 7,8 although longer follow-up may increase rates of normal tissue damage over time. 9
A variety of methods are currently employed to deliver APBI: external beam therapy may be applied using a conventional linac with 3-dimensional conformal radiotherapy (3D-CRT), 10 intensity-modulated radiotherapy (IMRT), 11 or arc therapy 12 ; by TomoTherapy 13 ; or using intraoperative radiotherapy with electrons (ELIOT) or photon therapy (IntraBeam [Carl Zeiss, Oberkochen, Germany]). 14 Brachytherapy methods include balloon-based single source devices (Mammosite [Hologic Inc., Bedford, MA]) 15 or hybrid multisource devices (Contura [SenoRx, Inc, Aliso Viejo, CA], SAVI [Cianna Medical, Aliso Viejo, CA], ClearPath [North American Scientific Inc, Chatsworth, CA]). 16 –18 Numerous studies have been carried out to compare and contrast the efficacy of these methods.
Despite its potential advantages, APBI with external beam techniques is not without controversy. Recht
CyberKnife (CK; Accuray Inc, Sunnyvale, California) has recently emerged as a possible alternative treatment modality for APBI. The CK has the ability to deliver circular fields ranging in size from 5 to 60 mm using either fixed aperture collimators or via the variable aperture Iris collimator (CK-Iris). Accuray recently announced the addition of a multi-leaf collimator (CK-MLC) to the system allowing treatments using fields ranging in size from a square of side 7.5 mm to 120 mm × 100 mm at 800 mm source-axis distance. The 41-leaf MLC allows 100% overtravel and interdigitation and promises improved plan quality, reduced treatment times, and a lower number of monitor units. One of the major advantages of CK as a treatment modality is its ability to track patient motion and adapt the treatment accordingly. According to Baglan
The goal of this study is to provide a comprehensive comparison between CK plans and plans from a well-established APBI modality. The 3D-CRT was chosen as the most useful comparison modality for our clinic, given our experience and expertise with it. This comparison is achieved by comparing these modalities from (1) a purely dosimetric viewpoint and (2) a clinically relevant viewpoint. The former is achieved by comparing plans with identical margins, while the latter recognizes the lack of motion tracking of 3D-CRT and thus applies appropriate margins. In both comparisons, treatment efficiency and monitor units are also considered. We believe this study is relevant because many clinics are already providing APBI treatments with CK-Iris without clear guidance on the benefits and detriments this may provide. In addition to considering the Iris collimator, the CK component of the study presents what we believe to be the first dosimetric evaluation of the CK-MLC for APBI. We outline the advantages this system has over CK-Iris for this treatment.
Materials and Methods
In this study, we considered 9 patients who were treated with whole breast CRT at our clinic. These included 5 left-sided and 4 right-sided patients, with tumor beds ranging in maximum extent from 0.6 to 1.8 cm. Five sets of plans were created for these patients and are compared in this study: 2 sets of 3D-CRT plans and 3 sets of CK plans. The margins for CK approach are 10 mm and 2 mm for tumor-bed to CTV and CTV to PTV, respectively, and follow the initial approach of Vermeulen
A Summary of the 5 Sets of Plans Created for the 9 Patients Under Consideration: the Treatment Modality, the Type of Collimator Used, the Dose Calculation Algorithm, the Expansion in mm From the Tumor to the CTV and the CTV to the PTV, and the Dose in Gy and the Fractionation.

Abbreviations: 3D-CRT, 3-dimensional conformal radiotherapy; CK, CyberKnife; MLC, multi-leaf collimator.
Target and normal structures were contoured in the Pinnacle treatment planning system (Philips, Andover, Massachusetts) following the guidelines of RTOG-0413. 33 For both sets of expansions, the CTV and PTV were limited to 5 mm from the skin surface and excluded the chest wall and pectoralis muscles. The ipsilateral breast tissue was defined by the prescription isodose volume of the whole breast treatment plans delivered to these patients. The contralateral breast tissue was contoured mimicking the shape and extent defined by the ipsilateral breast tissue. The skin was defined as ring of 3 mm thickness from the surface seen on the computed tomography scan. The chest wall of the ipsilateral breast was contoured as the thickness of a rib, with the sternum defining the medial border and the mid-axillary line defining the lateral border. This contour extended from the clavicle to the inframammary fold. The superior border of the heart was defined as just inferior to the level at which the pulmonary trunk branches into the left and right arteries, and the inferior border was the most inferior extent of the heart near the diaphragm. All the mediastinal tissue between these borders was contoured, including the great vessels and excluding the esophagus and pericardial fat. The smaller PTV and the normal structures were exported from Pinnacle to the CK Multiplan stations. The patients included in the study were simulated with the arm on the ipsilateral side raised above the head. For CK treatment, it is preferred to simulate the patient with the arms along the side of the body to prevent extending extremities outside the patient safety zone. This position is also more comfortable for the patient and helps minimizing target deformation and patient motion during treatment. In order to increase the potential number of lateral beams that can be used without concern for beams passing through the arms, CK patients can be simulated on a thick pad, so the arms fall below the level of the body.
Forward planning techniques were used for both sets of 3D-CRT plans in Pinnacle for a Siemens Primus conventional linear accelerator. This has a maximum 40 × 40 cm2 field collimated by independent jaws and an MLC with 1 cm wide leaves. Between 4 and 5 noncoplanar 6 MV beams were arranged following the description of Baglan
The CK planning system, Multiplan (v4.5 for CK-Iris and v5.1 for CK-MLC), employs inverse planning via sequential optimization, given a set of dose objectives and constraints. 34 The set of CK plans created for the MLC system was calculated with the finite-sized pencil beam algorithm (FSPB). 35 The CK-Iris plans were calculated using Monte Carlo (MC), as this provides the most accurate lung and skin dose. These plans were also recalculated using the ray-tracing (RT) algorithm to facilitate a comparison between CK-Iris and CK-MLC, as at the time of writing MC is not available for CK-MLC, and FSPB lacks a correction for lateral electron scattering. All available field sizes between 10 mm and up to the size of the PTV (maximum 60 mm) were selected for the CK-Iris plans. In all cases, the beams were constrained to avoid the contralateral breast, the thyroid, and the heart. Beam-reduction and time-reduction tools 24 were employed for all CK plans to reduce the treatment time while retaining the dose goals for target and critical structures.
Both the 3D and CK cases were planned with the aim of meeting the following dose guidelines 33 (summarized in Table 2): (1) the volume of ipsilateral breast receiving 100% for the prescription dose (V100%) should be less than 35%, and the V50% should be less than 60%; (2) the contralateral breast should not receive more than 3% of the prescription dose; (3) the ipsilateral/contralateral lung V30%/V5% should not exceed 15%; (4) the heart V5% should be less than 5% for right-sided patients and less than 40% for left-sided patients; and (5) the maximum dose to the thyroid should not exceed 3% of the prescription dose. In addition, the following goals were set for the skin and chest wall: (1) the volume of a 3 mm ring of skin receiving the prescribed dose should not exceed 5 cm3 and the chest wall V90% should not exceed 10 cm3; (2) the maximum dose to the skin and chest wall should not exceed 145% and 120% of the prescribed dose, respectively. The isodose lines covering 95% of the PTV should be at least 70%. Given the findings of the Recht and Jagsi studies, extra care was taken during planning to ensure that the ipsilateral breast and lung doses were kept as low as possible. Figure 1 shows an example of beam arrangement for a 3D-CRT, CK-Iris, and CK-MLC plan.
The Average Dosimetric Indices for 9 Patient Plans Using 3D-CRT for Small (10 + 2 mm) and Larger (15 + 10 mm) Margin, and CK for the Iris Collimator With Dose Calculated Using Both MC and RT and for the MLC Calculated With the FSPB.
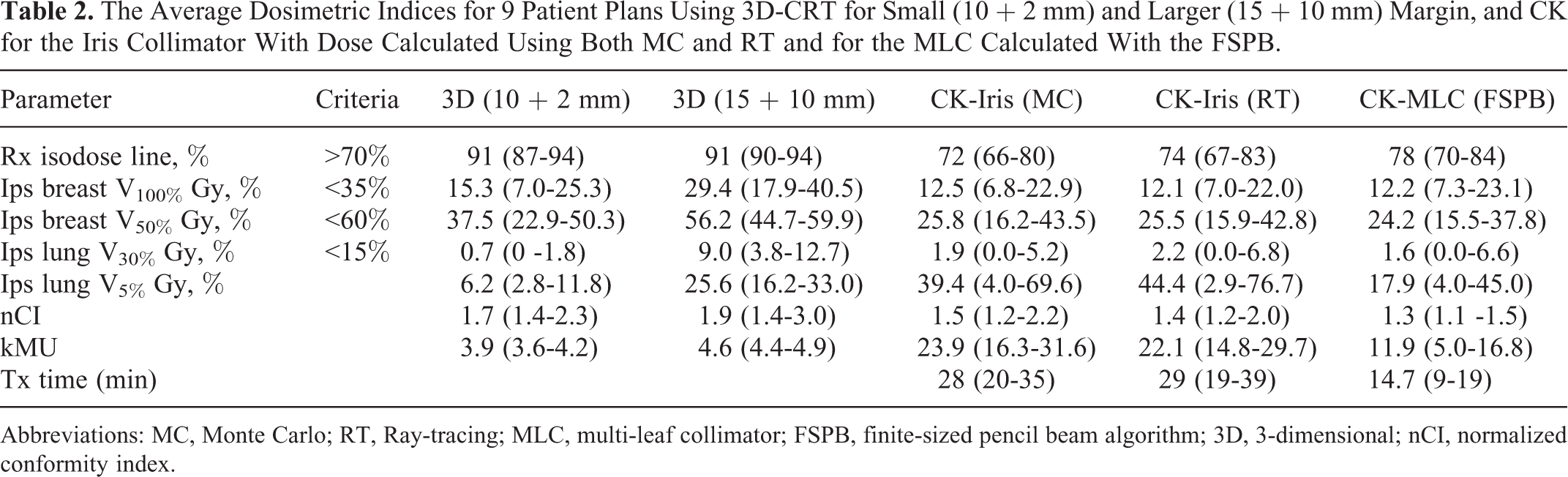
Abbreviations: MC, Monte Carlo; RT, Ray-tracing; MLC, multi-leaf collimator; FSPB, finite-sized pencil beam algorithm; 3D, 3-dimensional; nCI, normalized conformity index.

An example of the beam arrangement for a 3D (left), CK-Iris (middle), and CK-MLC (right) plan for 1 patient. CK indicates CyberKnife; MLC, multi-leaf collimator.
The approach taken to planning these cases was to meet the coverage constraint without exceeding the tolerance goal to the organs at risk. In cases where this was not possible, a lower coverage was accepted. Of the organs at risk, particular attention was paid to the ipsilateral breast normal tissue and the ipsilateral lung. Every effort was made to keep these as low as possible without exceeding the dose to the other organs at risk. In addition to constraining the ipsilateral lung V30%, tuning structures around the targets (shells) were created to control the dose fall-off away from the targets and the low dose levels in the normal tissue. The plans were compared by considering the ipsilateral breast V100% and V50%, the ipsilateral lung V30% and V5% the treatment time, the number of monitor units delivered, and the PTV normalized conformity index (nCI = TV × PIV/[TIV]2, where TIV is the target volume inside the prescription isodose volume; PIV is the prescription isodose volume and TV is the target volume). A conformity index equal to 1 represents perfect target conformity, and larger values indicate worse conformity. All plans were rescaled to have 95% coverage. An example of dose distribution in the axial and coronal plane for a small margin 3D-CRT, CK-Iris, and CK-MLC plan is shown in Figure 2. Figure 3 shows the corresponding dose distribution for the large margin 3D-CRT plan.

An example of dose distribution in the axial and coronal plane for a small margin 3D (left), CK-Iris (middle), and CK-MLC (right) plan for 1 patient. CK indicates CyberKnife; MLC, multi-leaf collimator.

An example of dose distribution in the axial and coronal plane for a large margin 3D plan.
Results
A summary of the comparison metrics (including ipsilateral breast V100% and V50%, ipsilateral lung V30% and V5%, treatment time, nCI, and number of monitor units [MU]) for each planning technique is shown in Table 2. As mentioned earlier, the purpose of the CK-Iris (RT) plans is to facilitate a comparison of the CK-MLC plans with CK-Iris plans, as both FSPB and RT algorithms are known to be less accurate than MC for air cavities. The implication is that the metrics that have similar results for the CK-Iris MC, and RT evaluations can be directly compared to the MLC plans; where they differ, the CK-MLC (FSPB) result should be compared to CK-Iris (RT) result.
The PTV 70% isodose prescription line was met in all of the 3D and MLC plans, and 2 CK-Iris (MC) plans were just below the threshold at 67% and 69%. Of the planning goals not included in the comparison metrics, we found that the contralateral breast, the contralateral lung, the heart, thyroid, and skin were all below the maximum allowed doses. The chest wall constraint was met in all of the small margin 3D-CRT plans but, due to the larger PTV expansion, failed in 8 of the 9 large margin 3D-CRT plans (19.7-59.8 cm3). The maximum dose was exceeded in three of the CK-Iris (RT) plans (120.5%-122.7%), and volume constraint was exceed in one CK-Iris (RT) plan (11.9 cm3), but this can be attributed to the inaccuracies of the dose calculation algorithm in the vicinity of an inhomogeneity (the lung), as these were all met when the plans were calculated with the MC algorithm. One CK-MLC plan barely exceeded the chest wall maximum dose constraint (121.0%), but again, we believe that this can be attributed to the inaccuracies of the dose calculation algorithm.
The mean ipsilateral breast V100% was comparable for the CK plans, 12.5%, 12.1%, and 12.2% for CK-Iris (MC), CK-Iris (RT), and CK-MLC (FSPB), respectively, as were the mean V50%: 25.8%, 25.5% and 24.2%. The V100% for the small margin 3D-CRT plans was comparable at 15.3%; however, the V50% was significantly higher than that of the CK plans at 37.5%. Not surprisingly, due to the larger margins, the 3D-CRT with the 15 + 10 mm expansion had a mean ipsilateral breast V100% of 29.4% and a V50% of 56.2%.
The small margin 3D-CRT plans had the lowest mean ipsilateral lung V30% and V5% (0.7% and 6.2% respectively). The mean ipsilateral lung V30% was 2.2% or less for the three CK sets of plans, and the mean ipsilateral lung V5% was significantly lower for CK-MLC than for CK-Iris (RT), 17.9% as compared to 44.4%. The corresponding CK-Iris (MC) dose was 39.4%. The large margin 3D-CRT plans had the highest V30% (9.0%) and an average V5% that was lower than that of the CK-Iris plans and higher than that of CK-MLC plans at 25.6%. The average dose volume histograms for the PTV and ipsilateral breast and lung for these three modalities is shown in Figure 4.

Average dose–volume histograms of the (A) PTV, (B) the ipsilateral breast, and (C) the ipsilateral lung for large and small margin 3D-CRT, CK-Iris (RT) and CK-Iris (MC), and CK-MLC. CK indicates CyberKnife; MLC, multi-leaf collimator; MC, Monte Carlo; CRT, conformal radiotherapy.
The CK plans resulted in a more conformal dose distribution around the target but required a larger number of MU. The nCI was higher for the 3D-CRT plans, 1.7 and 1.9 for small and large margin plans, respectively, than for the CK plans 1.5, 1.4, and 1.3 for CK-Iris (MC), CK-Iris (RT), and CK-MLC (FSPB), respectively. The mean number of MU required for the 3D plans was 3929 and 4611 for the small and large margin plans respectively, and 23861, 22059, and 11903 for CK-Iris (MC), CK-Iris (RT), and CK-MLC (FSPB) plans, respectively. A factor of approximately 2 reduction in the treatment time was observed in CK-MLC compared with CK-Iris (RT) plans, with the former averaging 14.7 minutes. The similarity in treatment time between the CK-Iris (MC) and CK-Iris (RT) stems from the fact that these 2 plans are identical in terms of robot position, beam orientations, beam apertures, and weighting. The only difference between the 2 sets of plans is a scaling of the dose due to the different calculation engine. Note that the CK-treatment time estimate includes the time interval when the aperture is reshaping at a single node and the travel time between robot position, as well as the imaging interval (every 60 seconds) and automatic intrafraction correction. This estimate does not include setup time. The mean 3D-CRT beam-on time is 15.3 minutes, considering the 300 MU/min dose rate Siemens linac for which these treatments were planned. (It should be noted, however, that higher dose rates are possible in more recently released conventional linacs.) An estimated 2 to 3 minutes per beam could be added for the therapist to enter the treatment room, rotate the couch for the next noncoplanar beam, and check the patient setup, adding an average of approximately 8 minutes to the average 3D-CRT treatment time.
Discussion
A very limited body of literature exists on APBI using CK. Recently published clinical results from one center have indicated a good outcome with no acute toxicities in the 9 patients treated, at a median follow-up of 7 months.
28
These results should be considered preliminary, however, as the length of follow-up is not sufficient to definitively prove a good outcome with this modality. Retrospective dosimetric studies including CK have provided promising results for the use of the modality for APBI. Heinzerling
In the current study, the 3D-CRT plans were created with a tumor cavity to CTV and CTV to PTV margin of 10 + 2 mm and 15 + 10 mm. The CK plans employed a 10 + 2 mm margin, and the dose was evaluated using MC and RT for CK-Iris and FSPB for CK-MLC. By using the same margins for both modalities, we are essentially comparing the dosimetric characteristics of the planning systems and ignoring the tumor-tracking advantage provided by CK. For these cases, the 3D-CRT plans have a significantly smaller low dose component to the ipsilateral lung than the CK plans, at the expense of higher ipsilateral breast V50% and worse PTV conformality. In practice, however, 3D-CRT lacks the tracking advantage of CK and, as such, the small margins are not appropriate for 3D-CRT. For the remainder of the study, we only consider 3D-CRT with margins as prescribed by the national protocol on APBI, that is, 15 + 10 mm.
Not surprisingly, with the greater margins the dose to the breast in the 3D-CRT increases, with the V50% and V100% more than double that of the CK plans. The ipsilateral lung V30% and Dmax for 3D-CRT plans also far exceed that of the CK plans, and the low dose component, V5% lies in between that of the CK-Iris and the CK-MLC plans with the latter having the lowest average value. For all modalities, the contralateral lung doses were low (less than 5% of the prescription dose), and the total lung doses were well below the threshold doses recommended for SBRT fractionations to preserve basic lung functions. 39 The average plan MU was lower for 3D than for CK by at least a factor of 2. When the time to set-up the noncoplanar 3D-CRT beams was considered, the treatment time was found to lie between that of the CK-Iris and the CK-MLC deliveries, with the CK-MLC plans once more having the lowest value.
Comparing the 2 types of CK collimation, the plans created using the MLC gave similar doses to most organs at risk as the Iris collimator with the exception of the lung V5%, which had a mean value of 17.9% for the CK-MLC plans and 44.4% for the CK-Iris (RT) plans. The MC calculated value for this parameter was 39.4% for the Iris collimator indicating that with this more accurate method, the CK-MLC value could be reduced even further. The most significant benefit provided by the CK-MLC over the CK-Iris collimator is the sizeable reduction in the number of MU delivered and in the treatment time. The mean number of MU delivered by the CK-MLC plans was 11 903, a factor of approximately 2 less than that for CK-Iris. However, this number is still 3 times larger compared to the mean number of MU delivered by the 3D-CRT plans. The number of MU is proportional to the leakage of radiation from the treatment unit, which contributes to the total body dose delivered to the patients. An increase in the patient integral dose is potentially associated with an increased risk of secondary malignancies, particularly in long-term survivors. Therefore, it is important to develop strategies for reducing the number of MU without compromising plan quality. An additional benefit to the CK-MLC system compared to CK-Iris is the associated reduction in average treatment time—14.7 minutes, half that of the mean treatment time for the CK-Iris plans.
Conclusion
For APBI, compared to 3D conformal approach, CK offers higher conformality due to the higher number of noncoplanar beams and less normal breast tissue exposure which is attributable to image-guided tracking. Additionally, the newly developed CK-MLC has the advantage of lower lung dose and comparable treatment times to 3D-CRT. A drawback of CK compared to 3D-CRT is the higher number of MUs. It is not clear how the pros and cons of each approach may translate to better outcomes clinically. The effect of low dose level to the lung and heart also require further investigation, as does the suitability of 10 + 2 mm margins. Careful evaluation on a patient-by-patient basis is therefore required when employing this emerging technology.
Footnotes
Acknowledgments
The authors thank Accuray Incorporated for providing access to MultiPlan 4.5 and 5.1 workstations and for valuable discussions regarding this work.
Note
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. L. M. Goggin is employed at Accuray Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
