Abstract
Aims
This review aims to synthesize the existing literature on palliative radiotherapy (RT) delivered in two daily fractions for patients with advanced cancer, focusing on its impact on symptom alleviation, treatment tolerance, and the implications for clinical practice and future research.
Methods
An international team conducted this narrative review, adhering to SANRA guidelines. Studies published in English on palliative RT delivered in two daily fractions were selected without date restrictions. The literature search, using a combination of specific key terms, led to a comprehensive examination of relevant studies. Data on study objectives, treatment approaches, palliative effectiveness, and toxicity were extracted and qualitatively analyzed.
Results
The review included 29 publications, showing consistent efficacy in symptom reduction (63.0%–100% palliative response rate) and general tolerability across various cancer types. These studies highlighted the potential radiobiological advantages and practicality of accelerated multi-fractionated regimens, which provide rapid tumor response with reduced late toxicity risks. Furthermore, the logistical benefits of such treatments, including shorter hospital stays and minimized travel requirements, were noted as particularly valuable during challenging times such as recent pandemics.
Conclusions
The evidence supports the integration of evidence-based, accelerated-hypofractionated RT into palliative care strategies, ensuring effective symptom management with minimal patient burden. Future research should focus on comparative studies on single versus multiple-cycle treatments, optimal intervals between treatment cycles, and the integration of advanced RT techniques.
Keywords
Introduction
Radiotherapy (RT) significantly impacts the relief of tumor-associated symptoms. As a result, palliative RT is frequently employed to manage symptoms in patients with advanced cancer.
In this context, preference is given to short treatment durations. In fact, the compromised clinical condition of these patients often precludes the feasibility of extended RT regimens. Additionally, the symptomatic relief achievable through relatively low RT doses, coupled with the minimal risk of toxicity, advocates for the adoption of accelerated treatments administered over a few sessions, known as accelerated-hypofractionated RT. This approach not only allows short treatment duration but is also theoretically linked to quick tumor responses, thereby potentially enhancing the speed and effectiveness of palliative outcomes.
Historically, in the 1980s, the Radiation Therapy Oncology Group (RTOG) conducted a phase II study involving twice-daily fractionation over 2 days, with each fraction being 3.7 Gy (resulting in 14.8 Gy per cycle). This regimen was repeated at 3–6 week intervals for a total of three cycles, delivering a cumulative RT dose of 44.4 Gy for pelvic malignancies, particularly tumors of the uterine cervix.1,2 Subsequently, this regimen, known as RTOG 8502 or the “QUAD shot,” was successfully adapted for palliative treatment of head and neck (H&N) cancers, showing promising results.3–5
These studies demonstrated the tolerability and symptomatic efficacy of such regimens, with doses around 14–15 Gy delivered twice a day (BID) over two days, with the possibility of repeating these treatments up to three times every 2–4 weeks.
Our investigation employed a similar approach (SHARON: SHort-course Accelerated radiatiON therapy), but delivered in a single cycle across various cancer types including multiple brain and complicated bone metastases, as well as advanced tumors of the H&N, thorax, and pelvis. Preliminary phase I trials validated the feasibility of delivering doses between 18–20 Gy BID over two days using 3D-conformal RT.6,7 Furthermore, subsequent phase II trials consistently reported response rates exceeding 80% across the aforementioned cancer scenarios,8,9 underscoring the regimen extensive applicability and efficacy.
Based on this background, and considering the lack of literature reviews on this topic, the aim of this report is to summarize and analyze the available evidence on palliative RT delivered BID over two days.
Materials and Methods
Design of the Literature Review
This narrative literature review was conducted by an international, multicenter and multidisciplinary team of experts, including radiation oncologists, medical physicists, and palliative care specialists, and was crafted in adherence to the Scale for the Assessment of Narrative Review Articles (SANRA) guidelines. 10 A structured narrative review checklist was employed to ensure an all-encompassing exploration of the subject matter (refer to Supplementary Table 1 for details).
Literature Search
For this review, we selected studies published in English, without any restriction on the publication date, focusing on palliative RT administered BID. On December 12th 2023, a search was performed on PubMed, utilizing a blend of key terms such as “Palliative Radiotherapy”, “Twice Daily”, “BID”, “two fractions per day”, “SHARON”, and “quad-shot”. Additionally, the snowball technique was employed to expand the search scope. It refers to the process of identifying additional relevant papers by examining the reference lists of initially selected articles from bibliographic databases.
Study Selection
The task of selecting relevant studies was independently undertaken by two authors (EG, CMD). The initial phase involved a screening of titles and abstracts to assess their pertinence to the application of the review topic. Articles deemed potentially relevant were then subjected to a thorough full-text review.
Extraction of Data
Authors LC and FCa independently performed the data extraction process. In case of discrepancies, the data extraction results were discussed with a third author (FCe). For every article included in the review, vital information such as the objectives of the study, therapeutic and evaluation approaches, outcomes concerning palliative effectiveness and toxicity, as well as key conclusions were gathered. This collated data was organized and summarized in Tables 1–4.
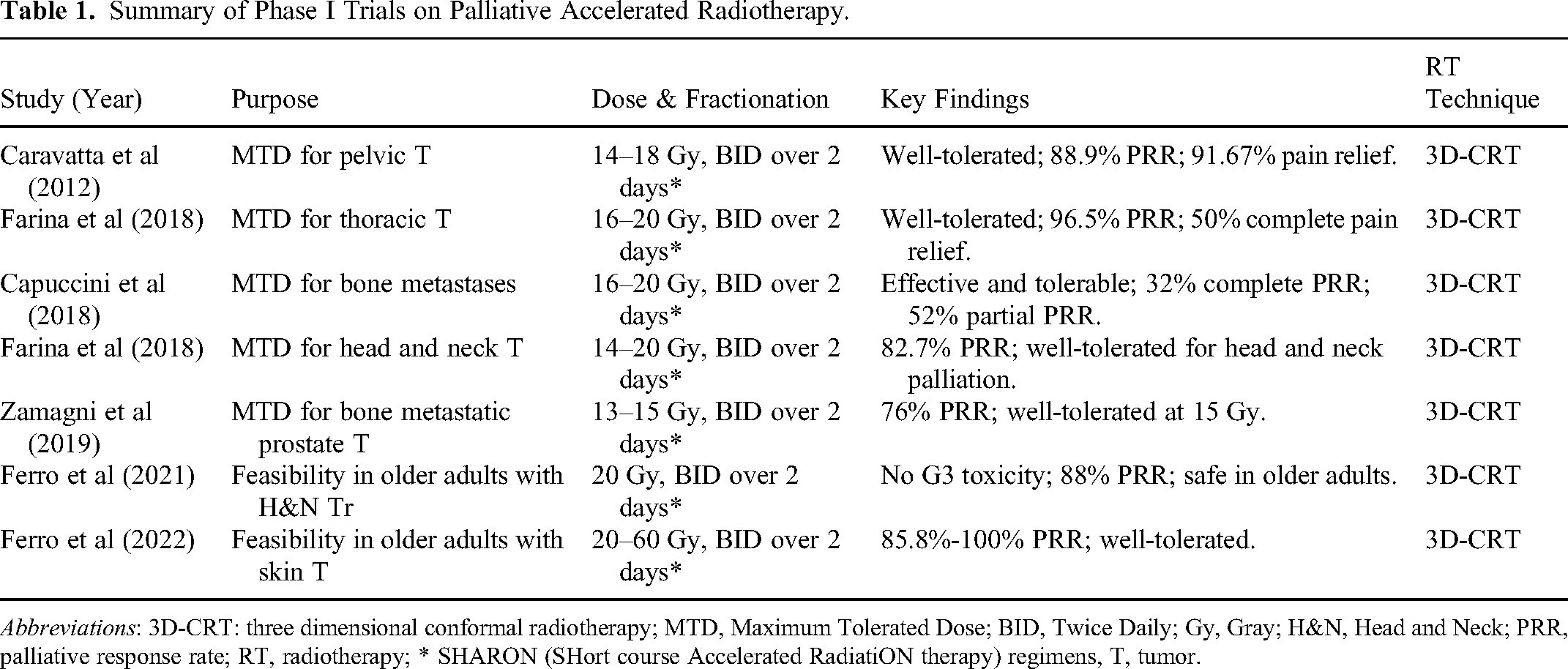
Summary of Phase I Trials on Palliative Accelerated Radiotherapy.
Summary of Phase I-II and Phase II Trials on Palliative Accelerated Radiotherapy.

Abbreviations: 2D-RT, two dimensional radiotherapy; 3D-CRT, three dimensional conformal radiotherapy; BID, Twice Daily; CR, Complete Response; CRR, clinical response rate; Gy, Gray; H&N, Head and Neck; MTD, Maximum Tolerated Dose; NR, not reported; PR, Partial Response; PRR, palliative response rat, Quad Shot, accelerated radiotherapy delivered in 4 fractions over two consecutive days; QoL, Quality of Life; RT, Radiotherapy; SHARON, SHort course Accelerated RadiatiON therapy, T, tumor.
Summary of Phase III Trials on Palliative Accelerated Radiotherapy.

Abbreviations: 2D-RT, two dimensional radiotherapy; BID, Twice Daily, Gy, Gray; H&N, Head and Neck; Quad Shot, accelerated radiotherapy delivered in 4 fractions over two consecutive days; QoL, Quality of Life; RT, Radiotherapy.
Summary of Other Studies on Palliative Accelerated Radiotherapy.

Abbreviations: 2D-RT, two dimensional radiotherapy; 3D-CRT, three dimensional conformal radiotherapy; BID, Twice Daily; CHT, Chemotherapy; CR, Complete Response; CRR, clinical response rate; Gy, Gray; H&N, Head and Neck; IMRT, Intensity-Modulated Radiotherapy; LPFS, Local Progression-Free Survival; LRRFS, Local Regional Recurrence-Free Survival; OS, Overall Survival; PFS, Progression-Free Survival; PR, Partial Response; PRR, palliative response rate; Quad Shot, accelerated radiotherapy delivered in 4 fractions over two consecutive days; QoL, Quality of Life; RR, response rate; RT, Radiotherapy, SHARON, SHort course Accelerated RadiatiON therapy; T, tumor; VMAT, Volumetric Modulated Arc Therapy.
Data Analysis
Given the diverse and limited nature of the available data in terms of methodologies and assessments, it was deemed inappropriate to conduct a statistical analysis. Instead, insights from the chosen articles were qualitatively articulated in a narrative style. This approach aimed to provide a thorough depiction of the current knowledge on the subject.
Results
Search Results
Our search yielded a total of 55 records, with 38 sourced directly from database queries and an additional 17 obtained through supplementary methods. Upon screening and application of our predefined selection criteria, 26 abstracts were deemed irrelevant and subsequently excluded from further consideration. Consequently, this resulted in a refined compendium of 29 publications, which were incorporated into our comprehensive analysis. Various radiotherapy techniques were used in the analyzed studies, including 2D radiotherapy, 3D-conformal radiotherapy, intensity-modulated radiotherapy, volumetric modulated arc radiotherapy, and proton therapy.
Phase I and Feasibility Trials
The phase I trials6,7,9,11–14 primarily focused on establishing the Maximum Tolerated Dose (MTD) of short-course BID RT regimens for palliative treatment in various types of advanced cancers, including pelvic, 6 thoracic, 11 bone metastases,9–12 and H&N cancers. 13 The methods varied across the trials, with some employing a single course of RT6,7,9,11,12 while others used multiple cycles.13,14 In several studies, escalating dose increments were tested, with total doses generally ranging between 14 and 20 Gy BID.6,7,9,11–13 The results consistently demonstrated that these regimens were well-tolerated, with doses up to 20 Gy being safe6,9,13 and associated with significant symptomatic response rates (76%-100%).6,7,9,11–13 In particular, the safety of the treatment is maintained even when the same fractionation is repeated a second time, as demonstrated in elderly patients. 13 Therefore, the effectiveness of these regimens in symptom control and improving patients’ quality of life (QoL) was highlighted, with minimal reports of acute and late toxicities (Table 1). Overall, these phase I trials were instrumental in defining safe and effective RT dosages for accelerated palliative RT in advanced cancers, allowing further research and optimization of treatment protocols.
Phase I-II and Phase II Trials
The phase I-II and II trials2,3,7–9,11,15–18 were primarily aimed at evaluating the efficacy and safety of various hypofractionated RT regimens for palliative treatment across different cancer types, including advanced pelvic,2,8 H&N3,7,15,18 and thoracic tumors,11,17 and brain 16 and bone metastases. 9 The studies explored the therapeutic benefits and potential toxicities of these regimens, often following promising results from phase I trials.7,9,11 The trials involved short-course RT, delivering total doses ranging from 12 to 44.4 Gy. The treatments were fractionated BID over two days.2,3,7–9,11,15–17 Results across these studies consistently demonstrated notable symptomatic response rates, ranging from 63.0% to 96.5%.7,8,9,11,15,16 Symptomatic improvement and palliation of presenting symptoms, such as pain and bleeding, translated in improved QoL in the studies reporting this end-point.3,8 The regimens were generally well-tolerated. In fact, most studies reported minimal acute toxicities,15,16,17 and the incidences of late toxicities were also low, 2 indicating the feasibility of these regimens for patients with advanced-stage cancers (Table 2). Furthermore, some studies showed a prolonged palliative benefit, with effective symptom control lasting beyond six months.8,16 This underscores the potential of these regimens to provide not only immediate relief but also sustained improvement over time. Overall, the phase II trials reinforced the findings from earlier studies, demonstrating that hypofractionated RT regimens can offer effective palliation with manageable toxicity profiles in various cancer types.
Phase III Trials
The phase III trials1,19–21 focused on assessing the comparative effectiveness and safety of established RT regimens for palliative treatment in patients with advanced pelvic1,19 and H&N cancers.20,21 These studies aimed to refine treatment protocols by evaluating different fractionation schedules and introducing concurrent chemotherapy (CHT) in certain cases (Table 3).
Spanos et al 19 aimed to assess the impact of rest intervals on tumor and normal tissue response in advanced pelvic malignancies through a phase III trial involving 136 patients. The study compared 2-week versus 4-week rest intervals, among 2 days courses of RT delivered BID, finding increased acute toxicity in the shorter rest interval group without significant differences in late toxicity or tumor response. Moreover, a higher completion rate and 12-month survival for patients with a Karnowski Performance Status ≥ 80 were noted in the 2-week group.
In a similar comparison, Spanos et al 1 evaluated the incidence of late complications from pelvic palliation using accelerated BID RT, analyzing 290 cases for late effects. The findings indicated a 6% late toxicity rate, with no significant difference between 2-week and 4-week rests between RT courses, with overall good tumor regression and symptom palliation and relatively low incidence of complications.
Soni A et al 20 compared the efficacy, tolerability, and toxicity of three palliative RT schedules in advanced H&N cancer. The Quad Shot schedule (accelerated rRT delivered in 4 fractions over two consecutive days) demonstrated better local control and acceptable toxicity, showing an improved QoL post-RT, positioning it as a favorable option in the Indian setting over conventional schedules (50 Gy in 16 fractions or 25 Gy in 5 fractions).
Lastly, Choudhary A et al 21 compared two hypofractionated palliative RT regimens for H&N cancers, finding similar symptom relief and overall response rates between conventional (30 Gy in 10 fractions) and Quad Shot regimens. The Quad Shot regimen was noted for its minimal toxicity and good symptom relief, making it an effective alternative to the conventional regimen. In conclusion, the phase III trials confirmed the efficacy and safety of hypofractionated BID RT regimens.
Other Studies
The studies in Table 44,5,22–30 primarily investigated the palliative effects of various RT regimens, delivered BID, focusing on advanced H&N,4,5,22–29 pelvic 22 and skin cancers, 29 and sarcomas. 28 These studies aimed to define optimal fractionation schemes and the potential benefits of integrating radiosensitizing agents to enhance the palliative outcomes. Results across these studies confirmed high response rates, with a significant proportion of patients experiencing complete or partial symptoms’ remission (65%-100%). In fact symptomatic improvement was a consistent finding, with many patients reporting substantial relief from distressing symptoms, which notably enhanced their QoL.4,5,22–28 Some studies explored the addition of radiosensitizing CHT to the RT regimen. This combination was generally well-tolerated and led to an enhanced tumor response (85.7%-94.7%)22,28 and symptom palliation (89.5%), 22 confirming a synergistic effect between CHT and RT also in the palliative setting.22,28 The safety profile of these regimens was favorable, with most studies reporting minimal acute and late toxicities.4,22,23,27,29 Notably, some studies confirmed the durable nature of the palliative benefit, with effective symptom control extending over significant periods.22,27
Discussion
Narrative
Our review analyzed a range of studies, generally identified by the equivalent terms Quad Shot or SHARON, on short-course hypofractionated palliative BID RT for patients with advanced cancer. Our goal was to understand if these treatments can really and quickly relieve symptoms without significant toxicity. The findings from our review consistently showed that these RT regimens were effective in reducing symptoms and were generally well-tolerated by patients.
However, it is important to note that while toxicity rates were uniformly low in the studies analyzed, 2D treatments were only used with doses ≤ 14.8 Gy (typically the Quad Shot regimen). In contrast, treatments with doses of 18–20 Gy were tested exclusively using the 3D technique (typically the SHARON regimen). Therefore, the tolerability of higher dose regimens delivered with the 2D technique cannot be considered demonstrated.
The treatment approach explored in these studies presents distinct radiobiological benefits. The adoption of a highly accelerated regimen is designed to prompt a swift and robust tumor response, leading to early symptom alleviation. Concurrently, conducting two sessions daily enables a considerable reduction in the dose per fraction, potentially reducing the risk of late toxicity. Although the treatment brief duration theoretically increases the risk of acute toxicity, particularly in instances where a total dose of 20 Gy was administered over two days, the findings, especially from the phase I studies, suggest good tolerance to this regimen, even among elderly patients.13,14,27 This is likely due to the delivery of palliative RT based on conformal techniques.7,8,9,11,16,17
Moreover, the logistical advantages of these accelerated RT protocols are pronounced. They offer significant convenience for hospitalized patients by potentially shortening their stay. This approach is also advantageous for patients requiring subsequent CHT, as it prevents delays in systemic therapy. Additionally, it benefits those residing far from treatment centers by minimizing the need for frequent travels. The utility of these regimens extends to complex logistical scenarios, as highlighted during the pandemic years of 2020–2022, 31 indicating their adaptability and relevance in various healthcare settings.
Ultimately, this treatment approach could offer significant benefits in low-resource environments, commonly seen in low- middle-income countries, where limited access to RT equipment often hinders patient care. By condensing the conventional 5–10 fraction treatment spread over 1–2 weeks into a short schedule of just 4 fractions delivered in two days, the workload of RT machines can be substantially decreased. This reduction in machine time can lead to shorter waiting lists, enabling more patients to receive timely RT.
Future research is essential to address certain unresolved questions in the field of palliative RT. In particular, forthcoming studies could focus on:
Assessing the potential advantages of single-cycle treatments, which involve higher doses per fraction, in comparison to multi-cycle treatments that employ lower doses per fraction. This analysis would provide insight into the efficacy and safety of different dosing strategies. Determining the optimal interval between courses in multi-cycle treatments to maximize therapeutic benefits while minimizing potential risks. Exploring the integration of these regimens with spatially fractionated RT techniques, which could potentially enhance treatment outcomes through more targeted dose distribution.32,33 Investigating the feasibility and potential advantages of administering doses higher than those evaluated in our reviewed studies. This would involve utilizing modern RT techniques, such as volumetric modulated arc therapy (VMAT), to deliver precise and intensified treatment doses safely.
Such studies would significantly contribute to refining palliative RT approaches, ensuring more effective and tailored treatment options for patients with advanced cancers.
Our study is not without limitations. The intrinsic nature of a narrative review, while providing a broad overview, may not capture the details of a systematic review or meta-analysis. Additionally, the variability in study designs, treatment protocols, and patient populations across the reviewed articles introduces a level of heterogeneity that may affect the generalizability of the findings. The absence of a quantitative synthesis, due to the diverse and limited nature of the data, means that our conclusions are primarily qualitative and interpretative.
Furthermore, while the review benefits from the inclusion of a range of studies, it is essential to acknowledge the possibility of publication bias, where studies with positive outcomes are more likely to be published than those with negative or inconclusive results. This factor could potentially influence the overall interpretation of the efficacy and tolerability of the treatment regimens.
Summary
This narrative review synthesizes a breadth of studies on palliative RT delivered BID, providing valuable insights for health professionals, policymakers, and researchers. For clinicians, the findings underscore the potential of accelerated-hypofractionated RT to offer effective symptom relief with a favorable safety profile, particularly beneficial for patients with advanced cancers who may not tolerate prolonged treatment courses. The review highlights the significance of personalized treatment approaches, considering the patient clinical condition, the tumor radiobiological characteristics, and logistical factors such as hospital stays and travel requirements.
For policymakers and healthcare administrators, the review underlines the importance of integrating advanced RT techniques into palliative care protocols. It advocates for the adoption of evidence-based practices that not only optimize patient care but also streamline healthcare resources. The clear logistical advantages of these RT regimens, including shorter hospital stays and reduced treatment burdens, align with the goals of efficient healthcare delivery and patient-centered care, especially pertinent in the context of strained healthcare systems or during crises like the recent pandemic.
From a research perspective, the review identifies relevant areas for future investigation, including the comparative efficacy of single versus multiple-cycle treatments, optimal intervals between treatment cycles, and the integration of these regimens with advanced RT techniques like spatially fractionated RT or volumetric modulated arc therapy. Addressing these research questions will refine our understanding of palliative RT, favoring protocols optimization and the development of tailored treatment strategies.
Conclusion
While our study provides valuable insights into the current state of palliative RT delivered BID, future research, ideally in the form of well-designed clinical trials, is needed to substantiate these findings and address the gaps identified in this review. Such studies will be useful in improving treatment protocols and ultimately enhancing the quality of palliative care for patients with advanced cancers.
Footnotes
Abbreviations
Authors Contributions
SC and AGM had the idea for the article; EG, CMD, LC, FCa and FCa performed the literature search and data collection and analysis; EG, FCe, RR, GM, FD, and MB drafted the manuscript; all authors critically revised the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
