Abstract
Keywords
Introduction
Mantle cell lymphoma (MCL) is an uncommon, uncurable B-cell non-Hodgkin lymphoma.1,2 According to its different morphological characteristics, MCL can be categorized into different subgroups, such as blastoid, pleomorphic, and classical MCL, and the former 2 subgroups have worse survival outcomes than the last subgroup. 3 Despite recent therapeutic advances in MCL, most treatment strategies, including chimeric antigen receptor T cell (CAR-T) therapy and bruton tyrosine kinase (BTK) inhibitors, provide limited durable response and survival due to drug resistance.4–7 After the failure of the management of BTKi and CAR-T, MCL patients have dismal survival with limited treatment options.8–10 Bispecific antibodies (also known as bispecific T-cell engagers, BiTE), including glofitamab, blinatumomab, and mosunetuzumab are being investigated in MCL. BiTEs result in attractive clinical responses even in CART-exposed lymphoma patients, but up to 30% of lymphoma patients do not respond to this treatment. Moreover, its cytokine release syndrome (CRS) and neurotoxicity are needed to be carefully monitored while using these agents, especially in elder patients.11–13 All these observations indicate an unmet medical need for novel therapeutic regimens in the field of MCL.
BCL2, an important antiapoptotic or prosurvival protein, is overexpressed in MCL via diverse mechanisms, including BCL2 gene amplification and protein degradation inability.14,15 Venetoclax is an oral selective BCL2 inhibitor that exerts its cytotoxicity by blocking the antiapoptotic function of BCL2 through its inhibitory interaction with proapoptotic BAX and BAK1 proteins.16,17 Treatment of MCL patients with venetoclax leads to promising results even in post-BTKi cases; therefore, this agent has been approved to treat patients with MCL who become resistant to BTKis. 18 However, resistance to venetoclax monotherapy occurs frequently after ibrutinib treatment in patients with MCL, suggesting the requirement of venetoclax-based combinational regimens.
XPO1, also termed CRM1, is a well-established nuclear exporter responsible for the transportation of many tumor suppressors and oncoproteins between the cytoplasm and nucleus. 19 It is crucial for the maintenance of cellular homeostasis. 20 Overexpression of XPO1 is observed in patients with MCL and is associated with high aggressiveness and worse clinical outcomes. 21 Therefore, XPO1 might be an excellent therapeutic target for the treatment of MCL. The XPO1 inhibitor selinexor has shown moderate activity in patients with MCL with tolerable safety profiles in early clinical trials. 22 In this study, we sought to investigate the therapeutic interaction of selinexor and venetoclax in MCL models and to elucidate the potential mechanism of both drugs in MCL.
Methods and Materials
Cell Culture and Reagents
Z138, a blastoid MCL cell line, and JEKO1, a classical MCL cell line, were purchased from Guidechem Biotechnology Company (Shanghai, China). REC1, another blastoid MCL cell line, was purchased from Anweisci Biotechnology Company (Shanghai, China). Z138, JEKO1, and REC1 cells were cultured in RPMI-1640 medium (Gibco, Life Technologies, NY, USA) supplemented with 10% fetal bovine serum (FBS; Gibco, USA) with 100 units/mL penicillin and 100 μg/mL streptomycin in a 5% CO2 incubator. Selinexor was obtained from Selleck (Shanghai, China), and venetoclax was purchased from MCE (Shanghai, China).
Cell Viability Assay
Cell viability was tested with Cell Counting Kit-8 (CCK-8, MCE, Shanghai, China). Z138, JEKO1, and REC1 cells (2-5 × 104 cells/well) were separately treated with selinexor and/or venetoclax, alone or in combination, for 72 h, followed by the addition and incubation of the CCK8 reagent (10 μL/well) for an additional 2 to 4 h. Subsequently, absorbance at 450 nm was assessed with a Bio-Rad microplate reader (Bio-Rad, CA, USA). Cell viability results were performed at least 3 independent times in triplicate. The IC50 values of selinexor in MCL cells were calculated by GraphPad Prism 8 software.
Cell Apoptosis Assay
An Annexin V/PI dual-staining kit was employed to detect the apoptosis of MCL cells treated with selinexor alone or in combination with venetoclax. Briefly, Z138, JEKO1, and REC1 cells (5 × 105 cells/well) were exposed to the indicated concentrations of selinexor in the absence or presence of venetoclax for 48 h and then stained with Annexin V and PI dye for 15 min in the dark. Finally, the Annexin V/PI-stained MCL cells were analyzed with a flow cytometer (BD, USA). Statistical differences were analyzed by analysis of variance (ANOVA).
Western Blot Analysis
Z138 and JEKO1 cells were treated with selinexor and venetoclax alone or in combination for 12 h. Protein was extracted from each treatment group using protein lysis buffer containing protease and phosphatase inhibitors. Subsequently, the cell lysates were subjected to SDS–PAGE on a 12% gel, followed by transfer onto PVDF membranes. Next, designated proteins were probed with specific primary antibodies and HRP-conjugated secondary antibodies (1:10 000, Cell Signaling Technology). Proteins were visualized using an ECL system (Amersham, Little Chalfont, UK). Antibodies against PARP (#9532S, 1:1000), BCL-XL (#2764S, 1:1000), MCL1 (#39224S, 1:1000), BCL2 (#15071S, 1:1000), NOXA (#14766S, 1:1000), BAX (#41162S, 1:1000), γ-H2AX (#80312S, 1:1000), Rad51 (#8875S, 1:1000), p-WEE1 (#4910S, 1:1000), WEE1 (#13084S, 1:1000), p-CHK1 (#2348S, 1:1000), CHK1 (#37010S, 1:1000), p-ATR (#2853S, 1:1000), ATR (#13934S, 1:1000), cleaved-caspase 3 (#9664S, 1:1000), and β-actin (#3700S, 1:1000) were obtained from Cell Signaling Technology (Danvers, MA, USA).
Statistical Analysis
Data are shown as the mean ± standard error of the mean (SEM). All significant differences were analyzed with GraphPad Prism 8.0 software. Two-tailed Student's t-test was used to assess differences between 2 groups, while one-way ANOVA followed by the Brown-Forsythe and Welch ANOVA test was used for comparisons among multiple groups. P < .05 was considered statistically significant.
Results
Selinexor is Cytotoxic in Both Classical and Blastoid Subtypes of MCL Cells
According to the morphological features of tumor cells, MCL can be classified into the classical and blastoid subtypes, and the latter has a worse prognosis than the former. In this study, selinexor showed dose-dependent suppression of cell viability in blastoid Z138 and REC1 cells as well as classical JEKO1 cells (Figure 1A and B). After treatment with selinexor for 72 h, the IC50 values were 78.23, 44.06, and 239.2 nmol/L for Z138, REC1, and JEKO1 cells, respectively. Based on the results of cell viability assays, a series of selinexor concentrations were selected to assess their proapoptotic activity in all 3 tested MCL cell lines. In agreement with the cell viability results, selinexor induced apoptosis in Z138 cells in a dose-dependent manner (Figure 1C to F). As shown in Figure 1C, the lowest dose of selinexor (62.5 nmol/L) led to an apoptosis rate of 25.56% in Z138 cells, and the apoptosis rate reached 58.48% at the highest dose (500 nmol/L). Similarly, dose-dependent proapoptotic activity of selinexor was observed in JEKO1 and REC1 cells (Figure 1D and F). In summary, the XPO1 inhibitor selinexor has the ability to decrease cell viability and increase cell apoptosis in both blastoid and classic MCL cells.

Selinexor potently kills classical and blastoid subtypes of mantle cell lymphoma (MCL) cells. (A, B) Z138, JEKO1, and REC1 cells were exposed to designated concentrations of selinexor for 72 h, and then selinexor-treated cells were assessed by CCK-8 assay to investigate the cytotoxicity of selinexor on MCL cells. (C to E) After treatment with selinexor for 48 h, cell apoptosis was investigated with the Annexin V/PI assay (C) in Z138, (D) JEKO1, and (E) REC1 cells. (F) Flow cytometry plots of MCL cell apoptosis obtained from (C to E). **, ***, and **** indicate P < .01, P < .001, and P < .0001, respectively. All experiments were performed in MCL cells in 3 times with triplicates.
Selinexor and Venetoclax Synergize to Lower Cell Viability and Induce Cell Apoptosis in MCL Cells, Regardless of Their Morphologic Characteristics
Venetoclax, a well-tolerated BCL2 antagonist, has demonstrated promising antitumor efficacy in clinical trials and has been approved as an effective treatment strategy for patients with recurrent MCL.18,23,24 We attempted to investigate the therapeutic interaction of selinexor in combination with venetoclax in blastoid and classic MCL cells. Fortunately, we found that selinexor and venetoclax cooperated with each other to lower cell viability in JEKO1, Z138, and REC1 cells (Figure 2A to C). The drug combination index (CI) was calculated using CompuSyn software based on the cell viability results. CI < 1 indicates synergy, =1 indicates addition, and <1 indicates antagonism. The CI values demonstrated that the combination of selinexor and venetoclax exerted synergistic effects on JEKO1, Z138, and REC1 cells (Figure 2D to F).

Selinexor and venetoclax synergize to decrease the viability of classical and blastoid MCL cells. After treatment with selinexor and venetoclax alone or in combination for 72 h, Z138, JEKO1, and REC1 cells were assessed with the CCK-8 reagent. (A to C) The combination of selinexor and venetoclax more strongly decreased the viability of (A) JEKO1, (B) Z138, and (C) REC1 cells. (D to F) CI values indicated that the combination of selinexor and venetoclax exerted synergistic viability-suppression effects on (D) JEKO1, (E) Z138, and (F) REC1 cells. The CI was calculated with CompuSyn software based on the cell viability results. CI < 1 indicates synergy, =1 indicates addition, and <1 indicates antagonism. The cell viabilities were performed in all MCL cells in 3 times with triplicates. The blue asterisks represent the statistical significance of the comparison between the combined treatment group and the selinexor group. The red asterisks represent the statistical significance of the comparison between the combined treatment group and the selinexor group. *, **, ***, and **** indicate P < .05 P < .01, P < .001, and P < .0001, respectively.
Next, we sought to investigate whether the 2 drugs have synergistic proapoptotic effects on MCL cells. Indeed, we observed that selinexor synergized with venetoclax to promote apoptosis in JEKO1, Z138, and REC1 cells (Figure 3A to C and G). The apoptotic cell proportions were 12.33% and 11.68% in JEKO1 cells treated with 125 nM selinexor and 12.5 nM venetoclax for 48 h, respectively, while the combination of the 2 drugs led to an apoptosis rate of 35.33% of JEKO1 cells (Figure 3A). A higher dose combination of selinexor (500 nM) and venetoclax (50 nM) led to an apoptosis rate of 57.77% of JEKO1 cells, while the rates were 16.73% and 21.1% with selinexor and venetoclax alone, respectively. CI values <1 in the 3 combined groups indicated that selinexor in combination with venetoclax had synergistic effects in promoting cell death in JEKO1 cells (Figure 3D). As expected, analog findings regarding the synergistic apoptosis-inducing activity of selinexor combined with venetoclax were noted in Z138 and REC1 cells (Figure 3E to G). Collectively, these results indicate that selinexor and venetoclax exert synergistic cell-killing activity in both blastoid and classic MCL cells.

Selinexor synergizes with venetoclax to promote apoptosis in MCL cells. (A) JEKO1, (B) Z138, and (C) REC1 cells were treated with selinexor and venetoclax alone or in combination for 48 h, followed by cell apoptosis detection with the Annexin V/PI assay. (D to F) CI values indicated that selinexor synergized with venetoclax to promote apoptosis in (D) JEKO1, (E) Z138, and (F) REC1 cells. (G) Flow cytometry plots of cell apoptosis of Z138, JEKO1, and REC1 cells treated with selinexor and venetoclax alone or in combination. *, ***, and **** indicate P < .05, P < .001 and P < .0001, respectively. The cell apoptosis assays were analyzed with the annexin V/PI kit in all MCL cells in 3 times with triplicates.
Dysregulation of DNA Damage and Repair Signaling Contributes to the Therapeutic Synergy of Selinexor and Venetoclax in MCL Cells
Next, we sought to investigate the potential mechanism of action for the synergy of selinexor in combination with venetoclax in MCL cells. In comparison to each single drug, the combined regimen had stronger power to induce apoptosis in Z138 and JEKO1 cells, as evidenced by higher levels of cleaved PARP and cleaved caspase 3, which are generally considered apoptotic biomarkers (Figure 4A and B). In Z138 cells, venetoclax treatment for 12 h induced an increase in MCL1 expression, while selinexor in combination with venetoclax decreased MCL1 expression (Figure 4A). In JEKO1 cells, either selinexor or venetoclax treatment led to MCL1 downregulation, and the downregulation effect was even stronger with the combination of the 2 drugs (Figure 4B). The expression levels of 2 other well-known BCL2 family antiapoptotic proteins, BCL2 and BCL-XL, were not affected by selinexor, venetoclax, or their combination after 12 h of treatment. Selinexor combined with venetoclax increased the expression of BAX protein, an important proapoptotic BCL2 family member, in MCL cells. However, selinexor and venetoclax alone or in combination did not impact the expression level of NOXA, another critical proapoptotic protein in Z138 (Figure 4A) and JEKO1 cells (Figure 4B).
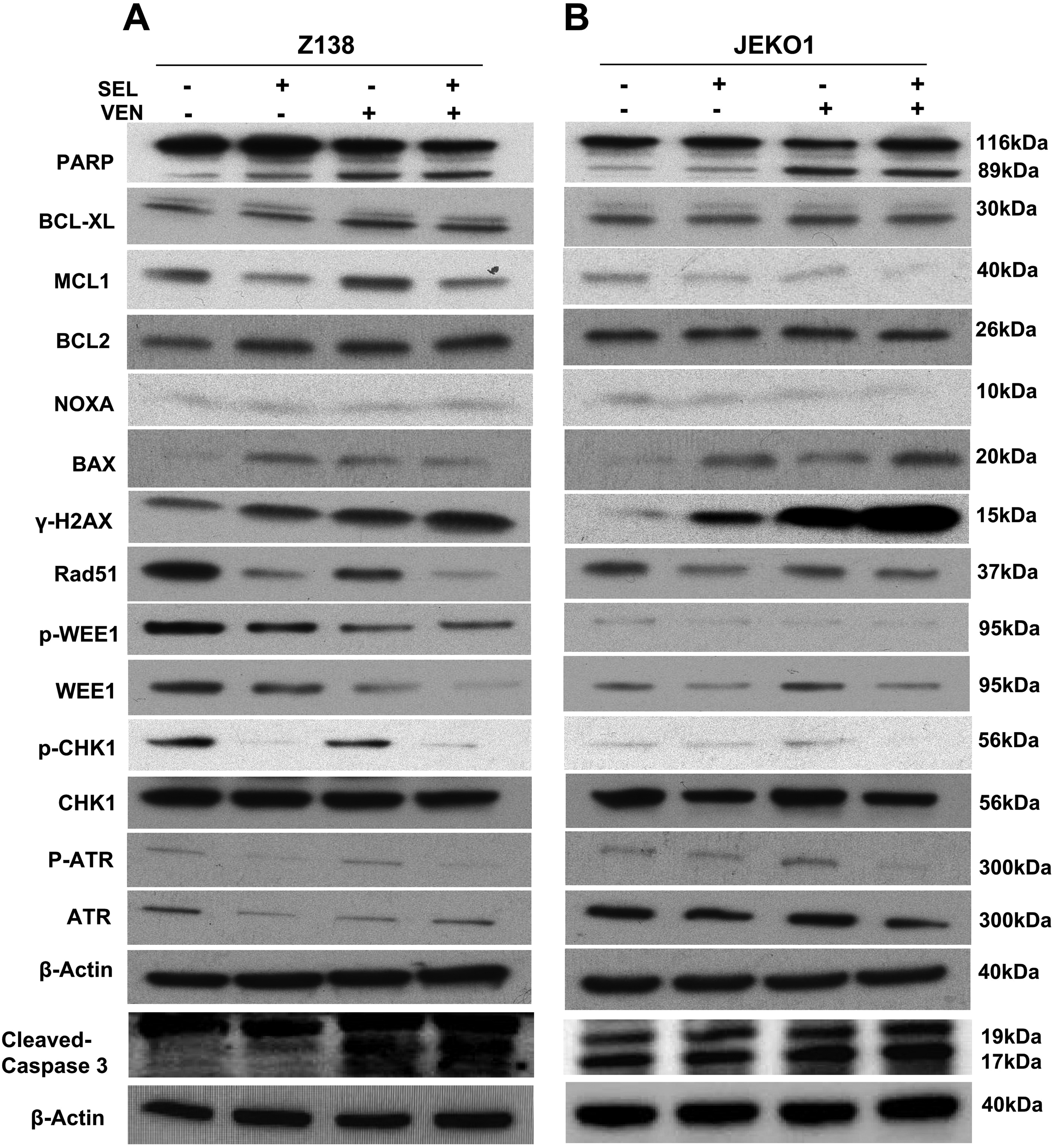
Dysregulation of DNA damage and repair signaling contributes to the therapeutic synergy of selinexor and venetoclax in MCL cells. Both (A) Z138 and (B) JEKO1 cells were exposed to selinexor (62.5 nM for Z138 and 125 nM for JEKO1) and venetoclax (6.25 nM for Z138 and 12.5 nM for JEKO1) alone or in combination for 12 h. Drug-treated MCL cells were lysed and subsequently probed with the indicated primary antibodies, including PARP, cleaved caspase 3, BCL-XL, MCL1, BCL2, NOXA, BAX, γ-H2AX, Rad51, p-WEE1, WEE1, p-CHK1, CHK1, p-ATR, and ATR. β-Actin was the loading control.
Previous studies have demonstrated that selinexor and venetoclax can influence DNA damage and repair pathways. Therefore, we sought to confirm whether treatment with both drugs could modulate the DNA damage and repair pathways to exert a synergistic effect in MCL. In this study, selinexor and venetoclax alone had the ability to induce the expression of phosphorylated H2AX (p-H2AX), also known γ-H2AX, which is an indicator of DNA damage, in Z138 and JEKO1 cells (Figure 4A and B). Moreover, the level of p-H2AX was further increased with the combined treatment of selinexor and venetoclax in Z138 and JEKO1 cells (Figure 4A). The expression of Rad51, a DNA repair protein, was decreased by selinexor and venetoclax alone, while its expression was further downregulated in the combined therapy group. Correspondingly, we noted that selinexor together with venetoclax had a greater capability to lower the levels of phosphorylated WEE1 (p-WEE1), WEE1, phosphorylated CHK1 (p-CHK1) and phosphorylated ATR (p-ATR) than selinexor and venetoclax alone in the 2 MCL cell lines (Figure 4A and B). Altogether, our findings reveal that perturbation of DNA repair processes and induction of DNA damage are involved in the therapeutic synergistic effects of selinexor and venetoclax in MCL1 cells.
Discussion
MCL remains incurable in spite of the revolutionary advancements in targeted therapy in recent years, necessitating new therapeutic strategies.1,2,25 In the current study, we demonstrated that the XPO1 antagonist selinexor, a promising antitumor drug with completely different mechanisms of action, showed significant cytotoxicity against blastoid and classical MCL cells in a dose-dependent manner. Our results were consistent with the findings of previous studies reported by Abeykoon et al 26 and Ming et al, 27 which together validate the antilymphoma potential of selinexor in MCL cells irrespective of its morphological subtypes. BCL2 abnormalities, including BCL2 protein overexpression and BCL2 gene amplification, are observed in patients with MCL. The BCL2 selective inhibitor venetoclax results in an impressive treatment response and improves prognosis in MCL patients and thereby has been licensed to be administered in this malignant disease.23,24 In this study, we found that selinexor and venetoclax synergistically decreased cell viability and promoted apoptosis in both blastoid and classical MCL cells. Given the efficacies, accessibilities, and acceptable safety profiles of venetoclax and selinexor, the combined regimen of both drugs might be a potentially promising approach in the management of patients with MCL. However, further patient-derived xenograft studies and clinical trials are mandatorily required to substantiate the conclusion.
The BCL2 family is composed of antiapoptotic and proapoptotic subfamilies, which have pivotal functions in governing cellular homeostasis.28,29 The apoptotic response of cells is mainly controlled by the relatively dynamic balance of pro- and antiapoptotic BCL2 proteins. The antiapoptotic components of BCL2, including BCL2, BCL-XL, and MCL1, play a key role in maintaining cell survival. NOXA and BAX proteins belong to the BCL2 proapoptotic family, and their activation targets the balance to the apoptotic side and then initiates cell apoptotic cascades. Previous studies have reported that upregulation of MCL1 contributes to BCL2 inhibitor resistance, whereas blocking MCL1 activity could rescue the antitumor sensitivity of BCL2 inhibitors.30,31 Selinexor has been confirmed to have the ability to reduce the expression of MCL1 in multiple tumor cells.32,33 Consistent with prior observations, selinexor decreased the expression level of MCL1 in blastoid and classical MCL cells. More notably, the downregulation of MCL1 was potentiated by the combination of venetoclax and selinexor. However, neither a single drug nor the combined regimen impacted the expression of BCL2 and BCL-XL. With regard to the apoptotic proteins, BAX rather than NOXA was upregulated by the combination of selinexor and venetoclax in MCL cells.
DNA damage occurs invariably in organisms and requires the DNA damage response (DDR) to repair or delete damaged DNA to prevent tumorigenesis.34,35 A DNA double-strand break (DSB) is a catastrophic DNA damage type and often leads to cell death. 36 In response to DSB lesions, histone H2A (H2AX) is immediately phosphorylated at its Ser 19 locus; therefore, H2AX phosphorylation (so-called γ-H2AX) is regarded as a biomarker of DNA damage. 37 In this study, selinexor and venetoclax monotherapy increased the level of γ-H2AX protein in blastoid and classical MCL cells, indicating that both drugs alone exert their cytotoxicity via induction of DNA damage. This observation was consistent with previous reports evaluating selinexor and venetoclax antitumor activity in other malignant diseases.38,39 More importantly, we found that the combined treatment of the 2 drugs led to the accumulation of more γ-H2AX protein than each single drug treatment, suggesting that inducing DNA damage is involved in the synergistic effect of selinexor and venetoclax on MCL cells.
The efficient repair of DSBs is essential for the maintenance of genome stability.34,35 Several DDR pathways have been developed to combat DNA damage in mammalian cells. The Rad51 protein is an important recombinase that plays a crucial role in DNA damage repair. 40 Overexpression of Rad51 is frequently observed in many malignancies and is associated with poor clinical outcomes.41,42 Our results indicated that selinexor and venetoclax alone have the capability to lower the level of Rad51 protein, and the Rad51 downregulation ability was further potentiated with the combined regimen. ATR is a main DDR modulator that is activated in response to DNA damage and phosphorylates its downstream targets, including CHK1 and WEE1.43,44 Numerous data have shown that ATR, CHK1, and WEE1 are overexpressed in multiple tumor types and positively correlate with worse prognosis, indicating that these DDR regulators are excellent antitumor targets. More interestingly, the blockade of ATR, CHK1, and WEE1 with their selective inhibitors has resulted in promising antitumoral efficacies.45–47 In this study, our findings revealed that selinexor synergized with venetoclax to inactivate the ATR–CHK1–WEE1 signaling cascade.
In conclusion, this study demonstrated that blastoid and classical MCL cells were sensitive to selinexor cytotoxicity in preclinical models. Selinexor synergized with venetoclax, an approved BCL2 inhibitor for the management of patients with MCL, to suppress cell viability and to induce apoptosis in both blastoid and classical MCL cells. In contrast to selinexor or venetoclax alone, the combined regimen more potently decreased MCL1 expression and increased BAX levels in MCL1 cells. Mechanistically, induction of DNA damage and inactivation of the DDR contributed to the synergy of selinexor in combination with venetoclax in MCL cells. Our results might provide a novel treatment strategy involving the combination of selinexor with venetoclax for the treatment of MCL. Because all results were obtained from in vitro models, additional in vivo and clinical studies are mandatory to draw a definitive conclusion.
Footnotes
Acknowledgments
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, Translational Research Grant of NCRCH (grant numbers 82260030 and 2021WWA02).
Ethical Approval
Our study did not require ethical board approval because it did not contain human or animal trials.
