Abstract
Keywords
Introduction
Coronavirus disease 2019 (COVID-19), a respiratory infection caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), first appeared in Wuhan, China, in late 2019 and was recognized as a pandemic by the World Health Organization (WHO) in early March 2020.1,2 Globally, as of 2:06 am CET, 22 November 2023, there have been 772166517 confirmed cases of COVID-19, including 6981263 deaths, reported to WHO (WHO COVID-19 Dashboard). 3 A temporary reduction in the infection rate of SARS-CoV-2 was achieved by isolating infected patients and close contacts and restricting mass gatherings, travel, and social distancing in high-risk areas. 4 However, the emergence of a more infectious variant of SARS-CoV-2 led to a rapid resurgence of SARS-CoV-2 infection, 5 posing a significant threat to human physical and mental well-being.
Cancer patients, especially those with lung tumors, are more susceptible to SARS-Cov-2 and are likely to experience worse outcomes during the COVID-19 pandemic.6–9 In addition to known risk factors such as age, smoking history, comorbidities, and physical fitness,7,8,10–12 patients with lung cancer may also have an increased risk of mortality due to systemic immunosuppression caused by cancer itself and anti-cancer treatment. 13 Several studies suggested that immune checkpoint inhibitors (ICIs) and chemotherapy may increase the risk of severe outcomes in COVID-19.10,11,14 As Maki et al showed, having a history of ICIs within 90 days could contribute to respiratory failure in patients with lung cancer and COVID-19 receiving chemotherapy. 10 Moreover, a study by the Veterans Health Administration found that the administration of immunotherapy was associated with a greater risk of death. 11 However, other researchers did not discover such an association and maintained that using ICIs, tyrosine kinase inhibitors (TKIs), or chemotherapy was safe during the pandemic.1,7,8,14 Consequently, there is conflicting evidence regarding the impact of cancer treatment modalities on the severity of COVID-19. Therefore, it is crucial to clarify the relationship between different cancer treatment modalities and the severe outcomes of COVID-19 in lung cancer patients. 13
In recent years, increasing studies have examined the role of anti-angiogenic therapies (ATs) in advanced non-small cell lung cancer (NSCLC). Bevacizumab, recombinant human endostatin (Rh-endostatin), and anlotinib are the most widely administered anti-angiogenic agents for advanced NSCLC. Bevacizumab, a humanized monoclonal antibody, effectively blocks vascular endothelial growth factor (VEGF) from binding to its receptor, limiting the supply of oxygen and nutrients to the tumor, thereby inhibiting angiogenesis and tumor growth.15,16 Rh-endostatin, a recombinant human endothelial inhibitor, could inhibit VEGF-induced tyrosine phosphorylation of KDR/Flk-1 in endothelial cells and the Wnt/β-catenin signaling pathway.17–19 This suppresses the formation of tumor blood vessels, nutrient supply, and metastatic pathways, leading to tumor apoptosis. In addition, Rh-endostatin may regulate growth factors, adhesion factors, and inflammatory mediators as well as hypoxia-inducible factor-1α/VEGF pathway in choroidal retinal endothelial cells to inhibit the proliferation and migration of tumor cells.20,21 It has been suggested that Rh-endostatin may also improve malignant tumor responsiveness to anti-cancer therapy by regulating the balance between VEGF A and platelet reactive protein 1, normalizing tumor vasculature and microenvironment, and temporarily increasing intra-tumoral blood perfusion to alleviate hypoxia condition.22–24 Anlotinib, a novel multi-target TKI, effectively inhibits vascular endothelial growth factor receptors (VEGFRs) 1 to 3, epidermal growth factor receptor (EGFR), fibroblast growth factor receptors 1 to 4, platelet-derived growth factor receptors α and β, stem cell factor receptors, and c-kit, which inhibits tumor angiogenesis and cell proliferation.25,26 Consequently, anti-angiogenic drugs could slow or prevent tumor growth by inhibiting regenerating blood vessels and improving the tumor microenvironment. 27 In addition, several studies have shown the favorable efficacy of anti-angiogenic drugs in the treatment of advanced NSCLC.26,28–33 To date, however, very few studies have confirmed the effect of anti-angiogenic drugs on the severity of COVID-19 in patients with NSCLC. To address this gap, we conducted this study to investigate the impact of anti-angiogenic drugs on COVID-19 severity in patients with NSCLC. We also examined the risk factors associated with severe outcomes to provide clinicians with better guidance about anti-angiogenic drug use during the pandemic.
Material and Methods
Study Design and Patient Selection
This retrospective study enrolled 166 consecutive NSCLC patients who tested positive for SARS-CoV-2 via RT-PCR and/or serology tests at the First Affiliated Hospital of Nanchang University between December 12, 2022, and January 15, 2023. Patients who received ICIs, chemotherapy, TKIs, AT, or different combinations of these treatment modalities within 60 days of COVID-19 diagnosis were eligible for this study. The other inclusion criteria were (1) the patient must be at least 18 years old and (2) a stage of IIIB/IIIC-IV NSCLC with confirmed histology. The exclusion criteria were (1) patients with suspected but unconfirmed COVID-19; (2) patients with other tumors; (3) patients undergoing clinical studies; and (4) patients without any information detailing the history and valid and complete follow-up data. This retrospective study was approved by the Research Ethics Committee of the First Affiliated Hospital of Nanchang University on May 16, 2023 with the approval number IIT2023131. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 34 We have de-identified the patient's details, ensuring complete identification confidentiality.
Data Collection and Treatment Group
The records of each patient were examined to identify information on demographics, smoking history, baseline clinical characteristics, comorbidities, pathology characteristics, treatments, symptoms, disease course, vital status, and chest CT scans. Additional details were also obtained from the medical history. The results of molecular testing were retrieved from institutional databases.
Patients were divided into two groups based on whether they received anti-angiogenic drugs during their latest anti-tumor treatment. The non-AT group received ICIs, TKIs, chemotherapy, or a combination of these treatments, while the AT group received anti-angiogenic drugs, either alone or in combination with other anti-cancer drugs. The anti-angiogenic medications involved in this study included bevacizumab, Rh-endostatin, and anlotinib.
Study Outcomes
Outcomes of concern included admission to the intensive care unit (ICU), intubation, development of severe or critical symptoms, and death. The disposition included the date of discharge from the hospital. The clinicians determined the patient's status (recovered, uncured, or dead) at the final follow-up date (February 15, 2023) by telephone follow-up. The clinical definition of severe/critical symptoms followed the 10th edition of the COVID-19 Diagnostic Criteria published by the National Health Commission in China, 35 which included one or more of the following criteria: documented hypoxemia (oxygen saturation by pulse oximetry ≤ 93%), tachypnea (respiratory rate ≥ 30 breaths/minute), respiratory failure (arterial partial pressure of oxygen/fraction of inspired oxygen ratio ≤ 300), septic shock, Chest imaging showed significant lesion progression > 50% within 24-48 h, and admission to the ICU for intubation.
Statistical Analysis
The Mann-Whitney U test, presented as median [interquartile ranges (IQRs)], was used to analyze continuous variables; the χ 2 test or Fisher's exact test, expressed as a number (%), was used for categorical variables. The Bonferroni method was used to perform comparisons among multiple groups. The RAWGraphs version 2.0 website was used to create the Sankey diagrams. Kaplan-Meier plots were used to analyze the time of patients from the positive SARS-CoV-2 RT-PCR and/or serology test to the date of follow-up or death. We used univariate logistic regression models to calculate odds ratio (OR) and 95% confidence interval (CI), with the aim of examining the effect of baseline characteristics and treatment approach on COVID-19 severity. The selection of variables in the multivariate logistic analysis was based on P < 0.05 in the univariate logistic regression analysis. SPSS version 26.0 (IBM SPSS Statistics, USA) and GraphPad Prism 9.0 (GraphPad Software Corporation) were used to perform the data analysis, and a P-value of 0.05 or less was considered statistically significant.
Results
Patients’ Characteristics and Clinical Course
One hundred and sixty-six hospitalized patients with NSCLC and COVID-19 were enrolled in our study from December 12, 2022, to January 15, 2023. Patients’ hospitalization and vital status information are shown in Figure 1a. The median follow-up period from diagnosis of COVID-19 was 48 days. Among them, 73 (44.0%) patients were in the AT group, while the remaining 93 (56.0%) were in the non-AT group. There was a significantly higher proportion of adenocarcinomas (78.1% vs 52.7%, P = 0.002) and incidence of brain metastases (19.2% vs 6.5%, P = 0.012) in the AT group than in the non-AT group; however, other baseline characteristics were comparable (Table 1). Among the participants, cough was the most frequently reported symptom (71.5%, 108/151), followed by fever (62.9%, 95/151), chest distress (53.6%, 81/151), and dyspnea (52.3%, 79/151) (Figure 1b). As of the follow-up period, 4.2% (7/166) of the patients were still uncured, 10.2% (17/166) had died, and the remaining 85.6% (142/166) had recovered (Figure 1c).
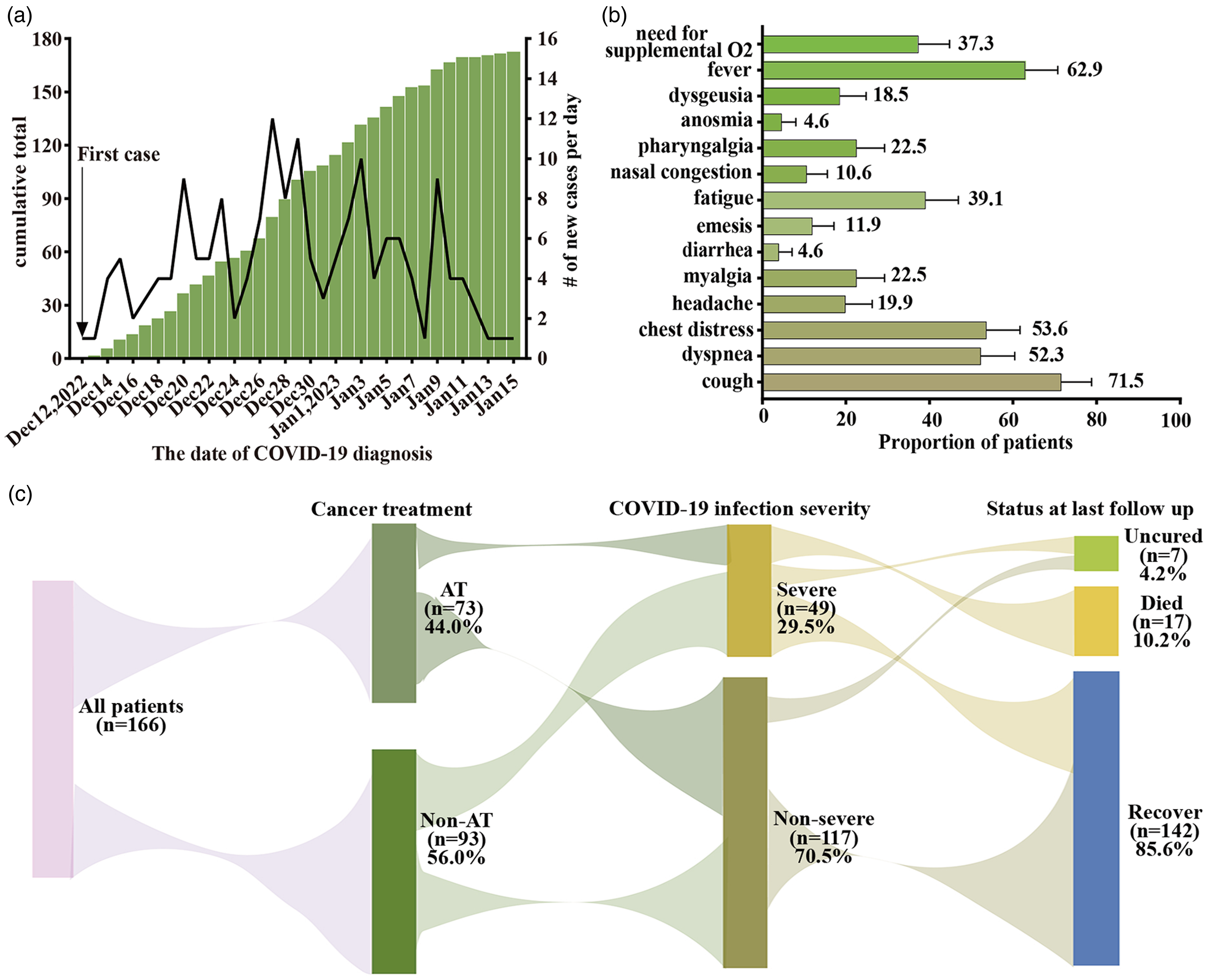
COVID-19 in patients with NSCLC. (a) Daily cases and cumulative incidence of positive SARS-CoV-2 RT-PCR and/or serology test in patients with NSCLC. (b) Patients with known information showed signs and symptoms of COVID-19 infection. Error bars show 95% CI estimates of the population proportion. (c) Patients were identified starting from the first case on December 12, 2022, to January 15, 2023, and followed until February 15, 2023. Cancer treatment, COVID-19 infection severity, and patients’ COVID-19 status at the time of final follow-up. By the end of the follow-up period, 4.2% (7/166) of patients had uncured, with one still hospitalized due to COVID-19; three patients had not returned to pre-COVID-19 clinical symptoms; and three patients had not returned to pre-COVID-19 physical condition. CI, confidence interval.
Baseline Characteristics of Patients with Non-small Cell Lung Cancer and Positive SARS-CoV-2 Test.

Data are median (IQR), n (%). SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; AT, anti-angiogenic therapy; BMI, body mass index; ECOG PS, Eastern Cooperative Oncology Group performance status; COPD, chronic obstructive pulmonary disease; PR, partial response; SD, stable disease; PD, progression disease; ICU, intensive care unit; IQR, interquartile range.
*P < 0.05.
Including large-cell neuroendocrine carcinoma, non-small cell lung cancer, and adeno-squamous carcinoma.
Data on cancer status was missing for five patients. There were four patients with missing data in the AT group and one patient in the non-AT group.
Including low-flow oxygen, high-flow oxygen, and mechanical ventilation.
Data on the timing of anti-cancer therapy was unknown for ten patients. There were two patients with missing data in the AT group and eight patients in the non-AT group.
Effects of Anti-angiogenic Drugs on COVID-19 Outcomes
The median follow-up period for patients in the AT group was comparable to that of patients in the non-AT group (46 days vs 48 days, P = 0.289). Additionally, there were no statistically significant differences in the incidence of severe or critical symptoms (21.9% vs 35.5%, P = 0.057), ICU admissions/intubations (6.8% vs 7.5%, P = 0.867), or mortality rates (11.0% vs 9.7%, P = 0.787) between the two groups (Table 2). Moreover, Kaplan-Meier survival analysis showed similar trends in overall survival for both groups of patients (Figure 2a). Furthermore, we compared patients who received AT during COVID-19 infection with those who did not and found that overall death rates were similar for the two groups (8.6% vs 10.7%, P = 1.000). Kaplan-Meier plots also supported that using anti-angiogenic drugs during COVID-19 infection had no impact on survival outcomes (Figure 2b).
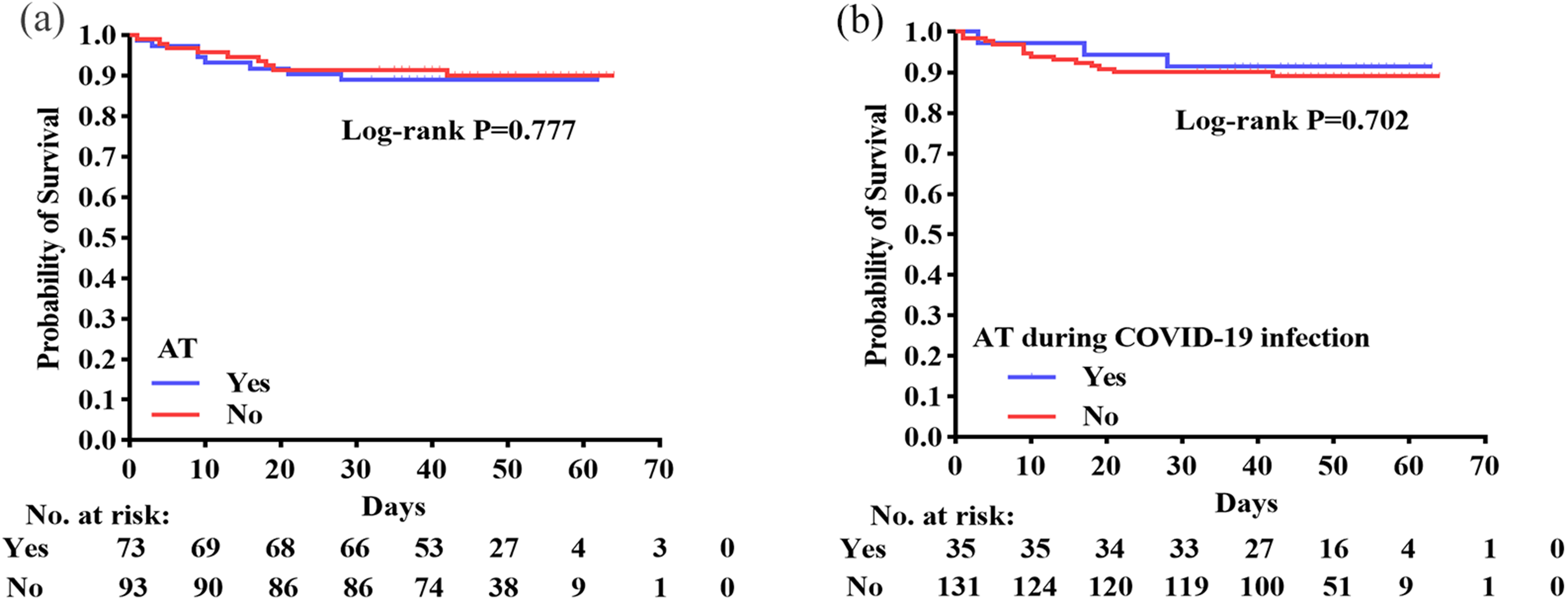
Effect of anti-angiogenic drugs on survival outcomes in NSCLC patients with COVID-19. (a) Kaplan-Meier survival curve of overall survival comparing the AT group and the non-AT group. (b) Kaplan-Meier survival curve for overall survival of patients receiving anti-angiogenic drugs during COVID-19 infection versus those who did not. AT, anti-angiogenic therapy.
COVID-19 Outcomes of Patients in the Two Groups.

AT, anti-angiogenic therapy.
Examination of Factors Influencing the Severe Condition
Univariable regression analyses revealed that Eastern Cooperative Oncology Group performance status (ECOG PS) was associated with an increased risk of severe COVID-19 outcomes, including death, ICU admission/intubation, and severe/critical symptoms (Table 3). Moreover, patients aged 60 years or older or those with comorbidities, such as COPD and hypertension, were at an increased risk of experiencing severe/critical symptoms (Table 3). Additionally, hypertension and COVID-19 treatment-related medications, such as immunoglobulins, hormones, and antibiotics, were associated with mortality (Table 3). As patients with severe/critical symptoms of COVID-19 and those requiring ICU admission/intubation are more likely to receive COVID-19 treatments, we didn’t perform a univariate regression analysis to investigate the effect of these treatments on severe/critical symptoms and ICU admission/intubation. In addition, we performed a within-group comparison of patients receiving COVID-19 treatment (Figure 3). As shown by univariate analysis, patients treated with COVID-19 had a significantly higher mortality rate than those who did not (P < 0.01), with the exception of those who received antiviral treatment (Figure 3). Multivariate regression analysis further demonstrated that age ≥ 60 years (OR: 2.52, 95% CI: 1.07-5.92), ECOG PS (OR: 5.87, 95% CI: 1.33-25.95), hypertension (OR: 2.98, 95% CI: 1.20-7.39), and COPD (OR: 7.25, 95% CI: 1.65-31.81) were related to an increased risk of severe/critical symptoms (Figure 4). Nevertheless, in the multivariate regression of mortality risk, only ECOG PS (OR: 9.77, 95% CI: 2.10-45.44) and use of immunoglobulin (OR: 5.26, 95% CI: 1.06-26.25) were significant risk factors for mortality (Figure 4).

Effect of different COVID-19 treatment drugs on mortality outcomes. Error bars show 95% CI estimates of the population proportion. *P < 0.05; **P < 0.01; ns, no significance; CI, confidence interval.

Multivariable model of factors associated with COVID-19 severity (severe/critical symptoms and death). Odds ratios were calculated by multivariate regression analysis based on univariate regression analysis with P < 0.05. Error bars represent 95% CIs. The x-axis is presented on a log10 scale. CI, confidence interval.
Univariate Regression Analysis of Prognostic Factors with COVID-19 Severity (Severe/Critical Symptoms, ICU/Intubation and Death) among all Patients.

ICU, intensive care unit; ref, reference; OR, odds ratio; CI, confidence interval; BMI, body mass index; ECOG PS, Eastern Cooperative Oncology Group performance status; COPD, chronic obstructive pulmonary disease; PR, partial response; SD, stable disease; PD, progression disease.
Odds ratio >1 suggests higher odds of severe/critical symptoms, ICU/intubation, death.
*P < 0.05.
To further validate the impact of anti-angiogenic drugs on COVID-19 severity, we performed a sensitivity analysis on the entire study population. After adjusting for ECOG PS, a critical risk factor for COVID-19 severity, as well as the type of cancer and brain metastases, which differed significantly between the AT group and the non-AT group, the sensitivity analysis showed that recent treatment with anti-angiogenic drugs did not have a significant impact on the severe outcomes of COVID-19.
Subgroup Analysis
Further subgroup analysis of the results showed that, in most of the identified categories, with the exception of the ECOG ≥ 2 and chest radiotherapy populations, the OR for severe/critical symptoms of COVID-19 was numerically favored in the AT group (Figure 5). Of interest, although it did not yield a statistically significant difference, the application of anti-angiogenic drugs did not increase the risk of severe/critical symptoms in patients aged ≥ 60 and with comorbidities. The data from subgroup analysis regarding ICU admission/intubation and deaths of COVID-19 are presented in Supplementary material figures 1 and 2. With the approval of various anti-angiogenic drugs for NSCLC, the choice of safer drugs became a new challenge during the COVID-19 epidemic. Therefore, we evaluated the impact of three different anti-angiogenic drugs on the severity of COVID-19. It was found that patients receiving anlotinib had a higher risk of severe/critical symptoms (26.7%, 4/15), but there was no statistical difference in severe condition compared with the other two anti-angiogenic drugs (Table 4).
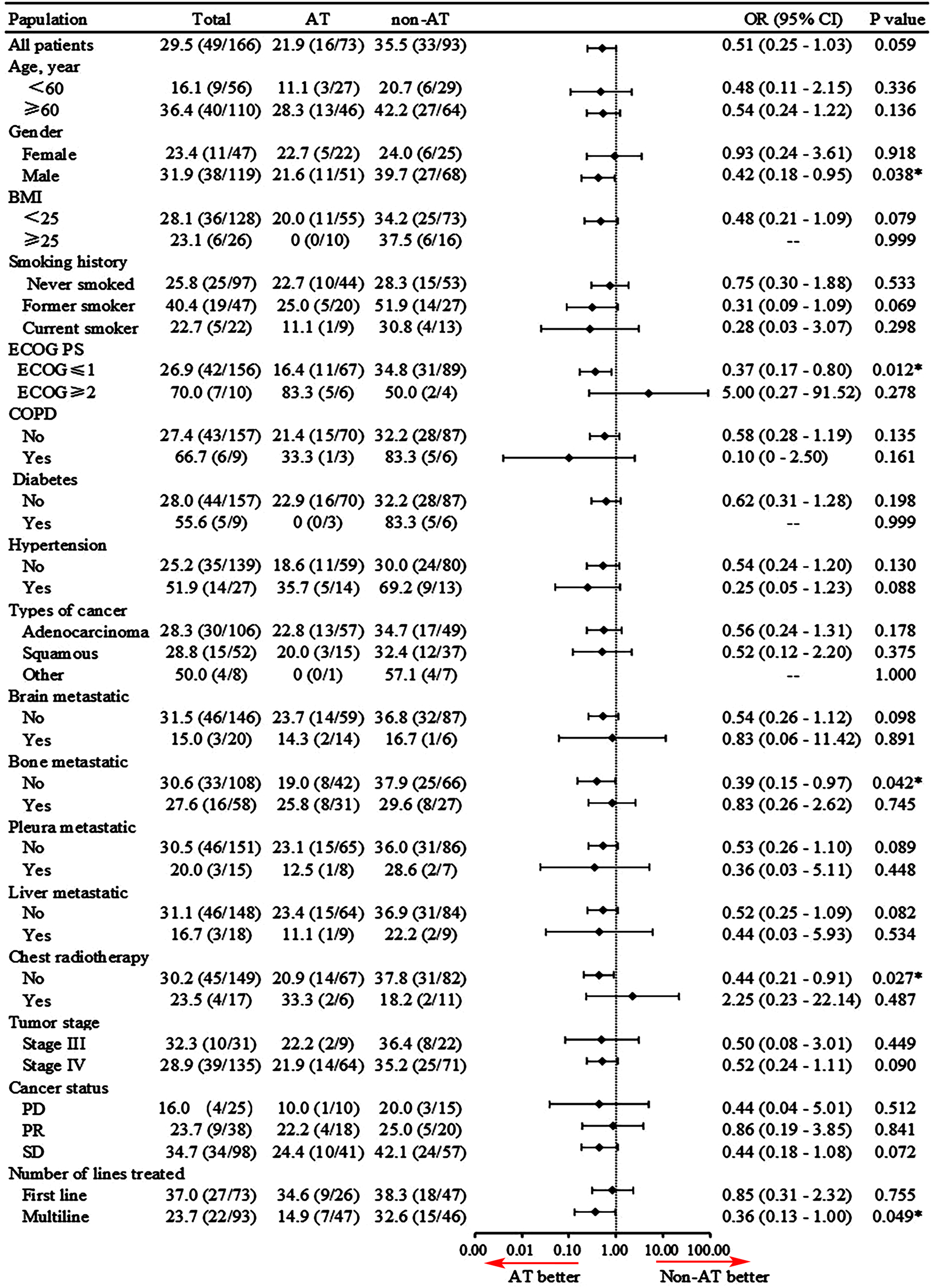
The odds ratio (OR) for severe/critical symptoms of COVID-19 was calculated based on baseline characteristics. *P < 0.05.
Effect of Different Types of Anti-angiogenic Drugs on COVID-19 Severe Condition.

Data expressed as n (%).
ICU, intensive care unit.
Discussion
COVID-19 has a serious impact on the health status of patients with NSCLC. Therefore, understanding how cancer treatment options affect the severity of COVID-19 in patients with NSCLC is essential for guiding clinical decisions. To our knowledge, this is the first retrospective cohort study to examine the impact of anti-angiogenic drugs on the severity of COVID-19 illness in patients with NSCLC. Our study found that there were no significant differences in COVID-19 outcomes between patients treated with anti-angiogenic drugs and those who were not. To further validate our results, we compared patients who received anti-angiogenic drugs during COVID-19 infection with those who did not and found no significant difference in overall mortality between these two groups. Accordingly, it appeared that exposure to anti-angiogenic drugs, whether prior to or during COVID-19 infection, did not negatively affect the severity or outcome of COVID-19.
In our study, the proportion of adenocarcinoma was higher in the AT group compared to the non-AT group, possibly for the reason that squamous cell carcinoma usually occurs in the proximal large bronchioles and the use of bevacizumab increases the risk of pulmonary hemorrhage.36,37 Therefore, the use of bevacizumab in squamous cell carcinoma of the lung has been limited.19,36 In addition, we observed a significantly higher proportion of brain metastases in the AT group than in the non-AT group. In our study, bevacizumab was the most commonly used anti-angiogenic drug for patients with brain metastases (85.7%, 12/14). The main fact is that bevacizumab, a recombinant human anti-VEGF monoclonal antibody, could effectively neutralize VEGF and block its binding to the VFGFR on endothelial cells, which may inhibit the formation of tumor neovascularization and alleviate peritumor edema.38,39 In addition, several studies have shown that bevacizumab has promising results in NSCLC patients with brain metastases.38,40–42 In addition, we performed subgroup analyses of cancer types and brain metastases between the AT and non-AT groups and found that there was no significant difference in COVID-19 outcomes between the two groups (Figure 6). Thus, although the ratio of cancer types and brain metastases was not balanced between the two groups, it had no significant effect on the outcome of COVID-19.

(a-c) Proportion of severe outcomes of COVID-19 (severe/critical symptoms, ICU admission/intubation, and death) among AT and non-AT groups for different types of cancer. (d-f) Proportion of patients with and without brain metastases who developed severe outcomes of COVID-19 (severe/critical symptoms, ICU admission/intubation, and death) in the AT and non-AT groups. AT, anti-angiogenic therapy; ICU, intensive care unit.
Similar findings were reported for the effect of other anti-cancer drugs on the severity of COVID-19 in patients with lung cancer.1,7,8 Two studies conducted in New York showed that patients with lung cancer who recently received chemotherapy, TKIs, and PD-(L)1 blockade did not experience increased severity of COVID-19.7,8 In addition, the TERAVOLT registry, which describes the impact of COVID-19 on thoracic cancer patients, also found that these treatment options had no effect on survival outcomes for patients with cancer and COVID-19. 1
Although specific anti-cancer drugs did not affect the severe outcomes of COVID-19 in patients with lung cancer, these individuals were more susceptible to the effects of SARS-CoV-2.6–9 Our study found that more than 25% (49/166) of NSCLC patients with COVID-19 had severe/critical symptoms, 7.2% (12/166) required ICU admission/intubation, and 10.2% (17/166) died. These findings contrasted with studies conducted in Hubei Province, China, and New York, USA, which showed a mortality rate of nearly 25% for patients with lung cancer and COVID-19.7–9 The reasons for this discrepancy may be associated with the prevalence of the vaccine and the evolution of SARS-CoV-2.43,44 Meanwhile, the morbidity and mortality rates reported in the TERAVOLT registry were 33% for thoracic tumors, with a higher proportion of patients having an ECOG PS of 2 or higher compared to the present study.1,14 In addition, geographical location plays a significant role in determining the mortality rates of patients with lung cancer and COVID-19. A meta-analysis of 13 studies showed that the mortality rates of lung cancer patients with COVID-19 in China were lower than those in developed countries, including the United States, France, Spain, Brazil, and Italy (23.0% vs 42%). 45 Another meta-analysis of 19 clinical studies from nine countries also found that the mortality rates for cancer patients with COVID-19, including lung cancer, varied across countries, with the highest in Spain (36.1%, 139/385) and the lowest in China (5.6%, 6/107). 46 Moreover, smoking may have a negative impact on COVID-19 outcomes, which may further explain differences in COVID-19 severity.1,7,8 However, our study found that patients who currently smoke did not have an increased risk of severity of COVID-19 (OR: 0.847, 95% CI: 0.283-2.535). It should be noted that this may reflect a small proportion of active smokers at the time of analysis.
Our study also observed a significant association between COVID-19 severity and ECOG PS47–50 as well as age47,48,51–54 among cancer patients, which was consistent with previous research findings. In addition to ECOG PS and age, COPD and hypertension could also increase the risk of COVID-19 severity in NSCLC patients. Previous studies showed that cell entry of coronaviruses depended on the binding of the viral spike (S) proteins to angiotensin-converting enzyme 2 (ACE2) receptors, as well as the priming of S proteins by the cellular serine protease TMPRSS2.55–57 In patients with COPD, the elevation of ACE2 and TMPRSS2 expression levels in lung tissue provided SARS-CoV-2 with an opportunity to enter target cells, which can lead to respiratory failure and even death.58–60 Moreover, patients with COPD are immunocompromised, making it more challenging to eliminate viruses and lead to a more severe systemic inflammatory response, exacerbating the severity of COVID-19. 61 While the underlying mechanism of hypertension's impact on the severity of COVID-19 remains unclear, previous studies showed that patients with hypertension had an activated renin-angiotensin-aldosterone system and that most patients required long-term use of ACE inhibitors (ACEI) or angiotensin receptor blockers (ARBs) medications, which may result in up-regulation of ACE2 expression. 62
Furthermore, the use of immunoglobulin was associated with a higher risk of death, which was supported by The ITAC (INSIGHT 013) Study Group 63 and Ali et al. 64 However, it is important to note that the increased risk of death may not be directly caused by immunoglobulins but rather by their higher frequency of use in patients with severe or critical symptoms. 65 Thus, it is necessary to conduct further research to determine the potential association between immunoglobulin use and an increased risk of death.
Subgroup analysis based on baseline characteristics supported AT treatment, except in patients with ECOG ≥2 and those receiving chest radiotherapy. In particular, the administration of anti-angiogenic drugs significantly reduced the risk of developing severe/critical symptoms of COVID-19 in males and the multiline therapy subgroup (P = 0.038, P = 0.049). Several studies have shown that male COVID-19 patients have a worse prognosis than their female counterparts, possibly due to estrogen levels, viral clearance, and the immune system.66–69 However, it is important to note that the use of anti-angiogenic drugs did not increase the risk of COVID-19 severity in either men or women in our study. The impact of gender on the severity of COVID-19 when anti-angiogenic drugs are used needs to be further investigated.
Our study has several limitations. First, our research was a retrospective and observational study with a limited sample size, which made it challenging to eliminate selection bias. Second, our study focused exclusively on hospitalized patients, which limited the generalizability of our findings to outpatients. Future investigations should include both inpatients and outpatients to improve the generality of our findings. Third, the SARS-CoV-2 strain mutates rapidly, and it is crucial to collect relevant information in order to better characterize the strain variability. However, the majority of the patients in our study did not receive this test. In future studies, we should actively advocate for the analysis and testing of SARS-CoV-2 strains in patients to explore the potential impact of different strains on the severity of COVID-19. In addition, patients in the non-AT group had previously received anti-angiogenic drugs, yet the effect of these drugs on the severity of COVID-19 in such patients remains unknown. Further clinical studies will be needed to confirm whether prior anti-angiogenic drug use may be associated with the severity of COVID-19. Finally, our study reported all-cause mortality instead of case fatality, so there was insufficient evidence on the cause of death, including COVID-19, cancer, or other comorbidities.
Conclusion
In summary, our findings confirmed that anti-angiogenic drugs may not be associated with the severity of COVID-19. Moreover, patients with NSCLC, especially those aged 60 or over with an ECOG PS of 2 or higher, hypertension, COPD, or receiving immunoglobulin treatment, had a higher risk of COVID-19 severity and death. It is, therefore, crucial to balance the severity of COVID-19, the risk of cancer progression, and the risk associated with cancer treatment to develop an individualized treatment plan for patients with NSCLC.
Supplemental Material
sj-tif-1-tct-10.1177_15330338241248573 - Supplemental material for Impact of Anti-angiogenic Drugs on Severity of COVID-19 in Patients with Non-Small Cell Lung Cancer
Supplemental material, sj-tif-1-tct-10.1177_15330338241248573 for Impact of Anti-angiogenic Drugs on Severity of COVID-19 in Patients with Non-Small Cell Lung Cancer by Sujuan Peng, Hongxiang Huang, Jinhong Chen, Xinjing Ding, Xie Zhu, Yangyang Liu, Li Chen, and Zhihui Lu in Technology in Cancer Research & Treatment
Supplemental Material
sj-tif-2-tct-10.1177_15330338241248573 - Supplemental material for Impact of Anti-angiogenic Drugs on Severity of COVID-19 in Patients with Non-Small Cell Lung Cancer
Supplemental material, sj-tif-2-tct-10.1177_15330338241248573 for Impact of Anti-angiogenic Drugs on Severity of COVID-19 in Patients with Non-Small Cell Lung Cancer by Sujuan Peng, Hongxiang Huang, Jinhong Chen, Xinjing Ding, Xie Zhu, Yangyang Liu, Li Chen, and Zhihui Lu in Technology in Cancer Research & Treatment
Footnotes
List of Abbreviations
Acknowledgments
Not applicable.
Author Contributions
ZL and LC researched literature and conceived the study. JC, XD, XZ, and YL were involved in protocol development, gaining ethical approval, patient recruitment, and data analysis. SP and HH wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data Availability Statement
The original contributions presented in the study are included in the article material. Further inquiries can be directed to the corresponding authors.
Declaration of Conflicting Interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Research Ethics and Patient Consent
The studies involving human participants were reviewed and approved by the research ethics committee of the First Affiliated Hospital of Nanchang University, approval/reference number IIT2023131, with an exemption from informed consent. It was performed in line with the guidelines of the Declaration of Helsinki (revised 2013). This was a retrospective clinical study, and statistical analysis of aggregated, deidentified data did not require specific consent. For this study, the raw data were first extracted from HIS, and patients’ identities, including names, screening IDs, patient IDs, and mobile phone numbers, were de-identified.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation for Young Scientists of China (grant number 82103339); the Medical Scientific Research Foundation from the Health Commission of Jiangxi Province, China (grant number 202210473); the Natural Science Fund for Youths of Jiangxi Province (grant number 20224BAB216067); and the National Natural Science Foundation for Regional Fund (grant number 82360507).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
