Abstract
Introduction
As the fourth most common malignant tumor worldwide, gastric cancer (GC) has a poor prognosis and a high mortality rate, representing the third leading cause of cancer-related death.
1
GC features a high degree of malignancy, rapid development, strong invasiveness, easy recurrence, and poor prognosis. Among these characteristics, invasion and metastasis are important malignant features of GC, and the main causes of death in patients with this disease.
2
A series of related studies of GC have shown that excessive salt intake, smoking, alcohol consumption, and
So far, common treatments for GC included surgical resection, radiotherapy and chemotherapy. Radiotherapy and chemotherapy are often used to treat patients with advanced gastric cancer who can't tolerate surgery or relatively low surgical value, which can improve the overall survival rate to some extent, but there are many reports of tumor recurrence or metastasis. For example, a review of primary tumor uveal metastasis reported that uveal metastasis originated from breast cancer, lung cancer, gastrointestinal tumors and other cancers. The overall prognosis of patients with uveal metastasis was poor, with a 5-year survival rate of 23%. The survival rate of pancreatic cancer with eye metastasis was the lowest, while the overall survival rate of patients with eye metastasis of lung cancer was relatively good. In addition, the basic research on the progression and metastasis of GC was reported. Exosomes, an important group of extracellular vesicles, have important roles in the initiation, progression, and metastasis of GC. 7 Lymph node metastasis is the main route of metastasis in the process of tumor migration. Most patients diagnosed with GC have lymph node metastasis; even in early GC, the proportion of patients with lymph node metastasis is 5.7%–29.0%. 8 In addition to lymphatic metastasis, other common metastasis pathways of GC include direct invasion, hematogenous metastasis, and peritoneal implantation metastasis. Common sites of GC metastases include the peritoneum, liver, spleen, lung, and lymph nodes; 9 however, the number of cases of ocular metastasis (OM) in GC is very small. One study reported metastasis of GC in the extraocular muscle; 10 in this case, the patient presented with poorly differentiated adenocarcinoma and the main ocular clinical manifestations were eyelid swelling, diplopia, and retroorbital pain. OM in patients with GC is often an indicator of poor prognosis, as the cancer cells may have metastasized to multiple sites throughout the body. Metastasis of GC is an independent prognostic risk factor for GC patients, and seriously influences treatment and prognosis.
The occurrence of OM in patients with GC has a great impact on their quality of life; however, the specific processes and mechanisms underlying of metastasis of primary GC remain unclear, despite frequently resulting in substantial patient suffering. The circular RNA, circUBA1, can promote GC proliferation and metastasis by regulating the effects of miR-375 and TEAD4; 11 however, this factor is seldom assessed in clinical practice, because of the technical difficulty and complexity of its method of measurement. Serological indictors (SI), as indicators routinely detected in clinical practice, have specific advantages for use in the diagnosis of GC metastasis, and can be important indicators for diagnosis of GC metastasis by the gold standard methods of auxiliary pathology. Cancer markers refer to substances produced directly by tumor cells or by non-tumor cells through tumor cell induction. The detection of tumor markers can predict the existence, pathogenesis, and prognosis of tumors. 12 In our study, we investigated whether changes in cancer biomarkers and SI can be used to differentially diagnose OM in patients with GC. In addition, by studying the correlations among various risk factors and the occurrence of OM in GC, we established a relatively accurate index to distinguish between patients with and without OM, to facilitate possible diagnosis at an early stage of GC metastasis, conduct timely intervention, and further improve the prognosis and quality of life for patients with GC.
Patients and Methods
Study Design
This retrospective cohort study was approved by the Medical Research Ethics Committee of the First Affiliated Hospital of Nanchang University and the approved number was cdyfy20170411. The study included older adult patients (age ≥ 60 years) diagnosed with GC between June 6, 2003 and May 15, 2019. A total of 1165 patients with GC were recruited to this study randomly. Patients with secondary GC were excluded. GC was confirmed in all patients by surgical resection and histopathological biopsy. Subjects were divided into OM and non-ocular metastasis (NOM) groups. The inclusion criteria for the OM group were eye metastases in patients with GC diagnosed by CT and MRI and confirmed by histology and cytology. Patients with primary eye tumors (benign or malignant) were excluded from the OM group. All the subjects understood the purpose and specific content of our study, and signed a paper informed consent form. The investigators have de-identified all patient details in this study. The reporting of this study conformed to STROBE guidelines. 13
Data Collection
Medical records of all participants were collected to acquire basic information about their age, sex, and pathological tumor type. In addition, serological data were collected from both groups of subjects, including: hemoglobin (HB), calcium, alkaline phosphatase (ALP), total cholesterol (TC), triglyceride (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), apolipoprotein A1 (ApoA1), apolipoprotein B (ApoB), alpha fetoprotein (AFP), carbohydrate antigen-724 (CA724), carcinoembryonic antigen (CEA), CA199, CA125, CA153, cytokeratin-19 fragment (CYFRA21-1), and lipoprotein(a) (Lp[a]) levels.
Statistical Analyses
The chi-square test was used to compare the sex distribution and pathological types between the OM and NOM groups. A two-tailed independent sample t-test was used to assess differences in age and levels of various SI between the two groups. A binary logistic regression model was used to analyze the significance of the associations of various SI with OM in older adult patients with GC. Finally, receiver operating characteristic (ROC) curves were used to analyze the significance of each indictor in differentiating OM and NOM. The statistical threshold used was
Results
Demographic and Clinical Characteristics of Participants
The total number of GC patients enrolled in this study was 1165, including 13 in the OM group and 1152 in the NOM group, with 1.1% of patients with GC diagnosed with OM. The mean age of patients in the OM group was 67.9 ± 6.5 years, while that in the NOM group was 68.3 ± 15.7 years (

Clinical characteristics of gastric cancer OM patients and NOM patients.

The HE staining and IHC images from gastric cancer OM patients. (A) gastric cancer (HE × 200). (B) Villin ( + ) (SP × 200). The tissue was collected from OM site of gastric cancer patients.

Imaging and pathological data of OM in GC patients. (A) Ultrasound examination of ocular. The relative shape of the hypoechoic mass is seen. The posterior echo of the mass is diminished, and there is an infiltration around the mass. (B) Fundus photography of an OM patient with GC. (C) Fluorescence leakage and low fluorescence were observed in the lesions. Blood vessels are visible in the field of vision.
The Clinical Characteristics in OM Group and NOM Group.

OM, including intraocular metastasis and eyelid metastasis.
Chi-squared test.
Student’ s t-test.
Comparison between the OM and NOM groups.
Data showed as mean ± standard deviation or n.
Differences in the Levels of Various Serological Indicators and Risk Factors Between the 2 Groups
Compared with the NOM group, among all SI, LDL, CA724, and CEA levels were significantly higher in the OM group, while those of ApoA1 were significantly lower (
Differences in the Concentration of Various Serological Indicators Between OM and NOM.

Independent sample
Abbreviations: OM, ocular metastasis; NOM, non-ocular metastasis.
Risk Factors of OM in Elderly Patients With GC.

Binary logistic regression analysis.
Abbreviations: B, coefficient of regression; OR, odds ratio; CI, confidence interval; OM, ocular metastasis; GC, gastric cancer.
ROC Analysis to Determine the Value of LDL, ApoA1, and CA724 for Diagnosis of OM
The cutoff value was the value corresponding to the Youden's index at the most approximate index, where the Youden's index was equal to the sum of sensitivity and specificity minus 1. Through ROC analysis, we determined cutoff values for LDL, ApoA1, and CA724 of 3.98 mmol/L, 1.71 g/L, and 14.08 U/mL, respectively; CA724 had the highest AUC value. ROC curves to assess each indicator as a risk factor for OM in patients with GC are shown in Figure 4A. We also assessed the significance of the ratios of LDL to ApoA1 and CA724 to ApoA1, as well as various combinations of indicators, including: LDL + ApoA1, LDL + CA724, CA724 + ApoA1, and LDL + ApoA1 + CA724, as risk factor indicators for assessment of OM occurrence in patients with GC. Analysis of combined indicators demonstrated that LDL + ApoA1 + CA724 had the highest AUC value (0.936). Further, the AUC value of LDL + CA724 was regimen also reached 0.918 (Figure 4B). Among the above indicators, the sensitivity of CA724 and CA724/ApoA1 was the highest, reaching 1, while the specificity of the combination of LDL + ApoA1 + CA724 was the highest, at 0.89. All the above results were statistically significant (

The receiver operating characteristics (ROC) curves of risk factors for detecting OM in elderly GC patients.
The Cutoff Value, Sensitivity, Specificity, and AUC of Risk Factors for the Prediction of OM in Elderly GC Patients.
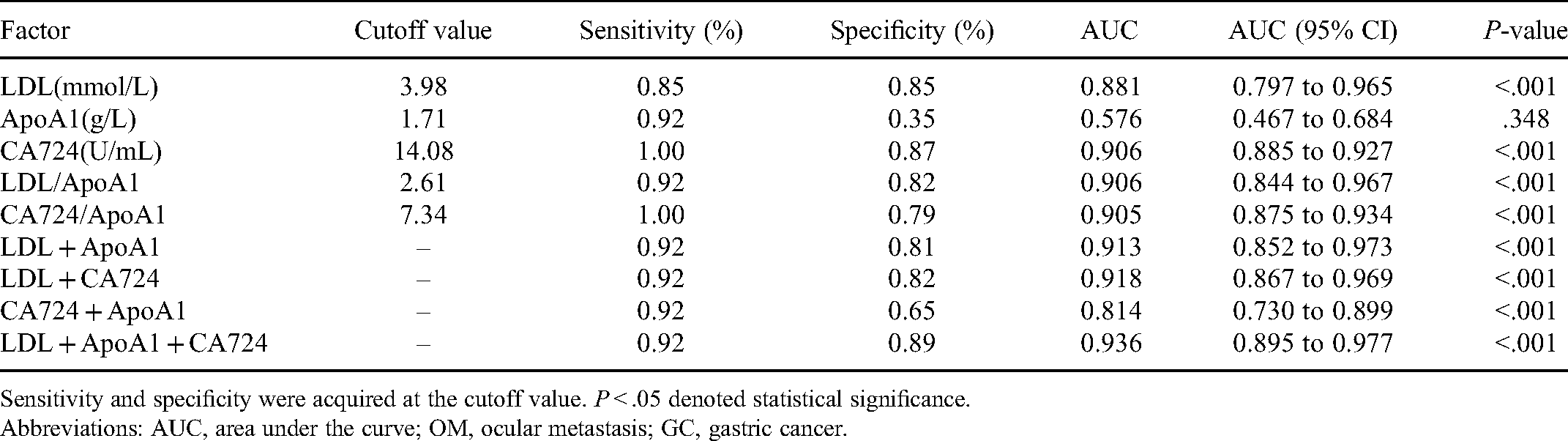
Sensitivity and specificity were acquired at the cutoff value.
Abbreviations: AUC, area under the curve; OM, ocular metastasis; GC, gastric cancer.
The AUC Value of Each Index Was Compared and Analyzed.

Statistical method: deLong test.
Abbreviation: AUC, area under the curve.
Discussion
In this study, we retrospectively analyzed data from 1165 patients with GC, and conducted a detailed study on pathological classification and risk factors for OM. Here, we found that the ratio of male to female patients with GC was unbalanced at 2.53:1. Based on relevant publications, we infer that factors influencing the high incidence in males include continuous stimulation of the cerebral cortex because of work pressure and mental stress, continuous excitation of the sympathetic nerve, and subsequent induction of chronic atrophic gastritis, gastric ulcer, and other precancerous lesions.
14
Moreover, the high concentrations of estrogen in females inhibits the expression of estrogen receptor ɑ36 (ER ɑ36) and the growth of GC cells, which has protective effects on the stomach.
15
At present, known risk factors for GC are primarily irregular diet and
For patients with early GC, the risk of metastasis is relatively low, and those without metastasis can undergo submucosal dissection or tumor removal using endoscopic technology. 17 In clinical practice, patients with GC are often at an advanced stage of disease on definite diagnosis, and a considerable number develop distant metastasis of cancer cells. Cancer-associated fibroblasts, an important component of various tumors, are involved in the invasion and metastasis of GC. 18 Numerous researchers have studied the risk factors for GC metastasis,19–25 and their findings are summarized in Table 6; however, there are few reports of OM of GC to date. Tadashi et al. reported a case of ptosis caused by orbital metastasis in a patient with advanced GC, diagnosed using CT and MRI technology, which are still widely used for clinical diagnosis of cancer metastasis. 26 In addition, another study reported a case of iris metastasis in a patient with GC undergoing chemotherapy. 27 In terms of clinical symptoms, iris metastasis of GC is relatively insidious, and easily misdiagnosed as iridocyclitis. In contrast, iridocyclitis is often accompanied by Tyndall's phenomenon in the cornea with anterior pus accumulation, blunted light response and post-pupillary rim adhesions. And some patients are also associated with systemic connective tissue disease. During clinical diagnosis and treatment, full integration of the patient's medical history and the results of examinations is required to ensure accurate judgments; however, in current clinical practice, it remains difficult to accurately identify OM of GC at an early stage. Further, CT and MRI are expensive examination methods, and there can be contraindications to their use, leading to limited sensitivity and specificity. In contrast, serological tumor markers are widely used in clinical practice, due to their advantages of good reproducibility, low cost, and easy access. To date, a considerable number of researchers have proposed various serological markers related to disease metastasis and prognosis including intraocular metastasis from bladder cancer; 28 however, there is a lack of specific SI for OM in older adult patients with GC. In our study, we collected serological data from 1165 patients with GC, including: HB, calcium, ALP, TC, TG, HDL, LDL, ApoA1, ApoB, AFP, CA724, CEA, CA199, CA125, CA153, CYFRA21 to 1, and Lp(a) and found that LDL, ApoA1, CA724, and CEA content differed significantly between the OM and NOM groups. Compared with the NOM group, LDL, CA724, and CEA levels were significantly higher in the OM group, while those of ApoA1 were significantly lower. In addition, LDL, ApoA1, and CA724 concentrations were independent risk factors applicable for differential diagnosis of OM by statistical analysis. Subsequently, we analyzed and determined the threshold values, sensitivity, specificity, and AUC values for each biomarker by constructing ROC curves, further confirming that LDL, ApoA1, and CA724 are important markers for OM in older adult patients with GC. Finally, we conclude that, at their respective thresholds of 3.98 mmol/L, 1.71 g/L, and 14.08 U/mL, LDL, ApoA1, and CA724 are indicators that patients with GC are at risk of developing OM. The ability to differentiate and diagnose OM could be further improved by combining changes in serological indicators with clinical imaging techniques, such as eye CT and MRI, to ensure timely treatment and improve patient prognosis.
The Risk Factors of Metastases of Gastric Cancer.

Abbreviations: NS, not specific; THBS4, thrombospondin 4; CEA, carcinoembryonic antigen; CA199, carbohydrate antigen-199; ENO1, alpha-enolase; HOXC10, homeobox C10.
LDL is a cholesterol-rich lipoprotein that carries cholesterol into peripheral tissue cells. Excess LDL is closely associated with atherosclerosis, myocardial infarction, stroke, and peripheral artery disease. In addition, study of a hyperlipidemia mouse model showed that LDL and tumor cell LDL receptor expression can mediate the growth and recurrence of breast cancer. 29 Further, LDL and VLDL can promote the proliferation, differentiation, and metastasis of breast cancer cells. 30 Moreover, LDL levels are reportedly positively correlated with the occurrence of liver metastasis of colorectal cancer, and LDL can promote inflammatory responses in the gastrointestinal tract and colorectal cancer progression by activating signaling pathways, including ROS and MAPK pathways. 31 In a study to explore the relationship between lipid levels and the risk and prognosis of GC, patients with GC had significantly increased LDL content, consistent with our results. 32
ApoA1 refers to the apolipoprotein attached to HDL and chylous glomeruli. ApoA1 plays an important role in physiological lipid metabolism, and is often regarded as a biomarker to predict the risk of coronary heart disease in individuals. 33 In a mouse MKN45 gastric cancer xenograft model, decreased plasma ApoA1 levels were closely associated with GC growth. 34 In addition, some prognostic studies of have demonstrated that the ApoB/ApoA1 ratio can be used as an independent prognostic factor for GC; the survival time of GC patients with ApoB/ApoA1 ratio ≥ 1 is significantly shortened. 35 Further, decreased ApoA1 levels have been associated with poor prognosis of patients with colorectal cancer and systemic inflammatory response. 36 In our study, the level of ApoA1 in the OM group was significantly lower than that in the NOM group, and the occurrence of OM in patients with GC often indicates poor prognosis, consistent with the conclusions of the above research.
As a GC antigen, CA724 is an important tumor marker for detection of GC and various gastrointestinal cancers. Due to its high sensitivity, CA724 is very commonly used as a clinical marker for detection of GC. In multivariate analysis based on the Chinese population, Chen et al. clearly demonstrated that, compared with other tumor markers, CA724 had the highest correlation with the occurrence of GC in the Chinese population. 37 Moreover, in a study of lymph node metastasis in patients with GC, lysyl oxidase, CEA, CA724, CA199, and CA125 were identified as significantly improving the sensitivity for detecting lymph node metastasis. 38 Monitoring alterations in CA724 concentration during chemotherapy in patients with advanced GC can be used as a predictor of tumor response. 39 In our study, we found that CA724 content in the OM group was higher than that in the NOM group. Based on our data, combined with previous reports, we conclude that patients in the OM group may have a relatively poor prognosis, since it is likely that many cancer cells have metastasized.
Finally, we tested combinations of LDL + ApoA1, LDL + CA724, ApoA1 + CA724, and LDL + ApoA1 + CA724, and the ratios of LDL to ApoA1 and CA724 and ApoA1, as indicators for ROC analysis, calculating AUC values for each combined indicator. LDL + ApoA1 + CA724 generated the maximum AUC value, followed by LDL + CA724. Hence, these two combination indicators can more accurately predict the occurrence of OM in GC.
Our study has some limitations. First, it is a retrospective cohort study; hence, there may be recall bias. Second, as eye metastasis of GC is very rare, only 13 cases of OM occurred among the 1156 patients with GC included our analysis. Further studies with larger sample sizes may provide more accurate results. Third, in our research results, only LDL, ApoA1 and CA724 were its risk factors, other indicators such as PGI, PGII, PGI/PGII were not included in our risk factor assessment, we will include more indicators to further improve the accuracy of the model. In addition, the data analyzed in our study did not include survival rates or follow-up findings, and specific patient prognoses were not analyzed. Finally, since all of our patients were from a single medical center, our results may not be representative of all populations with OM from GC.
Conclusion
In summary, risk factors for the occurrence of OM in GC include LDL, ApoA1, and CA724, and a combination of these three diagnostic indicators is most valuable for predicting the occurrence of OM in GC. Our research may assist in evaluation of the condition of patients over time, improve patient prognosis, and play a crucial role in the processes of disease screening, diagnosis, and treatment.
Footnotes
Acknowledgments
The preprint did not submit to other journal in the main document.
Ethical Statement
All research methods were approved by the committee of the medical ethics of the First Affiliated Hospital of Nanchang University (approval no. cdyfy20170411) and were in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All subjects were explained the purpose, method, potential risks and signed an informed consent form.
Data Availability Statement
All data is available on request.
Authors’ Note
WQS and SNW was involved in the data curation and writing of the original draft; TS and YS performed the data curation and formal analysis; HYS, QCY, and QYL was involved in the data curation, and writing, review, and editing of the manuscript; TS and YCP was involved in the study conceptualization and methodology design; WQS and RBL was involved in data validation and visualization; all authors have read and approved the final version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Key Research Foundation of Jiangxi Province (No: 20203BBG73059, 20181BBG70004); Excellent Talents Development Project of jiangxi Province (20192BCBL23020); Natural Science Foundation of jiangxi Province (20181BAB205034); Grassroots Health Appropriate Technology “Spark Promotion Plan” Project of Jiangxi Province (No: 20188003); Health Development Planning Commission Science Foundation of Jiangxi Province (No: 20201032); Health Development Planning Commission Science TCM Foundation of Jiangxi Province (No: 2018A060)
