Abstract
Radiotherapy is a multi-step process that includes planning, contouring, simulation, patient assessment, quality control, and treatment. Each step must be completed before moving on to the next. Numerous factors, including patient characteristics, disease type, management, radiotherapy personnel, equipment, treatment modality, and total/fractional doses, affect the overall duration of radiotherapy. Time is one of life's most valuable resources and should be well managed and utilized. In radiotherapy, eliminating factors that unnecessarily prolong the treatment period significantly benefits the institution, patient, and staff. This review article examines the variables that affect overall treatment time in current external beam radiotherapy routines and offers suggestions for reducing treatment time.
Introduction
Radiotherapy (RT) is an effective treatment method that uses radiation to destroy or inhibit cancer cell growth. 1 Advances in RT have enabled the precise protection of healthy tissues while increasing the treatment dose. 2 One of the main goals of developing these technologies is to streamline therapeutic procedures. Time is a critical factor in RT. 3 The total treatment time can be defined as the interval between the day the patient makes a decision to undergo treatment and the day the RT procedure is completed. Several steps during treatment, such as patient setup, image acquisition, image-guided online couch correction, and radiation delivery, contribute to the overall treatment time. 4 The duration of RT is influenced by numerous factors, including tumor type, size, location, prescribed dose, fractionation scheme, and plan complexity. 5 Treatment times may also vary based on individual factors, such as the complexity of the tumor, experience of the RT staff, and specific equipment and techniques used in a given medical facility. Patients with large or critically located tumors or those experiencing significant respiratory movement may require more complex treatment plans, potentially extending treatment time. 6
The length of the RT course is important for patients, clinical workflow, and overall treatment outcomes. 3 RT can be a drawn-out procedure that requires weeks or even months to complete. Patients and radiation staff are affected by long treatment processes. Patients may experience harmful psychological effects if their treatment is prolonged. 7 Nevertheless, long-term RT can lead to discomfort, a greater chance of treatment discontinuation, and greater treatment expenses. Therefore, research aimed at reducing the overall length of RT is necessary to improve patient quality of life, treatment compliance, and clinical effectiveness. Prolonging the duration of RT can increase tumor repopulation and affect tumor growth.8,9 The decrease in the total treatment time during RT is promising for patients and healthcare professionals. Providing safe and effective treatment within a short period can contribute to patients experiencing significant physical and emotional recovery compared with longer treatment regimens. 10 On the other hand, when tumor repopulation occurs, quick treatment procedures improve clinical outcomes and treatment compliance. 1 In addition, shortening the total treatment time is highly important for healthcare providers. Cost savings, easier planning, and better patient service can be achieved through time management.
New imaging systems that provide precise radiation delivery, rapid image acquisition, accurate target detection, and patient positioning with artificial intelligence (AI) support have been developed, minimizing the need for manual correction. More advanced treatment options are becoming possible with the introduction of new technological equipment. These capabilities include sophisticated dose calculation techniques, automated and AI-driven methodologies, enhanced target localization, and advanced treatment planning methods. Compared with conventional procedures, these advancements require less time for treatment because they involve more efficient processes. 11 Additionally, new treatment approaches, such as hypofractionation, stereotactic body radiation therapy (SBRT), intensity-modulated radiation therapy (IMRT), and planning algorithms, have transformed treatment planning by enabling the delivery of higher doses in fewer fractions while preserving therapeutic efficacy. 2 Recently, FLASH RT has emerged as a technique for very short treatment periods that can deliver ultrahigh doses at high speeds. 12
Modern external beam RT practices aim to enhance the comfort of both patients and staff while delivering effective treatment. Ideally, RT processes should proceed efficiently, continuously, and without unnecessary delays. This review aimed to identify the variables that influence the overall treatment time in current external beam RT regimens and provide recommendations for reducing treatment time from a comprehensive perspective. A detailed literature search was conducted to explore the concept of time in RT, using PubMed, Web of Science, and Google Scholar as the primary databases. The search was carried out without restrictions on publication date or geographic location, allowing for a broad perspective on the topic. The literature search involved a combination of specific keywords: “radiotherapy,” “time,” “time efficiency,” “time-saving processes,” “time-consuming,” “time reduction,” “current radiotherapy practices,” and “artificial intelligence in radiotherapy.” Additionally, articles were identified through reference lists of relevant studies and articles that cited them, ensuring a comprehensive inclusion of pertinent works.
The majority of the references were recent (published within the last five years) and written in English. Articles were selected based on their relevance to the core topics of time efficiency and technological innovations in RT. The process involved a thorough review of abstracts, conclusions, and key findings to determine inclusion. No statistical analyses or systematic protocols were performed in this literature review. The focus of the article is to synthesize and summarize broader trends in the literature.
Planning Time
RT planning time refers to the time required for patients to start RT. This process includes several steps.
Treatment Decision
The peer review of clinical decisions made during RT planning should be considered a standard of care. 13 The decision whether a patient will receive radiation therapy is based on evidence-based practices and international consensus treatment guidelines. The RT decision-making process includes both the radiation oncologist's recommendation and the patient's consent. Effective communication between the patient and clinician can expedite the decision-making process and facilitate timely treatment initiation. 14
Immobilization
Immobilization devices are commonly used to minimize patient movement during RT to ensure that the radiation dose is accurately targeted to the intended area. 15 Various disease-specific immobilization devices are available and are typically maintained unchanged throughout the treatment course. 16 These devices must be of high quality and should not become deformed during use; otherwise, replanning may be required. Imaging for treatment planning involves capturing images of the patient in the intended treatment position while the relevant immobilization devices are in place. The positioning time is defined as the duration between entry to the room and the initiation of imaging. During this phase, the patient is prepared for simulation or treatment. Delays in this process can arise from staff inexperience, unfamiliarity with the system, or patient non-cooperation. 17
Personalized immobilization during simulation can lead to extended time in the simulator room. 18 However, if the patient can be accurately positioned, this position can be replicated more easily in subsequent treatments, which is crucial for effective RT. Additionally, if patients feel comfortable during the procedure, the treatment time may be reduced. 19 The upper part of the simulation table, which is designed to accommodate the necessary treatment accessories, must also be compatible with the treatment device table. Advanced technology in these accessories enhances positioning accuracy, patient comfort, and can help reduce setup time.20,21
Simulation
Historically, RT images were acquired using traditional two-dimensional (2D) simulators, but today, 3D computed tomography (3DCT) simulators are commonly used. The primary goal of 3DCT simulation is to obtain detailed geometric information about the patient, which is then used to create a 3D model for treatment planning. 22 In some cases, magnetic resonance imaging (MRI) and positron emission tomography (PET) are used in conjunction with CT to more accurately define target volumes and organs at risk (OARs). 23 Modern CT scanners, which have replaced older models, offer significantly shorter scanning times. Newer CT scanners incorporate a fan beam and multiple detectors, resulting in faster scan times. The use of faster scanners can reduce the total RT treatment time. 24 However, the CT scanning process can still be time-consuming, and when combined with long treatment regimens, it can contribute to extended overall treatment times. Coupling AI and CT imaging can provide faster, more accurate, and more efficient imaging. 25 In addition, AI-based automatic positioning and cantering in CT imaging is a promising new technique and has been shown to reduce total positioning time by 28% compared with manual positioning. 26 Some studies have investigated non-simulation methods for palliative care to save time. 27
Faster CT scanners reduce motion artifacts because the patient has less time to move during the scan. Newer CT devices also have artifact-reduction techniques that save time by reducing CT repetition that can result from artifacts. 28
Tumor motions and internal structure changes due to respiratory and cardiac motion represent major sources of uncertainty, especially for the treatment of breast and abdominal cancers. To mitigate these uncertainties, recent advancements in 4DCT and 4DMRI techniques have been reported. While 4DCT is widely accepted in clinical settings because of its ability to track tumor motion, 4DMRI offers enhanced soft tissue contrast and fewer binning artifacts. 4DMRI generally requires longer acquisition times compared with 4DCT.29,30 Breath holding techniques are useful in moving at-risk organs away from the target area. However breath-holding techniques, although effective in reducing motion artifacts, require optimal communication between staff and patients and may significantly prolong treatment times. 31 Breath-hold procedures can extend treatment durations by 30 to 45 min, particularly in older or frail patients. 32 As a result, selecting suitable patients who can effectively coordinate using this method can help minimize time loss. 33
Contouring
In many clinics, tumor delineation and the identification of organs at risk prior to treatment planning are largely manual processes typically performed by radiation oncologists, dosimetrist, or physicists. 34 Accurate identification of the tumor and surrounding critical structures is crucial for effective treatment planning. One of the key factors contributing to the prolongation of the shaping process is the variability and complexity of the radiation oncologist's duties, 35 whereas high patient density in the clinic can further extend this process. The standard contouring workflow, which is typically managed solely by clinicians, is time-consuming and often inefficient.36,37 Many factors affect the time required for contouring, one of which is the need for peer review. 38 However, with advancements in technology, the automatic segmentation of tumor and organ volumes at risk has significantly improved because of developments in computational techniques. 39 AI encompasses a range of programs that simulate human intelligence. Machine learning, deep learning (DL), and convolutional neural networks (CNNs) are used for automatic contouring.40,41 There are several automatic segmentation methods. One such method is Atlas-based automatic segmentation (ABAS), where a software program is trained on a dataset in which the critical organs are pre-labeled to create an ‘atlas’ of the critical organs. Another automatic segmentation method is DL-based automatic segmentation (DLAS). DLAS uses machine learning to incorporate large datasets and produce an automatic solution. 42 Thus, the clinical application of AI tools in RT can increase contouring efficiency while reducing interobserver variation. The total time required for treatment preparation is greatly reduced owing to the fast and accurate automatic contour segmentation.43,44 Studies have reported that AI-based processes such as automatic contouring can reduce planning time by approximately 30% to 70% compared to manual approaches.34,36 However, the use of these systems requires rigorous evaluation of the clinical acceptability of the results and should be accompanied by appropriate quality assurance procedures.
Treatment Planning Time
Treatment planning is a critical process in radiation therapy (RT). Traditionally, treatment planning is a manual, time-consuming, and labor-intensive task, which in some cases can take hours or even days to complete. Multiple iterations of human interaction must balance tumor control with the protection of normal tissues. The iterative nature of manual treatment planning often makes the process tedious, particularly for complex plans, which necessitate the expertise of experienced. 45 Treatment planning time can be defined as the period from the initiation of the plan to the final optimization, completion, and approval of the dose calculation. With technological advancements, algorithms that can calculate plans more rapidly have been developed. 46 These systems significantly reduce the time required to generate high-quality treatment plans by enabling rapid contouring, dose calculation, and plan optimization. Additionally, incorporating automated plan evaluation tools, which assess plan quality and dose restrictions according to clinical guidelines, can accelerate the decision-making process and minimize time loss. 47 DL can completely automate planning from segmentation to optimization in hours, minutes, or even seconds. 48 The primary benefit of AI is reducing treatment planning time and improving plan quality. 49
Quality Assurance
The primary objective of quality assurance (QA) in radiation therapy (RT) is to ensure the stability and consistent performance of the RT system over time (eg, daily, weekly, monthly, and annually) and to identify any deviations or errors in system performance. 50 QA is essential to ensure that patients receive prescribed treatments accurately and safely. 51 Before the start of treatment, a customized quality assurance (QA) program must be developed to meet the specific needs of the treatment plan. 30 Achieving high-quality RT requires the integration of advanced treatment techniques and a robust QA program, both of which contribute to longer treatment times. 52 QA tasks, including administration, preparation, delivery, analysis, and reporting, represent a significant workload for RT teams, often resulting in machine downtime of up to 1–2 h per day. As a result, patient-specific QA processes are highly time-consuming and can lead to treatment delays. 53 QA management in RT is typically overseen by physics teams within RT departments. The efficient coordination of these teams can help optimize time management and minimize downtime. In addition, gamma analysis results measured using a 2D detector can be used to predict gamma analysis results for QA using machine learning research, thereby reducing QA time. 48 Novel treatment plan QA software can serve as an independent plan evaluation and dosimetry check. 54
In vivo dosimetry (IVD) is an important component of modern radiotherapy, enabling the safe delivery of radiation doses to patients by measuring dose parameters during treatment. However, integrating IVD into routine clinical workflow can present practical challenges. The additional steps required for dosimeter preparation, placement and data analysis can disrupt standard workflows and increase treatment times. To reduce the time spent in these processes, studies are being carried out on integrating artificial intelligence, Monte Carlo simulations and developing new materials such as nanowire-based OSLDs. 55
Setup Time
Advancements in technology have facilitated tighter control over the distribution of radiation fields, resulting in highly compatible dose zones. 56 Acquiring patient imaging data after the initial setup enables the comparison of the treatment position with the planning image, allowing for precise measurement of setup errors. 57 Ensuring precise and reproducible patient positioning is critical for maximizing treatment efficacy while minimizing side effects. By emphasizing the importance of lead time and implementing rigorous quality assurance protocols, RT departments can enhance treatment precision and overall success.
At the onset of treatment, a variety of setup errors may arise, including issues with the treatment table, patient movement, technician errors, data discrepancies, accessory malfunctions, and machine errors. 58 A daily online correction protocol that addresses both systematic and random errors can significantly extend treatment time. 59 The imaging time encompasses the duration required for delivering the initial imaging area, evaluating the details, applying the necessary table shifts, and calculating the corrections. 60 Expanding the setup margin can reduce the positioning time. 61 However, this may carry risks such as reducing the accuracy of radiation dose delivery. Clinics can develop rapid evaluation protocols for setup, and continued training of RT technicians during setup procedures can prevent unnecessary delays. Automated setup calculations and correction processes can further accelerate the treatment process. 62 Setup accuracy and speed are increasing thanks to real-time image monitoring systems supported by Al.
Adaptive Radiotherapy Time
During RT, various changes can occur, including alterations in weight, tumor size, organ deformation, filling variations, respiration, and peristaltic movement. Adaptive radiotherapy (ART) allows for treatment plan adjustments based on anatomical changes or patient responses. ART can be categorized into three main workflows: (I) offline between treatment fractions, (II) online before treatment fraction, and (III) real-time during treatment fraction. However, ART workflows introduce additional complexity, which may increase the uncertainty of radiation therapy planning and delivery. These processes are labor-intensive and time-consuming.63,64 Technological advancements in ART, such as volumetric imaging and automatic segmentation, facilitate the calculation of daily doses and allow adaptation decisions to be made based on dosimetric data rather than on geometric information. 65 Despite these advancements, ART still has technical limitations in clinical practice. This requires a multidisciplinary team consisting of radiation oncologists, dosimetrist, therapists, and medical physicists, all of whom invest considerable time in the process. Re-contouring, re-optimization, and treatment plans must also undergo re-examination for quality control purposes.
Online ART is performed when the patient is positioned in the treatment room, but maintaining patient stillness during replanning can be challenging. Achieving rapid and effective ART remains difficult.66,67 The key to overcoming these challenges lies in improving computational speed and automation. Recent studies indicate that with the latest technological developments, this process can be completed more efficiently.68,69 Time savings can also be achieved by developing protocols to guide patient selection for ART. 70 Significant research is ongoing to reduce ART time using AI-assisted workflow. 43
Treatment Time
The treatment time can vary based on the planning technique and treatment machine used. For example, complex treatment plans typically require longer planning and delivery times, including extended gantry or couch movements. Intensity-modulated radiation therapy (IMRT) is particularly time-consuming because it involves intricate planning, delivery, and quality assurance processes. Extended treatment times may increase the risk of intrafraction motion, impact patient comfort, and cause complicated positioning. Conversely, volumetric modulated arc therapy (VMAT) is associated with shorter delivery times compared with IMRT or tomotherapy.71,72 The treatment machine type also significantly affects the overall treatment time. For instance, pencil-beam scanning proton therapy tends to require longer treatment times, whereas traditional linear accelerators (linacs) provide faster treatment delivery. 60 Recent technological advancements, such as Flash-RT, enable the delivery of much higher doses of radiation, potentially reducing treatment times considerably. 12 The total treatment time includes the setup, imaging, and delivery stages. The imaging time varies depending on the treatment machine used, such as cone-beam CT (CBCT), megavoltage CT (MVCT), or magnetic resonance imaging.61,73 As treatment complexity increases, the plan must be adjusted accordingly to avoid prolonged treatment times. 74 Newer treatment devices with advanced software have lower error rates, thereby promoting time efficiency.
Interrupt/Error Time
One of the primary objectives of radiation therapy (RT) is to deliver the planned treatment dose without interruption or extension to the overall treatment duration. However, in clinical practice, treatment interruption may occur for a variety of reasons. 9 These interruptions, whether predictable or unpredictable, can lead to prolonged treatment times. 75 Such delays can disrupt the appointment scheduling system and cause scheduling timeline shifts. Some disruptions may be resolved within minutes, whereas others may persist for days. Common causes of treatment interruption include machine breakdowns, treatment plan changes, and immobilization errors. Additionally, issues related to the patient or treatment team, such as table movement and incorrect data entry, can also contribute to delays. 76 The introduction of systems that use advanced software and newer devices can help reduce the risk of errors. Many clinics are developing interruption management programs to address these challenges. The implementation of such programs improves RT treatment outcomes and enhances tumor control. 77 In addition, prompt field service engineer support and replacement of defective parts are important factors in reducing downtime.
The Role of Staff
The RT process comprises complex and detailed steps that require the coordinated efforts of a large team. The RT team includes radiation oncologists, medical physicists, dosimetrist, radiation therapists (RTTs) and nurses. 78 These professionals are involved in many stages of the process, including treatment planning, dose calculations, target area determination, equipment calibration, patient recruitment, and follow-up. In RT applications, careful monitoring is required to accurately define the target area, accurately position the patient, and deliver the correct radiation dose. Patient-specific checks, such as daily positioning or setup errors, tumor shrinkage, or weight loss over time, are time-consuming. 79 In particular, repeated CT imaging and re-planning processes contribute to increased time spent on re-contouring, dosimetry procedures, simulation, and treatment, as well as increased equipment uptime and RT staff workload. 80 A diagram of the time-consuming external beam RT workflow is presented in Figure 1. Each step is time-consuming and can significantly prolong the overall treatment process.

Time-Consuming External Beam RT Workflow (RO: Radiation Oncologist, P: Patients, N: Oncology Nurse, RT: Radiotherapy Technician, RP: Radiotherapy Physicist, D: Dosimetrist).
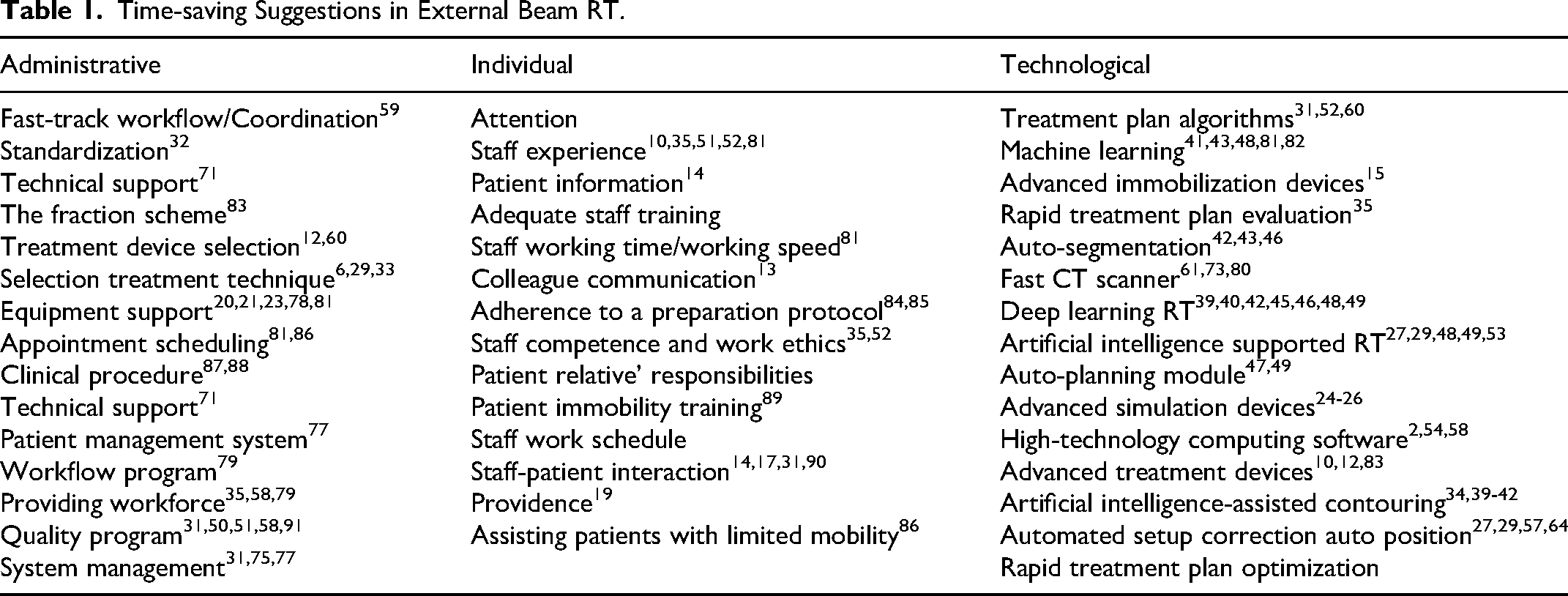
Considering the heavy workloads of RT personnel, errors arising from carelessness can be costly. Moreover, one potential strategy for reducing overall treatment time is to alleviate the workload of RT staff. As the demand for RT has increased over time, shortages in equipment and staffing limitations have contributed to longer treatment durations and increased patient waiting times. The burden on RT staff is heavy, and the cost of mistakes resulting from carelessness is high. In addition, the workload of RT staff should be alleviated to shorten the total treatment time. Because the demand for RT has increased over time, the lack of equipment and the limited number of employees have caused both the treatment time and the waiting time of patients to increase. 81 Adequate staffing is essential to ensure that patients can complete RT treatment in an efficient and timely manner. Time pressure in high-demand clinics can be detracted from the productivity of RT personnel. 58 An increase in the amount of RT personnel, coupled with greater availability of RT equipment, reduces patient waiting times. 35 In other words, expanding the workforce can lead to time savings. Furthermore, the competencies, experience, and work ethic of RT staff are critical factors influencing the efficiency of treatment execution. 52 Table 1. presents time-saving suggestions from administrative, individual and technological perspectives in external beam RT. Time efficiency is very important, but should not be to the extent that it compromises dosing accuracy. Any time-saving methods should be implemented after careful evaluation and used when clinically acceptable. Otherwise, they may compromise the possibility of tumor control or increase the possibility of normal tissue complications.
Time-saving Suggestions in External Beam RT.
The rapid advancement of RT technology has yielded significant and effective results. 87 Technological innovations, including enhanced simulation contouring and accelerated planning and treatment times, are likely to positively impact RT personnel by reducing the overall treatment duration. 31 Well-trained RT staff can mitigate time losses during the RT process by staying abreast of technological advances. Experienced practitioners are particularly pivotal in successful application of new methodologies. 10
The time allocated for each patient in the treatment room must account for correct positioning, verification of patient details and RT administration. Time allocation should be carefully calibrated according to the procedure, ensuring that it is neither rushed nor delayed. Time efficiency can be gained by optimizing appointment scheduling and considering multiple parameters to ensure the most efficient use of time.
The Role of Patients
Approximately half of all patients with cancer receive RT during tumor treatment. Making the RT decision on time and starting it without losing time increases the effectiveness of the treatment results. 91 Quick approval of treatment decisions reduces time costs. In addition, long waiting times for radiation therapy may have a negative clinical impact. For example, delayed radiation therapy may increase the risk of local recurrence92,93 and reduce the survival rate.94,95
The waiting period for the start of treatment after the decision to receive RT may lead to low treatment satisfaction.82,96,97 Waiting for a healthcare provider's waiting time can cause patients who are already sick and in pain to worry about when they will receive the necessary care. 98 Studies have shown that severe COVID-19 may cause additional psychological distress in patients who are exposed to delays in the start of treatment and long waiting times. 7 It is highly important to make patient appointment plans correctly in clinical operations and to comply with the timing to reduce unnecessary waiting time for patients. 99 An additional factor affecting the duration of treatment is the limitation of patient movement. 88 Patients who need assistance in moving take longer to receive treatment than those who do not. Therefore, considering that RT departments may need additional time to treat patients with limited mobility, treatment periods should be allocated appropriately. 86 The bladder-filling protocol, especially when applied before pelvic RT, is another factor that increases the waiting time of patients. Bladder-filling protocols specify a policy to void and drink water before RT to achieve consistent bladder volume at the time of RT each day. 84 Patients may be instructed to empty their bladder and drink 300-ml water 1 h before starting RT. 85 Patients must comply with these protocols. The expected duration of bladder fullness will affect the total treatment time. In addition, fever, blood loss, and weight before treatment may prolong the treatment process. In clinics with high patient density, crowded waiting rooms and the fear of missing an appointment or being late can create a stressful situation for patients. 58 Patient appointment plans should be created considering all these situations.
Shortening the treatment period can be advantageous for patients in many ways. For example, a shorter treatment duration for pediatric patients may also be beneficial when there are problems with treatment positioning that cause patient discomfort or difficulties accessing the airway when general anesthesia is required. 83 Time savings can reduce treatment costs and increase patient quality of life, access to radiation therapy, and RT department capacity. 100
RT is a very sensitive treatment method in which patients are required to take correct positions and remain motionless during treatment. A moving patient may cause radiation to spread to unwanted areas and damage tissues outside the treatment area. Patient movement can cause setup errors and organ movement. 89 If patients move during treatment, treatment is stopped, reimaging is performed, and the setup is repeated. During this process, patients and employees who interact and are sensitive about remaining inactive can prevent the treatment period from being extended. If the total treatment time can be reduced, the time patients and their families spend away from work and home can be reduced, and thus, economic benefits can be achieved. 90 Patients are expected to follow staff instructions during treatment, which can reduce treatment times.
Implementation of time-saving strategies in radiotherapy can improve treatment efficiency and patient throughput. Key approaches include the use of artificial intelligence for automated contouring and treatment planning, the adoption of standard protocols to streamline workflow, optimization of patient flow management through efficient planning systems, and the use of advanced imaging techniques for rapid and accurate planning.34,36 In addition, ART increases precision and efficiency by allowing treatment adjustments to be made in real-time. Implementing effective management principles to eliminate workflow inefficiencies, ongoing staff training, and the use of robotic technologies for precise patient positioning should also be included. 51 Combined, these methods help reduce patient preparation time, optimize resource use and improve overall quality of care.
Conclusion
The extension of the total treatment time in RT for various reasons is undesirable for patients, healthcare providers, and institutions. Technological developments in external beam RT and the integration of artificial intelligence into the system have significantly affected cancer treatment processes and led to the development of effective methods to reduce overall treatment times. The time savings achieved by these developed methods do not affect the accuracy of dose delivery. The current study determined the factors affecting the total treatment time and made recommendations to minimize such delays. In an age in which every second counts, it is essential to develop methods and programs that optimize the efficiency of RT.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
