Abstract
Ferroptosis is a novel cell death modality discovered in recent years that is different from apoptosis and necrosis. It is usually associated with changes in the regulatory signaling in multiple organelles and depends on iron. It is caused by an imbalance between the generation and degradation of intracellular lipid reactive oxygen species (ROS). In addition to increased levels of cytoplasmic ROS and lipids, decreased mitochondrial volume and thickened mitochondrial membranes are markers of ferroptotic death. Gastric cancer is a common malignant tumor, but few studies on the possible role of ferroptosis in gastric cancer have been reported. Although ferroptosis is involved in multifactor-induced carcinogenesis, studies have also shown the role of ferroptosis in the selective killing of tumor cells, thereby inhibiting tumor progression and metastasis. In this paper, the definition, characteristics, and regulatory mechanism of ferroptosis and its potential role in gastric cancer are discussed. Therefore, this review is expected to provide a reference for the treatment of diseases based on ferroptosis and provide a direction for future research on the pathogenesis and development of gastric cancer and the development of anticancer drugs.
Introduction
The types of cell death discovered thus far mainly include intrinsic apoptosis, extrinsic apoptosis, necroptosis, ferroptosis, pyroptosis, lysosome-dependent cell death, autophagy-dependent cell death, and immunogenic cell death.1–3 Cell death is an inevitable and important link under physiological and pathological conditions. 2 Research on the pathways of cell death has gradually revealed the main molecular mechanisms and biochemical characteristics of cell death. 4 Ferroptosis differs from necrosis and apoptosis, and as a recently discovered mode of atypical cell death, research into ferroptosis is valuable.5,6 An increasing number of studies have shown that ferroptosis is closely related to the regulatory growth of various tumor cells,7,8 such as gastric cancer, breast cancer, 9 head and neck squamous cell carcinoma, 10 cervical cancer, 11 pancreatic cancer, and clear cell renal cell carcinoma cells,12, 13 providing a series of potential targets for cancer treatment. Therefore, ferroptosis shows important translational significance in the treatment of gastric cancer and other digestive system tumors. 14 We present the following in accordance with the narrative review reporting checklist.
Methods
We prepared this review following the PRISMA 2020 statement. 15 This literature review and preliminary research were carried out at the library in Hebei Medical University. Key terms in a large number of research papers were searched, and the information collected was sorted, compiled, and summarized. Relevant research on molecular mechanisms, incidence data, and development process information was extracted from articles and combined with relevant descriptions of mechanistic molecules in gastric cancer tumors. Moreover, some data were combined to identify correlations and possible prognostic and related gene targets for use in future clinical diagnosis and treatment (Table 1).
The Search Strategy Summarized.

Definition of Ferroptosis
Ferroptosis is a novel iron-dependent programmed cell death modality that differs from apoptosis, necrosis, and autophagy. It is a result of an imbalance in cellular metabolism and redox regulation and is often associated with a variety of other forms of pathological cell death. 16 CD8+ T-cells play important roles in promoting iron-dependent death in tumor cells. 17
Ferroptosis Characteristics
In terms of cell morphology, the loss of membrane integrity is associated with nucleus swelling and mitochondrion shrinkage, increased mitochondrial membrane density, and outer mitochondrial membrane rupture. The nucleus shows no obvious morphological change in ferroptosis.
In terms of cell components, ferroptotic cells exhibit increased lipid peroxide and reactive oxygen species (ROS) levels. There are also some characteristic gene changes. The 3 important basic features are the activation of reduced iron (Figure 1), peroxidation of phospholipids containing polyunsaturated fatty acids, and loss of the repair signaling functions of lipid peroxides (Figure 2).
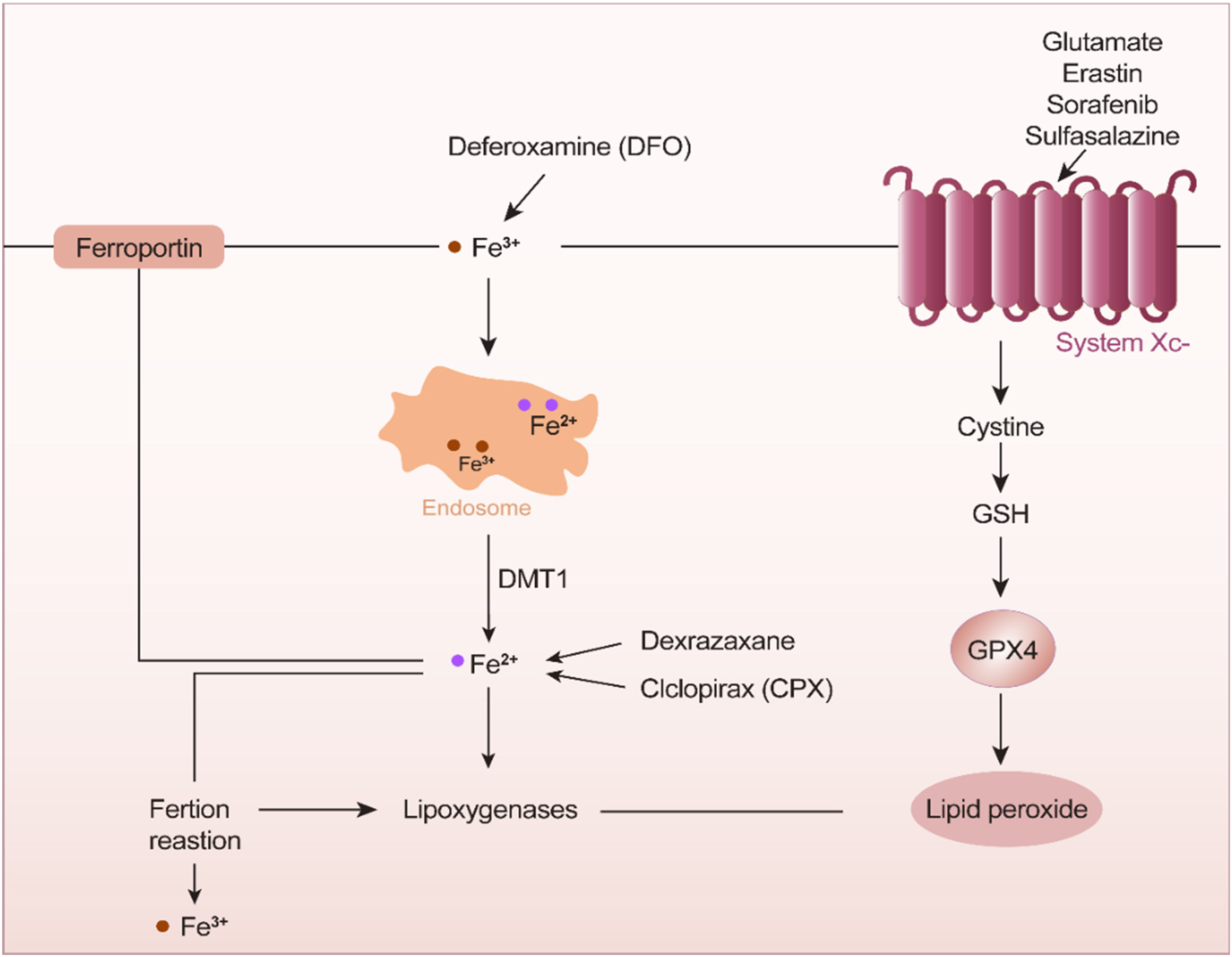
Ferric ions in endosomes undergo a reduction in the presence of deferoxamine. Iron reduction is mediated by the action of ciclopirox and System Xc−.

Under the action of transferrin, ionizing irradiation, and glutamic acid, iron is reduced, and glutathione (GSH) participates in these resulting ferroptosis-related reactions. After oxygen accepts electrons, the Fenton reaction proceeds in the presence of ferrous ions to form active hydroxyl radicals. Free radicals deprive unsaturated fatty acids of hydrogen atoms at long-chain double bonds, resulting in lipid peroxidation. Oxidative damage leads to leakage of liposome membranes.
Ferroptosis-Regulating Mechanism
Inactivation of GPX4 Caused by Glutathione Depletion
GPX4 is a key enzyme in lipid peroxidation. This key enzyme is critical for transport mediated by system Xc−. 18 The GPX family comprises many members, including glutathione peroxidase 1–8 (GPX1–8). Among these enzymes, GPX4 plays an important role in ferroptosis (Figure 3). Specifically, GPX4 significantly degrades small-molecule peroxides and plays a role in degrading complex molecules. Studies have shown that selenocysteine is an acidic component in the active center of the amino acids constituting GPX4, and the insertion of selenocysteine into GPX4 requires a special transporter, selenium-cysteine transfer RNA, which interferes with GPX4 activity, resulting in ferroptosis.17–20 Therefore, among the members in the large GPX family, GPX4 is inactivated, and along with glutathione (GSH) depletion, this inactivation has been found through research to be an important contributor to ferroptosis. 21

Ferroptosis mechanism.
GPX4 Inactivation
GPX4 is important for scavenging lipid peroxides, and its expression level exerts an important effect on the reduction of intracellular oxidized lipids. The regulation of GPX4 activity involves many important genes and proteins. As shown in Figure 4, in addition to the indirect action by GSH, which activates the GPX4 enzyme, GPX4 can be eliminated directly. For example, GPX4 inhibitors mediate a reduction in HMG-COA. GPX4 is a current hot spot in ferroptosis research, mainly because it is a key reductase in the ferroptosis pathway. 22 In addition to studies on its roles in metabolic pathways, other important targets of GPX4 and methods to identify them are important areas of exploration.
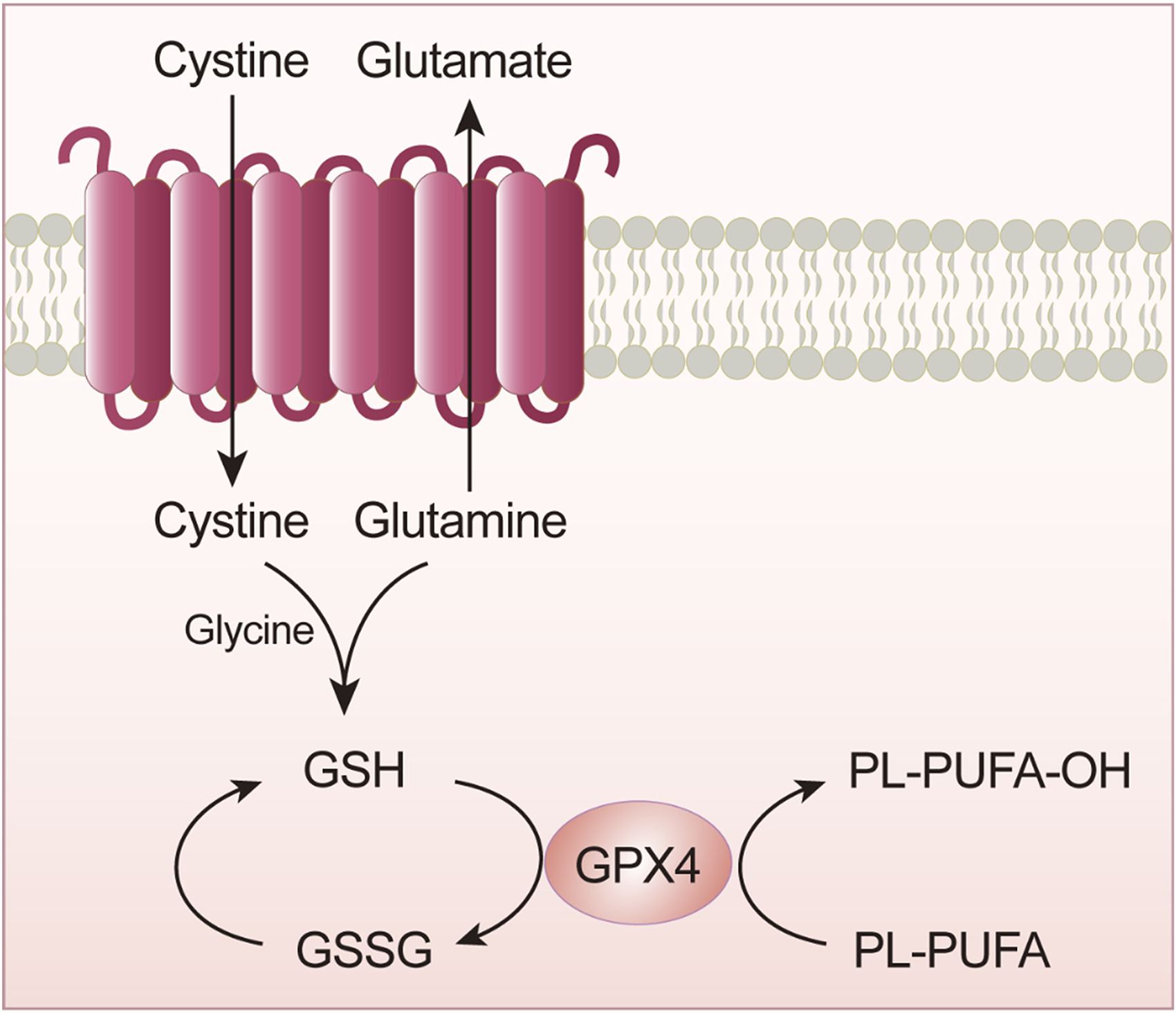
GPX4 inactivation leads to GSH depletion.
Iron Ion Influx and Reduction
Intracellular hydrogen peroxide reacts with free ferrous ions to form stable ferric ions and hydroxyl-free radicals. Iron ions are internalized into cells, guaranteed large quantities of the divalent iron, which can initiate liposomal peroxidation through the Fenton reaction. 23 During ferroptosis, a large amount of free Fe2+ is produced. Free Fe2+ shows high oxidizing ability and with H2O2 is protein to the Fenton reaction, which results in energy for the synthesis of DNA, proteins, and membrane lipids. 24 Hydroxyl radicals cause oxidative damage and promote lipid peroxidation damage to the cell membrane, leading to cell death 25 (Figure 5).

Imported iron ions into cells ensure large quantities of divalent iron, which initiates liposome peroxidation through the Fenton reaction. Oxidative damage leads to the leakage of liposome membranes.
Lipid Metabolism in Regulating Ferroptosis
Lipid metabolism leads to the accumulation of lipid peroxides and induces ferroptosis. 5 Dysregulation of lipid metabolism is the main cause of ferroptosis. In normal cells, lipid oxidation and reduction are regulated to maintain balance. As cells become cancerous, the expression levels of redox-related genes are upregulated, resulting in the accumulation of oxidized lipids in cells and the induction of ferroptosis. 26 Studies have shown that ferroptosis suppressor protein 1 is in a class of ferroptosis-inhibiting proteins that mediate the reduction of lipids, regenerating coenzyme Q10, and upregulating nicotinamide adenine dinucleotide phosphate oxidase can significantly increase the levels of oxidative-free radicals in cells and inhibit ferroptosis.19,27
p53-Mediated Ferroptosis
The p53 gene is a common tumor suppressor gene, and its mediation of cell cycle delays, senescence, and apoptosis plays important roles in the occurrence and development of various tumors. 28 Activation of p53 has been found to be required for ferroptosis in certain cancer cells. 29 In addition, p53 target genes, such as p21 and BAX, are involved in antiproliferative and proapoptotic activities, and ferroptosis mediates hyperactive p53 signaling to promote cell death.28,30
Other Pathways That Regulate Ferroptosis
In addition to the main mechanisms that induce ferroptosis, other pathways regulate ferroptosis. Under oxidative stress conditions, methionine is converted to cysteine through a transsulfuration pathway that leads to GSH synthesis. 31
Testing for Cell Death
Cell Counting Kit-8 for Assessing Cell Viability
Summary of Current Detection Indicators.

Abbreviations: CCK-8, cell counting kit-8; MDA, malondialdehyde; ROS, reactive oxygen species; TBA, thiobarbituric acid; GSH, glutathione; GLU, glucose; GR, glutathione reductase.
Iron Level Measurements
ROS Level Measurements
PCR/Western Blot Detection
Observation of Cell Morphology by Transmission Electron Microscopy
Mitochondrial Membrane Potential Detection
The mitochondrial membrane potential can be assessed with fluorescent dyes used to label active mitochondria; the fluorescence intensity decreases when the mitochondrial membrane potential decreases, as the fluorescence intensity is positively correlated with ferroptosis. 39
Cancer Cell Death Reveals the Role of Ferroptosis in Gastric Cancer
Overview
Iron is an essential element in humans, but only a small amount is absorbed by the body.16,40 Iron content in the body is usually assessed by measuring the free iron level, as 30% of iron is stored. 41 A study showed a link between body iron status and the risk of stomach cancer. 34 Moreover, ferritin is considered to be the molecule best suited for storing iron in the body. High iron levels increase the risk of cancer. 37 It is speculated that the lesions formed early in gastric cancer cause bleeding. 42
The Mechanism of Ferroptosis in Gastric Cancer Cells
Ferroptosis-Related RNA is Involved in the Occurrence and Development of Gastric Cancer
To date, preclinical studies have provided increasing evidence showing that abnormal regulation of ferroptosis-related microRNAs, long noncoding RNAs (lncRNAs), and circular RNAs (circRNAs) is closely related to the occurrence and development of gastric cancer. For example, miR103a-3p, as an oncogene with upregulated expression in gastric cancer and is closely associated with patient prognosis. 43 Moreover, miR103a-3p regulates ferroptosis in gastric cancer cells by altering intracellular GSH levels. In addition, currently available local anesthetics, such as bupivacaine, inhibit the proliferation of gastric cancer cells by inducing ferroptosis mediated via the miR-489-3p/SLC7A11 axis. 44 Ferroptosis-related lncRNAs and circRNAs are also involved in the occurrence and development of gastric cancer. It has been reported that lncLASTR mediates the proliferation and migration of gastric cancer cells by regulating ferroptosis. 45 Circ0000190 overexpression in gastric cancer cells inhibited the proliferation and migration of gastric cancer cells by inducing ferroptosis. 46 Ferroptosis-related RNA is involved in the expression of gastric cancer cells to varying degrees, affecting the occurrence and development of gastric cancer and suggesting a role for these RNAs in clinical applications for the diagnosis, treatment, and prognosis of gastric cancer.
The Regulation of Ferroptosis Functional Proteins Mediates the Occurrence and Development of Gastric Cancer
In the pathogenesis of gastric cancer, many functional proteins directly involved in the regulation of ferroptosis undergo significant changes. Recent studies have revealed that several functional proteins directly involved in the regulation of ferroptosis are involved in the occurrence and development of gastric cancer. For example, cysteine dioxygenase 1 plays an important role in the ferroptosis of gastric cancer cells. It can upregulate the expression of GSH and GPX4 in cells and inhibit ferroptosis in gastric cancer cells. 47 The low expression of cytoplasmic polyadenylation element-binding protein (CPEB) has been found to be associated with gastric cancer metastasis. 25 The low expression of CPEB can activate transcription factors that inhibit GSH-specific transferases, which are key to ferroptosis. By regulating functional proteins related to ferroptosis, the expression of related factors that participate in the occurrence and development of gastric cancer can be activated or inhibited, thereby regulating the expression of other related functional proteins, which is expected to provide new ideas for clinical diagnosis and treatment.
Ferroptosis is Involved in the Formation of the Gastric Cancer Microenvironment
Studies have found that malignant tumor cells such as gastric cancer cells and their surrounding microenvironment can be established through different rates of ferroptosis activation. 48 Moreover, there is evidence showing that macrophages, which maintain microenvironmental stability, can maintain the iron balance in the human body and are thus important participants in gastric cancer, promoting the progression of gastric cancer by increasing the activity of the metabolic pathways involved in ferroptosis. 38 Therefore, the macrophage content and iron levels in the gastric cancer microenvironment can be measured to provide a potential basis for better prediction of tumor progression.
The Clinical Value of Ferroptosis in the Detection of Gastric Cancer
Cell activity, iron metabolism, and GSH metabolism are determined by measuring iron ions (particularly ferrous ions), ROS, growth hormone, GSH levels, etc. To understand the relevant molecular indicators and evaluate the tumor molecular indicators related to gastric cancer, we objectively reported the levels of ferroptosis-related detection indicators in the stage evaluation and progression of gastric cancer. 48 The cytotoxicity of erastin at low doses in gastric cancer cells has been demonstrated; it results in ROS accumulation, reduced cellular activity, and ATP production, leading to cell death. 49 Perilipin2, also known as adipose-differentiation-associated protein, is significantly overexpressed in gastric cancer tissues, as determined by immunohistochemistry.27,33 The measurements of related protein expression levels provide references for finding potential therapeutic targets and regulatory genes in gastric cancer and ideas and directions for future antigastric cancer drug development.23,50
Problems and Perspectives
There are many questions to be answered and areas to be explored; they are summarized herein.
How can downstream signaling genes in ferroptosis or key pathways in iron-dependent ROS production be identified and their expression regulated? 50
What is the specific role of iron during ferroptosis?
It is necessary to further clarify whether GXP4 is an oncogene. 51 Second, immunotherapy therapy is a relatively new antitumor treatment method, and its relationship with ferroptosis is in the preliminary exploration stage. The related mechanisms are not completely clear, and further research is therefore needed. 52
There is still a lack of specific markers that can be intuitively used to detect ferroptosis.
How is ferroptosis related to other physiological processes in cells? 51
Tumor cells in different tissues vary widely in their sensitivity to ferroptosis and to ferroptosis-inducing agents. 36 Therefore, finding indicators that can reflect the sensitivity of cells, particularly in patients, and developing new ferroptosis-related drugs are of great significance for tumor treatment. 53 Ferroptosis is a newly discovered form of cell death.
This review summarizes the basic definition of ferroptosis and the mechanisms underlying ferroptosis. Progress in the research on ferroptosis in gastric cancer is presented. 54 Although it can be called GPX4-controlled cell death, ferroptosis is not regulated cell death in the traditional sense. 55 Research on the mechanism of ferroptosis has focused on the effects of oxidative damage, the direction of iron metabolism regulation, and related targets.47,56 Studies have shown that cell sensitivity to ferroptosis can be changed by regulating the intracellular iron content, and the combination of ferroptosis therapy with radiotherapy, chemotherapy, and targeted therapy for gastric cancer has shown good therapeutic effects in vivo and in vitro.16,57,58 Identifying the downstream signal transduction or iron-dependent ROS production pathways using specific indicators would further clarify the ferroptosis mechanism and distinguish ferroptosis from other types of cell death, which has recently become a research focus, and the ferroptosis mechanism can be distinguished from other mechanisms involved in iron metabolism, oxidative damage, and other diseases-related processes to suggest new research ideas and treatment, which shows important clinical significance.28,59,60 The sensitivity of gastric cancer cells to ferroptosis is controlled by mechanisms that regulate the consumption of GSH and ROS by ferroptotic pathways, inhibiting the proliferation of gastric cancer cells. 61 Moreover, ferroptosis is involved in both drug resistance and immune responses in the treatment of gastric cancer. All of these factors may make ferroptosis a new target in the treatment of gastric cancer.20,62 In-depth studies and clarification of the pathophysiological mechanism of ferroptosis in diseases can provide new ideas for finding drug targets and for clinical prevention and treatment.
Conclusion
Ferroptosis is a form of programmed cell death that is distinct from necroptosis and other death modalities, as indicated by differences in cell morphology, pharmacology, and gene expression. 30 Ferroptosis is a hot research topic, and many studies have been carried out on the biological mechanism of ferroptosis. However, the relationship between ferroptosis and gastric tumors needs to be further studied, especially in the fields related to gastric cancer. 63 Gastric cancer is the second leading cause of cancer morbidity and mortality in China. The mechanisms of gastric cancer occurrence and development and its diagnosis and treatment have attracted considerable attention. 64 Since the concept of ferroptosis was established, increasing evidence has shown that the induction and regulation of the ferroptosis pathway can effectively activate anticancer mechanisms and thus regulate cancer-related protein activity. 65 Moreover, ferroptosis plays a key role in human health. The tumor cell population is highly heterogeneous. An in-depth study into the general sensitivity of gastric cancer tumor cells to ferroptosis not only will increase the number of treatment methods for gastric cancer but will provide guidance and reference for resolving clinically refractory gastric malignant tumors through combination therapy. 66 Based on recent research hot spots, we expounded the definition, characteristics, regulatory mechanism, and progress of the research into ferroptosis in gastric cancer with the aim of facilitating the discovery of potential therapeutic targets and regulatory genes in gastric cancer by gaining a greater understanding of the regulatory mechanism of ferroptosis and related proteins; ultimately, we hope to provide a novel basis for the development of gastric cancer therapies. 67 The review provides a reference for diagnosis and treatment and ideas and directions for understanding the mechanisms underlying the occurrence and development of gastric cancer and the research and development of antigastric cancer drugs in the future. 68
In the near future, ferroptosis will be leveraged in the clinical treatment of human diseases. 69 More research is needed, especially studies on the tumor microenvironment and tumor immunity-related fields, to increase the understanding of ferroptosis. Ferroptosis is associated with gastric cancer, and studies have revealed that some tumor cells can be induced by iron concentration and that gastric cancer cells can leverage iron to inhibit tumor growth by inducing ferroptosis.45,68,70 Therefore, in the near future, ferroptosis will be applied to the clinical treatment of human diseases.
Footnotes
Abbreviations
Author Contributions
Zaibo Zhang wrote the paper. Bingyu Wang made important revisions to the paper. Wenbo Liu collected the literature. Bibo Tan approved the final version of the paper for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
