Abstract
Purpose
Owing to the mortality associated with metastatic prostate cancer and the shortcomings of the current parameters in predicting the disease prognosis, we require the identification of viable biomarkers, which would help in the diagnosis and prognosis of the disease. We aimed to determine whether the interleukin-8 level in the tumor microenvironment could serve as a potential clinical diagnostic marker and prognostic factor for prostate cancer.
Methods
The migration assay of prostate cancer cells was performed in an in vitro co-culture model. Cell lines PC3 and DU145 were divided into two groups and co-cultured with M0 and M2 macrophages, respectively. We used reverse transcription-quantitative polymerase chain reaction to detect M2 macrophage marker expression levels. Immunohistochemistry analyses of tissue microarrays were performed to analyze the correlation between the increased expression of interleukin-8 and the prognosis of prostate cancer. A retrospective analysis based on 142 residual serum specimens was performed to analyze the level of interleukin-8.
Results
We observed that M2 macrophages promoted the migration of prostate cancer cells and significantly increased the concentrations of interleukin-8 in the co-culture supernatants. We observed increased expression of CD163 and interleukin-8 in prostate cancer tissues. Furthermore, the levels of interleukin-8 in the serum of prostate cancer patients were higher than those in healthy controls. Untreated patients had higher levels of interleukin-8, which could be a predictor of a higher metastasis rate.
Conclusion
These results suggest that interleukin-8 produced via bidirectional communication between prostate cancer cells and M2 macrophages is a putative biomarker for prostate cancer diagnosis and treatment.
Introduction
Prostate cancer (PCa) is a critical disease that affects the health of men worldwide. 1 Despite the high long-term survival associated with localized PCa, metastatic PCa results in a poor prognosis. 2 Existing parameters, such as the Gleason sum or preoperative PSA, provide some prognostic information; however, estimating the prognosis in patients with PCa is challenging as two-thirds will have a 5–7 Gleason sum and PSA serum levels of 4–10 ng/dL. 3 Therefore, accurate prognostic markers must be developed to identify the biological potential of tumors.
Recent studies have shown that the tumor microenvironment (TME) plays a crucial role in the progression and metastasis of PCa.4,5 Several different stromal cells infiltrate the TME, including smooth muscle cells, adipocytes, fibroblasts, endothelial cells, and some inflammatory cells that influence tumor progression.2,6 M2 macrophages, comprising a phenotype of macrophage polarization, are major immune cells abundant in the TME. These macrophages release factors and enzymes, such as interleukin-8 (IL-8), interleukin-6 (IL-6), matrix metallopeptidase-2 (MMP-2), matrix metallopeptidase-9 (MMP-9), and transforming growth factor-β (TGF-β), to promote tumorigenesis, angiogenesis, and metastasis. 7
IL-8, a member of the CXC chemokine family, is a pro-inflammatory cytokine. The IL-8-regulated chemokine receptor CXCR7 stimulates epidermal growth factor receptor (EGFR) signaling to promote the growth 8 and invasion of PCa, 9 which promote PCa bone metastasis by enhancing bone sialoprotein (BSP) regulation. 10 However, the interactions between M2 macrophages and PCa cells that promote the expression of IL-8 and the subsequent progression of PCa remain unexplored. Additionally, the molecular mechanisms associated with IL-8 in PCa regarding their effects on diagnosis and prognosis have not been comprehensively explored.
Therefore, in the present study, we investigated whether M2 macrophages could promote the migration of PCa cells in an in vitro co-culture model. We further monitored the concentrations of IL-8 in the co-culture supernatants to identify any alterations. Finally, the IL-8 levels in the serum of PCa patients were compared with those in healthy controls who did not have the disease. We believe that our study would help assess whether IL-8 could serve as a putative diagnostic and prognostic biomarker for PCa progression.
Material and Methods
Serum Specimen Collection and Processing
According to the retrospective analysis of clinical data, 142 total residual serum specimens were collected from individuals from 2020 to 2022, including 55 patients with PCa (average age, 73), 67 patients with benign prostatic hyperplasia (BPH; average age, 72), and 20 healthy controls (average age, 50), which was approved by the Hospital Ethics Board on September 17, 2020 (NO.2020091701). Because the patient was no longer in the hospital when we collected the remaining samples, we applied for an informed consent waiver. A total of 19 treatment-naive PCa patients scheduled for their first blood samples were traced, and 36 PCa patients with any treatment (radiation or hormonal treatments) were also included in this study. Patients with active inflammation, other neoplasms, or autoimmune and chronic inflammatory diseases were excluded. The collected whole blood samples were placed at room temperature for 2 h and centrifuged at 1000 × g for 20 minutes, and the residual supernatant was collected after laboratory examination. The supernatant was aliquoted and stored at -80 °C to avoid repeated freezing and thawing.
Cell Culture and Polarization of Macrophages
All cell lines were purchased from the ATCC (American Type Culture Collection, Manassas, VA, USA). DU145 (HTB-81), PC3 (CRL-1435), and RAW264.7 (TIB-71) cells were cultured in Dulbecco's modified Eagle medium (DMEM, Gibco, Evansville, IN, USA), supplemented with 1% penicillin–streptomycin (Gibco) and 10% fetal bovine serum (FBS, Invitrogen, Carlsbad, CA, USA), maintained at 37 °C in a humidified atmosphere of 5% CO2. RAW264.7 cells were induced to form M2 macrophages by treating them with recombinant murine IL-4 (40 ng/mL; PeproTech, USA).
RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction
RNA was obtained using RNA extraction reagent (Takara, Dalian, China). A PrimeScript RT Reagent Kit (TaKaRa) was used for reverse transcription. A 7500-Fast Real-Time PCR system (Applied Biosystems, Waltham, MA, USA) was applied to perform quantitative real-time polymerase chain reaction (qPCR), using SYBR® Green RT-PCR Master mix (Takara). The relative expression levels were quantified and normalized to those of GAPDH, an endogenous control. The 2-ΔΔCt method was used for comparative quantification. All primer sequences were from GenBank, and all the operations were performed according to the manufacturer's instructions.
Transwell Migration and Co-Culture Experiments
All in vitro cell migration assays were performed using Transwell chambers (8 µm pore size, Corning, NY, USA) in 24-well plates. PCa cells (0.5 × 105 cells per 200 µL) were added in the upper inserts; then, RAW264.7 cells (M0) and M2 macrophages (1 × 105 cells per 450 µL) were placed into the lower wells, and both were cultured in DMEM containing 5% FBS. After incubation for 24 h, the upper chambers were rinsed three times with PBS after fixing with commercial methanol and then stained with crystal violet (Beyotime Biotechnology, Jiangsu, China) at a concentration of 0.1%. Microphotographs of the cells attached to the lower side of the membranes were obtained from at least three random fields (Inverted microscope IX71, Olympus, Tokyo, Japan, 20 × 10).
ELISA Cytokine Assays
IL-8 protein levels were measured using ELISA kits (Cloud-Clone Corp, Wuhan, China). Serum from patients with PCa or prostatic hyperplasia was collected and stored at -80 °C. The IL-8 concentration was determined according to the manufacturer's instructions and calculated on the basis of the standard curve of each ELISA.
High-Density Tissue Microarrays and Immunohistochemistry
Immunohistochemistry (IHC) was performed on commercially available high-density tissue microarrays (HProA150PG02, Shanghai Outdo Biotech, Shanghai, China). Three normal, 95 tumor, and 52 paracancerous non-tumor (NT) prostate tissue specimens were used. The classification of PCa grades followed a previous article. 11 Briefly, high-density tissue microarrays were dewaxed in xylene and rehydrated through gradient alcohol, antigen repair, and incubation with the primary antibody and secondary antibody (K5007, Dako, Glostrup, Denmark), followed by staining with 3,3′-diaminobenzidine staining and hematoxylin counterstaining. Primary antibodies were against IL-8 (ABP51621, Wuhan, China) and CD163 (XC8383, Outdo Biotech, Shanghai, China), each diluted to a concentration of 1:1000. The IHC staining assessment was independently conducted by two experienced pathologists as described previously. 12 The staining intensity was scored as follows: 0 (unstained), +1 (light yellow), +2 (brown–yellow), and +3 (brown), and the percentage of positive cell staining was graded as 0–100%. The two scores were multiplied to obtain a positive expression. The groups with a total CD163 score ≤ 14% was considered the low expression group, and that with a total CD163 score > 14% was considered the high expression group. Similarly, the group with a positive cell staining score >100% was considered the high expression group of IL-8.
Statistical Analysis
All values in vitro are expressed as the mean ± SD of at least three replicates. All statistical analyses were performed using the Statistical Package for the Social Sciences software (SPSS version 20.0) and GraphPad Prism V software. Statistical significance between groups was compared using unpaired two-tailed Student's t-tests. The Mann–Whitney U test was used to compare the expression of CD163 and IL-8 in prostate tissues where the assumption of normality was questionable. Two rates or two constituent ratios were compared using a Chi-squared test. Statistical significance was considered at p-values <0.05 for all experiments.
Results
M2 Macrophages Polarized from RAW264.7 Cells Promote PCa Cell Migration
To determine whether treatment with recombinant murine IL-4 for 48 h affected innate RAW264.7 (M0) differentiation into M2 macrophages, cells were collected to analyze the expression of M2 macrophage markers (CD206, Arg-1, and Mrc-2)13–15 using qPCR analysis. Subsequently, transwell migration assays using DU145 and PC3 cells were performed after co-culture experiments with M0 and M2 macrophages, respectively. The results revealed that M2 macrophage markers increased significantly after treatment with IL-4 for 48 h (Figure 1A, p < 0.05). Co-culture experiments showed that M2 macrophages increased the migration of PCa cells after 24 h (Figure 1B and C, p < 0.05). Moreover, to verify whether IL-8 affected the PCa environment in this study, the concentration of IL-8 in the supernatant of cell culture was determined after the co-culture of M2 macrophages and PCa cells. As depicted in Figure 3D, an increased IL-8 concentration was observed in the supernatants from the M2 macrophage/PCa cell co-culture system.

(A) M2 macrophage marker mRNA expression was detected. (B) Transwell migration assays using DU145 and PC3 cells were performed based on co-culture experiments with M0 and M2 macrophages. Representative micrographs of the cells attached to the lower side of the membranes were captured under a 20× objective and counted in five independent fields. (C) Quantitative results of the cell migration. (D) IL-8 protein levels, as determined from the conditioned media (Student's t-tests, *p < 0.05).
Increased Levels of M2 Macrophages and IL-8 in Prostate Cancer Tissues
To determine whether the IL-8 expression was associated with the number of M2 macrophages infiltrating during prostate carcinogenesis, human prostate tissue arrays were immunostained with antibodies against IL-8 and CD163, an M2 macrophage-specific cell marker. CD163 expression was low in normal acini and high in areas containing tissue stroma. High-resolution microscopic digital images were used to assess all immunohistochemical results. Representative IHC staining images depicting the expression of CD163 and IL-8 are shown in Figure 2A. Compared with that in normal prostate tissue, the expression of CD163 and IL-8 was significantly higher in PCa and adjacent tissues (Figure 2B and C, *p < 0.05). CD163 and IL-8 were overexpressed in the majority of PCa cases compared with levels in paracancerous NT samples (Figure 2, Tables 1 and 2, *p < 0.05). We also observed that IL-8 levels were not significantly correlated with age, but high-grade tumors exhibited significantly higher levels of IL-8 (Table 3).
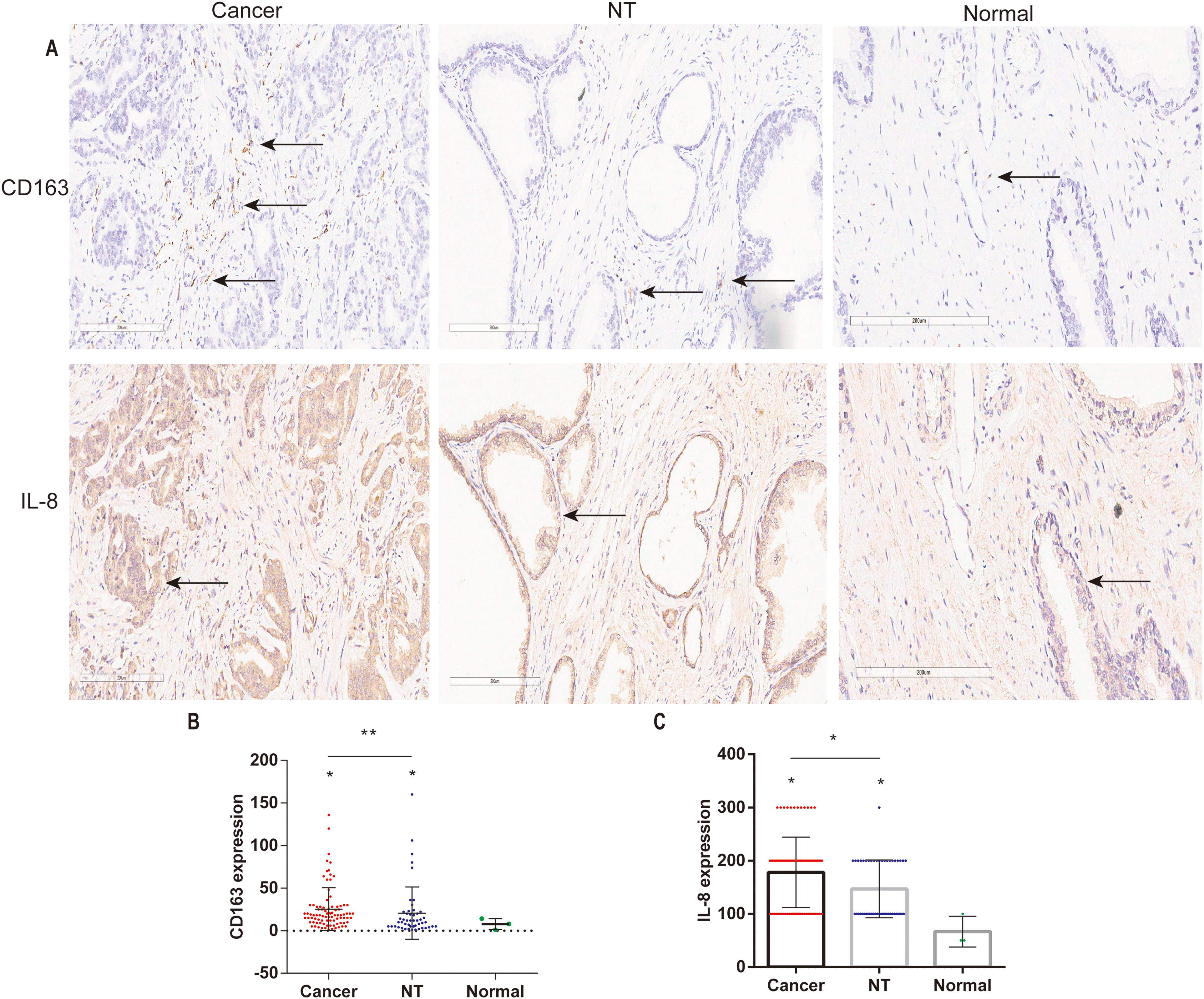
(A) Representative immunohistochemistry staining images depicting the expression of CD163 and IL-8 in prostate cancer, non-tumor (NT), and normal prostate tissues (image scope × 64 software, 20×). (B) Quantification of CD163 expression (staining intensity × number of positive cells; Mann–Whitney U test, *p < 0.05). (C) Quantification of IL-8 expression (staining intensity × positive rate; Mann–Whitney U test, *p < 0.05).

(A) Serum secretion of IL-8 in prostate cancer (PCa), benign prostatic hyperplasia (BPH), and healthy control (HC) cases. (B) Further examination of IL-8 expression in PCa cases of untreated patients and those with previous or current radiation or hormonal treatment. (C) The same procedure was performed for metastatic and non-metastatic patients (*p < 0.05, *compared with the control treatment, Student's t-test).
Differential Expression of CD163 in Cancer and Non-Tumor (NT) Tissues (Chi-Squared Test).

The actual analyzable data are shown in the table. Cases of tumor tissue loss and non-compliant glandular tissues were excluded after immunohistochemical staining.
Differential Expression of IL-8 in Cancer and Non-Tumor (NT) Tissue (Chi-Squared Test).

The actual analyzable data are shown in the table. Cases of tumor tissue loss and non-compliant glandular tissues were excluded after immunohistochemical staining.
Correlation Between IL-8 Expression and Clinicopathological Characteristics (Chi-Squared Test).

The actual analyzable data are shown in the table. Cases of tumor tissue loss and non-compliant glandular tissues were excluded after immunohistochemical staining.
Association Between serum IL-8 Concentrations and Prostate Cancer
The levels of IL-8 were significantly increased in patients with PCa (Figure 3A, p < 0.05) and those with a higher probability of tumor metastasis (Figure 3C, p = 0.07). We further examined IL-8 expression in untreated PCa cases and patients administered planed radiation or hormonal treatment, which exhibited an increased IL-8 concentration in the former (Figure 3B, p < 0.05). Moreover, the changing trend in serum IL-8 in each group was consistent with that of total prostate-specific antigen (tPSA) (Figure 3).
Discussion
Total PSA is a widely used screening tool for PCa worldwide. However, this screening technology, can result in over-diagnosis and over-treatment, which have harmful effects. 16 In a previous study, 15% of men with PSA less than 4.0 ng/mL were finally diagnosed as having PCa, and 15% of them had a high Gleason score. 17 There is also a risk of PCa in the PSA range <4 ng/mL.
M2 macrophages are evolved to facilitate the interaction between the tumor and human immune system.. 18 Moreover, previous studies have shown that increased M2 macrophage infiltration is related to poor pathological characteristics and worse prognosis of PCa.19–21 Also, IL-8 and IL-6 secreted by M2 macrophages promote tumor occurrence and development.22,23 The expression of IL-8 plays a pro-metastatic role,24,25 and its serum concentration is useful as a pharmacodynamic biomarker for early detection of response to immunotherapy. 26 Meanwhile, IL-8 could be an applicable surrogate marker of prostate inflammation,27,28 as a possible biomarker for diagnosis and treatment. The level of IL-8 secretion in normal prostate epithelial cells was detected and found to be closely related to cancer invasiveness, carcinogenesis, 29 and androgen receptor loss in metastatic disease. 30 We further verified this from serological and histopathological perspectives and found that M2 macrophages in the tumor microenvironment are also involved in this process, and that they induce the expression of IL-8 via bidirectional communication with PCa cells.
First, we demonstrated that M2 macrophages promote the migration of PCa cells using an in vitro co-culture model, and the concentrations of IL-8 in the co-culture supernatants were significantly increased. The expression of IL-8 was increased in PCa tissues, especially in the advanced stage, and was positively associated with tumor grade. Finally, the serum level of IL-8 was higher in patients with PCa than in patients with BPH. Moreover, the serum expression of IL-8 was significantly higher in some patients with PCa with low PSA, as confirmed in previous studies. 31 Meanwhile, untreated patients had a higher level of IL-8, which could predict a higher rate of metastasis. These data suggested that M2 macrophages and IL-8 play important roles in the development of PCa, IL-8 is a potential indicator for PCa diagnosis, and its serum level is a biological target for PCa treatment, which could help to extend the survival time of patients significantly.
However, this study had some limitations. First, although acute inflammation and other tumor diseases were excluded from the serum cases, other unknown diseases could have affected IL-8 serum concentrations. Second, the number of serum cases was relatively small, and differences between individual cases existed. Third, some patients were subjected to pathological examination at a different hospital, leading to incomplete pathological data, which made our analysis difficult. Further, due to geographical constraints, we did not collect serum from other races to verify this result, and future work should focus on this point. Despite these limitations, we believe that the identification of IL-8 in this context, from a new perspective of a co-culture system that simulates the in vivo tumor microenvironment complemented by validation using patient tissue samples, highlights the seminal contribution of this study both in terms of fundamental biology and its clinical and translational significance. The diagnostic use of serum IL-8 combined with PSA could reduce the number of patients with prostate disease being undiagnosed. We are currently attempting to study outpatients based on general screening to further evaluate the efficacy of IL-8 combined with PSA for the diagnosis and prognosis of PCa through continued follow-up. Future studies will focus on the mechanism underlying the effect of various cytokines or mediators related to the TME, as well as validation using a co-culture model of PCa cells.
In summary, we found that IL-8 secreted via bidirectional communication is associated with the malignant progression of PCa and is a potential biomarker for PCa diagnosis and treatment. However, whether IL-8 combined with PSA can reduce pathological examinations and improve the sensitivity and specificity of diagnosis needs to be further analyzed. More studies should be conducted in the future to address this.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Standards
This study was approved by Yongzhou Central Hospital Ethics Board on September 17, 2020 (NO.2020091701).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Hunan Province (grant number 2020JJ5558).
