Abstract
Background
This study aims to assess the efficacy and safety of Endostar in the management of locally advanced cervical cancer.
Methods
This retrospective, 2-center study enrolled 41 patients with locally advanced cervical cancer between June 2017 and December 2020. The patients were subjected to a combination of Endostar and chemoradiotherapy until they experienced disease progression or an unacceptable level of toxicity. The patients in the Endostar combined chemoradiotherapy (E + CRT) and CRT groups were matched 1:1 based on clinical features, including age, disease stage, and pathological type. The therapeutic efficacy and safety outcomes were compared between the 2 groups.
Results
Early treatment response: the CR rates in E + CRT and CRT groups were 48.8% and 26.8%, respectively (χ2 = 4.20, P < .05). The ORR and DCR were not significantly different between the 2 groups. Long-term efficacy: there was no significant difference in the 1-year and 2-year PFS rates and OS rates between 2 groups. However, in patients with stage IIB, subgroup analyses revealed a significant difference in PFS between the 2 groups (P < .05). Prognostic factors: stage, Eastern Cooperative Oncology Group (ECOG) score, and tumor size were independent predictive factors for PFS, while ECOG score and tumor size were those of OS in patients with locally advanced cervical cancer. Safety: The incidence of grade III-IV myelosuppression was significantly lower in E + CRT group than in CRT group (P < .05).
Conclusions
The combination of Endostar and concurrent CRT exhibited greater efficacy in treating locally advanced cervical cancer with no severe adverse reactions, when compared to simple CRT. It is expected that this approach will evolve into a new treatment alternative for patients with locally advanced cervical cancer.
Keywords
Introduction
Cervical cancer incidence and mortality rates are among the highest for female malignancies globally. 1 Surgery intervention is potentially curative for early-stage cervical cancer, while concurrent radiotherapy and chemotherapy are the standard treatment for locally advanced cases. Nevertheless, cervical cancer patients continue to encounter treatment challenges such as high recurrence and low survival rates.2,3 Following Dr Folkman's hypothesis that tumor growth is dependent on the process of angiogenesis, an antiangiogenic agent known as endostatin was subsequently identified. An appropriate amount of antiangiogenic agents can reconstruct tumor blood vessels in a short period of time and form vascular homeostasis, increase the function of blood vessel perfusion and reverse the multidrug resistance of chemotherapy, which is also called “vascular normalization.” However, clinical research stage endostatin exposed the preparation of difficult, poor drug stability shortcomings, and endostatin single drug efficacy is not obvious, usually cannot significantly shrink tumors, Endostatin finally failed to survive Phase II clinical trials and was discontinued in 2004. However, the researchers didn't stop at endostatin. A new 20-kDa recombinant human endostatin was purified from Escherichia coli by Chinese researchers in 2005 and named Endostar. The protein comprises 184 amino acids and is derived from human collagen X VIII, specifically the noncollagenous (NC1) domain, which contains the endostatin peptide. Endostar links 9 amino acids to the N-terminus of endothelial inhibitors, preventing protein aggregation and helping to correctly pair disulfide bonds by enriching the intermediate state at high concentration. This proprietary reconstitution technology is characterized by high concentration (>1 g/L), high reconstitution rate (>50%), and high purity (>99.9%). The modified protein is acid-resistant (pH 2.5), heat-resistant (100 °C), highly soluble (>15 mg/mL), and very stable (injection: 18 months × 4 °C). Structural modifications have increased the biological half-life by a factor of 2 to 5 and the activity by a factor of at least 2. 4 Endostar acts as an antiangiogenic agent, inhibiting tumor angiogenesis by blocking the proliferation and migration of vascular endothelial cells and inducing their apoptosis. 5 It has been extensively used via intravenous injection in combination with chemotherapy or radiotherapy for lung cancer, colorectal cancer, and other malignant tumors. 6 While Endostar has displayed potential in clinical practice, it may also have associated adverse reactions or side effects. In phases I to III clinical studies, the common adverse reactions (1% < incidence < 10%) of Endostar were mainly cardiac adverse reactions (6.38%), which were commonly seen in patients with a history of coronary artery disease and hypertension, but they were all reversible and did not require symptomatic treatment for relief. Rare adverse reactions (0.1% < incidence < 1%) were gastrointestinal reactions (occasional nausea, vomiting, diarrhea, abnormalities of hepatic function, mainly consisting of asymptomatic elevation of aminotransferases, and jaundice which was mainly mild2moderate and rarely severe), and cutaneous allergic reactions (manifested as generalized maculopapular rash with pruritus). In this multicentre clinical study, no deaths related to adverse drug reactions were observed in the 470 patients treated with this product. The aim of this trial is to assess the safety and effectiveness of Endostar in combination with chemoradiotherapy for treating locally advanced cervical cancer. This research provides novel evidence for further randomized controlled studies.
Materials and Methods
Study Design
The reporting of this study conforms to STROBE guidelines (insert reference number). 7 All patient details have been de-identified.
Patient Selection
This study retrospectively analyzed all patients who met the inclusion and exclusion criteria according to the 2009 International Federation of Gynecology and Obstetrics (FIGO 2018) for stage II B-IV A cervical cancer at the second People's Hospital of Changzhou, Nanjing Medical University and the First People's Hospital of Suzhou University from June 2017 to December 2020. All patients signed informed consent forms for the use of Endostar.
Eligibility Criteria
The following criteria were used to enroll patients: females aged 18 to 75 with pathologically confirmed cervical cancer, cervical squamous cell carcinoma, adenosquamous cell carcinoma, or adenocarcinoma at stage II B-IV A (FIGO 2018); Eastern Cooperative Oncology Group (ECOG) physical status score 0, 1 or 2; at least one measurable lesion according to RECIST 1.1; baseline blood panel and biochemical parameters in line with the following criteria: hemoglobin ≥ 90 g/L, neutrophil absolute count ≥ 1.5 × 109/L, platelet ≥ 100 × 109/L; alanine aminotransferase, aspartate aminotransferase ≤ 1.5 times normal upper limit, ≤ 5 times normal upper limit (with liver metastasis), serum total bilirubin ≤ 1.5 times normal upper limit, and serum creatinine ≤ 1.25 times normal upper limit or endogenous creatinine clearance ≥ 50 mL/min (using the standard Crockcroft-Gault formula); eligible patients should have adequate bone marrow, liver and kidney function, and heart function reserves for radiotherapy and chemotherapy, estimated survival ≥ 6 months, and no obvious coagulopathy. Patients were excluded if they had other primary malignant tumors, contraindications to radiotherapy and chemotherapy, serious infection, myocardial infarction, serious arrhythmia, serious cerebrovascular disease, psychosis, uncontrolled diabetes, were pregnant, breastfeeding or fertile women who had not taken contraceptive measures, received targeted therapy or were being tested with other drugs, or if investigators deemed other reasons for excluding the patient.
Treatment and Dose Adjustment
Radiotherapy: All patients underwent external beam radiation therapy (EBRT) combined with high-dose rate intracavitary brachytherapy (HDR-ICBT).
EBRT: Patients received 3-dimensional conformal/intensity modulated external irradiation, 5 times a week, 1.8 to 2 Gy each time, with a total dose of 45 to 50.4 Gy. If imaging examination indicates positive para-aortic or pelvic lymph nodes, an additional dose of 10 to 15 Gy would be given for positive lymph nodes. HDR-ICBT was given after 15 times of external irradiation. Point A was the dose reference point, with a single dose of 5 to 7 Gy given a week. External irradiation was not performed on the day of treatment. The total dose at point A in both groups was ≥ 80 Gy.
Endostar: Recombinant human vascular endostatin (Endostar, Shandong Siemcere Maidejin Bio-pharmaceutical) 30 mg/d was given via intravenous micropump maintenance at radiotherapy D1-7 with at least 1 cycle. 8
Chemotherapeutic regimen: (1) paclitaxel for injection (albumin-bound type) 260 mg/m2 or paclitaxel liposome 135 mg/m2 combined with nedaplatin 80 to 100 mg/m2 given every 3 weeks for at least 2 times. (2) Paclitaxel for injection (albumin-bound type) 260 mg/m2 or paclitaxel lipid 135 mg/m2 combined with 80 to 120 mg/m2 cisplatin given once every 3 weeks for at least twice. (3) Cisplatin 20 to 30 mg given once a week for at least 4 times. (4) Paclitaxel for injection (albumin-bound type) 260 mg/m2 or paclitaxel liposome 135 mg/m2 combined with Tegafur, Gimeracil and Oteracil Porassium Capsules 80 mg/m2/Bid, given continuously for 2 weeks and stop for 1 week, with at least 2 cycles. (5) nedaplatin 80 to 100 mg/m2 or cisplatin 80 to 120 mg/m2 combined with Tegafur, Gimeracil and Oteracil Porassium Capsules, given for at least 2 cycles. 9
Efficacy and Safety Outcome Measurements
The primary endpoints were objective response rate (ORR) and disease control rate (DCR). The secondary endpoints were overall survival (OS), progression-free survival (PFS), and safety.
Efficacy was assessed according to the response assessment criteria for solid tumors (RECIST 1.1). Efficacy indicators are as follows: Complete remission (CR, defined as disappearance of all target lesions with no new lesions; tumor markers returned to normal level and maintain for at least 4 weeks). Partial response (PR, defined as more than 30% reduction in target lesion diameter in the sum of all target lesions and maintain for at least 4 weeks). Stable disease (SD, defined as the sum total of target lesion diameter reduced but did not meet PR criteria or increased but did not meet disease progression criteria) and disease progression (Parkinson’s disease [PD], defined as an increase of at least 20% in the maximum diameter of the target lesion or the total number of new lesions appearing). ORR = (CR + PR)/ (total number of cases in each group). DCR = (CR + PR + SD)/ (total number of cases per group). PFS is defined as the time between first administration and objective evidence confirming disease progression or death from any cause. OS is defined as the time measured from first administration to death or last exposure. Adverse events (AEs) were assessed and documented according to the National Cancer Institute Standard for Common Terminology for Adverse Events (NCI-CTCAE version 5.0).
Statistical Analysis
SPSS 25.0 software was used for data analysis. Quantitative data were presented as mean ± standard deviation, median (95% confidence interval [CI]), or number of patients (percent). χ2 test or Fisher's exact test was used to analyze the efficacy and incidence of adverse events in both groups. Survival analysis was performed by Kaplan-Meier method and logarithmic rank test. Difference with P < .05 (2-tailed) was considered statistically significant.
Results
Patient Characteristics
A total of 82 patients with cervical cancer stage II B to IV A (FIGO 2018) were included in this retrospective study. Baseline characteristics are shown in Table 1. There were 41 patients in each group, with 1 patient in each group diagnosed with adenosquamous carcinoma or adenocarcinoma.
Baseline Characteristics of E + CRT and CRT Groups.
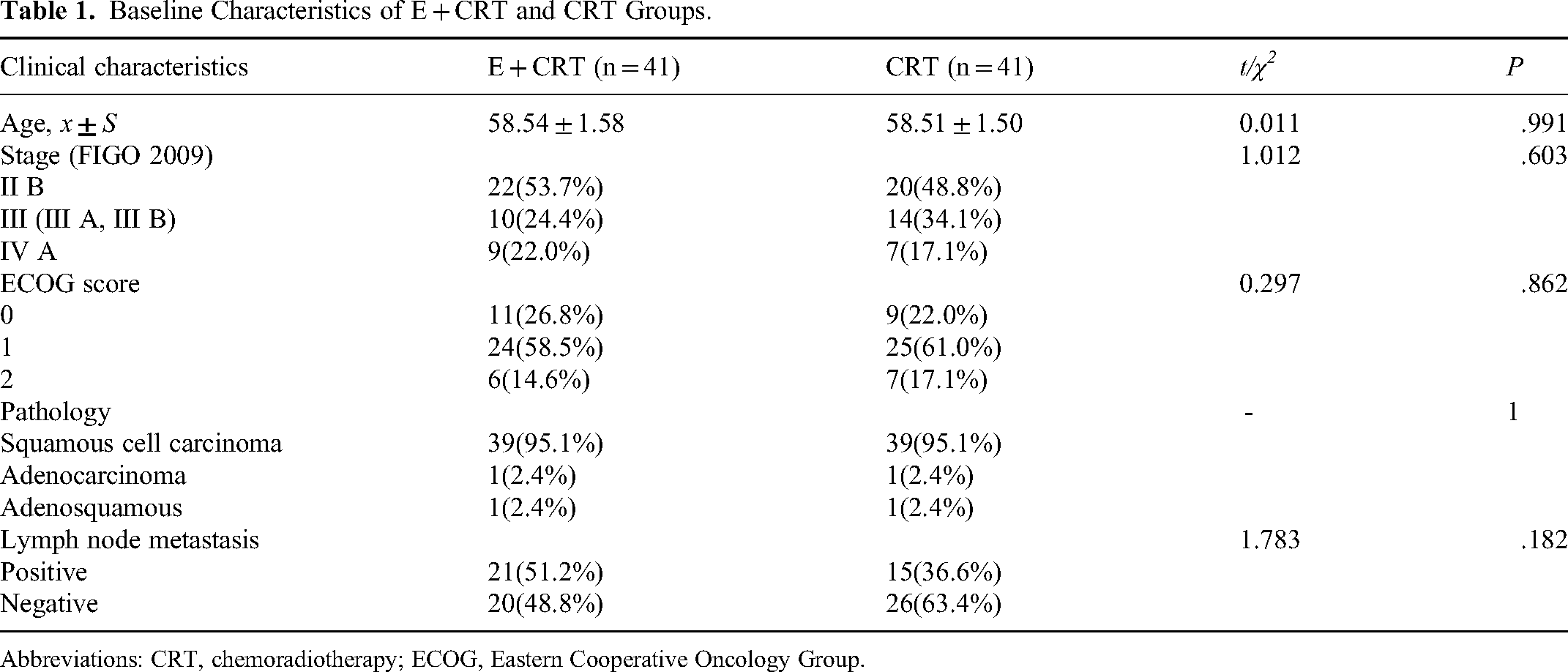
Abbreviations: CRT, chemoradiotherapy; ECOG, Eastern Cooperative Oncology Group.
Early Treatment Response
The statistical analysis of the 82 patients revealed a significant difference between the Endostar combined chemoradiotherapy (E + CRT) and the CRT groups (χ2 = 4.20, P = .04). However, there were no discernable differences in ORR (82.9% vs 70.7%) and DCR (92.7% vs 80.5%) between the 2 groups (P > .05) (Table 2).
Short-Term Efficacy of E + CRT and CRT Groups.

Abbreviations: CR, complete remission; CRT, chemoradiotherapy; DCR, disease control rate; ORR, objective response rate; PD, Parkinson’s disease; PR, partial response; SD, stable disease.
Survival Analysis
The median follow-up duration among all patients enrolled was 30 months (interquartile range [IQR]: 5-46 months). The median follow up of patients in the E + CRT and CRT groups were 32 (IQR: 6-46) and 28 (IQR: 5-36) months, respectively. A total of 23 patients experienced treatment failure (locoregional failure alone, distant metastases alone or both), including 11 patients in E + CRT group and 12 in CRT group. Eighteen patients, 9 in each group, died during follow up.
In this study, the Kaplan-Meier curves of PFS between the E + CRT group and the CRT group are shown in Figure 1(A). The 1-year PFS rates of the E + CRT and CRT groups were 80.5% and 68.3%, respectively. The 2-year PFS rates of the E + CRT and CRT groups were 68.3% and 46.3%, respectively. There was no significant difference between the 1-year and 2-year PFS rates of the 2 groups of patients (hazard ration [HR] = 0.677, 95% CI: 0.304-1.486, P = .323). Nevertheless, as illustrated in Figure 2(C), further subgroup analysis of PFS demonstrated that for locally advanced cervical cancer patients in stage IIB, the PFS of the E + CRT group was demonstrably superior to that of the CRT group, with statistically significant differences between the groups (P < .05). The findings indicate that Endostar may not have the same effect on patients with multiple cervical cancers. However, there are specific populations that may benefit from it.

Kaplan-Meier of PFS(A), OS(B)between E + CRT and CRT groups. Abbreviations: CRT, chemoradiotherapy; OS, overall survival; PFS, progression-free survival.

Kaplan-Meier of PFS(A) between E + CRT and CRT groups: age ≤ 60 (A), age>60 (B), stage II B (C), III A-IV A (D), ECOG score 0-1 (E), ECOG score 2 (F), tumor size ≥4 cm (G), tumor size <4 cm (H), lymph node metastasis (I), and no lymph node metastasis (J). Abbreviations: CRT, chemoradiotherapy; ECOG, Eastern Cooperative Oncology Group; PFS, progression-free survival.
The 1-year and 2-year OS rates were 87.8% and 73.2%, respectively, in E + CRT group and 82.9% and 48.8%, respectively, in the CRT group, with no significant difference between the 2 groups (HR: 0.553, 95% CI: 0.243-1.255, P = .154). As presented in Figure 3(A-J), the results of the further subgroup analysis of the OS show no significant difference of the 2 groups (P > .05). Although there was no statistically significant difference in OS between the 2 groups, a trend toward a difference was observed.

Kaplan-Meier of OS between E + CRT group and CRT group: age ≤ 60 (A), age>60 (B), stage II B (C), III A-IV A (D, ECOG score 0-1 (E), ECOG score 2 (F), tumor size ≥4 cm (G), tumor size <4 cm (H), lymph node metastasis (I), no lymph node metastasis (J). Abbreviations: CRT, chemoradiotherapy; ECOG, Eastern Cooperative Oncology Group; PFS, progression-free survival.
Multivariable Analysis
Age, FIGO staging, ECOG score, maximum tumor diameter, and presence or absence of lymph node metastasis were analyzed using Kaplan-Meier method and Log-rank test. Stage (P < .01), ECOG score (P < .01), and maximum tumor diameter (P < .01) were prognostic factors affecting patient PFS (Figure 4). Stage (P = .017), ECOG score (P < .01), maximum tumor diameter (P = .015), and lymph node metastasis (P = .022) were prognostic factors affecting patient OS (Figure 5).

Influences of age (A), stage (B), ECOG score (C), tumor size (D), and lymph node metastasis (E)on PFS. Abbreviations: ECOG, Eastern Cooperative Oncology Group; PFS, progression-free survival.

Influences of age (A), stage (B), ECOG score (C), tumor size (D), and lymph node metastasis (E) on OS. Abbreviations: ECOG, Eastern Cooperative Oncology Group; OS, overall survival.
Factors with statistical significance (P < .01) and clinical significance in univariate analysis were included in the Cox regression model. Prognostic analysis of PFS in locally advanced cervical cancer patients (Table 3) presented a significant difference in PFS between stage IIB patients and stage IIIA-IVA patients (HR = 3.751, 95% CI: 1.353-10.395, P = .011). Patients with an ECOG score of 0-1 had significantly better PFS compared to those with an ECOG score of 2 (HR = 5.888, 95% CI: 2.393-14.488, P < .01). Additionally, patients with a maximum tumor diameter of less than 4 cm had significantly better PFS than those with a diameter of 4 cm or greater (HR = 7.068, 95% CI: 1.594-31.341, P = .01).
Multivariable Analyses for Progression-Free Survival.

Abbreviations: CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ration.
It has been observed that the staging, ECOG score, and maximum tumor diameter are predictive factors for PFS in this study. Patients with stage IIB, an ECOG score of 0-1, and a maximum tumor diameter of less than 4 cm displayed significant improvement in PFS (Table 3). Table 4 exhibits the results of prognostic analysis on OS in patients with locally advanced cervical cancer. No significant differences were observed in OS between patients over 60 years old and those aged 60 years or younger (HR = 1.079, 95% CI: 0.360-3.230, P = .892), between patients at stage IIB and those at stage IIIA-IVA (HR = 2.256, 95% CI: 0.736-6.914, P = .154), nor between patients with or without lymph node metastasis (HR = 2.780, 95% CI: 0.862-8.965, P = .087). The OS of patients with an ECOG score of 0-1 was markedly higher compared to patients with an ECOG score of 2 (HR = 7.919, 95% CI: 2.793-22.451, P < .01). Moreover, the OS of patients exhibiting a maximum tumor diameter of less than 4 cm was superior in contrast to patients with a maximum tumor diameter of greater than or equal to 4 cm (HR = 5.898, 95% CI: 1.238-28.112, P = .026). The above outcomes verify the independence of ECOG score and maximum tumor diameter as prognostic determinants of patient OS in this study. Patients who have an ECOG score of 0-1 and a tumor diameter below 4 cm demonstrated a noteworthy boost in OS.
Multivariable Analyses for Overall Survival.

Abbreviations: CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ration.
Acute Adverse Events
According to Table 5, most acute adverse events in patients in both groups were of grade I-II, yet a small number of patients experienced grade III-IV or more severe adverse events. Significantly, there was a difference in the incidence of bone marrow suppression between the 2 groups(χ2 = 4.053, P = .044). No statistically significant difference was observed in the occurrence of nausea, vomiting, and radiation-induced enteritis between the 2 groups (P > .05). Four patients who received E + CRT treatment reported mild to moderate radioactive cystitis. Moreover, no adverse events, including hypertension, proteinuria and arrhythmia that could potentially be linked to Endostar, were reported during the trial.
Acute Adverse Reactions of E + CRT and CRT Groups.

Abbreviations: AE, adverse events; CRT, chemoradiotherapy.
Discussion
Currently, the optimal treatment for locally advanced cervical cancer is a combination of radiotherapy and chemotherapy administered concurrently. Compared to simple chemotherapy/radiotherapy, concurrent radiotherapy and chemotherapy can considerably enhance patients’ PFS and OS, while also significantly improving the prognosis and quality of life of patients with locally advanced cervical cancer. 10 A meta-analysis 9 investigating the management of locally advanced cervical cancer, comparing the effectiveness of concurrent radiotherapy and chemotherapy versus radiotherapy alone, demonstrated that concurrent radiotherapy and chemotherapy resulted in a 10.2% increase in patients’ CR rate (P = .027), an 8.4% improvement in local control (LRC) rate (P < .001), and a 7.5% increase in OS rate (P < .001) compared to radiotherapy alone. However, the simultaneous application of radiotherapy and chemotherapy resulted in a significant rise of 10.4% in the occurrence of acute toxic reactions of grade III/IV (P < .001).
Nevertheless, cervical cancer patients who have received standard treatment exhibit elevated recurrence rates and a suboptimal prognosis. 11 Furthermore, they may experience complications, such as chronic radiation enteritis, radioactive cystitis, and vaginal stenosis, which have a significant impact on their quality of life. 12 Therefore, it is imperative to develop novel treatment methodologies to enhance the survival rates and prognosis in patients with cervical cancer.
The development, spread, and invasion of malignant tumors are closely associated with the process of tumor angiogenesis. 13 Antiangiogenic medications can temporarily normalize abnormal tumor blood vessels over a specific period, support blood flow perfusion of tumor. This subsequently enables increased transportation of chemotherapy drugs to the tumor's interior, aiding in the tumor's elimination in conjunction with chemotherapy drugs. 14
Endostar, as a classical antiangiogenic medication, can enhance the effect of radiotherapy in cervical cancer when combined with it. Several factors may be responsible for its mechanism, including angiogenesis inhibition, vasculogenesis suppression, and endothelial cell apoptosis inducement. (1) Endostar can decrease the levels of vascular endothelial growth factor and hypoxia-inducible factor 1 (hypoxia inducible factor 1 α, HIF-1 α) in tumor tissue, inducing α inducing vascular endothelial cell apoptosis through proangiogenic factors, It also normalizes tortuous and deformed tumor blood vessels, improves the hypoxic state of tumor tissue, and thereby increasing the radiation sensitivity of the tumor tissue.15–17 (2) Endostar has the ability to improve radiation sensitivity in tumor tissue by inducing apoptosis in tumor cells and disrupting the tumor cell cycle during the radiation sensitive phase.14,18–20
A randomized controlled clinical trial 21 investigated the efficacy and adverse reactions of the combination of Endostar with chemotherapy in the treatment of advanced (IVb) and recurrent metastatic cervical cancer. Forty-four patients with recurrent and metastatic cervical cancer were randomly divided into an experimental group and a control group (22 cases in each group). The control group was given gemcitabine plus cisplatin (GP) or docetaxel plus cisplatin (DP) treatment, the experimental group was treated with Endostar on the basis of the control group. This study illustrates that the integration of Endostar with chemotherapy extends the median PFS significantly as compared to chemotherapy alone (7.2 vs 5.1 months, P = .036), while avoiding an increase in adverse reactions.
A retrospective clinical investigation conducted by Shu et al 22 on the combination of Endostar with chemoradiotherapy to treat cervical cancer, illustrated an increase in the CR rate in the E + CRT group compared to the CRT group (83.33% vs 65.12%, P < .05), with a higher CR rate reported in the E + CRT group than that in the present study. However, the variations in sample size of this study and differences in patient age, tumor size, and disease stage between the 2 studies might have contributed to this outcome. In another randomized controlled trial conducted by Lu et al, 23 the combination of Endostar with concurrent chemoradiotherapy for treating locally advanced cervical cancer resulted in the 1-year and 2-year PFS rates of the E + CRT and the CRT groups were 91.4% versus 82.1% and 80.8% versus 63.5%, respectively (HR: 0.496; 95% CI: 0.204-1.205, P = .091). Although there was no significant difference in PFS between the patients in the E + CRT and the control group, a trend toward PFS improvement was observed in the E + CRT group.
Similar findings were observed in this investigation. The 2-year PFS and OS rates in the E + CRT group exhibited an upward trend relative to the CRT group. Nevertheless, there was no statistical significance difference. In addition, a more precise subgroup analysis of PFS and OS between the E + CRT and the CRT groups showed that for patients with stage IIB locally advanced cervical cancer, the PFS of the E + CRT group was significantly superior compared to the CRT group. The difference between the groups was statistically significant (P = .047). Nevertheless, there was still no significant difference was found between the OS of the E + CRT and the CRT groups (P > .05). As the follow-up time increased and the number of the patients expanded, significant disparities in PFS and OS between the E + CRT and the CRT groups may emerge, given the trend toward PFS and OS enhancement.
Numerous studies had established links between the prognosis of cervical cancer patients and various characteristics including age, stage, ECOG score, pathological type, tumor size, lymph node metastasis, and chemotherapy cycle.24–29 Our own multivariable analysis revealed that ECOG score and tumor size were independent predictors of OS, while patient age, disease stage, and lymph node metastasis showed no association with OS. Meanwhile, disease stage, ECOG score and tumor size were identified as predictors of PFS, while patient age and lymph node metastasis did not show any significant association with PFS. This study found that factors such as age, FIGO stage, and lymph node metastasis were not independent predictors of OS in stage IIB-IVA cervical cancer patients, which were probably due to small sample size, short follow-up time, and selection bias.
Lu et al's previous study reported that the most frequent acute toxicities in patients receiving Endostar in combination with concurrent chemoradiotherapy as myelosuppression, nausea, diarrhea, and fatigue, which were mainly presented as grade I or II. 14 Similar results were obtained in this study. The most frequent acute negative effects were nausea, vomiting, myelosuppression, and radiation enteritis, mainly classified as grade I-II. Skin and subcutaneous tissue damage, edema, and pain in the lower extremities, radiation enteritis, radiation cystitis, and hydronephrosis were the most common late toxicities in this study. In general, Endostar did not increase the toxic side effects of concurrent radiotherapy and chemotherapy.
The current study is a retrospective clinical study, conducted across 2 centers. The primary endpoints, DCR and ORR, revealed only a trend in favor of E + CRT, due to the short follow-up period and small sample size. However, patients treated with E + CRT demonstrated a higher CR rate and a good safety profile, indicating the possibility of a promising future for combination therapy. In any case, this study is a meaningful attempt to serve as a significant effort to establish a foundation for subsequent large-scale, randomized controlled clinical studies conducted across multiple centers to confirm the safety, efficacy, and prognostic factors of Endostar when combined with chemoradiation as a treatment for cervical cancer.
Conclusion
When compared to simple chemoradiotherapy, the combination of Endostar and concurrent chemoradiotherapy exhibited greater efficacy in treating locally advanced cervical cancer with a higher CR rate and no severe adverse reactions. Nevertheless, while the entire group analysis did not indicate any substantial increment in terms of PFS and OS, subgroup analysis demonstrated a significant enhancement in PFS outcomes among patients with stage IIB (FIGO 2018). Patients with locally advanced cervical cancer in stage IIB (FIGO 2018), an ECOG score of 0-1, and a maximum tumor diameter of less than 4 cm displayed an improved prognosis. A large sample randomized controlled clinical study pertaining to Endostar in combination with concurrent chemoradiotherapy for locally advanced cervical cancer could be conducted to enhance the degree of evidence in evidence-based medicine. This approach is anticipated to develop into a new treatment alternative for patients with locally advanced cervical cancer.
Footnotes
Abbreviations
Data and Code Availability
The dataset analyzed during the current study is available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Studies involving human participants were reviewed and approved by the ethics Committee of Changzhou Second People's Hospital affiliated to Nanjing Medical University ([2017]KY027-01)and The First People's Hospital Affiliated to Soochow University ((2018) No. 092). The patient/participant provided written informed consent to participate in the study. Part of the authors who previously worked for the affiliations responsible for approving the study. However, after changing jobs, their current affiliation no longer aligns with the aforementioned affiliations.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
