Abstract
Study design
Circulating tumor cells is important in the clinical diagnosis of cancer and there are a number of circulating tumor cell detection systems associated with different isolation strategies being validated. There is a novel platform, the CytoBot 2000, which utilizes a combination of physical and immunological technologies to isolate and capture circulating tumor cells.
Methods
In this retrospective study, 39 lung cancer patients and 11 normal healthy individuals were enrolled and performed circulating tumor cell tests and immunofluorescence staining with CytoBot 2000. The performance of this device was assessed by receiver operating characteristic curve. The clinical relevance of circulating tumor cells was assessed by Chi-square. The correlations between circulating tumor cell number and blood lymphocytes and tumor biomarkers were analyzed by Pearson correlation coefficient.
Results
The number of circulating tumor cell is significantly increased in lung cancer patients (3.74 > 0.45,
Conclusions
This automatic platform showed excellent performance of circulating tumor cell detection by clinical sample. The tumor biomarkers increased with the number of circulating tumor cell in the lung cancer patients.
Introduction
Lung cancer (LC) is one of the most common cancers and is the leading cause of death from cancer. In 2020, almost 1 796 144 people died from LC, and this number is expected to increase to 238 million in the next 10 years (GLOBOCAN 1 ). LC has poor outcomes—it has a 5-year survival rate of 10% to 20%, 2 depending on the stage at diagnosis. However, for patients with advanced LC, therapy is of limited benefit. Hence, there is an urgent demand in the earlier diagnosis of LC with improved performance over tissue biopsy.
Liquid biopsy is a noninvasive and easily available method for cancer screening. 3 Numerous human body fluids and discharges are available for clinical diagnosis of cancers, such as blood, cerebrospinal fluid, saliva, pleural, ascites, stool, and urine. 4 In LC, peripheral blood (PB) is an optimal body fluid for liquid biopsy. The indicators detected in LC screening include circulating tumor cells (CTCs), 5 circulating tumor DNA (ctDNA), miRNA, blood tumor biomarkers, and microvesicles. 6
There has already been a lot of research in the detection of CTCs. 7 CTCs are disseminated from the tumor primary site or a metastatic site into the bloodstream. 8 The mechanisms by which CTCs evade immune cells are unknown. Many studies have reported that CTCs have a significant value in LC diagnosis and prognosis. Li et al 9 reported that the CTC number in LC patients is more than in healthy volunteers and patients with other lung diseases. Laura et al 10 confirmed that when the CTC baseline was greater than 5 per 7.5 mL blood, LC patients experienced worse progression-free survival and overall survival rates. Guo et al 11 found that there was 90% specificity but 52.94% sensitivity in malignant LC diagnosis with CTC detection by the platform of CellCollector, and not obtained CTCs from healthy normal.
At present, there are numerous platforms for CTC detection. Cellsearch, the first Food and Drug Administration (FDA) approved platform, isolates CTCs from peripheral blood mononuclear cell (PBMC) using the positive enrichment strategy by magnetic beads that EpCAM-conjugated and identifies CTCs using the biomarker of cytokeratin (CK). 12 ISET, a size-depend filtration platform that isolates CTC based on the expansile diameter of tumor cells and identifies CTCs using the method of cytopathological staining (hematoxylin–eosin). 13 By OncoQuick, which isolates cells based on a differential density gradient, the CTCs were identified by LUNX mRNA using reverse transcription polymerase chain reaction. 14 Microfluidics, an advanced technique, is increasingly used in liquid biopsy, such as microfluidic chip-based isolation of exosomes and extracellular vesicle for cancer diagnosis 15 and personalized medicine. 16 Magnetic affinity is one of the mostly used methods in microfluidic system that magnetic beads conjugated with anti-EpCam (E-type CTC) or anti-CD45 are mixed with blood samples for positive or negative enrichment, respectively. The sample mixture then passes through the microfluidic chip (Isoflux) for target separation. 13 Alternatively, the CTC is directly captured by the antibodies that attached to the wall of the microfluidic chip. 17 Obviously, the platforms that based on the microfluidic technique have improved the capture rate of CTCs, such as Parsortix 18 and Clearbridge system. 19 But the challenges of these platforms in clinical practice remain, like the limited throughput in clinical tests, time-consuming, and automaticity.
Here, we introduce a novel CTC isolation and enrichment system, CytoBot 2000. In this device, the CTCs isolation was performed using the combined strategies (physical and immunological) by an antibody-coated chip that with multichannels that improved the possibility of CTCs capture. Different from other microfluidic systems, only 4 mL of blood samples was needed and the whole process of CTC's capture finished in 10 min, then followed with immunofluorescence (IF) staining on the chip automatically. The determination of CTC tests will available in half day. In this study, we retrospective assessed the performance of this platform and compared our results with those of other platforms.
Materials and Methods
The reporting of this study conforms to STARD guidelines 2015. 20
Patients
In this study, 39 LC patients and 11 healthy subjects were randomly enrolled from September 2019 to November 2021. Patients’ inclusion: (1) ages > 18 years old; (2) confirmed LC association by clinical diagnosis; (3) without any treatment of anti-cancer drugs in recent 5 years; (4) without other kinds of cancers; (5) no history of infectious diseases, such as tuberculosis, hepatitis B, HIV, and syphilis; and (6) patients agree informed consent form. Cancers in the LC patients (33 glandular, 5 squamous, and 1 non-small cell) were confirmed through clinical examinations, including histopathological diagnoses (tissue biopsies) and ultrasound. The clinicopathological features, including cancer stage, metastasis, histological information, brain metastasis (BM), and smoking history of LC patients are presented in Table 1.
Baseline of Patients.

We obtained informed consent from all study participants. We declare that this study followed the Declaration of Helsinki (2013) and has been approved by the Ethics Committee of our institutions. All patient details have been de-identified.
Sample Collection
Peripheral blood (4 mL) was collected into vacutainer tubes containing EDTA (BD) from LC patients and healthy individuals, the blood from patients obtained prior to surgery and treatment. Blood samples were saved at 4 °C and processed within 6 h.
Cell Lines and Spiking Assay
Commercial cell lines of LC, A549 (iCell-h011) and 95-D (iCell-h233), were purchased from iCell (Shanghai, STR verified). Cells were cultured in RPMI-1640 (iCell-0002) medium, which contains 10% of fetal bovine serum (FBS) and 1% of penicillin and streptomycin, maintained with 5% CO2 at 37°C and 80% humidity.
For spiking assay, A549 and 95-D cells were digested with 0.25% of trypsin and then followed with centrifugation (1000 rpm) and resuspension. Cells were counted at the number of 5 to 10 and/or 25 to 30 with cell counter, and these cells were spiked in the 4 mL of whole blood. Then, the procedures of sample treatment and CTC enrichment were followed as below.
Lymphocyte and Tumor Biomarkers Test
Lymphocyte test
A 2-color direct IF method is used. Fluorescein-labeled CD3 monoclonal antibody (BD) was added to 100 μL of whole blood, well mixed, removed from light, and incubated at room temperature for 20 to 30 min to bind to the relative antigen on the leukocyte membrane. Hemolysis is performed by adding the red cell lysate. Wash and perform the assay with flow cytometry (BD). The neutrophile cells of whole blood were also analyzed using this method but with fluorescein-labeled CD16 monoclonal antibody (BD). The enzyme-linked immunosorbent assay for carcinoma embryonic antigen (CEA) and CA211 tests using LC patient's blood.
Briefly, blood was naturally clotted at room temperature for 10 to 20 min, then centrifuged for 20 min (2000-3000 rpm), and the supernatant was collected. Add 10 μL of the sample to be tested (5-fold dilution) and incubate at 37 °C for 30 min. Wash 3 times with ultra-pure water and then incubate for another 30 min with 50 μL of enzyme reagent. A color developer was added and incubated for 15 min at 37 °C. The reaction was finally terminated by the addition of termination solution and the absorbance (OD) of each well was measured at 450 nm.
CTC
Sample preparation and detection
The 4 mL of blood sample was transferred into 15 mL sterile centrifuge tube (Corning) and PBS added follow (4 mL, pH = 7.0, room temperature) (Biological Industries). The mixture was transferred into 15 mL sterile centrifuge tubes containing density gradient separation solution (4 mL) and then centrifuged at 700×
The PBMCs were resuspended with PBS at 300 µL and transferred to a unit of sample loading and performed CTC enrichment by CytoBot 2000 (as showed in Supplemental Figure 1A). Mechanismly, a functionalized capture chip with multichannels coated with a flexible polymer layer and anti-EpCam (Abcam) antibody was manufactured from Holosensor Medical Ltd, insert into the unit of sample loading. Once the program starts, first, most cells of PBMCs were passed the chip via the channels with dimensions of 15*20 μm, and meanwhile, the EpCam-positive cells in PBMCs were arrested by the capture chip. Then, the subprogram of IF assays was followed. The unit of sample loading was transferred to the IF kit by a robot arm, the cells-adherent chip was infiltrated with solutions of fixation (4% paraformaldehyde, P1110, Solarbio), permeation (Triton X-100, ×100-100 mL, Sigma), staining reagents and washes successively, as procedure of IF assay. Staining reagents (pan-CK ab215838 (Abcam), CD45 ab8216 (304001, Biolegend), and DAPI D9542 (Sigma)). When the program finished, the results of CTC test were observed by a fluorescence microscope (RX50M, Sunny Optics).
Statistical Analysis
Statistical analysis was performed with SPSS 24.0 (SPSS Inc) and Prime GraphPad 6.0 (GraphPad Software). Receiver operating characteristics curves were constructed for the assessment of LC diagnosis. The cutoff was determined by Youden Index, the Youden index = sensitivity + specificity-1. Unpaired
Results
CTC Detection in LC Patients and Analysis of Clinical Relevance
Before testing by clinical samples, spiked in assays were performed using commercial cell lines; 5 to 10 and 25 to 30 of A549 and 95-D cells were spiked into 4 mL PB, and the cell recovery rates were assessed. The data showed that the cell recovery rate of 75% and 78% when 5 to 10 and 25 to 30 of A549 cells spiked, respectively; in 95-D, the cell recovery rates of 68% and 75%, respectively (Supplemental Figure 1B and C). Then, the blood samples from 39 LC patients and 11 healthy were tested. As previously, the CTCs were identified with pan-CK+ CD45− DAPI+, and white blood cells (WBCs) were pan-CK− CD45+ DAPI+ (Figure 1A). CD45+-CTCs were also detected (double positive CTC, dual-positive CTC) (Figure 1A, right). CD45+-CTCs are reported to be fusions of tumor cells and macrophages, which fuel cancer metastasis. 21

Circulating tumor cell (CTC) count in lung cancer (LC) patients and healthy volunteers, and clinical relevance analysis. A total of 39 LC patients and 11 healthy volunteers were enrolled in this study, CTC was detected by CytoBot 2000. (A) Images of CTC, WBC, and dual-CTC indicated by red arrows, captured and stained by CytoBot 2000, pan-CK (green, arrow indication), DAPI (blue, arrow indication), and CD45 (red, arrow indication). 20×, scale bar = 20 μm. (B) Comparison of CTC count between LC patients and healthy volunteers. (C-E) Analysis of clinical relevance with CTC count. WBC, white blood cell; dual-CTC, double positive CTC (pan-CK + CD45+). Data are shown as mean + SEM, unpaired
The number of CTCs in LC patients (3.75; 95% CI, 1-9;
CTCs Count and Clinicopathologic Feature.
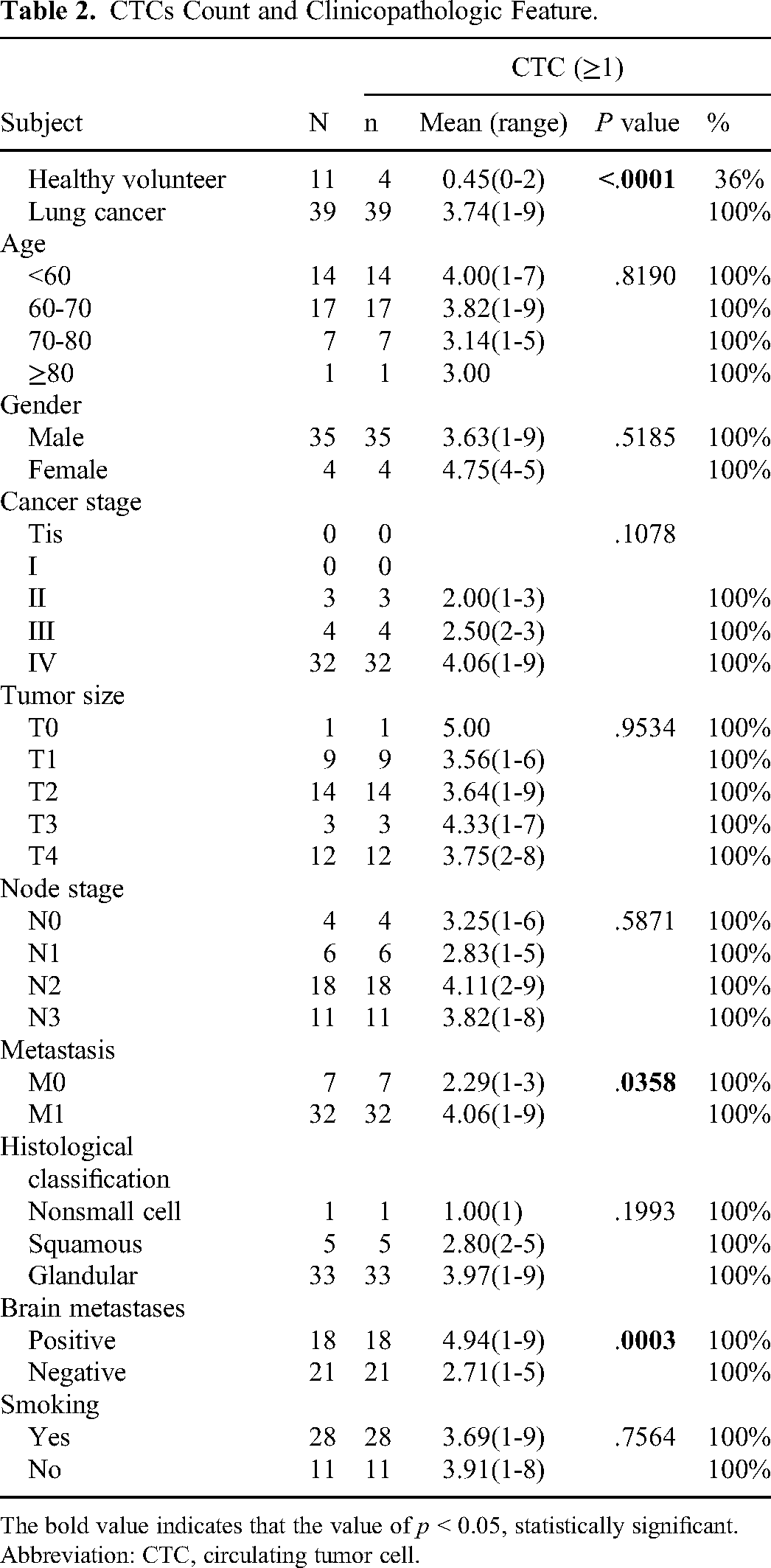
The bold value indicates that the value of
Sensitivity and Specificity of CytoBot 2000 in CTC Detection
A comparative analysis of data from healthy individuals and LC patients demonstrated that the system has excellent sensitivity and specificity for CTC enrichment (Figure 2A), with an AUC of 0.966. We determined the optimal cutoff value to be 1.5 by determining the Youden index. As shown in Table 3, when the cutoff is 1.5, the maximum Youden index is 0.806, and the sensitivity and specificity of the device for capturing CTCs are 89.07% and 90.9%, respectively. In combination with the CTC levels and clinical diagnosis, we think that a definite risk of LC association may propose when CTC ≥ 2 by this platform.

Performance of circulating tumor cell (CTC) detection by CytoBot 2000 and clinical relevance of CTC positive rate. (A) Receiver operating characteristic (ROC) of CTC detection by CytoBot 2000. (B) CTC positive rates in lung cancer (LC) patients and healthy volunteers when the cut-off was set as 2. (C, D) CTC positive rates in different tumor stages of LC patients when the cut-off was set as 2. Chi-square test was performed, *
Youden Index and Cutoff Determination.

The bold value indicates that the optimal pick of cut-off value in this study.
We analyzed the levels of CTCs at each stage of LC development at a cutoff value of 2. The results showed that the positive rate of CTCs in LC patients was 90% (35/39), which was significantly higher than healthy individuals of 9% (1/11) (95% CI,

Correlation of leukocyte levels and circulating tumor cell (CTC) counts in lung cancer (LC) patients. (A) Overview of leukocyte (brown), neutrophil (green), and CTC (red) in 39 LC patients. Comparison of leukocyte and neutrophil in II to IV tumor stages (B) and BM (brain metastasis) (C). Data are shown as floating bars (min to max, line at mean), 1-way analysis of variance (ANOVA),
CTCs Positive Rate in Patient's Subject.

The bold value indicates that the value of
Correlation Between Blood Leukocyte Levels and CTCs
We examined the levels of total lymphocytes (brown) and neutrophils (green) in the 39 LC patients by routine blood test (Figure 3A). Correlation and linear analyses showed that lymphocytes in the blood of LC patients did not increase with cancer progression (95% CI,
Correlation Analysis Between Blood Tumor Biomarkers and CTCs
Blood levels of CEA (Figure 4A, orange) and CA211 (Figure 4A, blue) were measured simultaneously in the LC patients. The levels of blood tumor biomarkers did not increase significantly with cancer progression (95% CI,
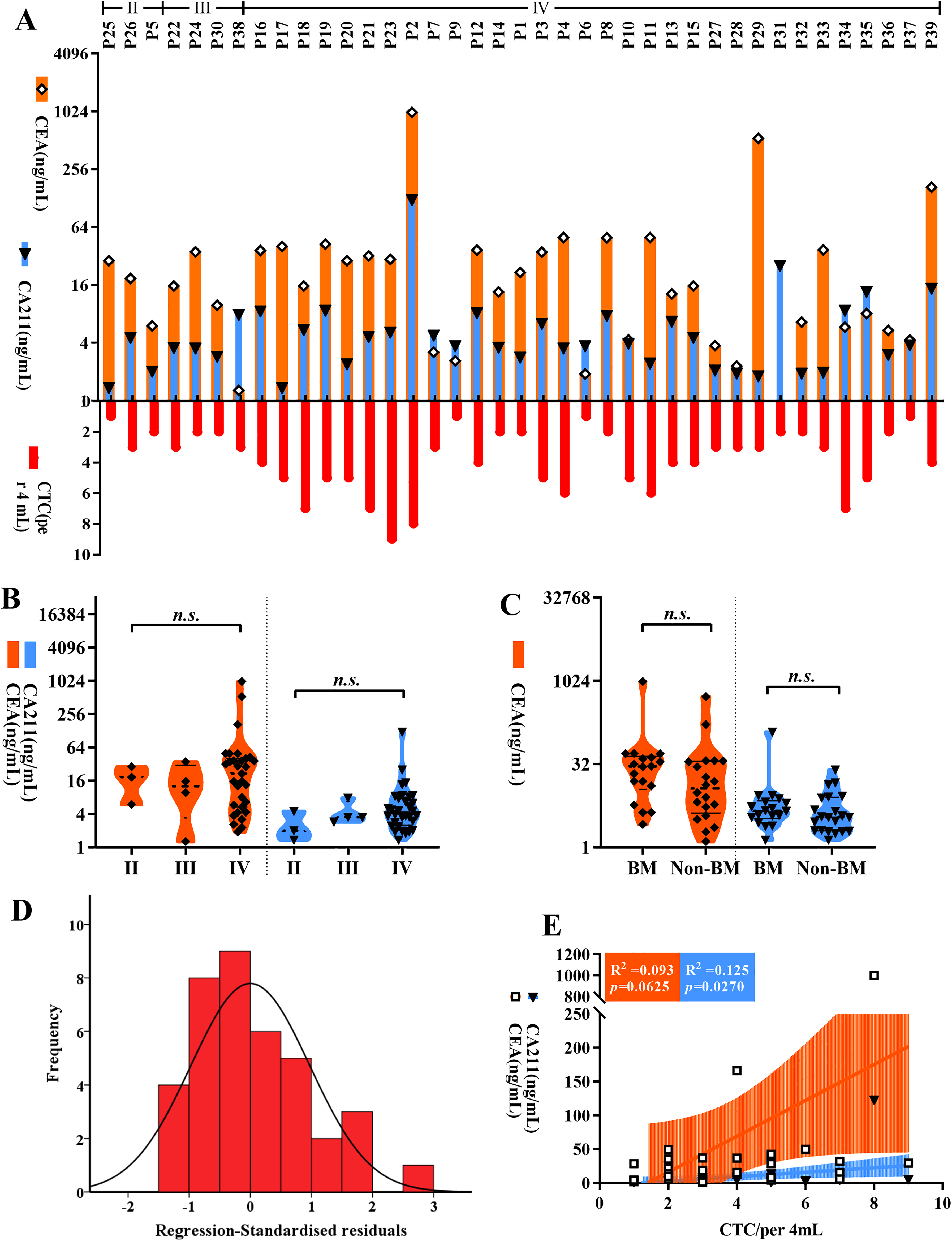
Correlation of tumor biomarkers and circulating tumor cell (CTC) counts in lung cancer (LC) patients. (A) Overview of carcinoma embryonic antigen (CEA) (orange), CA211 l (light blue), and CTC (red) in 39 LC patients. Comparison of CEA and CA211 in II to IV tumor stages (B) and BM (brain metastasis) (C). Data are shown as floating bars (min to max, line at mean), 1-way analysis of variance (ANOVA),
Discussion
The traditional methods of clinical testing for the presence of cancer are limited by sensitivity, the likelihood to cause physiological damage, the need for experienced practitioners, and higher costs. 22 The monitoring of CTC levels has become an important biomarker screen for cancer detection and diagnosis. 13 CTC tests are minimally invasive and have shown success in cancer detection and diagnosis. 3 In addition to detecting CTC levels, ctDNA, blood protein, and miRNAs are potential indicators in the bloodstream. The value of ctDNA in advanced LC diagnosis is well established. 23 It has been reported that the ctDNA test in LC patients has a sensitivity of 59% and a specificity of 99%. 24 However, the American Society of Clinical Oncology and College of American Pathologists declare that there is not enough evidence to demonstrate that ctDNA testing is useful in cancer screening and treatment monitoring, especially in early-stage tumors. 25 For example, the sensitivity of ctDNA testing in stage I non-small cell lung cancer patients are 15%, and overall (I-III) sensitivity is 48%. 26 In the profiling of cancer-associated proteins, CEA, CA-125, and CK are the most used biomarkers. Victoria et al 27 demonstrated that the sensitivity and specificity of using CEA, CA-125, CK21-1, and the antibody NY-ESO-1 in the PAULA test are 71% and 88%, respectively. Gerard et al 28 assessed malignant lung nodules using a 2-protein expression ratio (LG3BP and C163A) and demonstrated a sensitivity of 97% and a specificity of 44%.
Compared with other biomarkers in blood-based liquid biopsies, significant strengths are obvious in CTC detection: (a) Increased sensitivity. Numerous studies have verified that the diagnoses using CTC levels are more accurate and sensitive than those based on X-ray or tissue biopsy; 29 (b) Tumor origin exploration. Ruan et al 30 collected unknown CTCs from cerebrospinal fluid and identified the tissue-specific genes using next-generation sequencing (NGS); (c) Tumor development monitoring. CTC count dynamic in clinical therapy is a valid indicator of tumor response; 31 (d) Variety of downstream techniques. CTC culture for drug sensitivity tests, NGS, proteomics, methylation, and mutation detection technologies are possible. 32 These advantages provide CTC platforms with the potential to improve clinical cancer therapies.
The CytoBot 2000 collects CTCs using a microchannel chip. WBCs can pass through the microchannels, and CTCs are stopped because they are oversize. In addition, the CTCs also captured by the surface of the chip with high affinity depend on the capture antibody, anti-EpCAM. This “physical + immunological” dual capture mode increases the rate of CTC recovery significantly. There are many kinds of strategies that are used for CTC enrichment, like magnetic beads-based CTCs isolation. Although the cell recovery rates of these technique-based platforms are much higher, the detection rates are unstable, like the CellSearch system (the only FDA-approved CTC detection system). Fumihiro et al 33 detected CTCs with a detection rate of 30% in LC and 12% in the benign population. The CellCollector system, which inserts an EpCam-conjunct wire into a cannula and then implanted into a blood vessel of cancer patients, increased the detectable rate of CTC but decreased patient compliance. Guo et al 11 performed a CTC test using Cellcollector, and the CTC detection rates were 10%, 45%, and 67% in benign, in situ, and invasive tumors, respectively. The phenotype of CTCs in blood is quite different and the isolation of these different CTCs depends on the system that epitope free. Such as the ISET® filtration system, which is size-dependent, performed CTCs isolation with a filter and is identified by HE staining that is easier for determination. Galatea et al 34 found CTCs in 57% of LC patients only by the ISET® system. The application of microfluidics has dramatically promoted the development of liquid biopsy. CTCs flow at a certain speed in the microfluidic channels while being caught by capture antibodies that coat the inner walls of the channels or are differentially separated by cell gravity. 35 The results from spike assays and a clinical sample of pleural mesothelioma performed on a microfluidic chip platform showed a CTC positive rate of 68.8%. 36 Another microfluidic chip platform detected CTCs in metastatic prostate cancer patients with a much higher positive rate of 93%. 17 However, the time consumption is longer because it has to ensure the interaction between CTCs and capture antibodies in microchannels with the lower flow velocity. The shortcomings are also present in the CytoBot system, such as the phenomenon of chip clogging. The CytoBot declares that the lymphocytes could be deleted by the microchannels on the chip with a dimension of 15*20 μm. But the fact is that there still have many lymphocytes on the chip when observed under the microscope. Despite the clogging is not observed in our experiments, the possibility still makes the final determination more difficult. Additionally, the panel of this system is smaller, with only 4 tests per program, which needs to be amplified.
Our results do not provide a statistical difference in CTC levels by LC stage. This is most likely due to the small number of stage II and III patients in this study. We did observe that CTC levels were increased in stage IV LC patients compared to the early stage (Figure 1 and Table 2). We were able to demonstrate that the levels of CTCs in tumor metastasis and BM groups were greater than those in nonmetastasis patients, which is consistent with the results of previous studies. 37
Lymphocytes are one of the most important components of blood and are essential in blood immunity. Tumor cells in the circulatory system become potential targets for lymphocytes. We measured blood lymphocyte and neutrophil levels in LC patients, and the results showed no significant difference in blood lymphocyte and neutrophil levels in LC patients at different TNM stages. In addition, the level of CTCs in the blood of LC patients did not correlate with the level of lymphocytes. Lymphocytes, which include a wide range of immune cells, may not change dramatically associated with cancer development. There may be specific implications for the analysis of the specialized differentiation of lymphocytes. Neutrophils are a type of lymphocyte that inhibits inflammation by secreting certain cytokines. 38 It has been shown that CTCs are associated with neutrophils in the blood and thus evade immune monitoring and improve metastasis. 39 It has been reported that the association between neutrophils and CTCs drives cell cycle progression in the blood and expands the metastatic potential of CTCs. 40 Special identification of CTC clusters with neutrophils by this device may provide strong evidence for this research.
CEA is a broad-spectrum tumor biomarker that, although it cannot be used as a specific indicator for the diagnosis of a certain malignancy, still has important clinical value in the diagnosis of malignancy, disease monitoring, and evaluation of outcome. 41 Routine blood tests revealed that the levels of CEA and CA211 increase with cancer progression (Figure 4). Correlation and linear analyses showed a positive correlation between the number of CTCs and the level of CA211. These suggest that the diagnostic role of CTCs in cancer is similar to molecular biomarkers in blood. Considering the low specificity of CEA concentration, serum CEA is also elevated in 15% to 53% of patients with inflammatory conditions, CTCs are a more efficient indicator in cancer diagnosis. Also, in clinical practice, the combined testing of multiple indicators can increase the accuracy of cancer diagnosis and reduce misdiagnosis. 42
Conclusions
In this study, we demonstrate the improved detection and performance of the CytoBot 2000 in assessing CTC levels for LC. These results indicate that CTC tests will play a significant role in the clinical diagnosis of cancer. Furthermore, there is great possible for applications in cancer cell interactions and reference for immunotherapy treatment by this automatic and highly reprogrammable platform.
Supplemental Material
sj-docx-1-tct-10.1177_15330338231167827 - Supplemental material for Enrichment of Circulating Tumor Cells of Lung Cancer and Correlation With Serum Leukomonocyte and Tumor Biomarkers: A Retrospective Study
Supplemental material, sj-docx-1-tct-10.1177_15330338231167827 for Enrichment of Circulating Tumor Cells of Lung Cancer and Correlation With Serum Leukomonocyte and Tumor Biomarkers: A Retrospective Study by Zhou-Fang Mei, Jing Yan, Ling Qian, Yan-Chao He, Jing-Jing Feng, Jie Wang, Zhi-Jun Jie and Hua Zou in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgments
Many thanks for help and cooperation from Holosensor Medical Technology Ltd in this work.
Authors’ Note
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Ethics Committee of Daping Hospital of Army Medical University (No: 2020209) and Shanghai Fifth People's Hospital of Fudan university (No: 2023001), individual consent for this retrospective analysis was waived.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chongqing Science and Health Joint Medical Research Project (No.2021MSXM327); the Key Department of Shanghai Fifth People's Hospital (2020WYZDZK01)
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
