Abstract
Introduction
Uterine malignancy is one of the most common cancers in women worldwide, seriously threatening women's health. 1 Surgical treatment is the main option for these patients at an early stage; however, adjuvant radiation therapy is still necessary, especially for patients with positive resection margins, uterine invasion, pelvic or para-aortic lymph node metastasis, to improve the local control rate of lesions and survival rate of patients.
Considering the movement of the irradiated organ and the error of positioning, the Planning Target Volume (PTV) is commonly generated by expanding the clinical target volume (CTV) with 6–40 mm,2–4 which in turn increases the irradiated dose of a large number of normal tissues, i.e. the bladder, rectum, and intestine, incorporated into the PTV and the incidence of adverse reactions. Moreover, the small intestine after a hysterectomy is more easily falling into the pelvis space, leading to the increased amount of intestine treated with high doses.5–7
Controlling the consistency of bladder fullness during treatment can maintain stable bladder irrigation, thereby reducing target displacement errors and adverse reactions. JHINGRAN A et al 8 indicated that the position of the vagina could be shifted as much as 2 cm simply with variations in bladder filling, resulting in the integral dose delivered to the small intestine increasing dramatically. The radiation planning based upon an empty bladder predicted a larger volume of the small intestine receiving 45Gy compared to a full bladder, as described by Victor E. Chen et al, although an empty bladder reduced absolute variation in bladder volume. 9 Studies have shown that the volume of intestine irradiated correlates with bladder fullness, leading to the recommendation with a full bladder before radiation which can reduce irradiation of the small intestine by 72%,10–12 and that a constant bladder volume during radiotherapy can minimize the head-to-foot orientation error and the CTV-PTV boundary.13–15
To further define the magnitude of changes in target position as a result of bladder volume varying, it is necessary to monitor the changes in the position of the internal organs of the patient with a full bladder before each irradiation. Recently, various types of three-dimensional ultrasound system (3D-US), i.e. the Clarity system (Model 310C00, Elekta, Stockholm, Sweden), have been introduced as alternative image-guided radiation therapy (IGRT). The Clarity system is a 3D target positioning device that allows intra-modality verification by comparing the ultrasound images obtained before each treatment session with the ultrasound images obtained at the time of computed tomography (CT) simulation.
The clarity system has already been launched in the market and has been utilized worldwide in a series of diseases, i.e. prostate cancer, 16 lung and liver tumor17,18 for monitoring, particularly for radiation therapy. Very limited published data, however, are available on gynecological cancers. 19 Here we reported a primary workflow of 3D-US (the Clarity system) and cone-beam computed tomography (CBCT) dual-guided radiotherapy, which is introduced to maintain the consistency of bladder before CBCT during each treatment for postoperative uterine malignancy patients. In addition, the therapeutic potential and the early adverse events in this study were also assessed to highlight the adaptability of this technology in the real world.
Materials and Methods
Study Population
From April 2021 to December 2021, patients, who were referred to the Department of Radiation Oncology, the Affiliated Cancer Hospital of Nanjing Medical University (Jiangsu Cancer Hospital) for chemo-radiation therapy of uterine malignancy, were enrolled in this descriptive, Hospital Ethical Committee-approved study of pelvic IMRT (approval number: 2021-005). The reporting of this study conforms to STROBE guidelines.20,21 All patients signed well-informed consent forms before participating in the study. Patients were eligible for the study if they had undergone a radical hysterectomy as a result of carcinoma of the uterine and required postoperative radiation or chemo-radiation therapy because of positive pelvic lymph nodes, parametrial invasion, involvement of incisal edge, and/or other medium-high risks, i.e. Sedlis Criteria. On the other hand, exclusion criteria were defined as follows: ①Patients had disease outside the pelvis, mental status changes, or bladder control problems that made it difficult for them to comply with bladder-filling instructions, ②Patients’ bodyweight or lateral body diameter exceeded the limits of the treatment table or CT scanner. The basic characteristics including patients age, BMI, pathology description, etc were recorded and patients were followed-up after treatment for 1 year. All patient details have de-identified.
Treatment
Adjuvant Therapy
Same as in our previous study, the patients accepted concurrent chemotherapy (25 mg/m2 of cisplatin weekly for 4-6 weeks) but none was given with brachytherapy.
Radiotherapy
CT simulation (Siemens’ SOMATOM) was conducted as in our previous study, and CBCT scanning was performed using the Elekta XVI device. Through Monaco 5.1 Treatment Planning System (Elekta), Volumetric Modulated Arc Therapy was administered using Versa HD linear accelerator (Elekta) to the patient with the prescription dose DT 4500-4550cGy/24-25fractions.22,23
Preparation Before Radiotherapy
All the patients were trained with the purpose to obtain a reasonably full, but comfortable, bladder volume that they felt they could consistently reproduce during the whole period of their radiotherapy. Specifically, all the patients were educated and trained with written instructions to empty the bladder 60 min before simulation and then to drink 300–500ml of liquid during the subsequent 10 min (Each patient drinks a certain amount of liquid, but due to differences in the size of each person's bladder, etc, a range of rather than a fixed amount is given). Combined with the same hydration protocol, an additional portable ultrasound(Mindray, UMT-400) was also used to evaluate bladder volume to reach the range of approximately 200–300 ml before the initial 3D-US (Clarity system) and CT scan. For each patient, the first portable ultrasound measurement was performed once the patient felt comfortably full after pre-drinking the same amount of water, and multiple measurements were further needed until the bladder volume was consistent with the above optimal volume before simulation. Patients were also counseled to follow the same procedures before each daily radiation treatment. Normally, patients were recommended to empty their intestines before each fraction.
The Special Procedures of our Center
The figure below (Fig 1) summarized the system configuration of Clarity equipment in our center. The principal steps are as follows (Fig 2). Particularly, the patient needs to be prepared with a comfortably full bladder following the protocol as mentioned above and a portable ultrasound scan was performed to keep the bladder volume roughly the same (Fig 3A) before each fractional treatment.
① Procedure before radiotherapy: For simulation and treatment, patients were immobilized in a supine position using Vac-Loc devices to constrain the upper and lower body and were instructed to remain still throughout the subsequent scans.

System configuration of clarity in our hospital; this image is published with the patient/participant's consent. A: quality assurance in simulation room, QA Tools (quality assurance, a proprietary phantom, and related accessories); B: OTS (optical tracking system); C: C5-2/60 (curved probe); D: the specific process of US and CT simulation; E: mobile U/S console; F: quality assurance in treatment room; G: CPI (couch position indicator); H: the specific process of 3D-US and CBCT dual-guided radiotherapy. CBCT, cone-beam computed tomography; CT, cone-beam computed tomography.

Block diagram of the clarity technology roadmap.

A specific procedure. A CC patient (Squamous cell carcinoma-grade II, Stage of IIIC1p) accepting adjuvant postoperative radiotherapy was introduced here as an example of treatment under the guidance of the Clarity system. A. Bladder preparation. B. Ultrasound and CT fusion images. C. Delineation of radiotherapy plan in patients with cervical cancer. D. The representative motion amplitude of the patient's bladder in three directions for one fraction (The green line represents reference RPV, the red line represents daily RPV, and the blue line represents PTV). E. CBCT position verification. F. The bladder position in CT-simulation and each fractionated CBCT image. PTV, Planning Target Volume; CBCT, cone-beam computed tomography; CT, computed tomography; RPV, Reference Positioning Volume.
The scanning and contouring for each patient were administrated by the fixed corresponding operator, with the same US pressure. Finally, simulation CT and US were performed and fused further to guide the delineation of CTV (Fig 3B, C). Here, 3D-US data are first acquired with the Clarity Sim station. It is co-registered or fused with CT data on a Clarity AFC Workstation for treatment planning. A guidance structure in the workstation, defined as Reference Positioning Volume (RPV), is delineated on the Clarity Sim scan and used as a reference on the Clarity Guide scan.
② Contouring: Following the recommendations of ICRU Report 62/83, CTV includes upper vaginal 1/2 and stump, paravaginal soft-tissue, and pelvic lymphatic drainage area (including common iliac, external iliac, internal iliac, parauterine, obturator, and presacral lymph nodes). The upper boundary of the range is between the fourth and fifth lumbar vertebrae, and the lower boundary reaches the level of the lower edge of the obturator. PTV is generated with 1.0 cm extension from the CTV margin. 98% PTV accepts 95% of the prescribed dose, and 95% PTV accepts 100% of the prescribed dose. Organs at Risk (OAR) mainly include the rectum, small intestine, bladder, and left and right femoral heads (including the femoral neck). The OAR limit is: small intestine Dmax < 50Gy, V45 < 65cm3, V40 < 100cm3, V35 < 180cm3, rectum V60 < 30%, V50 < 50%, bladder V65 < 50%, femoral head Dmax < 50Gy, V45 < 25%, V40 < 40%. ③ Fractional radiotherapy guided by the Clarity system: Before each fractional treatment, a 3D-US scan of the patient's lower abdomen is performed to delineate the bladder (daily RPV), and the daily RPV is then aligned with the reference RPV (Fig 3D). When the alignment is considered optimal, the system automatically accounts for the final target displacement by manually adapting the couch in left-right (LR), anterior-posterior (AP), and superior-inferior (SI) directions with a maximum of 5 mm which is coincident with the limits of our center's quality control. Specifically, the bladder contouring is aligned in three directions, but mainly through the bottom wall close to the PTV as the main reference. ④ CBCT position verification: A CBCT is carried out for matching with the CT-Simulation at least three times a week (Fig 3E). The bladder position in each fractionated CBCT image is delineated using the commercial registration software (Offline Review, version 5.11.01, Monaco Medical Systems, Inc., Elekta) (Fig 3F), and the bladder centroid of the CBCT images and CT-simulation images is further compared to obtain the deviation.
Also, these aligning did not have any priority and would only be performed once the CBCT coverage requirements were satisfied. Otherwise, the couch will be shifted based on the result of matching CBCT with the CT-Simulation.
The coordinates of images exported to DICOM are converted to the DICOM coordinate convention. All displacements in the Clarity system follow the patient-centric nomenclature, relative to the patient head-first supine position. All LINAC geometric beam parameters used in the Clarity system follow the IEC 61217 coordinate convention. The figure below (Fig 1) shows a system configuration.
Participants
The study was performed by a team including experienced radiation oncologists, technologists, and physicists. Each of them must undergo rigorous and formal training before operating the Clarity system proficiently.
Displacement Distributions Assessment
By collecting intro-fractional data of involved patients, soft-tissue-based displacements resulting from the additional US-IGRT were acquired in the LT (left)/RT (right), ANT (anterior)/POST (posterior), and SUP (superior)/INF (inferior) directions of the patient before fractional treatment. After the completion of each fractionated radiotherapy for the individual patient, US and CBCT scan were performed again and aligned with the reference RPV and CT-Simulation to obtain displacements in the LT/RT, ANT/POST, and SUP/INF directions respectively. With the delivery of fractional dose, displacement distributions before and after treatment either from 3D-US or from CBCT were estimated during each fractionation for the same patient and were compared subsequently.
Adverse Events
The patients were followed-up and the early urinary and rectal toxicity was evaluated during treatment, and post-irradiation (quarterly) for 1 year according to the Radiation Therapy Oncology Group scale. 24
Statistical Analysis
Displacement distributions were expressed as mean and standard deviation (SD). According to Richter's report, 25 comparisons were performed using SPSS 19.0 statistical software (IBM Corp., USA) and a P-value < 0.05 was considered statistically significant.
Results
Patient Characteristic
Initially, our Center has carried out 3D-US and CBCT dual-guided postoperative radiotherapy for 13 uterine malignancy patients at a mean age of 47 years (range 32-65), and 23% were endometrial cancer, and 77% were cervical cancer. The Eastern Cooperative Oncology Group scores of all patients were 0–1. 38% of patients had a BMI over 24. FIGO I, II, or III accounted for 2(15%), 4 (31%), and 7 (54%) of patients, respectively. The baseline characteristics of eligible patients are summarized in Table 1.
Patient Characteristics.

Abbreviations: FIGO, International Federation of Gynecology and Obstetrics; ECOG, Eastern Cooperative Oncology Group.
All patients underwent a radical hysterectomy, 5 by laparoscopic and 8 by abdominal. Ten patients suffered from squamous cell carcinoma, and three patients suffered from adenocarcinomas. Only 1 case was grade I, and the rest were grade II or grade III. Postoperative radiation or chemo-radiation therapy is required mainly because of positive pelvic lymph nodes and/or other medium-high risks, ie, Sedlis Criteria (See Table 1 for details). Of all cases, only 1 patient did not undergo concurrent chemotherapy (the patient refused).
Soft-Tissue-Based Displacements Resulting from the Additional US-IGRT Before Treatment
After using hydration protocol combined with a portable ultrasound for the bladder preparation, the patients were well-trained, tolerated before simulation, and achieved planned bladder volumes comparable with simulation during fractional radiotherapies, as we can see that all patients’ relative bladder volume (fractional bladder volume relative to the simulation) were approaching to 1 (fig 4). Throughout the duration of the treatment, the use frequency would be gradually reduced or even waived for portable ultrasound.
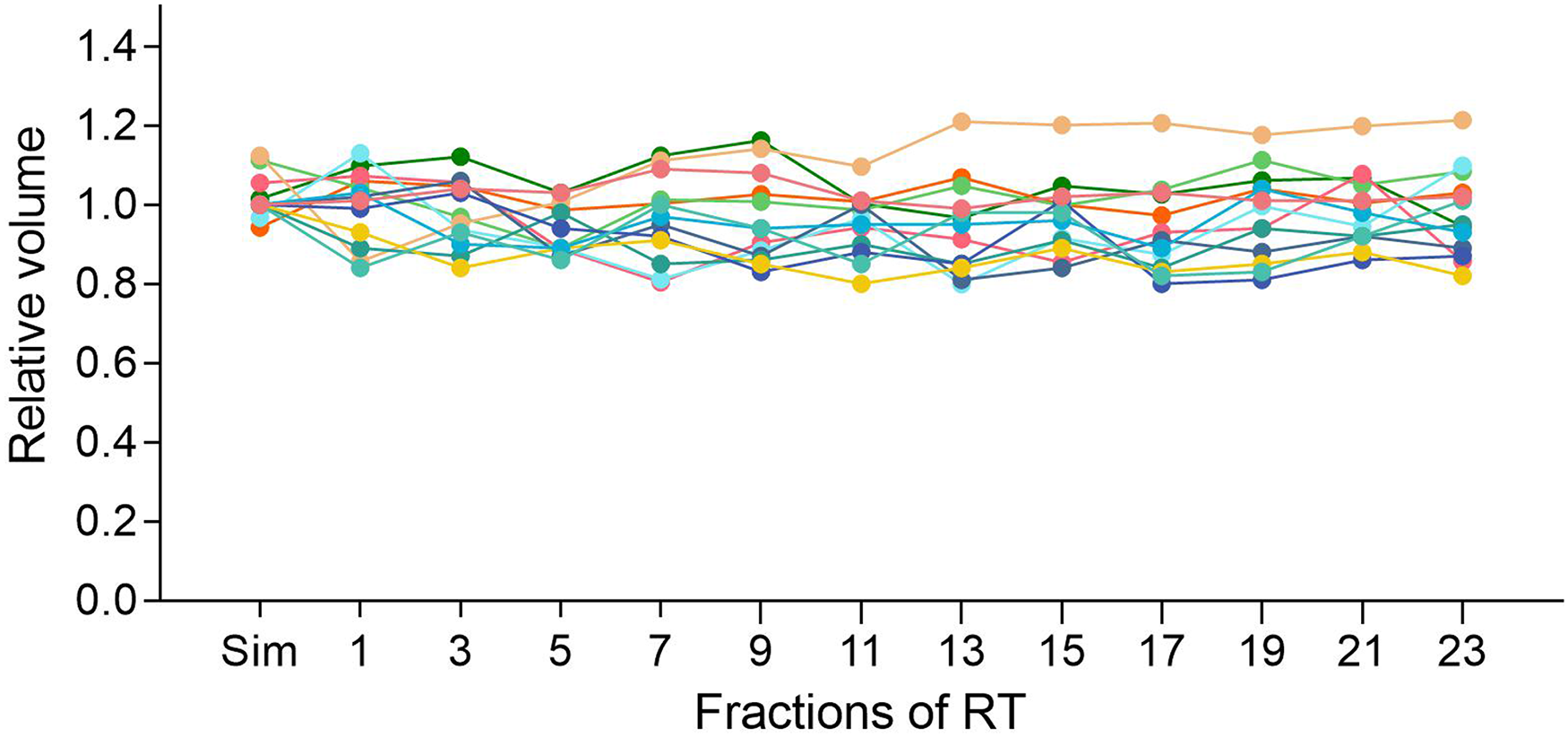
Relative bladder volume of 13 patients. Relative bladder volume: fractional bladder volume relative to the simulation. Each colored line represents a patient.
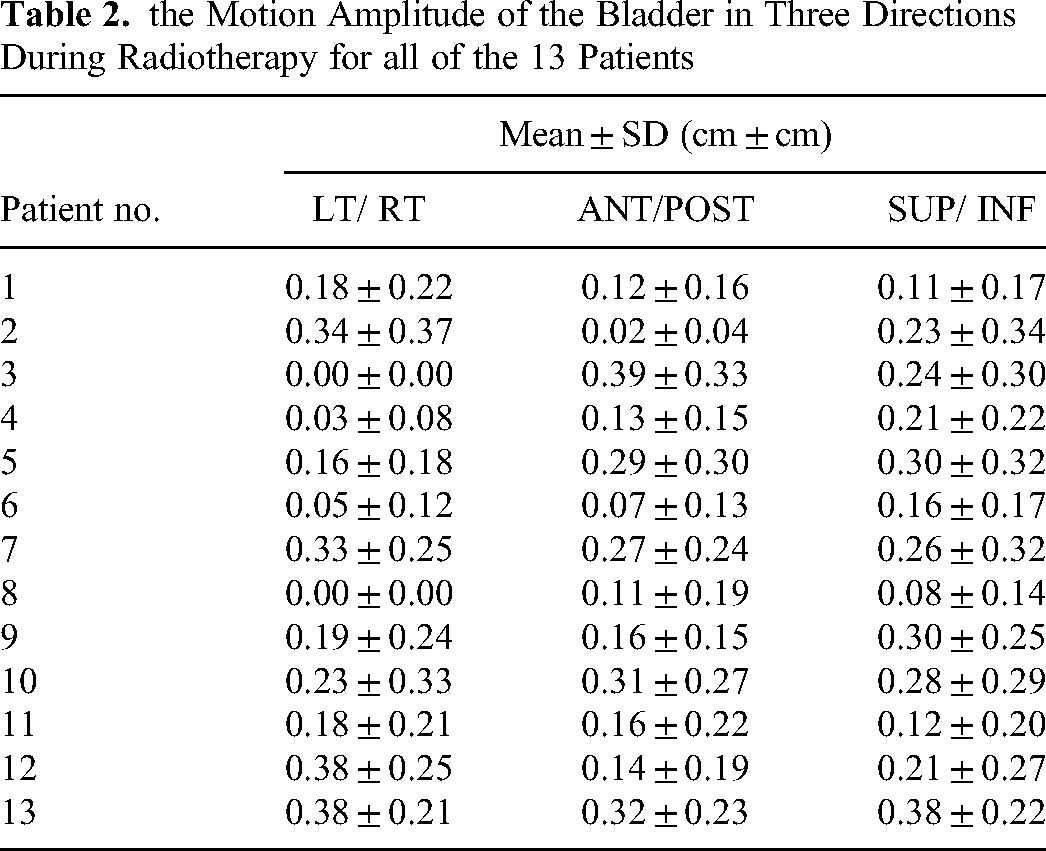
The bladder displacement of these patients has been obtained and presented in Table 2. As expected, the evaluation of 170 scans resulted in a mean displacement of (0.17 ± 0.24) cm, (0.19 ± 0.23) cm, and (0.22 ± 0.26) cm for bladder in LT/RT, ANT/POST, and SUP/INF directions, respectively.
the Motion Amplitude of the Bladder in Three Directions During Radiotherapy for all of the 13 Patients
Deviation of the Bladder Centroid
The bladder centroid of the CBCT images and CT-simulation images are also compared to get the deviation in three directions, and the results are shown in Table 3. As we can see, the evaluation resulted in a mean deviation of (0.26 ± 0.22) cm, (0.58 ± 0.50) cm, and (0.30 ± 0.23) cm in the LT/ RT, ANT/POST, and SUP/ INF directions, respectively. The average deviation of the LT/ RT and SUP/ INF axis of all 13 patients is less than 0.4 cm, and the deviation of the ANT/POST-axis is relatively larger (up to 2.5 cm).
The Average Deviation of the Bladder Centroid Between the CBCT Images and CT-Simulation Images in Three Directions for all of the 13 Patients CBCT, cone-beam computed tomography; CT, computed tomography.

Displacement Distributions After Treatment
Displacement distributions before and after treatment either from 3D-US or from CBCT were acquired for the same patient during the individual fractionation (see Table 4). Paired comparison between these two guidance shows that the variations from 3D-US are much smaller than those from CBCT in the LT/ RT, ANT/POST, and SUP/ INF directions, especially in ANT/POST, and SUP/ INF directions with significance (P = 0.000, 0.001, respectively).
Variations from 3D-US and CBCT in Three Directions for all of the 13 Patients. the Right is Obtained by 3D-US, and the Left t is Obtained by CBCT CBCT, cone-beam computed tomography.

Adverse Events
All patients completed radiation treatment with no more than moderate urinary or rectal toxicity and no patients reported any recurrence or metastasis so far. During treatment, there was no grade 3 or higher urinary and rectal toxicity happened. Five patients had G2 rectal toxicity during treatment, only one at the end of radiotherapy and at 6 months after treatment, and the rest were at G1 or lower toxicity (fig 5A). Simultaneously, only two patients experienced G2 urinary toxicity during treatment, one at the end of radiotherapy, and the developed urinary toxicity was G1 or lower at 3, 6, 9, and 12 months after treatment (fig 5B).

Percentage of patients with acute rectal (A) and urinary (B) toxicity (RTOG) observed during radiation treatment, and 0, 3, 6, 9, and 12 months after radiation treatment. RTOG, Radiation Therapy Oncology Group.
Discussion
Patients with uterine malignancy are generally treated with adjuvant radiation therapy after surgery to improve the local control rate of the lesions and the survival rate if necessary. Due to the variable filling and position of the bladder and/or rectum, CTV experiences underestimated movement and deformation during the whole treatment, which in turn reduces the accuracy of radiotherapy.
As Elekta's latest generation of ultrasound-based image guidance system, the Clarity system uses 3D image data, and the image quality is significantly improved. A series of studies have used the calibration phantom to quantify the ultrasonic imaging of the Clarity system and found that its imaging accuracy is less than 1mm in each direction, confirming the high accuracy of the Clarity system.26–28 Particularly, the Clarity system provides a true in-mold verification method which has proven to be more accurate than the cross-modal method in the prostate positioning (compared with CT-simulation). 29
To tackle the inaccuracy of radiation target as a result of bladder volume varying, a 3D-US (Clarity system) was introduced here as an alternative IGRT to monitor the changes in the position of the internal organs of the postoperative uterine malignancy patient with a full bladder before each CBCT irradiation. As most of these patients are poor in bladder function, and some even have urinary incontinence, we prefer to obtain a comfortably full bladder by which the patients could easily reproduce during treatment. Studies showed that uterine malignancy patients could easily achieve a full bladder in the 0–300 ml range of initial bladder volume even without ultrasound measurement 30 and that bladder volumes of 150–300 ml at planning were recommended for radiotherapy of uterine malignancy. 31 Also, in a study for prostate radiotherapy, a planned bladder volume greater than >200 ml reduced intrafraction organ motion, and the hydration protocol prior to treatment was well-accepted and tolerated. 32 In the current study, an optimal bladder volume of 200–300 ml together with hydration protocol was preferred before simulation.
Successfully, a primary workflow of 3D-US (Clarity system) and CBCT dual-guided radiotherapy has been established for postoperative uterine malignancy patients. Each patient was firstly counseled to follow the procedures mentioned above to get a comfortably full bladder before simulation CT and each daily radiation. As shown in Fig 4, these patients could be well-trained and would be tolerated these procedures. The bladder volume was consistent for each individual patient during fractional radiotherapies. Thereafter, the individual mold was subsequently adapted to the patients, and then simulation CT and US were performed and fused to guide the delineation of CTV. Before each fractional treatment, a 3D-US scan of the patient's lower abdomen is performed to delineate the bladder (daily RPV), and the daily RPV was further aligned with the reference RPV by adapting the couch with a maximum of 5mm and with no priority to CBCT verification. Finally, irradiation was done with precision along with our proposed workflow.
After maintaining the consistency of bladder volume, the centroid of the bladder was also compared in this study with the purpose to represent the position changes of the bladder. So far, the comparison of bladder centroids has been proposed in studies for uterine malignancy, 33 prostate cancer, 34 and bladder cancer 35 radiotherapy. Based on the results monitored by the Clarity system, it was found that the bladder displacement is clearly insignificant in most patients (see Table 3), providing the first evidence that the dual-guidance showed the therapeutic potential for postoperative uterine malignancy patients. At the same time, we had to admit some patients did experience considerable displacement, especially for patients with obesity, as the simulation error for these patients would be comparatively larger. Some studies noted strong correlations were observed between BMI and standard deviations of daily shifts, 36 and this could be part of the reason for the higher recurrence rate seen in obese patients who received IGRT for prostate cancer. 37 Similar results were reported in gynecological cancers as patients with a larger BMI have a higher set-up uncertainty and would benefit more from daily imaging. 38 Moreover, the measurement deviation would even become larger once the operators are less experienced in processing the Clarity system. Therefore, based on our experience, we strongly recommend that operators be systematically trained (to improve the accuracy of their imaging and positioning operations) before using the Clarity system, although this procedure itself consumes a lot of time and cost.
Meanwhile, several other factors which may decrease 3D-US accuracy also have been taken into consideration in this research. ①Each patient was suggested to empty their intestines prior to the 3D-US scan to minimize the impact of pelvic gas. ②A key factor restricting the accuracy of US guidance is the pressure exerted by the US probe on the skin, which may affect the imaging quality and may change the position of the bladder. Fortunately, a previous study has shown that a relatively low pressure (1cm displacement of the probe on the skin of the abdomen) using the Clarity system is sufficient to obtain excellent image quality and produce a small prostate displacement. 39 Considering this kind of displacement occurs in both US-simulation and US-guidance, these displacements would not affect the accuracy of bladder positioning correspondingly as long as the same pressure is applied by the fixed corresponding operators during the scan for this intra-modality verification.
Generally, Clipbox registration is routinely performed during fractional radiotherapy registration for postoperative patients with uterine malignancy, which mainly includes the target area and the surrounding pelvis, and the isocenter is the registration reference point. The conventional Clipbox only meets the correction of the positioning error, but it cannot correct the displacement of the soft-tissue. Oppositely, the Clarity system's bladder registration did make up for this deficiency. As we can see, the bladder deviation of the LT/ RT and SUP/ INF axis between the CBCT image and the positioning CT was successfully maintained within an acceptable range (less than 0.4 cm) after comparison, indicating the bladder consistency was fully ensured with Clarity's guidance and its therapeutic potential were certainly highlighted for the second time (see Table 2) However, a large deviation of the ANT/POST-axis is also observed, which may be due to the illegibility of the bladder boundary in the CBCT image, particularly the posterior wall. As long as manual delineation of the bladder is performed, the deviation is unavoidable so far and can be ignored in theory.
Reference from the above results, the displacement distributions before and after treatment either from 3D-US or from CBCT were estimated during each fractionation for the same patient. Paired comparison shows that the variations from 3D-US are much smaller than those from CBCT in the LT/ RT, ANT/POST, and SUP/INF directions, especially in ANT/POST, and SUP/ INF directions with significance (P = 0.000, 0.001, respectively, see Table 4), illustrating the probable cause that the CBCT (only) guidance deficiency, which thereby providing the third evidence verifying the therapeutic potential for the dual-guidance. Since the consistency of bladder was well maintained and the target displacement was reduced with the introduced 3D-US here, the PTV boundary (CTV-PTV margin) could assume to be minimized spontaneously.
Finally, all patients completed radiation treatment with mild to moderate toxicity. During treatment and follow-up, there was no grade 3 or higher urinary and rectal toxicity found (see Fig 5) and no recurrence and metastasis happened, highlighting again the adaptability and the therapeutic potential of this technology.
However, there are also several limitations to this study. Firstly, without adequate estimation and further follow-up, we need a greater deal of clinical outcomes regarding using the 3D-US technique for these patients. Secondly, this study is a descriptive study, and the small sample of patients involved in the study limited the value of this significant development in radiotherapy. In addition, 3D-US guided radiotherapy did prolong the treatment procedure, resulting in a larger amount of clinical workload for radiation oncologists, technologists, and physicists.
Conclusions
In summary, a primary workflow of 3D-US (Clarity system) and CBCT dual
Footnotes
Acknowledgments
We would like to thank Dr Jianghai Mei of Toronto, Canada for her professional help in polishing this paper.
Authors’ Contributions
Conception and design: Yang Li, Zhen Gong, Hanzi Xu, Xia He Administrative support: Yang Li, Zhen Gong, Hanzi Xu, Xia He Provision of study materials: Chenjing Zhu, Zhihua Sun, Li Sun Collection and assembly of data: Yang Li, Zhen Gong, Han Gao, Chang Guo, Le Yu Data analysis and interpretation: Yang Li, Zhen Gong, Mengyu Liu Manuscript writing: All authors Final approval of manuscript: All authors.
Availability of Data and Materials
The datasets generated during/or analyzed during the current study are not publicly available due to patient privacy concerns and institutional regulations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The study was approved by the ethics committee of the Affiliated Cancer Hospital of Nanjing Medical University (Jiangsu Cancer Hospital) (NO. 2021-005) and informed consent was taken from all individual participants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Clinical Research Fund of the Spark Program for Precision Radiation & China International Medical Foundation (No. 2019-N-11-12, HDRS2020030101), Jiangsu Province Association of Maternal and Child Health Grant (FYX202025) and National Natural Science Foundation of China (No.81872485).
