Abstract
Introduction
Gastric cancer (GC) is the fourth deadliest cancer worldwide. 1 It has a well-recognized infectious etiology 2 with Helicobacter pylori 3 (H pylori) and Epstein–Barr Virus (EBV) 4 being the most often linked agents. Gastric cancer pathogenesis also has an inflammatory component, and H pylori infection is considered the main cause of mucosal inflammation that drives the neoplastic pathway. 5 Over the last century, the declining prevalence of H pylori infection in many parts of the world has resulted in a declining incidence of GC. According to the Lauren classification, there are 2 major histologies of GC: the intestinal- and diffuse-types. These 2 types have different natural histories, the former evolving as progressive inflammatory lesions starting with a nonatrophic gastritis (NAG), which becomes atrophic gastritis (AG), intestinal metaplasia (IM), dysplasia (DYS), and cancer (a sequence known as Correa's cascade), 6 while for the latter there are not known intermediate pre-cancerous lesions, although it still originates from an inflammatory NAG.
Gastric cancer clinical course varies with stage at diagnosis. In the United States, cancer diagnosed before spreading outside the stomach has a 5-year survival rate of 70.3%. On the other hand, when it has already spread to surrounding tissues and/or regional lymph nodes, or to distant parts of the body, the 5-year survival decreases to 32% and 5.8%, respectively. 7 Unfortunately, most patients are diagnosed at advanced stages of the disease in both developed and developing nations. Early detection screening and H pylori eradication programs have been implemented in South-East Asian countries with a high incidence of GC to reduce its mortality. 8
Epstein–Barr Virus is a lymphotropic herpesvirus that infects about 95% of the world's population. The virus is transmitted orally through saliva droplets infecting both B lymphocytes and epithelial cells of the oronasopharynx. Primary infection usually happens during childhood and persists for the lifetime of the infected individual, with memory B cells being the site of lifelong persistence. 9 Infection of the epithelial cells increases the amount of viral particles released into saliva facilitating transmission to new hosts.10,11 It is not clear how EBV infects gastric epithelial cells and in which step of the Correa’s cascade EBV is already present in the progressing gastric lesion.
Epstein–Barr Virus exhibits a bipartite life cycle alternating between latent and lytic phases. During latent infection, most of the viral genome is epigenetically silenced, leaving the expression of only a handful of genes. Suppression of viral gene expression facilitates persistence since the virus can hide from antagonistic host immune responses. Latency is the preferred viral program upon infection of B lymphocytes, and it is the program found in EBV-associated neoplasms as several of the latent genes exhibit oncogenic activity. 12 Latent expression is limited to the EBV nuclear antigens (EBNA1, EBNA2, EBNA3A, 3B and 3C, and EBNA-LP (leader protein)) and the latent membrane proteins (LMP1, LMP2A, and LMP2B). There are also different latent transcriptional programs. There is a latency III program in which all latent proteins are expressed, while viral gene expression is more restricted in latencies II and I and completely absent in latency 0. EBNA1 remains expressed during latencies III to I, together with several noncoding transcripts. 11 The latency III program induces a strong opposing immune response and is mainly observed during primary infection or in immunocompromised individuals. In contrast, latency 0 is the least immunogenic and is the transcriptional program found in persistently infected memory B cells 9 and the one predominantly present in healthy EBV carriers. 9
Epigenetic repression is lifted during the viral lytic replication cycle. Here, viral gene expression is extensive and ends with the assembly and release of infectious viral particles. Lytic reactivation is regulated by the concerted expression of key genes. For instance, the immediate early genes are master transcription factors of the lytic cycle, such as ZEBRA (also known as BZLF1) and BRLF1. These transcription factors in turn activate early lytic genes, many of which encode proteins that replicate the viral genome. One example of an early gene is EA-D (diffuse early antigen, also known as BMRF1), the viral DNA polymerase processivity factor. Finally, late genes are structural proteins that form the infectious particle, including those present in the viral capsid or envelope, such as the viral capsid protein (VCA).
Serology-based studies support the causal role of EBV in associated malignancies. Epstein–Barr Virus antibodies serve as markers of disease or the risk that an individual carries to develop a particular neoplasm. Antibodies are also informative of the specific stages of the viral life cycle that are participating in the oncogenic pathway. For instance, anti-EBNA antibodies have been found to be associated with EBV-positive lymphomas, arguing the importance of latency programs in virally induced lymphomagenesis.13,14 On the other hand, antibodies against lytic cycle proteins are more often associated with nasopharyngeal carcinoma (NPC). 15 Indeed, studies conducted in regions of the world endemic for NPC have shown that individuals at risk present unusually elevated levels of anti-VCA IgA antibodies.16–18 To date, there are not known markers of GC diagnosis based on EBV infection. Here, we conducted a comprehensive review of the serological studies performed in patients with GC and associated gastric lesions to assess whether there are anti-EBV antibodies that can also serve as disease markers, uncover their identity, and better understand the participation of EBV in the genesis of this neoplasm.
Search Strategy
This study was carried out according to PRISMA reporting and has been registered in PROSPERO [CRD42022384462]. We performed a systematic search of the literature available on PubMed, SciELO, Scopus, and Google scholar using the terms (“Epstein–Barr Virus”) and (“Serology” or “Antibodies” or “Serum”) and (“Gastric Cancer” or “Stomach cancer”) up to January 2023. We also searched the reference lists of all retrieved studies without finding any additional research article. After removing duplicates, we screened the titles and abstracts of the remaining publications. Studies were included according to the following criteria: studies published in peer-reviewed journals, research articles that measured qualitatively or quantitatively any anti-EBV antibody in individuals with GC or precursor lesions, and studies written in English or Spanish language. To determine the EBV status of the tumor, only those studies that performed EBER-in situ hybridization (EBER-ISH) were taken into account, since this technique is the gold standard for determining an EBV-positive tumor. PCR-based studies to separate EBV-associated gastric cancer (EBVaGC) from EBV-nonassociated gastric cancer (EBVnGC) were not considered as this technique does not distinguish whether the virus is present in the tumor cell or in tumor-infiltrating B lymphocytes. Two researchers independently screened the studies selected and any disagreement on the eligibility criteria was settled through discussions among all 4 authors. After reading the articles, the same 2 researchers extracted the following information: first author, year of publication, study aim, number of patients analyzed, country where performed, gastric disease considered, method for assigning tumor EBV status, antibodies tested, and statistical significance of reported associations. See Figure 1 for the strategy for filtering the studies that did not meet the inclusion criteria.

Flowchart of the study selection process for the systematic review.
Results
Our systematic review identified 844 papers from the 4 databases. Based on title and abstract, we retrieved 21 full articles about EBV serology and gastric lesions. After full-text reading, we eliminated 3 papers because the authors did not disclose a statistical analysis or raw data supportive of significant or nonsignificant associations.19–21 Two studies22,23 were excluded because they duplicated patients reported in the subsequent publication by Varga et al, 24 ending up with 16 different articles from 12 different research teams for this review (Figure 1, Table 1). Overall the studies included 9735 individuals sorted in the following groups: 215 EBVaGC, 302 EBVnGC, 2571 GC of unknown EBV status, 814 noncancerous gastric diseases (AG/IM/Dys), and 5833 controls (820 NAG, 93 with nonspecified dyspeptic symptoms and 4920 healthy blood donors). The 5833 controls are the contrasting groups for the statistical analyses.
Characteristics of the 16 Studies Included.
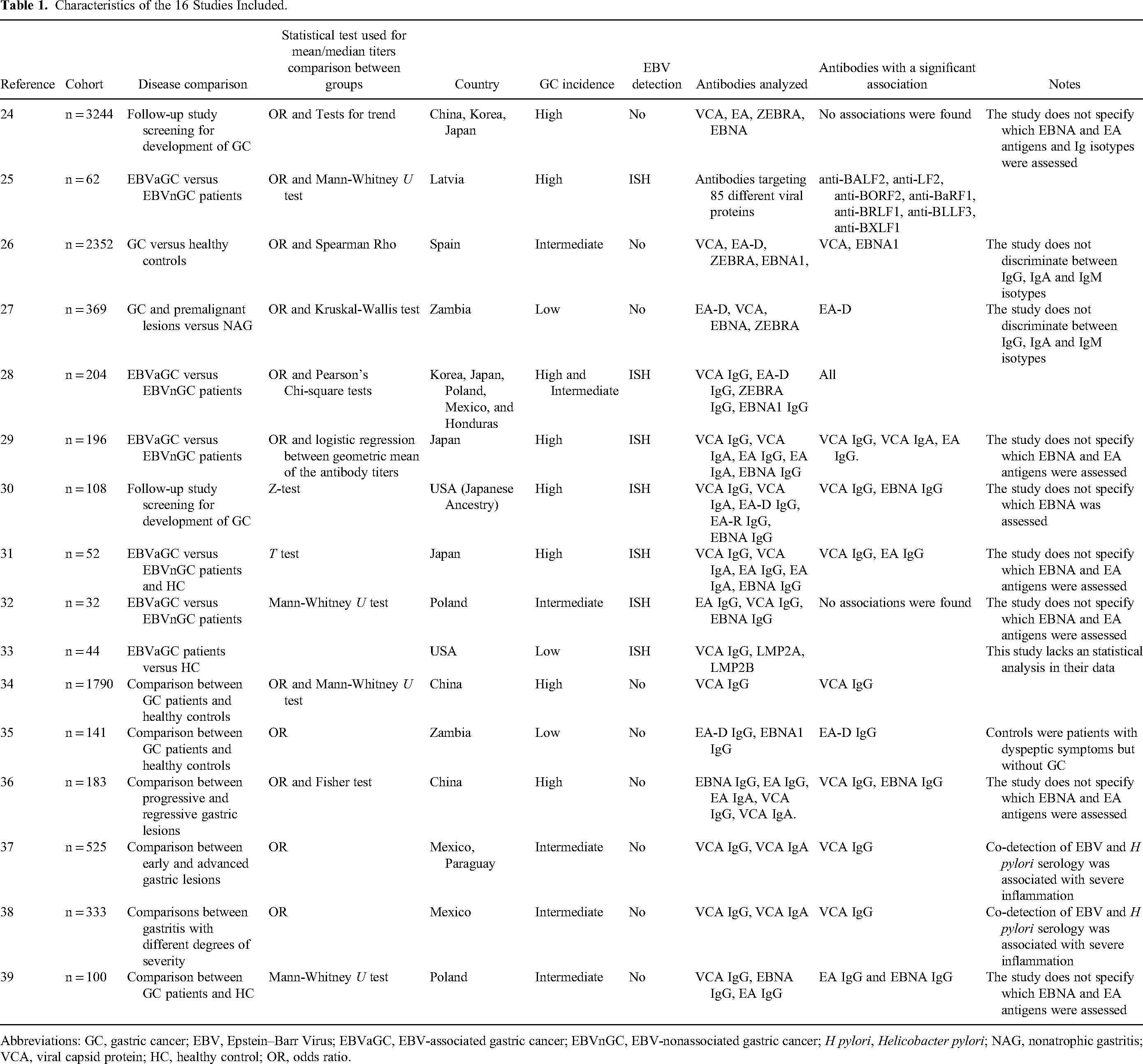
Abbreviations: GC, gastric cancer; EBV, Epstein–Barr Virus; EBVaGC, EBV-associated gastric cancer; EBVnGC, EBV-nonassociated gastric cancer; H pylori, Helicobacter pylori; NAG, nonatrophic gastritis; VCA, viral capsid protein; HC, healthy control; OR, odds ratio.
For a better understanding and management of the data, we grouped the selected studies into 2 categories: (i) studies comparing patients with gastric tumors of known EBV status (Figure 2), and (ii) studies comparing patients with GC or gastric lesions of unknown EBV status (Figure 3). The various antibodies tested in these studies were directed against lytic cycle antigens, including VCA, EA-D, EA-R (early antigen restricted, also known as BHRF1), and ZEBRA, as well as EBNA1 and LMP2A as markers of latency. One study 25 assessed the entire EBV immunoproteome encompassing all potential lytic or latent EBV antigens (see below). Some studies evaluated different antibody isotypes, but IgM was generally negative, so only IgA and IgG isotypes provided meaningful data. Three studies24,26,27 that did not specifically indicate the isotype tested were assumed to represent IgG data. When the authors did not specify which anti-EBNA was measured, we assigned it to EBNA1. Likewise for EA, when the study did not disclose if the antibody analyzed was targeting the diffuse or restricted antigen, we assigned it to EA-D. Those studies in which we inferred the immunoglobulin isotype or the EBV antigen are indicated in the legends of Figures 2 and 3 and in Table 1.

Association between anti-Epstein–Barr Virus (EBV) serology and EBV-associated gastric cancer. Two types of statistical tests are represented, comparisons between antibody levels and odds ratios based on either seropositivity or levels above specified cut-off values. Left panel: Circles and squares indicate a significant or nonsignificant difference in median antibody titers, respectively. Right panel: Forest plot showing reported odds ratios (ORs) and respective 95% confidence interval and symbol area representing the number of subjects included for associations with high antibody titers. The studies by Shinkura et al, 29 Levine et al, 30 Imai et al, 31 and Szkaradkiewicz et al 32 did not specify which EBNA or EA antigen was assessed and were assigned to EBNA1 and EA-D, respectively.

Association of anti-Epstein–Barr Virus (EBV) serology and gastric cancer or preneoplastic lesions of unknown EBV-status. Two types of statistical tests are represented, comparisons between antibody levels and odds ratios based on either seropositivity or levels above specified cutoff values. Left panel: Circles and squares indicate a significant or nonsignificant difference in median antibody titers, respectively. Right panel: Forest plot showing reported odds ratios (ORs) and respective 95% confidence interval and symbol area representing the number of subjects included for associations with high antibody titers. The studies by Aragones et al, 26 Varga et al, 24 and Kayamba et al 27 did not specify the antibody isotype and were assigned to IgG. Schetter et al, 36 Varga et al, 24 and Pachnia et al 39 did not specify which EBNA or EA antigen was assessed and were assigned to EBNA1 and EA-D, respectively.
Overall, of the 16 papers included, 14 showed statistically significant associations and 2 did not find an association. Most of the significant associations involved antibodies directed against lytic cycle antigens; however, the particular associated EBV antibody varied among studies (see text below and Figures 2 and 3).
We found 7 papers comparing EBVaGC versus EBVnGC or healthy controls (HCs) (Figure 2). A total of 698 individuals (215 EBVaGC, 302 EBVnGC, and 181 HC) from 7 countries (276 from Japan, 152 from the USA, 73 from Poland, 63 from Korea, 62 from Latvia, 45 from Honduras, and 27 from Mexico) were enrolled in these studies.25,28–33 Table 1 also specifies whether these studies were performed in countries with high or low GC risk and the type of statistical tests used for the analysis. Of note, the study from the USA by Levine et al involved men of Japanese ancestry, and it is considered to represent a high-risk population in the article, even though the USA is of low incidence. 30 Figure 2 presents EBV associations based on odds ratios (ORs) of antibody levels above a cutoff (right panel) and on statistical significance of differences between the medians of antibody titers (left panel). The latter were more informative for markers such as anti-EBNA1 or anti-VCA in which close to 100% of the subjects were seropositive, and thus, cutoff titers were not easily established to estimate ORs.
In general, antibodies against lytic antigens VCA and EA-D were consistently associated with EBVaGC. Two studies also found an association with anti-EBNA1.28,30 Also, the IgG isotype was more consistently associated than IgA. One study 25 used a novel protein microarray technology that allowed the investigators to screen a wider panel of antibodies against all open reading frames present in the EBV genome. They found antibodies against BALF2, LF2, BORF2, BaRF1, BRLF1, BBLLF3, BXLF1, BDLF2, and BOLF1 elevated in EBVaGC; all of them except anti-BDLF2 and BOLF1 were validated in an independent cohort of EBVaGC patients, and all with ORs > 5.0. The combination of anti-BALF2, anti-BORF2 and anti-LF2 efficiently discriminated between EBVaGC and EBVnGC with an area under the receiver operating characteristics curve of 0.88 (95% CI, 0.78-0.98). Interestingly, all these EBV antigens belong to the EBV lytic cycle.
In the study by Levine et al,
30
patients were followed up for a median of 13.7 years and serum samples taken at baseline were used to compare patients who did or did not develop GC. Although the ORs were not significant, patients who developed EBVaGC had higher anti-VCA IgG and anti-EBNA IgG titers than patients who developed EBVnGC or remained cancer-free (P
Several studies also compared GC of unknown EBV status versus other gastric lesions. There were 9 of these studies,24,26,27,34–39 which included NAG, AG, IM, DYS, GC, and HC (Figure 3). A total of 9037 individuals (2571 with GC, 1328 with gastritis, 246 with IM, 60 with DYS, 93 patients with unclassified dyspeptic symptoms but without evidence of GC, and 4739 HC) were recruited from 8 countries (3697 from China, 2352 from Spain, 804 from Japan, 716 from Korea, 642 from Mexico, 510 from Zambia, 216 from Paraguay, and 100 from Poland). In general, there was a significant association between EBV serology and advanced lesions; the antibody most frequently associated was anti-VCA IgG, although significant differences were sometimes reported for VCA IgA, EA-D IgG, and EBNA1 IgG (Figure 3).
Three studies26,34,39 compared GC versus HC, reporting associations with high titers to VCA IgG, VCA IgA, EA-D IgG, and EBNA1 IgG. The study by Wang et al 34 also found significantly higher levels of VCA IgG in GC patients than in those with NAG. Two studies34,37 only found associations with GC of the intestinal type but not with the diffuse type, suggesting the EBV and GC association may be restricted to Correa’s cascade pathway. The study by Wang et al 34 reported a very low frequency of EBV seropositive subjects (35.2%), contrary to the rest of the studies and the usual finding that about 95% of adults are EBV carriers.
Both studies by Kayamba and collaborators27,35 compared patients with GC and patients with either NAG or patients with unclassified dyspeptic symptoms. Both comparisons found a positive association with EA-D. One of these studies 35 also observed a correlation with EBNA1. Cárdenas-Mondragón et al 37 compared GC or premalignant lesions versus early NAG, reporting significantly increased titers of VCA IgG in both case groups. This group also found an association between VCA IgG and severe infiltration of immune cells in gastric lesions. Another study 38 from the same group documented a similar association between high VCA IgG and severe immune infiltration in pediatric patients with NAG.
Two studies had a longitudinal design. One study 36 associated anti-EBV antibodies with the progression of precancerous lesions in a Chinese high-risk cohort. The study reported that patients that progressed from NAG to IM or DYS had higher anti-VCA IgG and/or anti-EBNA IgG titers at baseline than patients that did not progress or that had lesions that regressed. The other study 24 had a similar design, also analyzing progression to GC in patients from several high-risk Asian countries and, notably, had the largest number of patients among the studies included in this review. However, the authors did not find any difference in the anti-EBV antibody titers at the beginning of the follow-up period (median 5.9 years) between those that developed GC and those that did not.
Discussion
Gastric cancer remains one of the deadliest cancer types. Because symptoms are unspecific and often confused with other gastric diseases, and we do not have noninvasive tools for early detection, GC is usually diagnosed at late stages with a dismal prognosis. High anti-EBV antibody titers have been found in patients with associated malignancies, especially NPC.14–17 Here, we conducted a systematic review of EBV serological studies in search of clues about its participation in the transformation of the gastric mucosa, and to recognize whether specific anti-EBV antibodies may mark individuals with or at risk of developing GC.
We found 16 different studies that included 9735 individuals. Antibodies targeting 6 different viral antigens were assessed in these studies, including 4 directed against the lytic cycle proteins ZEBRA, EA-D, EA-R, and VCA, and 2 against the latent proteins EBNA1 and LMP2. One additional study examined reactivity to 89 different EBV antigens derived from all open reading frames in the viral genome. Fourteen of these studies found that seropositivity or elevated antibody titers to EBV lytic cycle proteins were significantly associated not only with EBVaGC but also with GC and preneoplastic lesions of unknown EBV status. On the contrary, only 5 out of 12 studies found a positive association between anti-EBNA1 and EBVaGC or advanced lesions. These studies suggest that antibodies against lytic cycle proteins are indicative of GC and could be used as markers of the GC risk.
Lytic antibodies also mark individuals at risk to develop NPC. Although some evidence supports the importance of an abortive lytic cycle for lymphomagenesis, 40 VCA is of late expression implying a complete lytic cycle. Thus, evidence from patients with 2 different cancers suggests that EBV reactivation in B cells facilitates infection of epithelial cells. This concept is supported by in-vitro models of EBV infection, in which direct infection of epithelial cells with viral particles is highly inefficient, while co-cultures of EBV-infected B cells enhance the number of infected epithelial cells.41,42 This experimental observation aligns with the capacity of EBV to switch cellular tropism according to the protein composition of its envelope. EBV particles released from B cells lose the envelope protein gp42 and exhibit an enhanced tropism for epithelial cells. On the contrary, viruses released from epithelial cells retain gp42 and are more infectious for B cells. 43 In this scenario, antibody titers against lytic VCA and EA-D may mirror the level of viral reactivation in B lymphocytes, since EBV is persistently maintained in a latent state in these cells. 9 Thus, we can argue that viral reactivation in B lymphocytes is an important step leading to epithelial cell infection and the development of epithelial lesions.
A remarkable difference between NPC and GC is that, for the former, the best antibody markers are of the IgA isotype, whereas 3 of the 4 studies of IgA antibodies in the present report did not find an association with GC. There is not an obvious explanation for this difference. On the one hand, the oronasopharyngeal cavity presents an abundance of associated lymphoid tissue, while it is scarce in the gastric mucosa, which may limit the potential number of B lymphocytes reacting against EBV and switching the antibody isotype. On the other hand, the virus released in the oronasopharyngeal compartment is infective and the source of transmission to new hosts. Perhaps in the gastric mucosa, the virus released into the basolateral face of the gastric epithelial cells only generates IgG responses, while the virus released into the lumen of the stomach, which should preferentially generate IgA antibodies, may be rapidly inactivated by the gastric juice losing antigenicity. Another possibility is that the source of the antigenic stimulus is the B lymphocytes that are dying in the basolateral compartment of the gastric mucosa due to the EBV lytic reactivation, rather than the viral particles themselves.
Five out of 6 studies found higher antibodies in EBVaGC than EBVnGC. However, when EBVnGC or neoplastic and preneoplastic lesions of unknown EBV status were compared with NAG or with healthy blood donors, there was also a significant association with high EBV antibody titers. There are 2 potential explanations for this result: (1) EBV reactivation contributes to the inflammatory process that drives the appearance and progression of gastric lesions, or (2) it is the inflammatory process that drives EBV reactivation and enhances the production of antibodies directed against lytic proteins. In this latter scenario, anti-EBV lytic antibodies mirror inflammation but EBV does not contribute to it. There is some evidence supporting the former, Cárdenas-Mondragón et al found an association between EBV serology and the severity of the immune cell infiltrate in adult and pediatric gastric lesions.37,38 Also, Cárdenas-Mondragón et al, 37 Aragonés et al, 26 and Wang et al 34 reported an exclusive association between EBV serology and GC of intestinal type, which is the most dependent on chronic inflammation, but not with the diffuse type.
EBV has also been linked to benign gastric conditions; 2 different groups have reported associations between EBV serology and peptic ulcers. One study 44 found significant associations of VCA IgA and IgG antibodies with gastric and duodenal ulcer disease, and the other study 45 found higher VCA IgG in patients with ulcer disease than in patients with reflux and dyspepsia. Although these observations were noted with interest, we did not display these associations in tables and figures because ulcer diseases are not generally considered starting or intermediate lesion on the GC pathway.
Considering a potential causative role for EBV in the inflammatory process that drives the Correa’s cascade of gastric lesions, these data argue that EBV may also be contributing to the development of at least a fraction of the GCs that are classified as negative by EBER-ISH. Thus, EBV may contribute to the etiologies of EBVaGC and GC overall through 2 mechanisms that are not mutually exclusive; one is through a persistent epithelial cell infection with the expression of latency-restricted viral oncogenes, and the other is indirect through promoting an inflammatory microenvironment similar to the H pylori-promoted damage of the gastric epithelium. The former may be more common in the diffuse type of GC, and the latter in the intestinal type. More studies using in vitro models and patient cohorts are needed to elucidate the role of EBV in the inflammatory process that drives Correa’s cascade of gastric lesions.
Study Limitations
The main limitation of this systematic review is the relatively limited number of studies addressing EBV serology and gastric lesions. Another limitation is the heterogeneity found in each study, for instance, the gastric lesions studied and the groups used as controls for the statistical comparisons. Also, most studies do not disclose the H pylori infection status, the anatomical location of the lesions, the history of the patients’ treatments, whether there were immunosuppressed individuals in the cohorts, etc. These limitations are particularly significant for the studies of EBVnGC and preneoplastic lesions. More rigorous approaches with more homogeneous cohorts are needed to validate the observations to date, together with in vitro mechanistic molecular studies. Although antibodies provide evidence of exposure, they are not indicative of the anatomic site of infection. Additional understanding may be gained by combining serology with molecular methods assessing viral load and location in gastric lesions.
Conclusions
Anti-EBV antibodies directed against lytic antigens are associated with EBVaGC as well as with GC and other advanced gastric lesions irrespective of EBER-ISH positivity. These observations suggest that EBV antibodies may be markers of advanced gastric disease, neoplastic, and preneoplastic, regardless of whether EBV plays a direct causative role in the development of the lesion. Determining the mechanisms underlying these associations would be additionally interesting and important, H pylori pharmacological eradication has been proposed to stop the progression of preneoplastic lesions to GC but we do not have antiviral drugs to treat EBV-associated cancers. Hypothetically, the observed association with markers of viral reactivation may support a potential benefit from members of the gancyclovir family of drugs to prevent the progression of gastric lesions to advanced irreversible stages. More studies are needed to corroborate and extend these ideas and to establish the antibody combinations and thresholds indicative of elevated risks for EBVaGC and for GC overall.
Footnotes
Abbreviations
Acknowledgments
Fontes-Lemus JI is a master student from Programa de Posgrado en Ciencias Biológicas, Universidad Nacional Autónoma de México (UNAM) and acknowledges CONACYT for the fellowship (800902. CVU 1025658).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was funded by CONACyT “FORDECYT- PRONACES” (project number 303044) “PRONAII-7-VIRUS Y CÁNCER,” Fondo de Apoyo a la Investigación, Hospital Infantil de México Federico Gómez (project number HIM/2016/002) and by the Intramural Research Program of the National Cancer Institute, NIH.
