Abstract
AHNAK nucleoprotein 2 (AHNAK2) has been emerged as a crucial protein for neuroblast differentiation and cell migration, thereby involving in the development of various cancers. However, the specific molecular mechanism of AHNAK2 in lung adenocarcinoma is inconclusive. By accessing to the Oncomine dataset and GEPIA website, a higher expression level of AHNAK2 was observed in lung adenocarcinoma tissue samples. Overall survival (OS) curve plotted by Kaplan-Meier method showed that up-regulation of AHNAK2 was related with poor prognosis of lung adenocarcinoma patients. Quantitative reverse transcription polymerase chain reaction (qRT-PCR) analysis and western blot were conducted to examine the expression level of genes in lung adenocarcinoma cells. Through functional in vitro experiments, cell proliferation, migration and invasion were all suppressed after AHNAK2 knockdown using Cell counting kit-8 (CCK-8) assay, wound-healing and transwell analysis. Reduction of AHNAK2 decreased the apoptosis rate using flow cytometry analysis. Moreover, the key markers of MAPK pathway, p-MEK, p-ERK and p-P90RSK were decreased due to the transfection of si-AHNAK2 in A549 cells. U0126, a MEK inhibitor, showed the similar effects on MAPK-related protein levels with si-AHNAK2. To sum up, AHNAK2 is significantly increased in lung adenocarcinoma and plays a carcinogenic role by activating the MAPK signaling pathway, providing a novel insight and raising possibility for lung adenocarcinoma treatment.
Instruction
Lung cancer is the most common type of respiratory system cancer and is notorious for its dismal prognosis, with a 5-year overall survival rate under 15%, especially in China. 1,2 A total of 85% of lung cancer cases are diagnosed as lung adenocarcinoma, 3 a subtype of non-small cell lung cancers (NSCLC), 4,5 and its high mortality shows a major public health challenge. 6 Despite great advances of treatment have been made, prognosis of lung adenocarcinoma patients remain still pessimistic. 7 Thus, expounding full understanding of molecular mechanism related with lung adenocarcinoma and identifying novel targets will be essential to improve the treatment of lung adenocarcinoma.
AHNAKs are well known as desmoyokins consisting of 2 members — AHNAK1 and AHNAK2. 8,9 Previous studies have determined that AHNAK1 is involved in cell migration, cytoarchitecture and barrier properties. 10 -12 AHNAK1 is responsible for development of tumors owing to its location is in the nuclear of tumor cells. 13 Besides, the motility and mechanical properties are also modulated by AHNAK1 in Schwan cells. 14 Wang et al. have indicated that AHNAK2 is highly regulated in clear cell renal cell carcinoma (ccRCC) and correlates with cancer progression as well as patients survival rates. 15 Meanwhile, AHNAK2 exerts a promoting role in the proliferation and migration of uveal melanoma cells. 16 But up to now, there is no further investigation on the biological function of AHNAK2 and its molecular mechanism in lung adenocarcinoma.
Here, the expression of AHNAK2 in lung adenocarcinoma tissues and cells was examined. Knockdown of AHNAK2 can lead to a decrease of cell proliferation, migration, invasion and apoptosis, which is related with the inactivation of the MAPK signaling pathway. Our results indicate that AHNAK2 is a promising robust biomarker and therapeutic target in lung adenocarcinoma.
Materials and Methods
Patients Samples
The gene expression profiles of 3 different kinds of lung tissues, Bhattacharjee Lung, Su Lung and Landi Lung, were downloaded from the Oncomine database (https://www.oncomine.org/resource/login.html) to analyze the expression of AHNAK2. Differential expression of AHNAK2 and its correlation with pathological stage in lung adenocarcinoma were obtained from GEPIA website(http://gepia.cancer-pku.cn/index.html) as well as AHNAK1.
Cell Culture and Transfection
Normal lung epithelial cells BEAS2B was purchased from American Type Culture Collection (ATCC; Manassas, VA, USA). Two human lung cancer cell lines, NCI-H460 and A549, were obtained from the cell bank of the Chinese Academy of Medical Sciences (Shanghai, China). All cells were incubated in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS; Gibco, Grand Island, NY, USA) and 1% penicillin-streptomycin (Gibco) at 37°C with 5% CO2. Cell transfection was conducted when the cell confluence was over 80% using Lipofectamine 2000 as the manufacturer’s instructions. Sequences of AHNAK2-siRNAs are as follows: AHNAK2-siRNA#1:F: 5’-AUAUUUUAUGUUUUCAAAGAA-3’, AHNAK2-siRNA#2:F: 5’-UAUGGAUUGAAAUUUUGGCCA-3’. The si-con group was considered as an internal control. These above productions were synthesized by GenePharma Co., Ltd (Shanghai, China).
To explore whether the regulatory effect of AHNAK2 in A549 cells is associated with MAPK pathway, A549 cells were treated with a MEK inhibitor U0126 (10 μM, San Diego, CA, USA). Cells were grouped as: control, U0126, si-AHNAK2, si-AHNAK2+U0126.
RNA Isolation and Quantitative RT-PCR
Trizol reagent (Invitrogen; Thermo Fisher Scientific, Inc.) was used to isolate total RNA of tumor cells, according to the manufacturer’s instructions. Then cDNA was reversed transcription using SuperScript II reverse transcriptase (Invitrogen; Thermo Fisher Scientific, Inc.) and subjected to qRT-PCR with specific AHNAK2 primers. qRT-PCR analysis was conducted by SYBR® Premix Ex Taq™ (Takara Biotechnology Co., Ltd., Dalian, China) via the system of ABI 7500 Sequence Detection System (Applied Biosystems; Thermo Fisher Scientific, Inc.). The primers of qRT-PCR were shown as follows: AHNAK2: F, 5’-CGCGATGTGCGACTGC-3’, R, 5’-TGGTCATCTTCCGTTTCTGC-3’; GAPDH: F, 5’-GGAGCGAGATCCCTCCAAAAT-3’, R, 5’-GGCTGTTGTCATACTTCTCATGG-3’. The qRT-PCR was run as the following conditions: 95°C for 5 min followed by 40 cycles of 95°C for 30 s, and 60 °C for 45 s, 72 °C for 30 min. The relative expression of AHNAK2 was calculated by the 2-ΔΔCt method. Data were presented at least 3 independent experiments.
Western Blotting
48 hours later, transfected cells were placed in RIPA lysis buffer on the ice to extract proteins. The concentration of proteins was detected by BCA Protein Assay Kit (Beyotime, Shanghai, China). Equal amounts of protein extracts (20 μg/lane) were loaded in 12% SDS-PAGE and transferred onto a PVDF membrane (Millipore, Boston, MA, USA). The membrane was then incubated with 5% non-fat milk for 1 h at room temperature and the indicated primary antibodies (1:1,000; Abcam, Cambridge, UK) overnight at 4°C against AHNAK2 (ab70053), Bcl-2 (ab32124), Bax (ab32503), p-MEK (ab214445), MEK (ab33918), p-ERK (ab73209), ERK (ab53277), p-P90RSK (ab32203) and P90RSK (ab32114), as well as secondary antibody (1:10,000; Abcam, Cambridge, UK) for 1 h at room temperature. Finally, all bands were probed with an enhanced chemiluminescence (ECL). The gray values of protein bands were scanned with QUANTITY ONE software and recorded by a Bio-Rad camera system (Bio-Rad, USA).
Cell Proliferation Assay
CCK-8 assay was performed to detect cell proliferation ability. Transfected cells were seeded into 96-well plate (1 × 103) and then incubated in a carbon dioxide incubator. We investigated the cell viability every 24 h. At the specific time point, 10 µL of CCK-8 reagent was added into per well for another 1.5 h at 37°C. OD value of cells was measured at 450 nm on an enzyme-labeled instrument.
Cell Migration and Invasion Assay
Cell migration and invasion were assessed by wound-healing assay and transwell analysis. The migration rates of cells were explored by wound-healing assay. To get a cell monolayer with over 80% confluence, cells were placed in a 6-well plate with a density of 5 × 105 cells per well at 37 °C for 8 h or more. A pipette tip was to draw a line to destroy the cell layer and the floating cells were removed by the washing with PBS. Cells were subsequently cultured in serum-free medium and the wound closure distance was measured under a microscope at a time range of 0 and 24 h. Transwell assay was carried out to measure the invasion capacity as well as migration. At first, the Matrigel was melted overnight and diluted with serum-free medium at a 1:6 ratio. Then 100 µL of mixture was added to the upper surface of transwell chamber. To hydrate the basement membrane, 500 µL of serum-free medium was placed to the lower chamber. 100 µL of cell suspension (1 × 105) was seeded in the upper chamber and incubated overnight. The un-invaded cells were removed and the transwell chamber was washed with 1 × PBS for 3 times. Next, 4% paraformaldehyde was to fix cells and 0.1% crystal violet was to stain cells, respectively. The detection of migration capability was explored without the Matrigel.
Flow Cytometry Assay
Cell apoptosis was determined using a Cell Apoptosis Detection kit on the basis of the manufacturer’s protocols. Briefly, transfected A549 cells were re-suspended by the mixture of 100 µL binding buffer, 10 µL Annexin V-fluorescein isothiocyanate (FITC) and 5 µL propidium iodide (PI), and then maintained for 15 min at 25 °C under the dark. At last, the apoptotic rate was analyzed using a flow cytometer (BD Biosciences, San Jose, CA, USA).
Statistical Analysis
Data was analyzed by SPSS22.0 statistical analysis software and GraphPad Prism 5.0 (La Jolla, CA, USA). The Student’s t-test was used to determine the difference of 2 groups; the mean comparison between the larger groups was confirmed by 1-way analysis of variance with post-hoc analysis of Dunnett. Kaplan-Meier method was conducted to plot the overall survival curve with the log-rank test. All results were presented as mean ± standard deviation (SD). P < 0.05 in all cases was considered statistically significant.
Result
AHNAK2 Is Remarkably Up-Regulated in Lung Adenocarcinoma Tissues
To explore the biological effect of AHNAK2 in lung adenocarcinoma, we firstly downloaded the genomic profiles from the Oncomine database. Comparing with the normal human samples, we found that AHNAK2 expression was significantly increased in lung adenocarcinoma tissues (Figure 1A-C). In consistent with these results, Gene Expression Profiling Interactive Analysis (GEPIA) verified that AHNAK2 was highly expressed in lung adenocarcinoma (Figure 1D and E).

Expression of AHNAK2 in lung adenocarcinoma. A, AHNAK2 expression in Bhattacharjee Lung containing 17 normal lung cases and 132 lung adenocarcinoma cases. B, AHNAK2 expression in Landi Lung containing 49 normal lung cases and 58 lung adenocarcinoma cases. C, AHNAK2 expression in Su Lung containing 30 normal lung cases and 27 lung adenocarcinoma cases. D and E, Box plot of the mRNA expression levels of AHNAK2 in lung adenocarcinoma and normal controls. D, Match TCGA normal and GTEx data including 347 normal tissues and 483 tumor tissues; E, Match TCGA normal data including 59 normal tissues and 483 tumor tissues. P < 0.05 is considered significant.
Upregulation of AHNAK2 Is Associated With Poor Prognosis in Patients With Lung adenocarcinoma
Next, we analyzed the relationship between the expression level of AHNAK2 and clinicopathological characteristics of lung adenocarcinoma patients. According to the data from GEPIA website, we found that pathological stage in lung adenocarcinoma revealed a close association with evident AHNAK2 expression (Figure 2A). It can be clearly seen from Figure 2B that lung adenocarcinoma patients with high AHNAK2 expression had a poorer prognosis in the short term compared with patients carrying the low AHNAK2 expression. However, in the long term, lung adenocarcinoma patients with upregulation of AHNAK2 could live longer.
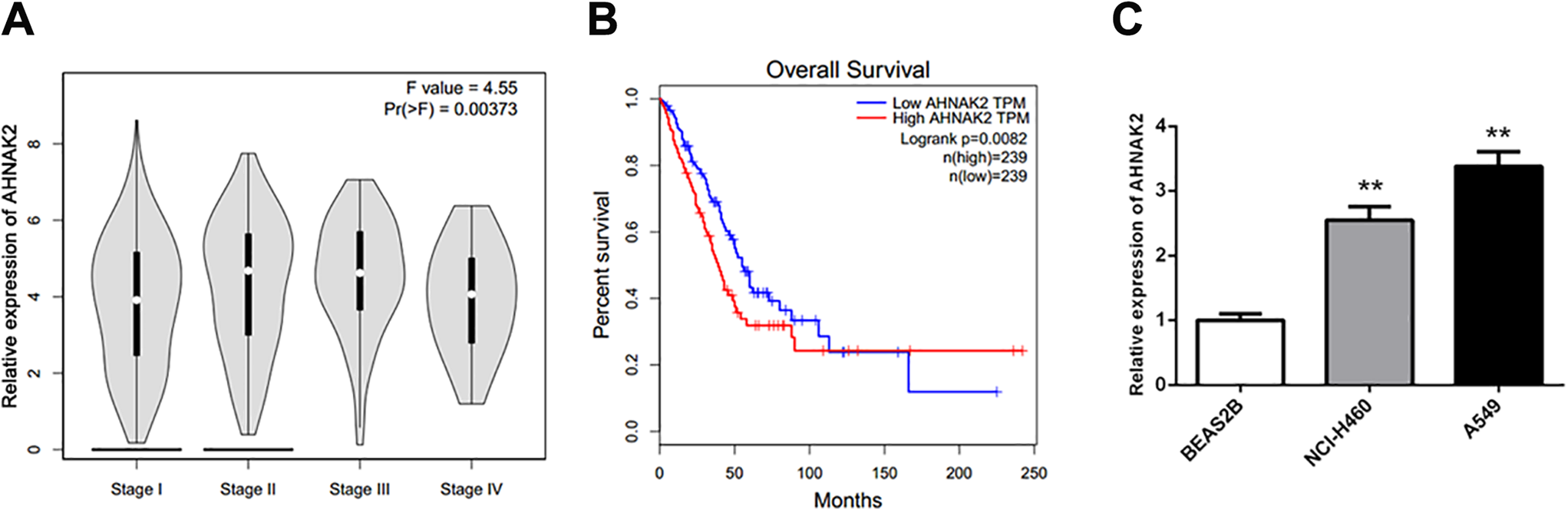
The association between AHNAK2 expression and overall survival rates of lung adenocarcinoma patients as well as the expression level of AHNAK2 in lung adenocarcinoma cell lines. A, Relationship between pathological stage and AHNAK2 expression was shown in pathological stage plotter. B, The overall survival curve exhibited the overall survival of patients with low (n = 239) or high (n = 239) expression levels of AHNAK2 using Kaplan-Meier method, P = 0.0082. C, The mRNA expression levels of AHNAK2 in BEAS2B, NCI-H460 and A549 cell lines were detected by real-time PCR, this experiments were repeated 3 times, **P < 0.01 versus BEAS2B.
Reduction of AHNAK2 Inhibits Proliferation of A549 Cells
To investigate the biological function of AHNAK2 in lung adenocarcinoma cells, 3 cell lines were employed to detect the expression of AHNAK2. According to the result of qRT-PCR, we discovered that among the 3 cell lines, both NCI-H460 and A549 showed higher expression levels of AHNAK2 compared with the other human normal cell line BEAS2B (Figure 2C, P < 0.01). Due to the aforementioned results, we selected A549 cells with the highest expression level of AHNAK2 as a subject in the following experiments. siRNA technology was conducted to knockdown the expression of AHNAK2 in A549 cells. Results of qRT-PCR and western blot showed that AHNAK2 was obviously reduced at the mRNA and protein levels after si-AHNAK2 transfection (Figure 3A-C, P < 0.01). Subsequently, we performed CCK-8 assay to evaluate whether the reduction of AHNAK2 affects the proliferation of A549 cells. As shown in Figure 3D, AHNAK2 knockdown aberrantly suppressed the proliferative ability of A549 cells in comparison to the si-con group.

Reduction of AHNAK2 significantly inhibited lung adenocarcinoma cell proliferation. A, The mRNA expression level of AHNAK2 in A549 cells transfected si-AHNAK2 for 24 h was assessed by qRT-PCR, **P < 0.01 versus si-con group. B, The protein levels of AHNAK2 were detected by western blot analysis. C, The quantification of B, **P < 0.01 versus si-con group. D, The CCK-8 assay showed that the proliferation of A549 cells was inhibited after AHNAK2 knockdown at 0, 24, 48, 72 and 96 h, *P < 0.05 and **P < 0.01 versus si-con group. All results were performed in triplicates.
Silencing AHNAK2 Suppresses Migration and Invasion, and Induces Apoptosis in A549 cells
We then implemented wound-healing assay and transwell assay to explore the role of down-regulation of AHNAK2 in migration and invasion capacities. After scratching 24 h, the migration of A549 cells transfected with si-AHNAK2 was inhibited compared with si-con group (Figure 4A and B, P < 0.01). Transwell assay results indicated that knockdown of AHNAK2 repressed the migration and invasion of A549 cells (Figure 4C and D, P < 0.01). Flow cytometry analysis showed that AHNAK2 inhibition significantly enforced the apoptotic capability of A549 cells (Figure 4E and F, P < 0.01). Furthermore, the anti-apoptosis protein Bcl-2 level was repressed whereas the protein expression level of pro-apoptosis protein Bax was increased after si-AHNAK2 transfection (Figure 4G and H, P < 0.01).

Knockdown of AHNAK2 suppressed migration and invasion, while facilitated cell apoptosis of A549 cells. A, The migratory capacity of A549 cells transfected with si-AHNAK2 for 24 h was examined using the wound-healing assay. B, The cell migration rate of A549 cells after AHNAK2 knockdown, **P < 0.01 versus si-con group. C, Transwell assay showed the migration and invasion of A549 cells at 24 h-post transfection. D, The numbers of migratory and invaded cells, **P < 0.01 versus si-con group. E and F, Cell apoptosis was detected using flow cytometry, **P < 0.01 versus si-con group. G and H, The apoptosis-related proteins Bcl-2 and Bax levels were measured by western blotting in A549 cells transfected with si-AHNAK2, **P < 0.01 versus si-con group. Each experiment was performed in triplicates.
Down-Regulation of AHNAK2 Attenuates the Activity of the MAPK Signaling Pathway in A549 Cells
To study the underlying molecular mechanism of AHNAK2 in lung adenocarcinoma, we detected the MAPK-related proteins expression levels using western blot. The MAPK signaling pathway has vital effects in many cellular processes including cell growth and metastasis. 17 The results of western blot illustrated that there was no detectable change at the aspect of expression of MEK in the si-AHNAK2 group, but the phosphorylated MEK protein level was significantly reduced. Meanwhile, ERK showed the same expressional pattern as MEK (Figure 5A, P < 0.01). Furthermore, the quantified results of these proteins in Figure 5B clearly exhibited that the phosphorylated MEK and ERK were all decreased after si-AHNAK2 transfection (P < 0.01). p-P90RSK, the direct downstream molecule of p-ERK, was also attenuated after the transfection of si-AHNAK2 (Figure 5C and D, P < 0.01). In addition, U0126 stimulation revealed a similar effect to si-AHNAK2 on the MAPK pathway-related proteins levels in A549 cells (Figure 6, P < 0.05).
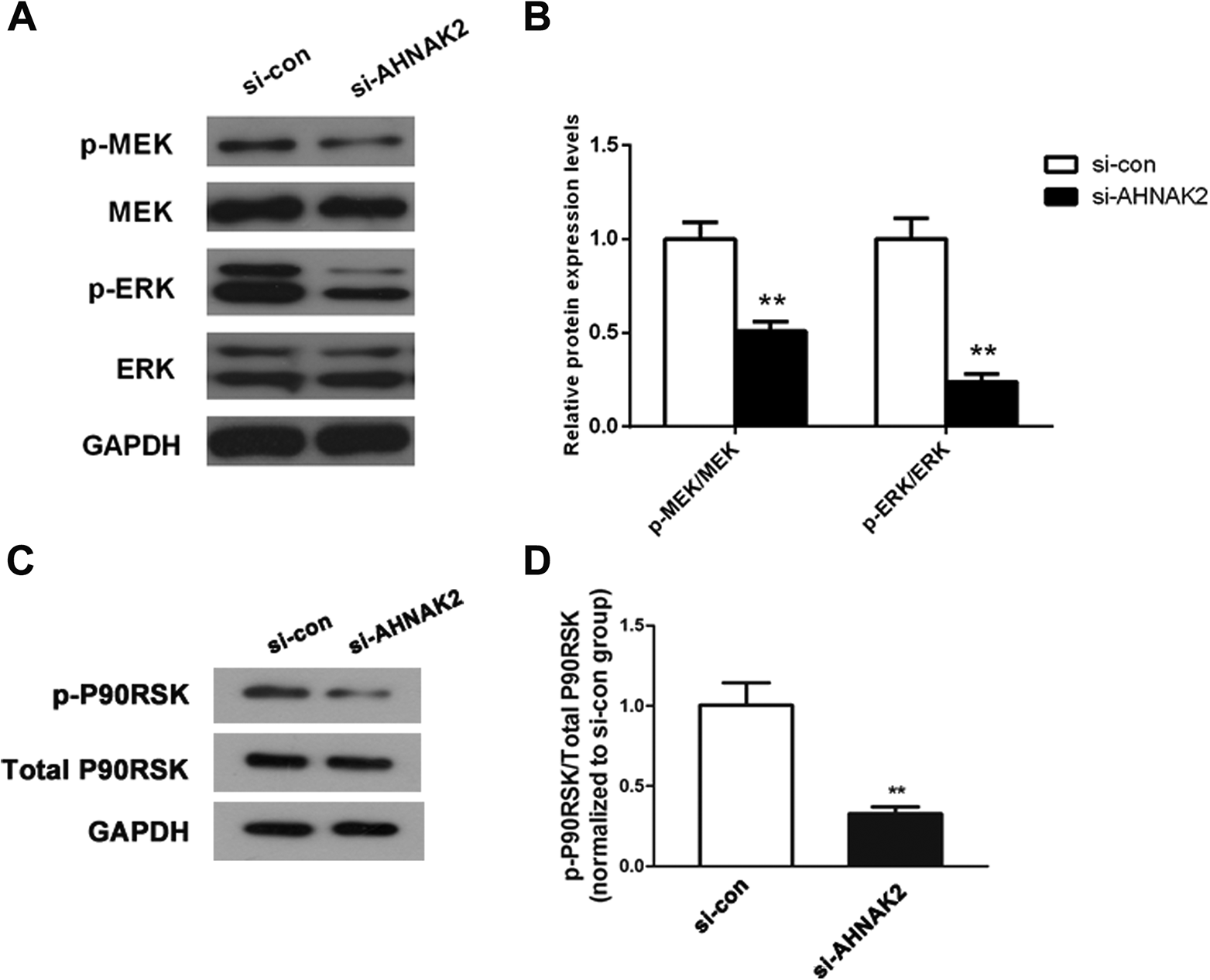
Down-regulation of AHNAK2 inactivated the MAPK signaling pathway in lung adenocarcinoma cells. A, Western blot analysis determined that the proteins expression level of MEK/ERK and p-MEK/p-ERK. B, The gray values of protein bands were quantified, **P < 0.01 versus si-con group. C and D, After transfection with si-AHNAK2 for 48 h, p-P90RSK/P90RSK expressions were measured by western blotting. The protein bands intensity was quantified, **P < 0.01 versus si-con group. These experiments were all conducted at least 3 times.

The effects of si-AHNAK2 on the MAPK pathway were consistent with the impacts of U0126, a MEK inhibitor. The MAPK pathway-related proteins expression levels were assessed and quantified, **P < 0.01 versus si-con group, #P < 0.05 versus si-AHNAK2 group. Every assay was repeated in triplicates.
Discussion
In this present study, we observed that AHNAK2 was significantly upregulated in lung adenocarcinoma tissues and cell lines. The highly expressed AHNAK2 was associated with poor outcome of lung adenocarcinoma patients. Moreover, the pathologic stage and overall survival rates were identified to be linked with AHNAK2 expression. NCI-H460 is an non-small cell lung cancer cell line 18 ; A549 is a adenocarcinoma human alveolar basal epithelial cell line. 19 A previous study has showed that lung adenocarcinoma is a subtype of non-small cell lung cancer. 20 Moreover, NCI-H460 and A549 cell lines have been widely used for the detection of lung adenocarcinoma. 21,22 Thus, to investigate the expression of AHNAK2 in lung adenocarcinoma cells, we selected NCI-H460 and A549. Given that A549 cells showed a higher level of AHNAK2 than NCI-H460 cells, A549 was utilized in the functional in vitro experiments (Figure 2C). Down-regulation of AHNAK2 arrested cell proliferation, migration and invasion, and promoted cell apoptosis via inhibiting the activity of MAPK signaling pathway in A549 cells.
AHNAK2 is a large protein (> 600 kDa), which is firstly identified in 2004. Previous study has suggested that AHNAK2 is remarkably increased in pancreatic ductal adenocarcinoma (PDAC) tissue samples and cell lines.
23
In consistent with that, we found that the expression of AHNAK2 was highly regulated in lung adenocarcinoma and its upregulation could induce poor prognosis of lung adenocarcinoma patients. According to the current publications, most of researchers focus on the investigation to the biological function of AHNAK1. For example, Shtivelman et al. discovered that AHNAK1 is expressed at higher levels in promyelocytic leukemia.
24
AHNAK1 is essential in calcium signaling during T cell activation as a modulator to measure calcium channel activity.
25,26
Moreover, the C-terminal of AHNAK1 could strengthen the connection between AHNAK2 and other proteins.
27
-29
Thus, we also explored the expression level of AHNAK1 in lung adenocarcinoma and found that AHNAK1 expression was significantly reduced in tumor tissues (
AHNAK proteins have a closely connection with large protein complexes at the plasma membrane that linked the extracellular matrix to the cytoskeleton. 33 AHNAK2 is also implicated in the hypoxia-related pathways. 15 Based on which, we detected the underlying molecular mechanism of AHNAK2 in lung adenocarcinoma using western blot analysis. A previous study performed by Gao et al. indicated that the activation of MAPK signaling pathway is responsible for 76% occurrence of lung adenocarcinoma. 17 At the early stage, the MAPK pathway is active in cancers as a notable sign. 34 Several researches have determined that ERK, a known molecule in MAPK pathway, is activated in tumor cells and then promotes cell growth, inhibits cell apoptosis. MEK has a similar role in tumorigenesis to ERK, and its active form p-MEK could activate the ERK. 35 P90RSK is directly phosphorylated and activated by ERK in response to various stimuli, which plays a crucial role in the development of lung adenocarcinoma. 36 Therefore, in order to examine whether AHNAK2 affects the MAPK pathway in lung adenocarcinoma, we measured the protein levels of MEK/ERK/P90RSK and their phosphorylated forms in A549 cells after si-AHNAK2 transfection. We found that down-regulation of AHNAK2 induced a decrease level of p-MEK, p-ERK and p-P90RSK. Hence, we speculated that AHNAK2 might contribute to the progression of lung adenocarcinoma via activating the MAPK signaling pathway. In addition, U0126 37 (an inhibitor candidate of MEK) treatment further intensified the inhibitory effect of si-AHNAK2 on the protein levels of p-MEK, p-ERK and p-P90RSK, which validated the correlation between AHNAK2 and MAPK pathway in lung adenocarcinoma.
Nevertheless, a few limitations of this investigation must be acknowledged. The different time points in cell viability assay and wound-healing experiments was a potential problem. Based on our results, no changes in cell proliferation at 24 hours were observed while we found a change in cell migration at 24 hours. The wound-healing test with serum-free medium actually ruled out the effect of cell proliferation on the migration capacity, which discloses that there might exist other potential reasons for the regulation of AHNAK2 on the proliferation. All these data indicate that the choice of time points would affect the final conclusions, even may completely subvert our conclusions. This maybe act as a potential systemic error in this present study. To avoid this impact, further experiments should be conducted by measuring the same time points in cell viability and wound-healing assays. However, the serum-free medium for wound-healing test cannot support the cell growth for a long time. In addition, it seems difficult to detect the significant difference in a short time in the proliferation experiment. Therefore, how to coordinate this problem in future experiments will become a relative key challenge.
In conclusion, AHNAK2 was significantly up-regulated in lung adenocarcinoma tissues and cells, and its highly expression was associated with the poor prognosis of patients with lung adenocarcinoma. Reduction of AHNAK2 could hinder the cellular aggressive behaviors, including cell proliferation, migration, invasion, and apoptosis via inactivating the MAPK signaling pathway, which sheds a new insight to treat lung adenocarcinoma treatment.
Supplemental Material
Supplemental Material, Supplementary_S1 - Down-Regulation of AHNAK2 Inhibits Cell Proliferation, Migration and Invasion Through Inactivating the MAPK Pathway in Lung Adenocarcinoma
Supplemental Material, Supplementary_S1 for Down-Regulation of AHNAK2 Inhibits Cell Proliferation, Migration and Invasion Through Inactivating the MAPK Pathway in Lung Adenocarcinoma by Dong-Wei Wang, Hai-Zheng Zheng, Na Cha, Xiao-Jie Zhang, Min Zheng, Ming-Ming Chen and Li-Xiang Tian in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Authors’ Note
Our study did not require an ethical board approval because it did not contain human or animal trials.
Acknowledgments
We thank Changchun Obstetrics-Gynecology Hospital and Changchun Medical College for support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
